Back to Journals » International Journal of Nanomedicine » Volume 11
Upregulation of Akt/NF-κB-regulated inflammation and Akt/Bad-related apoptosis signaling pathway involved in hepatic carcinoma process: suppression by carnosic acid nanoparticle
Authors Tang B, Tang F, Wang Z, Qi G, Liang X, Li B, Yuan S, Liu J, Yu S, He S
Received 25 November 2015
Accepted for publication 7 March 2016
Published 30 November 2016 Volume 2016:11 Pages 6401—6420
DOI https://doi.org/10.2147/IJN.S101285
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Lei Yang
Bo Tang,1,2,* Fang Tang,1,2,* Zhenran Wang,1,2,* Guangying Qi,3 Xingsi Liang,1,2 Bo Li,1,2 Shengguang Yuan,1,2 Jie Liu,1,2 Shuiping Yu,1,2 Songqing He1,2
1Department of Hepatobiliary Surgery, Guilin Medical University Affiliated Hospital, 2Laboratory of Liver Injury and Repair Molecular Medicine, Guilin Medical University, 3Department of Pathology and Physiopathology, Guilin Medical University, Guilin, Guangxi, People’s Republic of China
*These authors contributed equally to this work
Abstract: Primary liver cancer is globally the sixth most frequent cancer, and the second leading cause of cancer death and its incidence is increasing in many countries, becoming a serious threat to human health. Many researches focused on the treatment and prevention of liver cancer. However, due to the underlying molecular mechanism of liver cancer still not fully understood, the studies and development of treatments were forced to be delayed. Akt has been suggested to play an essential role in the progression of inflammation response and apoptosis. Hence, in this study, Akt-knockout mice and cells of liver cancer were used as a model to investigate the molecular mechanism of Akt-associated inflammatory and apoptotic signaling pathway linked with NF-κB and Bcl-2-associated death promoter (Bad) for the progression of liver cancer. Carnosic acid (CA), as a phenolic diterpene with anticancer, antibacterial, antidiabetic, as well as neuroprotective properties, is produced by many species from Lamiaceae family. Administration of CA nanoparticles was sufficient to lead to considerable inhibition of liver cancer progression. The results indicated that, compared to the normal liver cells, the expression of Akt was significantly higher in liver cancer cell lines. Also, we found that Akt-knockout cancer cell lines modulated inflammation response and apoptosis via inhibiting NF-κB activation and inducing apoptotic reaction. Our results indicated that the downstream signals, including cytokines regulated by NF-κB and caspase-3-activated apoptosis affected by Bad, were re-modulated for knockout of Akt. And CA nanoparticles, acting as Akt-knockout, could inhibit inflammation and accelerate apoptosis in liver cancer by altering NF-κB activation and activating caspase-3 through Bad pathway. These findings demonstrated that the nanoparticulate drug CA performed its effective role owing to its ability to reduce inflammatory action and enhance apoptosis for the overexpression of NF-κB and Bad via Akt signaling pathway, playing a direct role in liver cancer progression. Thus, nanoparticle CA might be an important and potential choice for the clinical treatment in the future.
Keywords: liver cancer, Akt-knockout, carnosic acid nanoparticle, inflammation, apoptosis
Introduction
Liver cancer, also known as hepatic cancer, is a cancer that originates in the liver. Higher rates of liver cancer occur where hepatitis B and C are common, including East Asia and sub-Saharan Africa.1 Viral infection with either hepatitis C virus or hepatitis B virus is the chief cause of liver cancer in the world today, accounting for 80% of hepatocellular carcinoma, with >600,000 deaths each year. The viruses cause hepatocellular carcinoma because massive inflammation, fibrosis, and eventual cirrhosis occur within the liver. The current treatment strategies of liver cancer include surgical resection, radiotherapy, and especially chemotherapy.2 However, many patients with liver cancer fail to respond to initial chemotherapy or acquire drug resistance during the following treatment.3 Normal cells divide as many times as needed and stop.4 They attach to other cells and stay in place in tissues. Cells become cancerous when they lose their ability to stop dividing, attach to other cells, stay where they belong, and die at the proper time. Normal liver cells will commit cell suicide (apoptosis) when they are no longer needed. Until then, they are protected from cell suicide by several protein clusters and pathways.5–7 However, as a disease with high mortality rate, we still have not had the most effective measures to treat it. Therefore, the development of novel therapies for liver cancer is urgently needed.
The Akt pathway is a signal transduction pathway that promotes survival and growth in response to extracellular signals.8 Activated Akt mediates downstream responses, including cell survival, growth, proliferation, cell migration, and angiogenesis, by phosphorylating a range of intracellular proteins.9 Studies have found that Akt pathway plays an important or protective role in many tumors, such as lung cancer, intestine cancer, and pancreatic carcinoma.10 Sometimes, the genes along these protective pathways are mutated, rendering the cell incapable of committing suicide when it is no longer needed. This is one of the steps that causes cancer in combination with other mutations. Thus, the Akt pathway is stuck in the position, and the cancer cell does not commit suicide.11,12 Akt can phosphorylate and activate the IκB kinase IKK-α, causing degradation of IκB and nuclear translocation of NF-κB where it promotes expression of cytokines, including interleukin (IL)-1β, IL-18, as well as tumor necrosis factor-α, accelerating inflammation response. And also, Akt negatively regulates pro-apoptotic proteins by direct phosphorylation.13 For example, phosphorylation of Bcl-2-associated death promoter (Bad), the Bcl-2 family member, causes translocation from the mitochondrial membrane to the cytosol. Akt phosphorylates caspase-9, preventing a caspase cascade leading to cell death, modulating apoptosis. Thus, the association of Akt and NF-κB and Bad signaling pathways plays a critical role in inflammatory formation and apoptotic development.14 However, it is still far to know the specific molecular mechanism of Akt in liver cancer. For these aforementioned reasons, we investigated the progression of liver cancer from Akt/NF-κB and Akt/Bad signaling pathway, revealing Akt-regulated inflammation and apoptosis during the development of liver cancer.
Carnosic acid (CA) has been known as a natural benzenediol abietane diterpene detected in rosemary and common sage.15 CA is used as a preservative or antioxidant in food and nonfood products, including toothpaste, mouthwash, and chewing gum. Currently, CA has been reported to have several properties in antitumor, such as colonic cancer, mammary tumors, as well as skin tumors, through regulating cancer cell growth, apoptosis, reactive oxygen species, and also Akt activation.16–20 And also, previous studies have suggested that nanoparticulate drug delivery systems have been known to better the bioavailability of drugs on intranasal administration compared with only drug solutions.21,22 In addition, a study has found that nanoparticulate drug delivery system for CA intranasal administration needed less amounts of administrations to induce the required pharmacological reaction due to its ability to locate on the olfactory mucosal region and supply controlled CA delivery for prolonged time periods.23,24 Generally, for the potential anti-inflammatory and pro-apoptosis of CA in cancer cells and easier absorbance of nanoparticles, CA nanoparticles could be used as a candidate, which is likely to improve anticancer effects. And the focus will be on evaluating the effect of CA nanoparticles on liver cancer cells in vitro and in vivo experiments through regulating inflammation response and apoptosis.
Hence, in this study, in the first part, we investigated whether Akt-knockout could affect liver cancer by interfering with NF-κB pathway activation as well as Bad signaling pathway from inflammation and apoptosis level, which might serve as a potential therapeutic target and prognostic marker for the treatment of liver cancer. And then, anticancer activity of CA nanoparticles was investigated to clarify the underlying molecular mechanism, providing an effective method to facilitate clinical treatment.
Materials and methods
Animals and CA preparation
Five-week-old BALB/c Akt-knockout mice and nude mice, purchased from Vital River Laboratories (Beijing, People’s Republic of China), a leading company in commercial production of laboratory animals in the People’s Republic of China, were used for the liver cancer cell lines MHCC97-H and Bel7402 xenograft model. An amount of 0.2 mL medium containing 5×106 MHCC97-H and Bel7402 was injected subcutaneously into the left and right posterior flank regions of each mouse. Mice were housed in a pathogen-free environment and tumor growth was examined every 1 week. After the tumor growing for 5 weeks, animals were assigned to intraperitoneal injection of CA nanoparticles (10 and 20 mg/kg) on the following 4 weeks. The CA nanoparticles were prepared according to a previous study.24 Briefly, CA nanoparticles were developed by ionotropic gelation technique using response surface methodology. CA loaded chitosan nanoparticles were prepared by ionic interaction with tripolyphosphate as per the experimental design. An amount of 0.1% (w/v) concentration of chitosan solutions was prepared by dissolving required quantity of chitosan in Krebs-Ringer Modified buffer with 1% glacial acetic acid. Simultaneously, CA was then dissolved in the second portion of the buffer containing 20% hydroxy propyl-beta-cyclodextrin. Then the two solutions were mixed together. The resulting solution was ultrasonicated using a very high intensity microprobe with a diameter of 3.2 mm and a 100 W high intensity processor (Microson XL-2000; VWR International, Bridgeport, NJ, USA). The probe was then immersed into the solution and ultrasonication was carried out to produce CA loaded chitosan nanoparticles. The nanoparticles produced were characterized immediately. Animals were euthanized and tumors excised and subjected to pathologic examination. All the animal experiments were reviewed and approved by the Review Board of Guilin Medical University. All the experiments were performed following the Care and Use of Laboratory Animals of the university.
Cell culture
Human liver cancer cell lines (MHCC97-H and Bel7402) and normal liver cells (hhl-5) purchased from American Type Culture Collection (ATCC, Manassas, VA, USA) were grown and maintained in Roswell Park Memorial Institute (RPMI) 1640 medium containing 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin. Recombinant lent viral particles encoding Akt and short hairpin RNA targeting Akt were produced, concentrated, and titrated. Cells were seeded at 3×104 cells/mL and infected with recombinant lent virus twice with an interval of 12 hours and incubated for 24 hours. After 24 hours, the medium was refreshed and the cells were cultured for another 24 hours.
ELISA measurement
At the end of experiments, blood was extracted from the eyeball, and the different serum concentrations of the inflammatory cytokines, such as tumor necrosis factor-α, IFN-γ, IL-2, IL-1β, IL-10, IL-18, and IL-6, were measured using enzyme-linked immunosorbent assay (ELISA) kits according to the manufacturer’s instructions (R&D Systems Inc., Minneapolis, MN, USA).
Inflammatory cell counts
The cell samples were centrifuged (4°C, 3,000 rpm, 10 minutes) to pellet the cells. The cell pellets were resuspended in phosphate-buffered saline (PBS) for the total cell counts using a hemacytometer, and cytospins were prepared for differential cell counts by staining with the Wright–Giemsa staining method.
Histopathologic examination of cells and tissues
Histopathologic evaluation was performed on mice and cells that were collected. Samples were fixed with 10% buffered formalin, imbedded in paraffin, and sliced. After hematoxylin and eosin staining, pathological changes of tissues were observed under a light microscope. Some of these samples were subjected to immunohistochemical staining with Akt, NF-κB and Bad antibodies (Cell Signaling Technology, Danvers, MA, USA) and this assay was performed by the Shanghai Zhenda biotechnology, Co., Ltd (Shanghai, China).
Immunofluorescence staining
After induction by conditioned culture medium, the cells were fixed in 4% paraformaldehyde, permeabilized with 0.1% Triton X-100 in PBS containing 0.5% bovine serum albumin (BSA) for 30 minutes. The cells were subsequently incubated with Akt, NF-κB, and Bad for 30 minutes, followed by labeling with Alexa Fluor 488-conjugated rabbit antimouse or goat antirabbit IgG antibody. The cells were viewed under a fluorescent microscope.
Apoptosis analysis by TUNEL
Apoptosis assay of samples was determined by terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) using an In Situ Cell Death Detection Kit, fluorescein (Hoffman-La Roche Ltd., Basel, Switzerland), according to the manufacturer’s protocol. The number of TUNEL-positive cells was counted under a fluorescence microscope. The percentages of apoptotic cells were calculated from the ratio of apoptotic cells to total cells counted. Tissue sections were counter-stained with hematoxylin and observed under microscopy. The experiment was performed independently three times for each cell line.
Western blotting analysis
Cell proteins were extracted using T-PER Tissue Protein Extraction Reagent Kit (Thermo Fisher Scientific) according to the manufacturer’s instructions. Protein concentrations were determined by BCA protein assay kit, and equal amounts of protein were loaded per well on a 10% sodium dodecyl sulfate-polyacrylamide gel. Subsequently, proteins were transferred onto polyvinylidene difluoride membrane. The resulting membrane was blocked with Tris-buffered saline containing 0.05% Tween-20 (TBS-T), supplemented with 5% skim milk (Sigma-Aldrich) at room temperature for 2 hours on a rotary shaker, and followed by TBS-T washing. The specific primary antibody, diluted in TBS-T, was incubated with the membrane at 4°C overnight. Subsequently, the membrane was washed with TBS-T followed by incubation with the peroxidase-conjugated secondary antibody at room temperature for 1 hour. The immunoactive proteins were detected by using an enhanced chemiluminescence Western blotting detection kit. Western blot bands were observed using GE Healthcare ECL Western Blotting Analysis System and exposed to X-ray film of Kodak.
Real-time quantitative PCR analysis
The total RNA isolated from individual mouse kidney with Trizol reagent (Thermo Fisher Scientific) following the manufacturer’s protocol was to evaluate mRNA expressions of Akt, NF-κB, Bad, IKK-α, IκB-α, IL-1β, IL-18, Bcl-Xl, Cyto-c, Apaf-1, caspase-9, caspase-3, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH). The reverse transcription were performed with SuperScript First-Strand Synthesis kit (Thermo Fisher Scientific). The primers used for Real-Time PCR are summarized in Table 1. All primer sequences were checked in GenBank to avoid inadvertent sequence homologies. They were designed and synthesized by BioGenes GmbH. Reactions were performed using SYBRGreen PCR master mix (Thermo Fisher Scientific, Waltham, MA, USA) in a BioRad iCycler iQ Detection System. As an internal control, GAPDH levels were quantified in parallel with target genes. Normalization and fold change for each of the genes were calculated using the 2-Delta Delta C (T) method. The sequences of these genes are displayed in Table 1.
  | Table 1 Primer sequences of RT-PCR analysis |
Secondary colony-forming assay
Briefly, cancer cells were suspended in 0.9% methylcellulose-based semisolid medium MethoCult H4100 (STEMCELL Technologies, Vancouver, BC, Canada). After 14 days, individual primary clones (450 cells) were trypsinized and replated in the same conditions to examine the secondary colony-forming ability for self-renewal.
Flow cytometry analysis
For flow cytometry analysis, the hepatocytes were obtained through shearing liver tissue, separating by collagenase type II (Thermo Fisher Scientific) digestion and suspended in RPMI 1640 medium (Thermo Fisher Scientific). Cell suspensions were centrifuged at 1,000 rpm for 5 minutes to remove cellular debris and impurities. Then, the hepatic mononuclear cells were harvested and resuspended in 70% percoll (Sigma). Mononuclear cells were collected from the interphase and washed twice in Hank’s buffer (Gibco, BRL). The experiments were performed according to the protocol of R&D kit systems (R&D Systems Inc.). Briefly, anti-CD4 FITC and anti-CD8 FITC antibody were added to the flow cytometry tube containing single-cell suspension, and these tubes were analyzed by Cytomics™ FC 500 MCL of Beckman Coulter, Inc (Sharon Hill, PA, USA).
Matrigel invasion assay
The cells in serum free media were seeded in the rehydraded matrigel invasion chamber with 10% FBS containing media added as chemoattractant. Cells were then allowed to incubate for 48 hours. The chambers were then removed and washed twice with PBS. The matrigel was cleaned with a cotton plug and cells were fixed with methanol and stained with crystal violet. The membranes were cut, removed, and then mounted with DPX mountant and observed under the Axio-Observer microscope.
Statistical analysis
Data are expressed as mean ± standard error of the mean. Treated cells, tissues, and the corresponding controls were compared using GraphPad PRISM (version 6.0; GraphPad Software, Inc., La Jolla, CA, USA) by a one-way analysis of variance with Dunn’s least significant difference tests. Differences between groups were considered significant at P<0.05.
Results
Akt is overexpressed in liver cancer cell lines and tissues
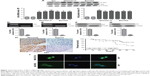
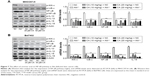
Here, we tested the situation of the Akt expression in liver cancer cell lines and normal liver cells. As it is shown in Figure 1A–C, we found that Akt is overexpressed in liver cancer cell lines compared to the normal cells via Western blot and reverse transcription-polymerase chain reaction (RT-PCR) analysis, demonstrating that Akt is likely to play an essential role in the progression of liver cancer. Thus, we aimed at Akt-knockout, and studied Akt expression in MHCC97-H and Bel7402 (Figure 1D and G). In order to certify Akt role on liver cancer, immunohistochemical staining analysis was used to demonstrate the difference between the normal group and tumor group, which showed higher survival percent of low Akt levels (Figure 1H). And, finally, immunofluorescence staining was performed to further indicate Akt expression in negative control (NC) group and Akt−/− group of MHCC97-H and Bel7402 (Figure 1I). These results suggested that Akt indeed expressed highly in liver cancer cell lines and after knockout of Akt, the survival percent of cells was improved compared to the control cells, suggesting that Akt could be taken as a target for the treatment of liver cancer.
The effects of Akt on the progression liver cancer
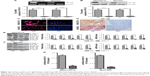
In this regard, matrigel invasion assay was applied to reveal the role of Akt-knockout in MHCC97-H and Bel7402 (Figure 2A–C), which showed us that Akt-knockout significantly decreases the number of invading cells. And also, loss of Akt activity attenuated liver cancer cell growth in vitro, and the cloned number of cells have been shown in Figure 2D and E. What is more, nude mice were used to test the role of Akt on cancer development directly, which indicated that compared to the NC group, mice treated with Akt-knockout of MHCC97-H and Bel7402 lines showed lower tumor volume markedly (Figure 2F). After treatment, we tested the cell counts of per miter. The results showed that the number of cell counts cultured from 12 to 96 hours was highly reduced in Akt−/− cells, both in MHCC97-H (Figure 2G) and Bel7402 (Figure 2H). The earlier results proved that Akt is of great importance for the progression and development of liver cancer.
The effects of Akt on NF-κB-regulated inflammation response
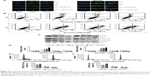
The treatment of knockout of Akt inhibited NF-κB expression via Western blot and RT-PCR analysis with significantly statistical difference compared to the NC group (Figure 3A and B). The immunofluorescence staining (Figure 3C) and immunohistochemical staining (Figure 3D) analyses showed to us that NF-κB-marked liver cancer cells were decreased in both the cell lines. And also, the transcriptional activity of NF-κB was determined by the luciferase assay when Akt was knocked out. And it is shown in Figure 3G, NF-κB transcriptional activity was reduced markedly compared to the NC group. In addition, the upstream and downstream signals of NF-κB were also determined via Western blot and RT-PCR, which demonstrated that cytokines, involving IL-1β and IL-18, were downregulated greatly, in accordance with the trend of NF-κB alteration (Figure 3E and F).
What is more, we tested the levels of inflammatory cytokines in the serum of animal models. As shown in Figure 4A, tumor necrosis factor-α, IL-1, IL-6, IL-10, IL-1β, IL-2, IL-18, IL-4, IL-17, IFN-γ, TGF-b1, TGF-b2, and VEGF were tested through ELISA kits. From the results, it was obvious that these cytokines were reduced significantly in Akt-knockout mice compared to the normal liver cancer mice. These results indicated that Akt regulated liver cancer progression via NF-κB activation and its downstream signals contributing to inflammation response.
And also, we investigated TLR4/MyD88 signaling pathway, contributing to the activation of NF-κB. The results showed significant reduction of TLR4 and MyD88 via Western blot and RT-PCR analysis in both MHCC97-H and Bel7402 (Figure 4B). In addition, the downstream signals of TLR4/MyD88 pathway were decreased, including IRAK1, IRAK4 as well as TRAF6 (Figure 4B and C), which were consistent with the expression trend of NF-κB and inflammation-related cytokines. In this regard, we finally tested the expression level of PI3K in both cells, displaying reduced phenomenon (Figure 4B and C). These results possibly suggest to us that TLR4/MyD88 signaling pathway was regulated, in turn, after the overexpression of inflammation-associated factors. And, then, activated PI3K phosphorylated Akt, lead to aggravation of inflammation response and apoptosis, which was likely to form a vicious cycle. Thus, knockout of Akt is related to the progression of inflammation response.
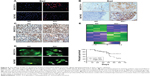
The effects of Akt on Bad-regulated apoptosis in liver cancer
We supposed that apoptosis accelerates the progression of liver cancer. Thus, Bad, which is essential to apoptosis, was tested via immunofluorescence staining (Figure 5A) and immunohistochemical staining (Figure 5B) analyses. The results showed that cells expressing Bad were higher in Akt-knockout cells. And also, mice without Akt expression displayed serious cell death in liver cancer cells, which is shown in Figure 5C and D. TUNEL assay was performed here to analyze the effects of CA on apoptosis (Figure 5C). Here, we found that the number of TUNEL-positive cells was higher in Akt-knockout cells. And also, the cell structure was damaged significantly (referred with white arrows in Figure 5D) in hepatocellular carcinoma cells without Akt expression. Hierarchical clustering of microarray data from MHCC97-H and Bel7402 cells treated with Akt-knockout were analyzed. And Kaplan–Meier survival plot of liver cancer animals comparing a positive correlation (green lines) with the Akt-knockout gene signature or negative correlation (blue lines) with the Akt-knockout gene signature was done as shown in Figure 5E, demonstrating that the gene signature produced by Akt-knockout conferred a marked improvement in survival.
Furthermore, RT-PCR and Western blot were used to analyze Bad expression (Figure 6A and B). The trends of Bad expression were consistent with immunofluorescence staining (Figure 5A) and immunohistochemical staining (Figure 5B), further indicating that Akt-knockout played a critical role in regulating Bad. On the other, we investigated the downstream signals of Bad, including Bcl-2, Cyto-c, Apaf-1, caspase-9, and caspase-3 (Figure 6C and D). Akt-knockout induced significant reduction of these genes displayed earlier, except the upregulation of Bcl-2, which plays an important role in anti-apoptosis in MHCC97-H and Bel7402 cells. Determined by Annexin V/ propidium iodide (PI) double staining analysis, we found that knockout of Akt could suppress overexpression of Akt-induced apoptosis in liver cancer cells, showing higher apoptosis ratio in indicated cancer cells (Figure 6E). And also, caspase-3 activation assay with the caspGLOW fluorescein active caspase-3 staining kit with fluorescence-activated cell sorting (FACS) analysis was applied to determine the effect of Akt on apoptosis development, showing that Akt-knockout cells displayed higher expression of caspase-3, which resulted in apoptosis in liver cancer cells (Figure 6F). These data demonstrated that Akt-knockout serves as a survival signal and induction of apoptosis in cancer cells.
The effect of CA on NF-κB pathway in Akt-deficient liver cancer cells
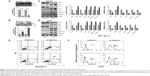
As shown in Figure 7A, we found that the phosphorylated expression of IKK-α and IκB-α was enhanced in MHCC97-H cells. And after the administration of CA, these activated protein expressions were downregulated, which is similar to the treatment of Akt-knockout. Also, p-NF-κB was inhibited by the use of CA, subsequently decreasing inflammation-related IL-1β and -18 expressions. We further investigated the results via RT-PCR analysis. From Figure 7A (left), IKK-α, IκB-α, NF-κB, IL-1β, and IL-18 mRNA expression were reduced after the treatment of CA with significant difference. However, there was no obvious difference in cells treated with Akt deficiency alone and Akt-knockout binding with CA. But there was no significant dose-dependent manner from the results. In addition, in order to certify the accurate effect of CA, the evaluation of these inflammatory factors was performed in Bel7402 cells, which was consistent with the results in MHCC97-H cells, both through Western blot (Figure 7B, right) and RT-PCR analysis (Figure 7B, left). The earlier results indicated that CA might have roles in inhibiting the expression of inflammation-related proteins and genes via NF-κB pathway in liver cancer cell lines.
The effect of CA on apoptosis in Akt-deficient liver cancer cells
In this regard, TUNEL analysis was used to analyze the apoptosis in liver cancer cells after the treatment of 30 μM CA. We found that the counts of cells detected via immunofluorescent method were lower with 30 μM CA compared with NC group in the cancer cell lines (Figure 8A). However, CA did not accelerate the reduction of cancer cells compared with Akt-knockout cancer cells. What is more, the degree of apoptosis in MHCC97-H cells and Bel7402 cells (Figure 8B) was investigated via Annexin V/PI double staining and representative flow cytometry profiles, and percentages of apoptotic cells were displayed in the lower right quadrant in which the number of apoptotic cells was significantly higher after the use of CA than that in NC group. Next, the effects of CA on cell proliferation were examined by Western blot. From Figure 8C and D, proliferating cell nuclear antigen (PCNA) was found to be downregulated for Akt deficiency. And also, after administration of CA in cells treated with or without Akt-knockout, PCNA protein expressions were also reduced, especially Akt-knockout combined with CA treatment together. However, P21, cyclin-dependent inhibitor (CKI), has been suggested to lead to the G1 arrest and proliferation inhibition of breast cancer cells.25 In our study, P21 was found to be upregulated for CA treatment in both two liver cancer cell lines, which was consistent with a previous study26 (Figure 8C and D). And conversely, Cyclin-D1 and Cyclin-D3, as two essential regulators for cell cycle, were also determined. The expression levels of Cyclin-D1 and Cyclin-D3 were lower in Akt-knockout mice and CA-administered mice compared to the NC group (Figure 8C and D), suggesting the role of CA in proliferation inhibition in liver cancer cells. In order to further confirm the role of CA in the cell proliferation suppression, flow cytometry assays were conducted to reveal the cell cycle. Interestingly, we found that CA treatment induced cell cycle arrest in G0/G1 phase in liver cancer cell lines. When compared to the NC group, the percentage of G0/G1 phase in Akt-knockout mice and CA-treated mice combined with Akt-knockout mice was increased significantly (Figure 8E and F). The results indicated that CA treatment might induce cell cycle arrest at G0/G1 phase. Taken together, our results demonstrated that knockout of Akt and administration of CA could induce apoptosis and inhibit proliferation of liver cancer cells.
The effect of CA on apoptosis via Bad pathway in Akt-deficient liver cancer cells
Bad pathway is essential to the development and progression of apoptosis. Thus, we tested the effects of CA on Bad signaling pathway by Western blot and RT-PCR analysis in different liver cancer cell lines shown in Figure 9A and B. From the results, we found that CA could inhibit the activation of Bad, reducing its phosphorylation and also cutting down Bcl-2 expression. Then, the protein expression of downstream signals, including Cyto-c, Apaf-1, caspase-9 and caspase-3, were elevated, leading to the occurrence of apoptosis in liver cancer cell lines. Furthermore, RT-PCR analysis suggested that mRNA expression of Bad, Bcl-2, Cyto-c, Apaf-1, caspase-9, and caspase-3 could be reduced from the gene levels after receiving CA both in 20 and 30 μM (Figure 9A and B [ii]). These suggested that apoptosis is promoted by CA in liver cancer cells.
The effect of CA on apoptosis in Akt-deficient liver cancer cells in vivo
To further assess the effect of CA on apoptosis, we determined its function in vivo by inoculating nude mice with tumor cells. After being treated with CA, the volume of tumors were markedly lower compared to the tumors formed in NC groups, indicating the induction of CA on apoptosis in liver cancer cells in vivo (Figure 10A and B). Consistently, induction of CA cut down tumor size (Figure 10A and B), which was much smaller compared with that in NC groups. Taken together, these results demonstrated that CA inhibits apoptosis of liver cancer cells in vivo.
Discussion
Liver cancer is a cancer that develops from liver tissue, making it the third leading cause of cancer death after lung and stomach. There is no doubt that liver cancer, as a critical illness syndrome, has been emphasized as a serious threat for the people of the world.2,27 Survival rates in the developed world are high. However, in developing countries, survival rates are poorer. Worldwide, liver cancer is the leading type of cancer in women, accounting for 25% of all cases. Moreover, the formation and development of liver cancer is associated with high morbidity and mortality.3,4 A previous study reported the application of some natural production to treat liver cancer in cultured cells, animal models and clinical investigations. This provided useful data for cancer research.5 However, the detailed underlying molecular mechanisms controlling liver cancer are still not fully explained. And we are looking for the most effective treatment for liver cancer-related diseases.
Akt is known to be implicated in several cancers, including glioblastoma, ovarian, pancreatic, and particularly liver. And studies also found that Akt is upregulated in terms of mRNA production in liver.6 First of all, human liver cancer cell lines (MHCC97-H and Bel7402) and normal liver cells (HHL-5) were used to investigate the expression of Akt in different cell lines.7 We found that the expression of Akt in liver cancer cell lines was higher compared to the normal liver cells (Figure 1A–C). And also, Akt-knockout liver cancer cells displayed lower expression of Akt (Figure 1D–I), which was used to further indicate the effect or importance of Akt in the treatment of liver cancer. Moreover, we found that the invading cells, clone number of liver cancer cells, and cell counts cultured for different time were downregulated after treatment of knockout (Figure 2A–H). And also, xenograft in nude mice analysis suggested that Akt-knockout mice were ameliorated for the volume of tumor in Figure 2F. The earlier results showed that Akt is of great importance in the development or progression for liver cancer. The activation of Akt will further enhance the NF-κB pathway phosphorylation levels. As the previous study reported, the increase of NF-κB pathway phosphorylation leads to inflammatory responses and cytokines production.28 Inflammatory abnormalities are a large group of disorders that underlie a vast variety of human diseases, especially regulated by ILs. Although the processes involved are identical to tissue inflammation, systemic inflammation is not confined to a particular tissue but involves the endothelium and other organ systems. The RT-PCR, Western blot, immunofluorescence, immunohistochemical, and luciferase activity analysis also give more evidence for our ideas (Figure 3A–G). The results showed that the expression of NF-κB in Akt−/− liver cancer cells was lower than in the liver cancer cells, demonstrating that inhibition of overexpression of Akt could suppress the abnormal NF-κB expression, subsequently inhibiting cytokines expression, including IL-1β and IL-18 (Figure 3E and F). Furthermore, as shown in Figure 4A, many cytokines were tested to further clarify the effect of Akt on the levels of inflammatory factors, which play an essential role in the progression of liver cancer. And then, overexpression of cytokines, in turn, further activated TLR4/MyD88 signaling pathway, contributing to the downstream signal expression. Subsequently activation of PI3Kand phosphorylation of Akt lead to the phosphorylation of NF-κB and intensified inflammation response, this is a feature of cancer progression. A previous study has shown that the upregulation of NF-κB promotes cell survival.29 However, in our study, we found that decreasing Akt reduced NF-κB expression and lessened p-NF-κB level. Accordingly, it decreased cancer cell survival rate and inhibited the feature of inflammation response in liver cancer cells. This is similar to the previous report.
The Bad protein is a pro-apoptotic member of the Bcl-2 gene family which is involved in initiating apoptosis. BAD is a member of the BH3-only family, a subfamily of the Bcl-2 family.30 It does not contain a C-terminal transmembrane domain for outer mitochondrial membrane and nuclear envelope targeting, unlike most other members of the Bcl-2 family.31 When Bad is phosphorylated by Akt, it forms the Bad-(14-3-3) protein heterodimer. Bax proteins are believed to initiate apoptosis by forming a pore in the mitochondrial outer membrane that allows cytochrome c to escape into the cytoplasm and activate the pro-apoptotic caspase cascade.32 The anti-apoptotic Bcl-2 and Bcl-xL proteins inhibit cytochrome c release through the mitochondrial pore and also inhibit activation of the cytoplasmic caspase cascade by cytochrome c.33,34 Previous studies have suggested that activated Akt could phosphorylate Bad, contributing to cell survival, otherwise apoptosis is carried out.35,36 Finally, caspase-9 and caspase-3, playing essential roles in apoptosis, are regulated.37–39 The results in our study, similar to the previous reports, found that Akt-knockout liver cancer cells displayed lower p-Bad expression and higher Bad expression, cutting down Bcl-xL expression and promoting the downstreaming signals, such as cytochrome c, Apaf-1, caspase-9, and caspase-3 expression, subsequently contributing to apoptosis in cancer cells (Figures 5A–E and 7). And Western blot and RT-PCR analysis further certify the results that Akt−/− enhanced Bad expression. In addition, Annexin V and caspase-3 staining kit with FACS analysis (Figure 6E and F) demonstrated Akt-knockout might affect caspase-3 in an indirect manner. As we mentioned earlier, the Akt signaling pathway might be associated with the progression of liver cancer. These data indicated that Akt as an important upstream of NF-κB and Bad might impact inflammatory responses and apoptosis (Figure 10C). Thus, inhibitory Akt activation might be an essential strategy to suppress liver cancer progression via inflammation response and apoptosis.
In addition, we investigated the effects of CA, which is an essential antioxidant compound in Rosmarinus officinalis L., on liver cancer. CA has been suggested to have anticancer effects in colon cancer, acute myeloid leukemia, and skin cancer via anti-inflammation, antioxidant, and antimicrobial effects.15–20 However, the molecular mechanism revealing improving liver cancer remains poorly understood. And few previous studies have reported that nanoparticle of CA could be better absorbed for animals. Also, CA nanoparticle has a more effective role on anticancer.40,41 Thus, in this study, the nanoparticle of CA was used to underlie the molecular effects or mechanism of CA against liver cancer in vitro and in vivo. In the first part of our study, we found the important role of Akt overexpression on promoting liver cancer. Thus, in the second part, we investigated whether CA could suppress the aggravation of liver cancer through Akt/NF-κB and Akt/Bad signaling pathway. As is known, the occurrence or the extent of inflammatory response is closely associated with the activation of NF-κB signaling pathway.42 From our study, p-NF-κB was inhibited by CA in cancer cells. Subsequently, cytokines of IL-1β and IL-18 were decreased both in protein and gene levels without obvious dose-dependent manner (Figure 7), displaying lower feature of inflammation in cancer cells in case of damaging the normal adjacent cells, which is consistent with former studies.15–17 Hence, it is deduced that CA plays an essential role in inflammation response via regulating NF-κB signaling pathway. In another, the anti-apoptosis effect of CA via Bad pathway was also investigated to further reveal the specific role on anticancer. We observed accelerated apoptosis after the use of CA in liver cancer cells via immunofluorescence, flow cytometry, Western blot, RT-PCR, as well as inoculating nude mice with tumor cells. The counts of apoptotic cells were elevated due to CA treatment via Annexin V/PI double staining and representative flow cytometry profiles. What is more, protein expression of regulators mainly for apoptosis in liver cancer cells, such as Bad, Cyto-c, Apaf-1, caspase-9, and caspase-3, was accelerated. Caspase-3, as the main regulatory factor contributing to apoptosis, was evaluated finally, displaying strong protein expression and weakening mRNA levels after the use of CA nanoparticle, which suggested CA inhibited liver cancer through apoptosis pathway. Further, it was notable that CA could inhibit liver cancer development not only relying on apoptosis induction but also cell proliferation inhibition. Cyclin-D1 and Cyclin-D3 were both found to be blocked for CA administration. It has been suggested that cell cycle development includes activation of CDKs. Cyclin-D1, as an essential regulator for G1 to S transition, could be increased in cancer cells, leading to controlled growth advantage.43,44 On the other hand, the CDKI, P21, regulates cell progressions in the G0/G1 phase of the cell cycle. P21 induction results in a blockade of the G0 to S transition, thus leading to a G0/G1 phase arrest in the cell cycle.45,46 Previous study has indicated that loss of CDKI in human cancer causes uncontrolled cell proliferation for an increase of CDK–cyclin complex.47 In our study, CA administration led to a significant enhancement of P21 in both liver cancer cell lines (Figure 8C and D). These results indicated that CA might play a vital role in cell cycle in human liver cancer via modulating the levels of cyclins–CDKs–CDKIs.
As for mRNA levels, we found these genes could be inhibited from the gene levels (Figure 9). And in our study, we found that nanoparticle of CA was able to obviously induce the reduction of tumor volume as well as tumor size (Figure 10A and B). Consequently, we highly suspected that the administration of nanoparticle of CA could effectively induce apoptosis via Bad pathway in liver cancer cells. Currently, many studies are involved in CA, regulating effects of inflammatory reaction and apoptotic activity.48,49 Therefore, regarding the significant ability of inflammatory inhibition and apoptotic induction in liver cancer cells of our nanoparticle CA, it is illustrated that the natural compound of nanoparticle CA has high potential to suppress the liver cancer growth and could be used as a promising anticancer drug. However, prospective researches should be conducted for full understanding of the functional mechanism of CA nanoparticle at the molecular level.
Taken together, liver cancer has been emphasized as a threat to human health and life, particularly in women. Nowadays, many risk factors contribute to the occurrence and progression of liver cancer.1–3,50 However, the underlying mechanism of liver cancer and treatments for liver cancer is far from known and thus further strategies or studies are required for patients with such disease. Thus, in our study, we determined to investigate the liver cancer animal model and cells from the point of Akt-regulated NF-κB and Bad signaling pathway and its downstream signals. Also, the Akt activation could be significantly upregulated in liver cancer cell lines, and knockout of Akt could reduce inflammation-related cytokines expression and apoptosis-associated signals, which might be a potential indicator for the clinical treatment. Our results confirmed that CA nanoparticle has a good prevention of liver cancer through remission of liver cancer cell inflammation progression by regulating NF – κB, which damage the normal cells, and induction of apoptosis in liver cancer by Bad-regulated caspase-3, as well as cell proliferation inhibition (Figure 10C). Thus, the current study plus previous researches demonstrated that CA nanoparticle possesses effective ability and function, making CA nanoparticle a new and promising material in clinical application.
Disclosure
The authors report no conflict of interest in this work.
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90. | ||
Kim YK, Lee GS, Jung EM, Hyun SH, Hwang WS, Jeung EB. Generation of fibroblasts overexpressing liver-specific PEPCK in a miniature pig model of human type 2 diabetes mellitus. Mol Med Rep. 2012;6: 45–50. | ||
Rampone B, Schiavone B, Martino A, Viviano C, Confuorto G. Current management strategy of hepatocellular carcinoma. World J Gastroenterol. 2009;15:3210–3216. | ||
Xu G, Qi FZ, Zhang JH, Cheng GF, Cai Y, Miao Y. Meta-analysis of surgical resection and radiofrequency ablation for early hepatocellular carcinoma. World J Surg Oncol. 2012;10:163. | ||
Salhab M, Canelo R. An overview of evidence-based management of hepatocellular carcinoma: a meta-analysis. J Cancer Res Ther. 2011;7:463–475. | ||
Zhou XD. Recurrence and metastasis of hepatocellular carcinoma: progress and prospects. Hepatobiliary Pancreat Dis Int. 2002;1: 35–41. | ||
Guan M, Zhou X, Soulitzis N, Spandidos DA, Popescu NC. Aberrant methylation and deacetylation of deleted in liver cancer-1 gene in prostate cancer: potential clinical applications. Clin Cancer Res. 2006;12:1412–1419. | ||
Lizarraga IM, Sugg SL, Weigel RJ, Scott-Conner CE. Review of risk factors for the development of contralateral breast cancer. Am J Surg. 2013;206:704–708. | ||
Tang JM, He QY, Guo RX, Chang XJ. Phosphorylated Akt overexpression and loss of PTEN expression in non-small cell lung cancer confers poor prognosis. Lung Cancer. 2006;51:181–191. | ||
Chapuis N, Tamburini J, Cornillet-Lefebvre P, et al. Autocrine IGF-1/IGF-1R signaling is responsible for constitutive PI3K/Akt activation in acute myeloid leukemia: therapeutic value of neutralizing anti-IGF-1R antibody. Haematologica. 2010;95:415–442. | ||
Muders MH, Zhang H, Wang E, Tindall DJ, Datta K. Vascular endothelial growth factor-C protects prostate cancer cells from oxidative stress by the activation of mammalian target of rapamycin complex-2 and AKT-1. Cancer Res. 2009;69:6042–6048. | ||
Opel D, Poremba C, Simon T, Debatin KM, Fulda S. Activation of Akt predicts poor outcome in neuroblastoma. Cancer Res. 2007;67:735–745. | ||
Chagpar RB, Links PH, Pastor MC, et al. Direct positive regulation of PTEN by the p85 subunit of phosphatidylinositol 3-kinase. Proc Natl Acad Sci U S A. 2010;107:5471–5476. | ||
Lopez-Carballo G, Moreno L, Masia S, Perez P, Barettino D. Activation of the phosphatidylinositol 3-kinase/Akt signaling pathway by retinoic acid is required for neural differentiation of SH-SY5Y human neuroblastoma cells. J Biol Chem. 2002;277:25297–25304. | ||
Huang MT, Ho CT, Wang ZY, et al. Inhibition of skin tumorigenesis by rosemary and its constituents carnosol and ursolic acid. Cancer Res. 1994;54:701–708. | ||
Anderson D, Cheng Y, Duan RD. Ursolic acid inhibits the formation of aberrant crypt foci and affects colonic sphingomyelin hydrolyzing enzymes in azoxymethane-treated rats. J Cancer Res Clin Oncol. 2008;134:101–107. | ||
Moran AE, Carothers AM, Weyant MJ, Redston M, Bertagnolli MM. Carnosol inhibits beta-catenin tyrosine phosphorylation and prevents adenoma formation in the C57BL/6J/Min/ + (Min/+) mouse. Cancer Res. 2005;65:1097–1104. | ||
Singletary K, MacDonald C, Wallig M. Inhibition by rosemary and carnosol of 7,12-dimethylbenz[a]anthracene (DMBA)-induced rat mammary tumorigenesis and in vivo DMBA-DNA adduct formation. Cancer Lett. 1996;104:43–48. | ||
Amagase H, Sakamoto K, Segal ER, Milner JA. Dietary rosemary suppresses 7,12-dimethylbenz(a) anthracene binding to rat mammary cell DNA. J Nutr. 1996;126:1475–1480. | ||
Sharabani H, Izumchenko E, Wang Q, et al. Cooperative antitumor effects of vitamin D3 derivatives and rosemary preparations in a mouse model of myeloid leukemia. Int J Cancer. 2006;118:3012–3021. | ||
Betbeder D, Spérandio S, Latapie JP, et al. Biovector nanoparticles improve antinociceptive efficacy of nasal morphine. Pharm Res. 2000;17:743–748. | ||
Fernández-Urrusuno R, Calvo P, Remun-López C, Vila-Jato JL, Alonso MJ. Enhancement of nasal absorption of insulin using chitosan nanoparticles. Pharm Res. 1999;16:1576–1581. | ||
De Oliveira MR, Ferreira GC, Schuck PF. Protective effect of carnosic acid against paraquat-induced redox impairment and mitochondrial dysfunction in SH-SY5Y cells: Role for PI3K/Akt/Nrf2 pathway. Toxicol In Vitro. 2015;32:41–54. | ||
Vaka SR, Shivakumar HN, Repka MA, Murthy SN. Formulation and evaluation of carnosic acid nanoparticulate system for upregulation of neurotrophins in the brain upon intranasal administration. J Drug Target. 2013;21:44–53. | ||
Macleod KF, Sherry N, Hannon G, et al. p53-dependent and independent expression of p21 during cell growth, differentiation, and DNA damage. Genes Dev. 1995;9:935–944. | ||
Lodygin D, Menssen A, Hermeking H. Induction of the Cdk inhibitor p21 by LY83583 inhibits tumor cell proliferation in a p53-independent manner. J Clin Invest. 2002;110(11):1717–1727. | ||
Yang XW, Wang XL, Cao LQ, et al. Green tea polyphenol epigallocatechin-3-gallate enhances 5-fluorouracil-induced cell growth inhibition of hepatocellular carcinoma cells. Hepatol Res. 2012;42:494–501. | ||
Hoesel B, Schmid JA. The complexity of NF-κB signaling in inflammation and cancer. Mol Cancer. 2013;2(12):86. doi: 10.1186/1476-4598-12-86. | ||
Luo JL, Kamata H, Karin M. IKK/NF-κB signaling: balancing life and death – a new approach to cancer therapy. J Clin Invest. 2005;115(10): 2625–2632. | ||
Berg M, Soreide K. EGFR and downstream genetic alterations in KRAS/BRAF and PI3K/AKT pathways in colorectal cancer: implications for targeted therapy. Discov Med. 2012;14:207–214. | ||
Radisavljevic Z. AKT as locus of cancer angiogenic robustness and fragility. J Cell Physiol. 2013;228:21–24. | ||
Balogova L, Maslanakova M, Dzurova L, Miskovsky P, Stroffekova K. Bcl-2 proapoptotic proteins distribution in U-87 MG glioma cells before and after hypericin photodynamic action. Gen Physiol Biophys. 2013;32:179–187. | ||
Danial NN. BAD: undertaker by night, candyman by day. Oncogene. 2008;27:S53–S70. | ||
Levine B, Sinha S, Kroemer G. Bcl-2 family members: dual regulators of apoptosis and autophagy. Autophagy. 2008;4:600–606. | ||
Stauffer SR. Small molecule inhibition of the Bcl-X(L)-BH3 protein-protein interaction: proof-of-concept of an in vivo chemopotentiator ABT-737. Curr Top Med Chem. 2007;7:961–965. | ||
Hojabrpour P, Waissbluth I, Ghaffari M, Cox ME, Duronio V. CaMKII-gamma mediates phosphorylation of BAD at Ser170 to regulate cytokine-dependent survival and proliferation. Biochem J. 2012;442:139–149. | ||
El-Awady RA, Semreen MH, Saber MM, Cyprian F, Menon V, Al-Tel TH. Modulation of DNA damage response and induction of apoptosis mediates synergism between doxorubicin and a new imidazopyridine derivative in breast and lung cancer cells. DNA Repair (Amst). 2015;37:1–11. | ||
Vanajothi R, Srinivasan P. An anthraquinone derivative from Luffa acutangula induces apoptosis in human lung cancer cell line NCI-H460 through p53-dependent pathway. J Recept Signal Transduct Res. 2015;20:1–11. | ||
Rajamani U, Essop MF. Hyperglycemia-mediated activation of the hexosamine biosynthetic pathway results in myocardial apoptosis. Am J Physiol Cell Physiol. 2010;299:C139–C147. | ||
Paasch U, Grunewald S, Fitzl G, Glander HJ. Deterioration of plasma membrane is associated with activation of caspases in human spermatozoa. J Androl. 2003;24:246–252. | ||
Beyret E, Lin H. Pinpointing the expression of piRNAs and function of the PIWI protein subfamily during spermatogenesis in the mouse. Dev Biol. 2011;337:215–226. | ||
Yu XN, Chen XL, Li H, Li XX, Li HQ, Jin WR. Reversion of P-glycoprotein-mediated multidrug resistance in human leukemic cell line by carnosic acid. Chin J Physiol. 2008;51:348–356. | ||
Lin SY, Xia W, Wang JC, et al. Beta-catenin, a novel prognostic marker for breast cancer: its roles in cyclin D1 expression and cancer progression. Proc Natl Acad Sci U S A. 2000;97:4262–4266. | ||
Bahnassy AA, Zekri AR, Alam El-Din HM, et al. The role of cyclins and cyclins inhibitors in the multistep process of HPV-associated cervical carcinoma. J Egypt Natl Canc Inst. 2006;18:292–302. | ||
Yang L, Zhang HW, Hu R, et al. Wogonin induces G1 phase arrest through inhibiting Cdk4 and cyclin D1 concomitant with an elevation in p21Cip1 in human cervical carcinoma HeLa cells. Biochem Cell Biol. 2009;87:933–942. | ||
Soria G, Gottifredi V. PCNA-coupled p21 degradation after DNA damage: The exception that confirms the rule? DNA Repair (Amst). 2010;9:358–364. | ||
Beckerman R, Donner AJ, Mattia M, et al. A role for Chk1 in blocking transcriptional elongation of p21 RNA during the S-phase checkpoint. Genes Dev. 2009;23:1364–1377. | ||
Wang R, Li H, Guo G, et al. Augmentation by carnosic acid of apoptosis in human leukaemia cells induced by arsenic trioxide via upregulation of the tumour suppressor PTEN. J Int Med Res. 2008;36:682–690. | ||
Min HK, Kim SM, Baek SY, et al. Anthocyanin extracted from black soybean seed coats prevents autoimmune arthritis by suppressing the development of Th17 cells and synthesis of proinflammatory cytokines by such cells, via inhibition of NF-κB. PLoS One. 2015;10:e0138201. | ||
Barni MV, Carlini MJ, Cafferata EG, Puricelli L, Moreno S. Carnosic acid inhibits the proliferation and migration capacity of human colorectal cancer cells. Oncol Rep. 2012;27:1041–1048. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.