Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Two-Year Outcomes for the Double-Blind, Randomized, Sham-Controlled Study of Targeted Lung Denervation in Patients with Moderate to Severe COPD: AIRFLOW-2
Authors Valipour A, Shah PL , Herth FJ , Pison C, Schumann C , Hübner RH, Bonta PI , Kessler R , Gesierich W , Darwiche K , Lamprecht B, Perez T , Skowasch D , Deslee G, Marceau A, Sciurba FC, Gosens R, Hartman JE , Conway F , Duller M, Mayse M, Norman HS, Slebos DJ
Received 12 June 2020
Accepted for publication 7 October 2020
Published 5 November 2020 Volume 2020:15 Pages 2807—2816
DOI https://doi.org/10.2147/COPD.S267409
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Arschang Valipour,1 Pallav L Shah,2 Felix J Herth,3 Christophe Pison,4 Christian Schumann,5 Ralf-Harto Hübner,6 Peter I Bonta,7 Romain Kessler,8 Wolfgang Gesierich,9 Kaid Darwiche,10 Bernd Lamprecht,11 Thierry Perez,12 Dirk Skowasch,13 Gaetan Deslee,14 Armelle Marceau,15 Frank C Sciurba,16 Reinoud Gosens,17 Jorine E Hartman,18 Francesca Conway,2 Marina Duller,1 Martin Mayse,19 Holly S Norman,19 Dirk-Jan Slebos18 On behalf of the AIRFLOW-2 Trial Study Group
1Department of Respiratory and Critical Care Medicine, Karl-Landsteiner-Institute for Lung Research and Pulmonary Oncology, Krankenhaus Nord-Klinik Floridsdorf, Vienna, Austria; 2Royal Brompton & Harefield NHS Trust, Chelsea & Westminster Hospital and Imperial College, London, UK; 3Thoraxklinik, Department of Pneumology and Critical Care Medicine and Translational Lung Research Center Heidelberg (TLRCH), University of Heidelberg, Heidelberg, Germany; 4CHU Grenoble Alpes, Service Hospitalier Universitaire Pneumologie Physiologie; Université Grenoble Alpes, Grenoble, France; 5Clinic of Pneumology, Thoracic Oncology, Sleep- and Respiratory Critical Care, Klinikverbund Allgaeu, Kempten and Immenstadt, Germany; 6Charité Universitätsmedizin Berlin, Medizinische Klinik m. Schw. Infektiologie und Pneumologie, Campus Virchow, Berlin, Germany; 7Department of Respiratory Medicine, Academic Medical Center, University of Amsterdam, Amsterdam, the Netherlands; 8Service de Pneumologie, Nouvel Hôpital Civil, Université de Strasbourg, Strasbourg, France; 9Asklepios-Fachkliniken Munich-Gauting, Comprehensive Pneumology Center Munich, Gauting, Germany; 10Department of Pulmonary Medicine, Section of Interventional Pneumology, Ruhrlandklinik - University Hospital Essen, University of Duisburg-Essen, Essen, Germany; 11Department of Pulmonary Medicine, Kepler Universitatsklinikum GmbH, Linz, Austria; 12CHU de Lille – Hôpital Calmette, Lille, France; 13Department of Internal Medicine II - Cardiology/Pneumology, University of Bonn, Bonn, Germany; 14CHU de Reims, Hôpital Maison Blanche, Service de Pneumologie, Reims, France; 15Service de Pneumologie, Hôpital Universitaire Bichat, Paris, France; 16University of Pittsburgh School of Medicine, Pittsburgh, PA, USA; 17Department of Molecular Pharmacology, University of Groningen, Groningen, the Netherlands; 18Department of Pulmonary Diseases, University of Groningen, University Medical Center Groningen, Groningen, the Netherlands; 19Nuvaira, Inc., Minneapolis, MN, USA
Correspondence: Dirk-Jan Slebos
Department of Pulmonary Diseases/Interventional Bronchoscopy AA11, University Medical Center Groningen, PO Box 30001, Groningen, the Netherlands
Tel +31 503612357
Fax +31503619320
Email [email protected]
Purpose: COPD exacerbations are associated with worsening clinical outcomes and increased healthcare costs, despite use of optimal medical therapy. A novel bronchoscopic therapy, targeted lung denervation (TLD), which disrupts parasympathetic pulmonary innervation of the lung, has been developed to reduce clinical consequences of cholinergic hyperactivity and its impact on COPD exacerbations. The AIRFLOW-2 study assessed the durability of safety and efficacy of TLD additive to optimal drug therapy compared to sham bronchoscopy and optimal drug therapy alone in subjects with moderate-to-severe, symptomatic COPD two years post randomization.
Patients and Methods: TLD was performed in COPD patients (FEV1 30– 60% predicted, CAT≥ 10 or mMRC≥ 2) in a 1:1 randomized, sham-controlled, double-blinded multicenter study (AIRFLOW-2) using a novel lung denervation system (Nuvaira, Inc., USA). Subjects remained blinded until their 12.5-month follow-up visit when control subjects were offered the opportunity to undergo TLD. A time-to-first-event analysis on moderate and severe and severe exacerbations of COPD was performed.
Results: Eighty-two subjects (FEV1 41.6± 7.4% predicted, 50.0% male, age 63.7± 6.8 yrs, 24% with prior year respiratory hospitalization) were randomized. Time-to-first severe COPD exacerbation was significantly lengthened in the TLD arm (p=0.04, HR=0.38) at 2 years post-TLD therapy and trended towards similar attenuation for moderate and severe COPD exacerbations (p=0.18, HR=0.71). No significant changes in lung function or SGRQ-C were found 2 years post randomization between groups.
Conclusion: In a randomized trial, TLD demonstrated a durable effect of significantly lower risk of severe AECOPD over 2 years. Further, lung function and quality of life remained stable following TLD.
Clinical Trial Registration: NCT02058459.
Keywords: COPD exacerbation, targeted lung denervation, bronchoscopy, COPD
Background
The burden of COPD is well documented with exacerbations associated with worsening clinical outcomes1–3 and increased healthcare costs.4,5 In fact, COPD is associated with an elevated mortality risk as high as 14-fold the year following a severe exacerbation.6 Despite the known risks and growing healthcare burden,7 therapeutic options which impact exacerbation rate for COPD patients are limited. In fact, it has been reported that 70% of GOLD D patients continue to exacerbate despite optimal pharmaceutical care (triple therapy, LABA-LAMA-ICS).8 Current inhaled therapeutic options, such as ICS, target symptom relief during periods of disease stability and reduction of exacerbation risk; however, limitations due to patient compliance, delivery mechanism, and dose response,9,10 or known associations with increased rate of pneumonia11,12 result in a population of patients with high rates of exacerbations despite optimal inhaled pharmaceutical care.8
Acute exacerbations of COPD are predominately driven by viral and bacterial respiratory infections, which have been shown to cause increased reflex signaling via the pulmonary branches of the vagus nerve.13,14 The resultant parasympathetic overstimulation drives the release of acetylcholine15 and in COPD patients is associated with changes in airway inflammation, mucus hypersecretion, and airway hyperresponsiveness.2,16,17 Indeed, acute increases in symptoms in response to respiratory infection (viral or bacterial) are in part secondary to changes in the function of the nervous system.18,19 Accordingly, pharmacologic blockade of the parasympathetic airway system is a known therapeutic target for both acute and chronic management of COPD using long-acting muscarinic antagonists.20
A novel bronchoscopic therapy, targeted lung denervation (TLD), ablates the bronchial branches of the vagus nerve with the aim to permanently decrease parasympathetic stimulation, resulting in decreased airway hyperresponsiveness and reduced inflammation, as evidenced through attenuated inflammatory markers.21 Further, preclinical studies have demonstrated that the TLD ablation is durable,22 and prior clinical results have indicated it is additive to optimal medical therapy treatment.23,24 The positive safety profile of TLD,23,25,26 as well as clinical impact on severe COPD exacerbation rate following targeted lung denervation (TLD) has been previously reported.27 Here we present two-year follow-up data for the randomized sham-controlled AIRFLOW-2 clinical trial.22
Patients and Methods
Study Design and Oversight
This study is the follow-up of a randomized, sham-controlled, double-blind, prospective, multicenter study designed to evaluate the safety of TLD in patients with moderate to severe COPD (AIRFLOW-2 trial – NCT02058459). The study design, procedures, and full methodology of the AIRFLOW-2 trial has been described.27 Following 12.5 months of blinding, sham participants were unblinded and offered the opportunity to undergo TLD therapy. No preset window of crossover eligibility was defined. Results from subjects who crossed-over are limited to those results collected prior to the cross-over procedure. All subjects are followed for up to 3 years.
All adverse events were defined by the study investigator. This study was conducted in accordance with the amended Declaration of Helsinki. All local ethics committees of participating institutions were approved, and all subjects provided written informed consent.
Study Subjects
Subjects aged between 40 and 75 years with a diagnosis of moderate to severe symptomatic COPD (post-bronchodilator ratio of FEV1/FVC <0.70 and FEV1 30–60% of predicted), a modified Medical Research Council (mMRC) dyspnea grade ≥2 or COPD assessment test (CAT) score ≥10 were enrolled (GOLD B or D subjects), as previously described.27 Major exclusion criteria were more than two respiratory-related hospitalizations within the past year, gastroparesis cardinal symptom index (GCSI) ≥18,28 and previous lung or chest procedure.27
All patient inhaler usage was documented at screening. A wash-out period for long-acting muscarinic antagonists (LAMA) for 7 days, long-acting beta-agonist (LABA) and short-acting muscarinic antagonist (SAMA) and short-acting beta-agonist (SABA) for 12 hours were employed. Following washout, all subjects were placed on inhaled tiotropium 18μg per day, and could continue with the prior inhalers, including inhaled corticosteroids, at the discretion of their physician, for 7–21 days prior to their Run-In procedure testing. On the day of study pre-procedure testing, subjects were evaluated prior to the daily tiotropium dose (24 hours) and post-tiotropium administration for both trough and peak measures, ie, Run-In Trough and Run-In Peak. All subjects were encouraged to continue LAMA and other maintenance medications throughout the study follow-up period.
Study Procedures
As previously described, subjects underwent spirometry, body plethysmography, and health-related quality of life questionnaire (eg, COPD-specific St. George’s Respiratory Questionnaire (SGRQ-C)) at baseline and pre-defined follow-up appointments at 6.5 months, 12.5 months and yearly out to 3 years.27 Briefly, subjects allocated to the TLD arm received Nuvaira lung denervation therapy (Nuvaira, Inc.), which deploys radiofrequency (RF) energy to ablate the bronchial branches of the vagus nerve while protecting the bronchial mucosa utilizing a dual-cooled catheter. Subjects allocated to the sham arm underwent a mock procedure to maintain blinding. Following the 12.5-month follow-up visit, all participants in the sham group had the opportunity to undergo TLD therapy, ie, cross-over. Cross-over could occur at any timepoint during follow-up after the 12.5-month follow-up visit. COPD exacerbations severity for the AIRFLOW-2 was defined previously.27 Briefly, moderate or severe COPD exacerbations were defined as COPD exacerbations requiring treatment with systemic steroids and/or antibiotics, with or without hospitalization. All adverse events were reported and tracked throughout the study.
Statistical Methods
Statistical hypothesis tests are based on t-tests for continuous data that are normally distributed. Non-parametric tests were performed when there was evidence of non-normality. Fisher’s exact test was used to compare categorical data. Annualized rate comparisons were performed using negative binomial regression analysis. Comparisons between groups of time to event data, such as time to first COPD exacerbation, were accomplished using a standard Log rank statistic. Analysis was performed using Minitab (Version 18.1, 2017) and SAS (Version 9.4, 2020). P<0.05 was considered statistically significant.
Results
Patient Disposition
As described previously,27 the study inclusion was conducted between July 2016 and May 2017. One hundred and ninety-four subjects were screened and 82 subjects were randomized. At enrollment, 95% of subjects were either managed via dual or triple therapy and complied with the tiotropium protocol requirement.27 Demographics of the control and TLD groups at baseline and 2 years are provided in Table 1. In total, the TLD arm had a retention of 88% at 2 years (n=36), whereas the control arm group was reduced (n=14) as 49% of control subjects (n=20) underwent an optional cross-over procedure and 5 subjects voluntarily exited due to crossover ineligibility or other reasons (Figure 1). Control patients who underwent a cross-over procedure were censored from the adverse event annualized rate and time-to-event analysis at the time of cross-over and are not included in other variable analysis as they did not complete a full 2-year follow-up time period.
 |
Table 1 Characteristics of Subjects at Baseline27 and 2 Years |
Long-Term Safety Assessment: All Adverse Events
Differences in the percent of patients with severe AEs were non-significant in year 2 (p=0.72) between the Control and TLD arms with a lower, but non-significant difference in respiratory SAEs favoring the TLD group (p=0.19). In the treatment arm, a single severe gastrointestinal AE was reported 507 days post-procedure in the treatment arm with an inconclusive relationship to the TLD procedure (Figure 2). In the TLD arm, respiratory (n=4) and gastrointestinal (n=1) severe AEs were reported, as well as three patients with severe AE classifications which were unrelated to TLD: musculoskeletal and connective tissue disorders (n=1), reproductive system (n=1), and skin and subcutaneous tissue disorder (n=1; Figure 2). Additionally, the control arm had respiratory (n=4), cardiac disorder (n=1) and psychiatric severe AEs (n=1; Figure 2). In the second year of follow-up, 11.1% of TLD subjects (n=4) experienced a respiratory-related hospitalization, compared to 28.6% of control subjects (n=4). These events resolved. Hospitalizations in the year prior to randomization were 24% for each group.27
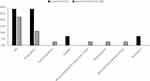 |
Figure 2 Percent (%) of patients experiencing a serious adverse event during the second year post-randomization for control (n=14; black bars) and TLD (n=36; diagonal stripe bars). |
Long-Term Safety Assessment: COPD Adverse Events
The risk of a severe COPD exacerbation requiring hospitalization was significantly lower in the TLD group than the control group at 2 years post-randomization, as assessed in a time-to-first event analysis (Figure 3, p=0.04, HR=0.38; 95% CI 0.15–0.99). There was no statistical difference in risk of first moderate or severe COPD exacerbation from baseline to 2 years (Figure 4, p=0.18, HR=0.71, 95% CI 0.42–1.18).
AEs were also evaluated in terms of annualized rate. There was not a statistically significant difference between groups for the annualized rate of COPD moderate and severe exacerbations (Control=0.81 vs TLD=0.64; p=0.54, Table 2) or for the severe COPD AEs (Control=0.15 vs TLD=0.11; p=0.71, Table 2) annualized rate at 2 years.
 |
Table 2 Annualized Rate of AECOPDs |
Lung Function and Quality of Life Assessment
To assess lung function, pulmonary function test results performed at the 2-year follow-up visit were compared to initial Run-In visit test results. Approximately 94% of TLD (n=34) and 79% of control (n=11) subjects completed the in-person follow-up visit; remote visits were attempted for patients unable to complete the in-person follow-up visit to collect disease severity and quality of life assessment. All results were analyzed prior to daily drug administration, ie, trough. Lung function remained stable out to 2 years in the treatment arm. Specifically, FEV1 remained stable (Table 3; p=0.44),29 as well as FVC (p=0.51), TLC (p=0.66), and RV (p=0.84). The control group had increased variability at 2 years, with trends of improvements in lung function noted for FVC, RV, and TLC, likely due to the adjusted population demographic at the 2-year visit due to cross-over design. Despite the trends, no changes were statistically significant (Table 3). Additional lung function measurements are included in Table 3. Further, Quality of Life as measured by SGRQ-C assessment remained stable out to 2 years for the TLD (p=0.93) and control groups (p=0.78) compared to Run-In visit results. No differences in SGRQ-C were found between groups (p=0.69; Table 3).
 |
Table 3 Pulmonary Function and SGRQ-C Assessments |
Discussion
The current study was the open-label follow-up of a randomized, double-blinded, full sham bronchoscopy controlled study of targeted lung denervation in subjects with symptomatic COPD, with primary and secondary endpoints at 12.5 months previously reported.27 Here we presented long-term safety and durability of TLD’s impact on severe COPD exacerbations on top of maintenance inhaler therapy in a high retention 2-year follow-up study. TLD was associated with a lower percentage of patients experiencing a severe COPD exacerbation over the second year of follow up, compared to control subjects who underwent a sham procedure, and a significant reduction in the risk of severe COPD exacerbations over the two years of follow up in a time to event analysis.
Safety
The safety profile of the TLD system and procedure was confirmed out to 2 years. Respiratory and gastrointestinal adverse events were decreased during the second year of follow-up, with only one gastrointestinal SAE reported 507 days post-procedure. This is a meaningful drop of gastrointestinal SAEs in comparison to the 1st year of follow up and suggests that severe gastrointestinal events related to the procedure present during the periprocedural period (within 1 month) and are transient in nature.27 Additionally, a reduction of severe AECOPD out to two years post treatment with TLD is reported (Figure 3). These data suggest a sustained impact of the TLD therapy on COPD exacerbation out to two years post procedure in the treatment arm. A regression to the mean in the treatment arm represented by a converging of the time-to-event curves or increase in rate of COPD exacerbations in the second year would have been expected if the treatment effect was transient. There was no regression to the mean in the observed data set which demonstrated continued separation of the treatment and control arms in the second year of follow-up. This data is additive to the previously reported reduction in severe AECOPD following 1-year post-TLD therapy.27
Clinical Impact
COPD exacerbations are known to have an impact on overall population health and healthcare costs, despite optimal pharmaceutical therapy.1,5,30,31 Further, COPD exacerbations are associated with an increased mortality rate,2 with one severe COPD event associated with a 14-times increase in risk of death and moderate COPD exacerbations associated with varying increased risk of death depending on frequency.6 In recent pharmacologic trials, while subjects were on dual or triple inhaler therapy, 50–65% of GOLD D subjects had 1 or more moderate or severe exacerbations in one year of follow-up, representing an unmet clinical need.8,32,33
A reduction in COPD exacerbations following TLD therefore may have the potential to decrease both healthcare costs and mortality risk. While marked improvements in lung function were not observed, TLD was performed in COPD subjects who experienced a high exacerbation rate despite optimal medical therapy. It is known that pharmacologic therapy attenuates daily symptoms and improves lung function, whereas TLD prevents exacerbations through a blunted vagal response to stimuli. Therefore, early data suggest optimal clinical outcomes may result through a combination of pharmacologic therapy and TLD.
Lung Function and Quality of Life
The temporal progression of lung function for COPD subjects is highly variable;34 however, a standard decrease of approximately 47–59 mL per year in FEV1 is anticipated for GOLD 2 and 3 patients, with a standard decrease of ~35 mL anticipated for GOLD 4 patients.35 Despite this anticipated decline in lung function in similar populations, FEV1 and FVC values remained stable out to 2 years.29 Similarly, SGRQ-C remained stable out to 2 years. Over two years of follow-up in this patient population it would be expected that the SGRQ-C measures would have deteriorated by ~2 pts per year.36 A larger clinical study is needed to determine the impact of TLD on patient quality of life.37 The variability in lung function in the control group was not surprising due to the small sample size (n=11) able to complete the in-person 2nd year follow-up visit due to cross-over.
Limitations
In addition to the strengths and weaknesses previously documented,27 the small sample size was an enhanced limitation in this report. While an improvement in the time-to-first event is seen in the TLD group, this result was found in a relatively small study size. This sample size was further decreased after approximately 50% of control subjects crossed over before the 2-year follow-up visit, which likely resulted in a healthier group, ie, subjects who had a higher symptomatic disease characteristic were more likely to undergo TLD, which is a well-known challenge of cross-over design.38
Conclusion
Here we report that the clinical impact of TLD is durable, as it is found to reduce the risk of serious COPD adverse events over 2 years with stabilized lung function. The reduction of severe COPD exacerbations has the potential to improve long-term clinical outcomes and reduce healthcare costs. Further study in a large population is warranted to confirm the healthcare impact of TLD.
Abbreviations
AE, adverse events; BMI, body mass index; CAT, COPD assessment test; COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; ICS, inhaled corticosteroid; GCSI, Gastroparesis Cardinal Symptom Index; GOLD, Global Obstructive Lung Disease; LABA, long acting beta agonist; LAMA, long acting muscarinic antagonist; MCID, minimal clinically important difference; mMRC, modified Medical Research Council; RF, radiofrequency; RV, residual volume; SABA, short acting beta agonist; SAMA, short acting muscarinic antagonist; SGRQ-C, St. George’s Respiratory Questionnaire; TLC, total lung capacity; TLD, targeted lung denervation.
Ethics Approval and Consent
The AIRFLOW-2 study was approved by the following ethics committees: Ethics Committee of the Federal Land of Upper Austria (B-106-15); Medizinischen Fakultat Heidelberg (Mzmu-171/2014); Protection of Persons Committee (CPP) 2014-A00530-47/7; Health Research Authority 14/EE/1193; Medical Ethics Review Committee (METc; NL48849.042.14); and Protection of Persons Committee (CPP; 2014-A00530-47/70). The authors do not plan to share individual deidentified participant data. The study protocol has been made available with the publication of the 1-year data (Slebos et al. AJRCCM 2019).
Acknowledgments
The authors thank Alexander Peterson for his role in the data analysis and the entire AIRFLOW-2 research team for their dedication to this trial. Additionally, the authors thank NAMSA for statistical analysis support. All clinical trial expenses were reimbursed by the study sponsor (Nuvaira, Inc., Minneapolis, MN, USA). Versions of this data were presented at the European Respiratory Conference (European Respiratory Journal 2019; 54: Suppl. 63, OA1615, https://erj.ersjournals.com/content/54/suppl_63/OA1615) and the American Thoracic Society Conference (American Journal of Respiratory and Critical Care Medicine 2020;201:A2489, https://www.atsjournals.org/doi/abs/10.1164/ajrccm-conference.2020.201.1_MeetingAbstracts.A2489) as a poster presentation/conference talk with interim findings.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
DJS reports grant, consultancy fees paid to the institution, and nonfinancial support from Nuvaira, Inc. during the conduct of the study. HSN reports employment with Nuvaira, Inc. MM reports consultancy fees and employment with Nuvaira, Inc. during the conduct of the study. MM owns stock from Nuvaira, Inc in exchange for multiple patents with Nuvaira. AV reports speaker fees from Nuvaira during the conduct of the study. PLS reports personal fees from Nuvaira during the conduct of the study; personal fees for consultancy from Olympus, PneumRX/BTG, Creo Medical, and from CSA medical outside the submitted work. FJH reports personal fees for serving at the advisory board for Olympus, Pumonx, and Uptake outside the submitted work. Christophe Pison reports grants, personal fees, non-financial support, and fees to CHUGA to conduct RCT IPSII, AF2 and AF3 from Nuvaira during the conduct of the study; grants, personal fees, non-financial support in the field of COPD and Asthma from GSK France, Boehringer Ingelheim, Chiesi, and AstraZeneca outside the submitted work. PIB reports non-financial support from Nuvaira during the conduct of the study. RK reports grants, personal fees from Nuvaira during the conduct of the study. WG reports personal fees as speaker and receives travel supports from PulmonX and Astra Zeneca, and travel support from PneumRX outside the submitted work. TP reports costs of clinical trial from Holaira-Nuvaira during the conduct of the study; personal fees served in the COPD advisory board from Novartis, Chiesi, and Boehringer Ingelheim, grants for clinical research from Astra Zeneca outside the submitted work. GD reports personal fees from Nuvaira during the conduct of the study; personal fees from AstraZeneca, Boehringer Ingelheim, Chiesi, BTG-PneumRx, and Novartis outside the submitted work. RG was scientific advisory board member for Nuvaira until 2019; reports grants from Boehringer Ingelheim, Aquilo, Novartis, and Chiesi outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Celli BR, Wedzicha JA Update on clinical aspects of chronic obstructive pulmonary disease. N Engl J Med. 2019;381(13):1257–1266. doi:10.1056/NEJMra1900500
2. Wedzicha JA, Seemungal TAR COPD exacerbations: defining their cause and prevention. Lancet. 2007;370(9589):786–796. doi:10.1016/S0140-6736(07)61382-8
3. Soler-Cataluña JJ, Martínez-García MA, Román Sánchez P, Salcedo E, Navarro M, Ochando R Severe acute exacerbations and mortality in patients with chronic obstructive pulmonary disease. Thorax. 2005;60(11):925–931. doi:10.1136/thx.2005.040527
4. Halpin DM, Miravitlles M, Metzdorf N, Celli B Impact and prevention of severe exacerbations of COPD: a review of the evidence. Int J Chron Obstruct Pulmon Dis. 2017;12:2891–2908.
5. Guarascio AJ, Ray SM, Finch CK, Self TH The clinical and economic burden of chronic obstructive pulmonary disease in the USA. Clinicoecon Outcomes Res. 2013;5:235–245.
6. Rothnie KJ, Müllerová H, Smeeth L, Quint JK Natural History of chronic obstructive pulmonary disease exacerbations in a general practice-based population with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018;198(4):464–471. doi:10.1164/rccm.201710-2029OC
7. Ho T, Cusack RP, Chaudhary N, Satia I, Kurmi OP Under- and over-diagnosis of COPD: a global perspective. Breathe. 2019;15(1):24–35. doi:10.1183/20734735.0346-2018
8. Pavord ID, Chanez P, Criner GJ, et al. Mepolizumab for eosinophilic chronic obstructive pulmonary disease. N Engl J Med. 2017;377(17):1613–1629. doi:10.1056/NEJMoa1708208
9. George J, Kong DCM, Thoman R, Stewart K Factors associated with medication nonadherence in patients with COPD. Chest. 2005;128(5):3198–3204.
10. Newman SP Drug delivery to the lungs: challenges and opportunities. Ther Deliv. 2017;8(8):647–661.
11. Suissa S Inhaled corticosteroids preventing pneumonia mortality: paradox or selection bias? Eur Respir J. 2019;53(2). doi:10.1183/13993003.02112-2018
12. Morjaria JB, Rigby A, Morice AH Inhaled corticosteroid use and the risk of pneumonia and COPD exacerbations in the UPLIFT study. Lung. 2017;195(3):281–288. doi:10.1007/s00408-017-9990-8
13. Sapey E, Stockley RA COPD exacerbations. 2: aetiology. Thorax. 2006;61(3):250–258. doi:10.1136/thx.2005.041822
14. Criner GJ, Eberhardt R, Fernandez-Bussy S, et al. Interventional bronchoscopy: state-of-the-art review. Am J Respir Crit Care Med. 2020.
15. Kistemaker LE, Gosens R Acetylcholine beyond bronchoconstriction: roles in inflammation and remodeling. Trends Pharmacol Sci. 2015;36(3):164–171. doi:10.1016/j.tips.2014.11.005
16. Zanini A, Cherubino F, Zampogna E, Croce S, Pignatti P, Spanevello A Bronchial hyperresponsiveness, airway inflammation, and reversibility in patients with chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis. 2015;10:1155–1161. doi:10.2147/COPD.S80992
17. van den Berge M, Vonk JM, Gosman M, et al. Clinical and inflammatory determinants of bronchial hyperresponsiveness in COPD. Eur Respir J. 2012;40(5):1098–1105.
18. Zaccone EJ, Undem BJ Airway vagal neuroplasticity associated with respiratory viral infections. Lung. 2016;194(1):25–29. doi:10.1007/s00408-015-9832-5
19. Canning BJ Reflex regulation of airway smooth muscle tone. J Appl Physiol. 2006;101(3):971–985.
20. Beeh KM, Burgel PR, Franssen FME, et al. How do dual long-acting bronchodilators prevent exacerbations of chronic obstructive pulmonary disease? Am J Respir Crit Care Med. 2017;196(2):139–149. doi:10.1164/rccm.201609-1794CI
21. Kistemaker LE, Slebos DJ, Meurs H, Kerstjens HA, Gosens R Anti-inflammatory effects of targeted lung denervation in patients with COPD. Eur Respir J. 2015;46(5):1489–1492. doi:10.1183/13993003.00413-2015
22. Mayse ML, Norman HS, Peterson AD, Rouw KT, Johnson PJ Targeted lung denervation in sheep: durability of denervation and long-term histologic effects on bronchial wall and peribronchial structures. Respir Res. 2020;21(1):117. doi:10.1186/s12931-020-01383-3
23. Slebos DJ, Klooster K, Koegelenberg CF, et al. Targeted lung denervation for moderate to severe COPD: a pilot study. Thorax. 2015;70(5):411–419. doi:10.1136/thoraxjnl-2014-206146
24. Koegelenberg CF, Theron J, Slebos DJ, Klooster K, Mayse M, Gosens R Antimuscarinic bronchodilator response retained after bronchoscopic vagal denervation in chronic obstructive pulmonary disease patients. Respiration. 2016;92(1):58–60. doi:10.1159/000447641
25. Valipour A, Asadi S, Pison C, et al. Long-term safety of bilateral targeted lung denervation in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2018;13:2163–2172. doi:10.2147/COPD.S158748
26. Valipour A, Shah PL, Pison C, et al. Safety and dose study of targeted lung denervation in moderate/severe COPD patients. Respiration. 2019:1–11.
27. Slebos DJ, Shah PL, Herth FJ, et al. Safety and adverse events after targeted lung denervation for symptomatic moderate to severe COPD (AIRFLOW): a multicenter randomized controlled trial. Am J Respir Crit Care Med. 2019. 200 12 1477–1486 doi:10.1164/rccm.201903-0624OC
28. Revicki DA, Rentz AM, Dubois D, et al. Gastroparesis Cardinal Symptom Index (GCSI): development and validation of a patient reported assessment of severity of gastroparesis symptoms. Qua Life Res. 2004;13(4):833–844. doi:10.1023/B:QURE.0000021689.86296.e4
29. Jones PW, Beeh KM, Chapman KR, Decramer M, Mahler DA, Wedzicha JA Minimal clinically important differences in pharmacological trials. Am J Respir Crit Care Med. 2014;189(3):250–255. doi:10.1164/rccm.201310-1863PP
30. Pasquale MK, Sun SX, Song F, Hartnett HJ, Stemkowski SA Impact of exacerbations on health care cost and resource utilization in chronic obstructive pulmonary disease patients with chronic bronchitis from a predominantly Medicare population. Int J Chron Obstruct Pulmon Dis. 2012;7:757–764.
31. Rehman AU, Hassali MAA, Muhammad SA, Harun SN, Shah S, Abbas S The economic burden of chronic obstructive pulmonary disease (COPD) in Europe: results from a systematic review of the literature. Eur J Health Econ. 2019. doi:10.1007/s10198-10019-01119–10191.
32. Lipson DA, Barnhart F, Brealey N, et al. Once-daily single-inhaler triple versus dual therapy in patients with COPD. N Engl J Med. 2018;378(18):1671–1680. doi:10.1056/NEJMoa1713901
33. Martinez FJ, Calverley PMA, Goehring U-M, Brose M, Fabbri LM, Rabe KF Effect of roflumilast on exacerbations in patients with severe chronic obstructive pulmonary disease uncontrolled by combination therapy (REACT): a multicentre randomised controlled trial. Lancet. 2015;385(9971):857–866. doi:10.1016/S0140-6736(14)62410-7
34. Vestbo J, Edwards LD, Scanlon PD, et al. Changes in forced expiratory volume in 1 second over time in COPD. N Engl J Med. 2011;365(13):1184–1192. doi:10.1056/NEJMoa1105482
35. Tantucci C, Modina D Lung function decline in COPD. Int J Chron Obstruct Pulmon Dis. 2012;7:95–99. doi:10.2147/COPD.S27480
36. Oga T, Nishimura K, Tsukino M, Sato S, Hajiro T, Mishima M Longitudinal deteriorations in patient reported outcomes in patients with COPD. Respir Med. 2007;101(1):146–153. doi:10.1016/j.rmed.2006.04.001
37. Slebos DJ, Degano B, Valipour A, et al. Design for a multicenter, randomized, sham-controlled study to evaluate safety and efficacy after treatment with the Nuvaira(R) lung denervation system in subjects with chronic obstructive pulmonary disease (AIRFLOW-3). BMC Pulm Med. 2020;20(1):41. doi:10.1186/s12890-020-1058-5
38. Weinstein GS, Levin B Effect of crossover on the statistical power of randomized studies. Ann Thorac Surg. 1989;48(4):490–495 doi:10.1016/S0003-4975(10)66846-4
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.