Back to Journals » Open Access Journal of Sports Medicine » Volume 8
Treatment strategies for the female athlete triad in the adolescent athlete: current perspectives
Authors Thein-Nissenbaum J, Hammer E
Received 22 July 2016
Accepted for publication 24 December 2016
Published 4 April 2017 Volume 2017:8 Pages 85—95
DOI https://doi.org/10.2147/OAJSM.S100026
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Andreas Imhoff
Jill Thein-Nissenbaum,1 Erin Hammer2
1Department of Family Medicine, University of Wisconsin-Madison, Madison, WI, USA; 2Department of Orthopedics and Rehabilitation, University of Wisconsin-Madison, Madison, WI, USA
Abstract: Since the passage of Title IX in 1972, female sports participation has dramatically increased. The benefits of physical activity, including decreased risk for heart disease and diabetes as well as improved body image and self-esteem, far outweigh the risks. However, a select population of adolescent and young adult females may experience symptoms related to the female athlete triad (Triad), which refers to the interrelatedness of energy availability, menstrual function, and bone mineral density (BMD). These conditions often manifest clinically as disordered eating behaviors, menstrual irregularity, and stress fractures; an individual may suffer from 1 or all of the Triad components simultaneously. Because of the complex nature of the Triad, treatment is challenging and requires a multidisciplinary approach. Team members often include a physician, psychologist or psychiatrist, nutritionist or dietitian, physical therapist, athletic trainer, coach, family members, and most importantly, the patient. A thorough physical examination by a primary care physician is essential to identify all organs/systems that may be impacted by Triad-related conditions. Laboratory tests, assessment of bone density, nutritional assessment, and behavior health evaluation guide the management of the female athlete with Triad-related conditions. Treatment of the Triad includes adequate caloric consumption to restore a positive energy balance; this is often the first step in successful management of the Triad. In addition, determining the cause of menstrual dysfunction (MD) and resumption of menses is very important. Nonpharmacologic interventions are the first choice; pharmacologic treatment for MD is reserved only for those patients with symptoms of estrogen deficiency or infertility. Lastly, adequate intake of calcium and vitamin D is critical for lifelong bone health. For this review, a comprehensive search of relevant databases from the earliest dates to July 2016 was performed. Keywords, including female athlete triad, adolescent female athlete, disordered eating, eating disorder, low energy availability, relative energy deficit, anorexia, bulimia, menstrual dysfunction, amenorrhea, oligoamenorrhea, bone mineral density, osteopenia, osteoporosis, stress fracture, and stress reaction, were utilized to search for relevant articles. Articles that directly addressed assessment and management of any 1 or all of the Triad components were included in this comprehensive review. The purpose of this narrative review is to provide the reader with the latest terms used to define the components of the female athlete triad, to discuss examination and diagnosis of the Triad, and lastly, to provide the reader with the latest evidence to successfully implement a multidisciplinary treatment approach when providing care for the adolescent female athlete who may be suffering from Triad-related components.
Keywords: energy availability, menstrual dysfunction, bone mineral density, treatment
Introduction and history
Title IX of the educational amendments requires that any federally funded programs or institutions (such as high schools and universities) provide equal opportunity for participation in sports and extracurricular activities regardless of gender.1 Since the implementation of Title IX in 1972, the number of females participating in sports has dramatically increased. In the 2014–2015 school year, there were 7.8 million high school athletes participating in sports; females comprised ~42% of the population.2 Sport participation and physical activity in females boasts numerous benefits, including improved self-esteem and body image, higher academic success, and greater bone mineral density (BMD) as compared to nonactive females.3 The benefits of sport participation far outweigh the risks. However, in the 1980s and 1990s, clinicians and researchers identified a unique condition in a subset of female athletes, and in 1997, the American College of Sports Medicine (ACSM) established a position stand on the female athlete triad (Triad). The initial terms used to define the Triad were disordered eating (DE), amenorrhea, and osteoporosis.4 However, researchers and clinicians soon realized that these terms, with strict definitions and inclusion criteria, were too restrictive; many females suffering from components of the Triad were not successfully identified. It was discovered that Triad symptoms occur along a spectrum, and not as “absolute” ones.5 As such, an updated position stand was published in 2007, utilizing the terms low energy availability (EA) (with or without an eating disorder [ED]), menstrual dysfunction (MD), and low BMD.3 With the newly established definitions, as well as the understanding that the conditions occur along a spectrum, more females are successfully identified as suffering from the Triad and receive proper care.3 In addition, a consensus statement, endorsed by the Female Athlete Triad Coalition, ACSM, and the American Medical Society for Sports Medicine (AMSSM), was recently published utilizing the terms set forth in the 2007 position stand.6 Recently, other terms, such as Relative Energy Deficiency in Sports (RED-S), have gained attention in the literature.7 RED-S addresses a multisystems approach and discusses the effect of energy deficiency on numerous systems, including the gastrointestinal, metabolic, endocrine, and immunological systems. Advocates of RED-S theorize that Triad components are only a small portion of what is affected by lack of appropriate caloric intake.7 Although numerous systems may indeed be affected by a negative energy balance, this review employs terms related solely to the Triad: low EA, MD, and low BMD.3
The purpose of this narrative review is to define Triad-related components and discuss examination and diagnosis of the Triad and multidisciplinary management of Triad-related components. To achieve this, a comprehensive search of relevant databases, including MEDLINE, Cochrane Database of Systematic Reviews and Controlled Trials, and Scopus, from the earliest dates to July 2016 was performed. Keywords that were used in the search include the following: adolescent female athlete, disordered eating, eating disorder, low energy availability, relative energy deficit, anorexia, bulimia, menstrual dysfunction, amenorrhea, oligoamenorrhea, bone mineral density, osteopenia, osteoporosis and stress fracture, and stress reaction. All articles were reviewed for relevance. Those articles that directly addressed identification, assessment, and management of any 1 or all of the Triad components in the adolescent and young adult female were included in this comprehensive review.
The female athlete triad
The components of the female athlete triad are low EA (with or without an ED), MD, and low BMD.3 These conditions can occur in isolation or in combination. Although the estimated prevalence of the triad is low (0%–1.2%), the prevalence of any 2 or any 1 of the triad conditions ranges from 2.7% to 27.0% and 16.0% to 60.0%, respectively.8 As such, it is imperative that if 1 condition is identified, the clinician screen the athlete for the other conditions.
Low EA
EA is simple math: energy intake (in the form of calories) minus energy expenditure (utilized during exercise, normal activities of daily living, and normal bodily functions).3 EA occurs along a spectrum from optimal EA to low EA (Figure 1). Energy is first used for exercise; the remaining energy serves to regulate bodily functions, such as tissue healing, cardiovascular functioning, and menstruation.3,9 When EA falls into a negative balance, the body reacts by reducing the amount of energy needed for bodily functions, such as cellular maintenance, thermoregulation, and reproduction.3 This “shift” in energy distribution is not without negative consequences, as it impairs overall health.
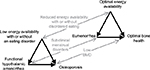  | Figure 1 Female athlete triad. Notes: The spectrum’s of energy availability, menstrual function, and bone mineral density along which female athletes are distributed (narrow arrows). An athlete’s condition moves along each spectrum at a different rate, in one direction or the other, according to her diet and exercise habits. Energy availability, defined as dietary energy intake minus exercise energy expenditure, affects bone mineral density both directly via metabolic hormones and indirectly via effects on menstrual function and thereby estrogen (thick arrows). Reproduced with permission from Nattiv A, Loucks AB, Manore MM, Sanborn CF, Sundgot-Borgen J, Warren MP; American College of Sports Medicine. American College of Sports Medicine position stand. The female athlete triad. Med Sci Sports Exerc. 2007;39(10):1867–1882. Promotional and commercial use of the material in print, digital or mobile device format is prohibited without the permission from the publisher Wolters Kluwer. Please contact [email protected] for further information.3 Abbreviation: BMD, bone mineral density. |
Low EA can occur with or without an ED. Unintentional low EA is due to a lack of knowledge on the part of the athlete; she is simply unaware of the amount of daily calories needed to offset her training and normal bodily functions.10,11 Other athletes intentionally fall into low EA by dramatically reducing caloric intake, exercising excessively, or through a combination of both. Purposeful caloric restriction often manifests as DE and may involve restrictive eating, elimination of specific food groups, fasting, skipping meals, or use of diet pills, laxatives, diuretics, or binge-eating followed by purging.3,11,12 The most recent Diagnostic and Statistical Manual of Mental Disorders fifth edition (DSM-V) identifies specific subcategories of DE.13 The most prevalent category is Eating Disorder-Not Otherwise Specified (ED-NOS), where the individual manifests with some DE behaviors, but fails to meet the criteria of anorexia nervosa (AN) or bulimia nervosa (BN). Common signs and symptoms associated with DE behaviors are listed in Table 1.3,10,11 Research suggests that, regardless of when unhealthy eating attitudes and behaviors manifest, the behaviors are formed during adolescence.14–16 The prevalence of DE (AN, BN, and ED-NOS) has been examined in the high school population, in both athletes and sedentary individuals. The Eating Disorder Examination-Questionnaire (EDE-Q) is a self-report questionnaire reflecting eating behaviors over the previous 28 days. It has high internal consistency and moderate-to-high concurrent and criterion validity.17–19 Using the EDE-Q, the prevalence of DE behaviors in adolescent and young female athletes has been found to range from 18.0s% to 35.0%.20–22 Using an energy expenditure calculation, Hoch et al23 determined the prevalence of DE in high school athletes and sedentary controls to be 36% and 39%, respectively.
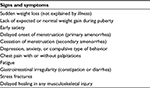  | Table 1 Signs and symptoms commonly seen with disordered eating behaviors Note: Data from Nattiv et al,3 Beals and Hill,10 and Beals.11 |
The negative consequences, both short- and long-term, of DE are substantial. Females with DE report suffering from low self-esteem, depression, and anxiety. They may also experience cardiovascular, endocrine, gastrointestinal, and renal complications as well as poor sleep patterns.24 The long-term effects are significant; DE in adolescence is negatively associated with socioeconomic achievement during early adulthood and actually predicts DE behavior in adulthood.25 In addition, DE behaviors affect sport performance. Junior elite female swimmers with low EA had poor sport performance compared to healthy peers; high school athletes with DE behaviors were more than twice as likely to sustain a musculoskeletal injury during their competitive season.22,26
Another avenue in which athletes can fall into low EA is by exercising excessively. In a study of over 300 high school female athletes, almost 60% of females reported training outside of scheduled practice sessions.22 The clinical significance of this is substantial: in females with DE behaviors, “overexercising” is the variable most strongly associated with suicidal behaviors.27
MD
Similar to EA, menstrual function occurs along a spectrum, ranging from eumenorrhea (normal menses) to amenorrhea (Figure 1).3 The overarching term of MD includes subcategories of primary amenorrhea, secondary amenorrhea, and oligomenorrhea.3 Primary amenorrhea is a delay in the onset of menses, where the female has not started menstruating by age 15.3,28 Secondary amenorrhea is a cessation of menstruation for 3 consecutive months in a female who has started menstruating. Lastly, oligomenorrhea is menstrual cycles occurring regularly, but >35 days apart.3
Determining the prevalence of MD comes with challenges, particularly in the adolescent population. Menstrual irregularity immediately after the onset of menses is very common. However, normalization of menstruation occurs within 2 years of menarche in 90% of females; as such, researchers exclude the first 2 years postmenarche when considering MD.29 The prevalence of MD in adolescent females has been studied via self-report. In the high school population, MD (excluding the 2 years after the onset of menses) ranges from 18.8% to 54%.20–23 Lean build athletes and esthetic athletes (cheerleading, pom-pon squad, dance team, diving, and gymnastics) have a significantly higher prevalence of MD (26.7% and 28.2%, respectively) when compared to general high school athletes.20,22
Although many high school athletes feel that a lack of menstruation is a sign of “being in shape” and consider their period an inconvenience while participating in sport, the negative consequences of MD are significant. Amenorrheic women experience infertility, as they do not develop ovarian follicles or ovulate.3 However, with proper treatment, ovulation can resume and often does prior to menstruation. As such, the female athlete may be ovulating, but is unaware of it because she has not resumed menstruating. Therefore, there is a risk of an unexpected pregnancy if a form of birth control is not utilized during this time. Other consequences of MD include luteal deficiency, which may also cause infertility due to poor follicular development or failure of implantation of a fertilized egg. In addition, low estrogen levels, which are commonly seen with MD, can result in impaired endothelium and dependent arterial vasodilation, which reduces the perfusion of working muscle.30
Low BMD
BMD occurs across a spectrum, ranging from optimal bone health to osteoporosis (Figure 1).3 BMD is diagnosed via dual-energy X-ray absorptiometry (DXA). In postmenopausal women, the terms osteopenia and osteoporosis have clearly established definitions. Osteopenia is defined as a BMD T-score between −1.0 and −2.5, and osteoporosis is a T-score of <−2.5.31 However, these normal values were established using data from Caucasian, postmenopausal woman; as such, they cannot be used when assessing adolescents and premenopausal females. Therefore, the International Society for Clinical Densitometry (ISCD) recommends avoiding the terms osteopenia and osteoporosis in this population. In addition, it is recommended that BMD in this population be expressed as Z-scores, as these scores compare individuals to age- and sex-matched controls. In adolescent and premenopausal females, any Z-score <−2.0 is defined as “low bone density for chronological age”.32
BMD is reflective of numerous other variables, including EA, menstrual status, genetic composition, and environmental factors. Because of this, a 1-time “snapshot” of BMD in the adolescent or young adult female does not provide as much information as does changes in BMD that occur longitudinally.3,33 Therefore, there are a limited number of studies that examine BMD in adolescent athletes.21,23,34,35 In a study by Nichols et al,21 the prevalence of low bone mass in 170 high school females was determined to be 21.8%. In 80 high school female athletes, 13% had a Z-score between −1.0 and −1.9, and 3% had a Z-score of ≤−2.0.23 Ackerman et al34 demonstrated that lumbar BMD Z-scores were lower in amenorrheic athletes as compared to eumenorrheic athletes and nonathletes. In addition, amenorrheic athletes also had impaired bone microarchitecture compared to the other groups.34
The long-term consequences of decreased bone acquisition during adolescence are substantial. Adolescence and young adulthood is a time of maximal bone mass accrual; a lack of maximal bone acquisition may result in osteoporosis later in life.36 Therefore, achieving maximal BMD during adolescence is critical for the overall long-term health of the female’s skeletal system.36,37
Examination and diagnosis
Early detection of signs and symptoms related to the Triad is critical as early identification and early intervention are associated with better outcomes.38 Screening for the Triad should be undertaken as part of the pre-participation physical evaluation on an annual basis, at both the high school and collegiate levels.6 There are multiple screening questionnaires noted in the literature, including the Low Energy Availability in Females Questionnaire,39 RED-S Risk Assessment Model,7 and the Female Athlete Triad: Cumulative Risk Assessment.6 Currently, a “gold standard” screening tool does not exist; however, screening questions should address all Triad-related components (Table 2).6
  | Table 2 Triad consensus panel screening questions Note: Reproduced from Br J Sports Med. 2014 female athlete triad coalition consensus statement on treatment and return to play of the female athlete triad: 1st International Conference held in San Francisco, California, May 2012 and 2nd International Conference held in Indianapolis, Indiana, May 2013. De Souza MJ, Nattiv A, Joy E, et al; Expert Panel. 2014;48(4):289, with permission from BMJ Publishing Group Ltd.6 |
Screening questionnaires can be utilized at any time if a health care professional suspects that an athlete has developed Triad-related conditions outside of the preseason setting. These tools can also be used to assess any female, regardless of whether she participates in organized sport. In the high school and collegiate setting, struggling athletes are often first identified by their athletic trainer or teammates and then encouraged to seek help. Progressive weight loss, DE behaviors, and irritability can be clues that an athlete suffers from 1 or all of the Triad-related conditions. Some athletes present due to concerns about declining performance or are identified after sustaining an overuse injury, such as a stress fracture. If 1 component of the Triad exists, a more thorough examination of other Triad components should ensue. The care of the athlete should be managed by a health care professional knowledgeable of Triad-related conditions. Often, this is a primary care physician who can perform the appropriate studies and identify team members essential in the treatment process.
Low EA
The workup for low EA should be tailored to the patient’s severity of illness and resources available. Adolescent and young adult females who might be suffering from low EA should undergo a comprehensive physical examination combined with appropriate laboratory tests. A body mass index (BMI) of <17.5 kg/m2 or <85% of expected body weight may indicate low EA, but is a flawed measure. Absolute BMI cutoffs should not be used for adolescents.6 BMI can be an imprecise measure in the athletic population with higher percentage of lean body mass compared to nonathletes.40 Furthermore, a state of low EA can occur in athletes with a normal BMI and in those whose weight is stable if they do not consume enough calories to account for energy used in exercise. The assessment of EA, determined by detailed dietary intake and energy expenditure records, is time intensive and requires the expertise of a trained sports dietitian or exercise physiologist.6 As such, this may not be possible in environments with minimal access to these resources. Dietary intake and energy expenditure records also rely on accurate self-reporting by the athlete, which is time intensive.
Signs of low EA on physical exam include bradycardia, lanugo (course hair) on the extremities, and evidence of dehydration.26 Based on the findings from the physical examination, the provider may order the following labs: serum electrolytes; renal function (BUN, Cr); calcium; liver function tests; thyroid-stimulating hormone (TSH); complete blood count, differential, and platelets; urinalysis; iron studies; vitamin D; vitamin B12; magnesium; and phosphorous.26 The results of these tests will give the provider a profile of the patient’s hydration status and kidney and liver function and identify if anemia or other medical conditions are present. Lastly, in patients who present with overt AN, heart palpitations, syncope, and a resting heart rate in supine <50 bpm, an electrocardiogram is recommended.26
MD
Functional hypothalamic amenorrhea due to low EA is a diagnosis of exclusion; athletes should be evaluated for other causes of primary or secondary amenorrhea including pregnancy and endocrine system disorders. First, pregnancy should be excluded in all cases. In athletes who have yet to reach menarche, outflow tract obstruction is an important consideration. Congenital causes of primary amenorrhea include imperforate hymen and Müllerian agenesis; a pelvic ultrasound may be obtained.6,41 The provider should consider causes of primary ovarian insufficiency such as Turner syndrome, which can be suggested by dysmorphic features such as a webbed neck or low hairline.41 Disruption of the hypothalamic or pituitary function can be evaluated with the following laboratory tests: serum luteinizing hormone, follicle-stimulating hormone, prolactin, total and free estradiol, total and free testosterone, TSH and free T4, and dehydroepiandrosterone and its sulfate.26 These tests evaluate for thyroid dysfunction, hyperprolactinemia, primary ovarian insufficiency, hypothalamic and pituitary disorders, and polycystic ovary syndrome. They should be performed based on clinical suspicion due to symptoms and physical exam findings such as hirsutism, acne, fatigue, and dry skin.6,42 In the case where the diagnosis of MD is due to a medical condition other than negative energy balance, the athlete may be referred to an endocrinologist for further evaluation and management.
Low BMD
Low BMD is often first identified when an athlete presents with a stress fracture. A DXA scan is the preferred method for evaluating bone density. Indications for obtaining a DXA scan are multifactorial and include factors such as BMI ≤17.5 kg/m2, onset of menses ≥16 years of age, a history of DSM-V-diagnosed ED, and a history of stress fracture(s).6 A detailed algorithm is reported elsewhere in the literature.6 As previously stated, a 1-time snapshot of an adolescent female’s BMD will not be as helpful as monitoring bone acquisition over time. However, a baseline should be established in at-risk athletes. The recommended BMD sites in the adolescent female include total body less head and the posterior–anterior spine.43 The reader is referred elsewhere for details regarding BMD sites in the adolescent female.43
Management of the triad
Management of Triad-related components is complex and multifactorial. As such, a multidisciplinary approach is vital.44 Each team member provides a unique perspective and has a well-defined role within the team (Table 3). Because every athlete has unique needs, not every health care professional listed in Table 3 may be involved in the care of the athlete; the primary care physician typically oversees the multidisciplinary team and makes recommendations related to which team members are necessary in the care of the athlete.
The need for an individualized program to provide appropriate care for the athlete cannot be overemphasized. Research has shown large intraindividual variability in physical characteristics and physiological attributes in female athletes who play the same sport and even large variability in the same position within that sport.45 Nikolaidis et al examined 61 female volleyball players from the top 2 leagues in Greece. Athletes were divided into 3 groups: under 14, 14–18, and over 18 years. The authors found significant large intraindividual variability, regardless of age, level of play, and experience.45 As such, a training program for 1 middle blocker may not be effective for another middle blocker on the same team. Similarly, 1 program to address low EA in a basketball player may be ineffective in another player on the same team. Therefore, each program must meet the unique needs of the athlete.
Low EA
The goal of treatment for females with low EA is to resume a positive energy balance. Management of low EA is dependent upon how the athlete fell into negative energy balance in the first place. Recall, some athletes have low EA simply because they are unaware of the caloric demands of their sport combined with normal bodily functions. If this is indeed the case, a referral to a registered dietitian (RD) or sports nutritionist is adequate. Females with unintentional low EA typically do not present with the psychosocial components seen in individuals with intentional DE behaviors, such as depression, anxiety, and obsessive compulsive disorder. As such, a referral to a psychologist is not indicated.6
In the case where low EA is due to intentional restrictive caloric intake and/or excessive caloric expenditure (in the form of “overexercising”), treatment requires a multidisciplinary team. In this case, reversal of low EA will not be successful without the input of a mental health professional.46,47 The physician, RD or sports nutritionist, psychologist, or other mental health professional each has a role. The goals for this athlete include normalizing her eating behaviors and altering negative beliefs related to food and body image.48
If the ED is mild, or low EA is unintentional, the overseeing physician may allow the athlete to continue to practice and compete with restrictions. In this situation, the athletic trainer or physical therapist will play a key role in the design of the athlete’s training program. Typically, the athletic trainer has more contact with the coaching staff and athlete, and therefore, is best suited to design a training program for the athlete. However, in situations where there is no athletic trainer present or the athletic trainer works for limited hours, the physical therapist in the outpatient setting may be charged with training program design. Regardless, the physical therapist or athletic trainer will need to work closely with the RD or sports nutritionist to design a program that maintains a positive energy balance for the athlete. As caloric consumption changes, the athlete’s training program must mirror these changes to maintain adequate EA. As such, the physical therapist or athletic trainer and RD or sports nutritionist must maintain constant communication. In addition, the athlete must have a thorough understanding that training is restricted to the program designed for them; “overtraining” or training outside of the designated program is forbidden.
Pharmacologic management of EDs may be considered in patients with conditions resistant to non-pharmacologic means or with certain psychological comorbidities. There is no evidence that selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants, or antipsychotics are effective in treating AN.49,50 However, a Cochrane review in 2003 found that antidepressant agents were effective for the treatment of BN compared to placebo.51 In particular, the SSRI, fluoxetine, has been the most studied and has been found to be superior to placebo for reduction of binging and vomiting frequency, eating attitudes, and depression.50 If other comorbid conditions, such as anxiety, depression, and obsessive compulsive behavior, are identified in the female with DE, psychotropic medications can be beneficial to treat those particular conditions.6
Measurements of weight and BMI should not be a focus when working with the female athlete who is trying to achieve a positive energy balance. However, weight gain and an increase in BMI will most likely be observed once positive EA is achieved. Athletes may perceive this as a negative consequence. On the contrary, research has proven otherwise. In a study by Nikolaidis, 54 female soccer players were divided into 3 groups by BMI: <21, 21–23, and >23 kg/m2.52 A battery of fitness tests was subsequently performed. The author found that soccer players with average BMI (21–23 kg/m2) were stronger than those with low BMI and had better mean power than those with high BMI. The author concluded that achieving an optimal BMI may lead to improvements in power, which may result in enhanced performance.52 This information is an essential component of patient education when working with the young female athlete who is trying to improve overall performance.
MD
If other causes of MD have been ruled out by the gynecologist or endocrinologist, resumption of menses should occur through restoration of EA without pharmacologic intervention. In a retrospective study of 51 female collegiate athletes with MD who resumed menses without pharmacologic intervention, percentage weight gain was identified as the strongest predictor of resumption of menses.53
Caloric consumption is typically increased gradually, with enough of an increase to result in a gain of approximately 1 pound/week.6 Because of this slow, gradual increase, menses may not resume immediately. In a study by Arends et al, the average time to resumption of menses without pharmacologic intervention was 15 months.53 Therefore, the athlete must be educated that, even though she has not yet resumed menses, she may be ovulating, and some form of contraception is required if the athlete does not desire to become pregnant. Careful monitoring of EA and weight gain is paramount to the resumption of menses.
Pharmacologic treatment for MD is reserved only for those patients with symptoms of estrogen deficiency or infertility.6 Oral and nonoral contraceptives for MD have been studied extensively and are not recommended.6,54,55 Hormone-containing contraceptives do induce withdrawal bleeding, but this does not restore spontaneous menses, nor is it associated with improvement in the function of the endocrine system. Furthermore, contraceptive-induced bleeding might provide false reassurance to providers and patients that the hormonal milieu is improving and delay efforts to improve nutrition and exercise behaviors.6 While not indicated for amenorrhea, providers should not restrict hormone-containing contraceptives for contraception when needed.
Low BMD
The goal of treating females with low BMD is to prevent further bone loss and attempt to recover lost BMD. Research has shown that weight gain and resumption of menses are critical to prevent further bone loss and to improve overall bone health.56–60 A few studies have shown that weight gain in isolation has a positive effect of BMD.61,62
If amenorrhea goes untreated, females will continue to lose bone mass at a rate of 2–3% per year.57,58 It is critical for the young female athlete to understand the ramifications of amenorrhea: a lack of menstruation for as little as 6 months during adolescence, which is a critical time for bone acquisition, can result in a loss of bone of 1–2%.57,58
In addition to weight gain and resumption of menses, weight-bearing exercise is a proven nonpharmacological strategy for increasing BMD.63 Bone is highly responsive to loading activities seen with impact and resistance training.64 Interestingly, weight-bearing exercise combined with resistance training increases bone mass in the premenopausal years; however, the results are mixed when each activity is performed in isolation.57,65 In addition, fat-free mass has been shown to be a significant positive predictor of changes in bone and was even a stronger predictor of BMD change than the change in fat mass or body weight.57,65 As such, both weight-bearing activity and resistance training, which improve fat-free mass, play a role in bone acquisition in the premenopausal female. Most importantly, any training program geared toward increasing BMD needs to have both components.
Athletic trainers and physical therapists play a key role in educating the athlete on interventions to improve BMD in the female athlete with Triad-related conditions. Once resistance training and weight-bearing activities are allowed, the athletic trainer or physical therapist will design an exercise program that includes the appropriate amount of impact and resistance training while concurrently meeting the sport-specific goals of the athlete. For example, a female gymnast who was recently suffering from low EA and secondary amenorrhea may have recently resumed menstruation after a weight gain and completing sessions of cognitive behavioral therapy with a psychologist. She has been cleared by her physician to resume training. Her goal is to compete in floor exercise and vault. Combining weight-bearing exercise and resistance training with her goal of competing in a floor routine and vault can be done with relative ease; her sport-specific training has a weight-bearing component. On the other hand, designing a training program for a swimmer proves to be more challenging. Because her primary activity for training does not include weight-bearing, the athletic trainer or physical therapist must carefully design a training program that affords her adequate time in the pool to train, but has components of land-based weight-bearing and resistance training to increase BMD. All of this must be done while maintaining a positive energy balance.
Regarding pharmacologic strategies, estrogen-containing oral contraceptives are not effective for increasing BMD and should not be used for that purpose.6,54,55,66,67 There is no evidence regarding the use of transdermal estrogen or testosterone in the treatment of low BMD in amenorrheic athletes. A Cochrane review in 2014 regarding the risk of fractures in women who use hormone-containing contraceptives suggested that depot medtroxyprogesterone acetate was associated with decreased BMD, though this was not specific to the athletic or ED population.68
Vitamin D supplementation is recommended to maintain serum 25-hydroxyvitamin D levels between 32 and 50 ng/mL, and calcium intake should be between 1000 and 1300 mg/day.6,69 Serum 25-hydroxyvitamin D levels <30 ng/mL are associated with increased incidence of stress fracture.70 There is evidence that vitamin D and calcium supplementation can prevent stress fractures in the athletic population.71
There is currently no strong recommendation for the use of bisphosphonates in the prevention or treatment of low BMD for premenopausal athletes. While BMD Z-scores <−1.0 in athletes may confer increased fracture risk,3 it is not clear that pharmacotherapy is effective in preventing fracture or improving bone health outcomes. Furthermore, bisphosphonates have long half-lives, which is of concern in the premenopausal population given the teratogenic risks. However, if all nonpharmacologic treatments have been exhausted for the treatment of low BMD in the athletic population, it is appropriate to pursue consultation with an endocrinologist to discuss the initiation of bisphosphonate therapy.
Conclusion
The components of the female athlete triad are low EA (with or without an ED), MD, and low BMD. These conditions can occur collectively or in isolation. Signs and symptoms commonly observed include rapid weight loss, restrictive eating or fasting, and use of diet pills; cessation of menstruation in a eumenorrheic female; and stress fractures and overuse injuries that are slow to heal. Coaches, parents, health care professionals, and fellow teammates should be aware of any of these signs and symptoms and discuss any concerns with the athlete privately. Screening for the Triad should occur during the preparticipation examination or any time components of the Triad are suspected. Specific test and measures, including laboratory testing and assessment of BMD, should be ordered at the discretion of a primary health care provider who is knowledgeable in the assessment and treatment of the Triad. A multidisciplinary team, spearheaded by the primary care physician, must work closely together to provide optimal care for the athlete. All team members must be consistent in the information provided to the athlete; inconsistent information and “mixed messages” will lead to a lack of trust and confidence in the team by the athlete. Non-pharmacologic interventions are the first choice in treating Triad components; pharmacologic intervention should be used with scrutiny. Throughout the recovery process, the athlete needs to feel respected and that her feelings, thoughts, and opinions about the recovery process plan are valued. With a team of experts working collaboratively with each other and the female athlete, the goal of resuming a healthy lifestyle, including successful participation in sport, can be achieved.
Disclosure
The authors report no conflicts of interest in this work.
References
U.S. Department of Justice. Overview of Title IX of the Education Amendments of 1972. Available from: https://www.justice.gov/crt/overview-title-ix-education-amendments-1972-20-usc-1681-et-seq. Accessed May 25, 2016. | ||
The National Federation of State High School Associations. 2014–15 high school athletics participation survey. Available from: http://www.nfhs.org/ParticipationStatistics/PDF/2014-15_Participation_Survey_Results.pdf. Accessed May 25, 2016. | ||
Nattiv A, Loucks AB, Manore MM, Sanborn CF, Sundgot-Borgen J, Warren MP; American College of Sports Medicine. American College of Sports Medicine position stand. The female athlete triad. Med Sci Sports Exerc. 2007;39(10):1867–1882. | ||
Otis CL, Drinkwater B, Johnson M, Loucks A, Wilmore J. American College of Sports Medicine position stand. The Female Athlete Triad. Med Sci Sports Exerc. 1997;29(5):i–ix. | ||
Matzkin E, Curry EJ, Whitlock K. Female athlete triad: past, present, and future. J Am Acad Orthop Surg. 2015;23(7):424–432. | ||
De Souza MJ, Nattiv A, Joy E, et al; Expert Panel. 2014 female athlete triad coalition consensus statement on treatment and return to play of the female athlete triad: 1st International Conference held in San Francisco, California, May 2012 and 2nd International Conference held in Indianapolis, Indiana, May 2013. Br J Sports Med. 2014;48(4):289. | ||
Mountjoy M, Sundgot-Borgen J, Burke L, et al. The IOC consensus statement: beyond the Female Athlete Triad – Relative Energy Deficiency in Sport (RED-S). Br J Sports Med. 2014;48(7):491–497. | ||
Javed A, Tebben PJ, Fischer PR, Lteif AN. Female athlete triad and its components: toward improved screening and management. Mayo Clin Proc. 2013;88(9):996–1009. | ||
Rumball JS, Lebrun CM. Preparticipation physical examination: selected issues for the female athlete. Clin J Sport Med. 2004;14(3):153–160. | ||
Beals KA. Eating behaviors, nutritional status, and menstrual function in elite female adolescent volleyball players. J Am Diet Assoc. 2002;102(9):1293–1296. | ||
Beals KA, Hill AK. The prevalence of disordered eating, menstrual dysfunction, and low bone mineral density among US collegiate athletes. Int J Sport Nutr Exerc Metab. 2006;16(1):1–23. | ||
Beals KA. Eating disorder and menstrual dysfunction screening, education, and treatment programs. Phys Sportsmed. 2003;31(7):33–38. | ||
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Available from: http://www.dsm5.org/about/Pages/Default.aspx. Accessed May 26, 2016. | ||
Ackard DM, Peterson CB. Association between puberty and disordered eating, body image, and other psychological variables. Int J Eat Disord. 2001;29(2):187–194. | ||
Kotler LA, Cohen P, Davies M, Pine DS, Walsh BT. Longitudinal relationships between childhood, adolescent, and adult eating disorders. J Am Acad Child Adolesc Psychiatry. 2001;40(12):1434–1440. | ||
Vohs KD, Heatherton TF, Herrin M. Disordered eating and the transition to college: a prospective study. Int J Eat Disord. 2001;29(3):280–288. | ||
Binford RB, Le Grange D, Jellar CC. Eating disorders examination versus eating disorders examination-questionnaire in adolescents with full and partial-syndrome bulimia nervosa and anorexia nervosa. Int J Eat Disord. 2005;37(1):44–49. | ||
Carter JC, Stewart DA, Fairburn CG. Eating disorder examination questionnaire: norms for young adolescent girls. Behav Res Ther. 2001;39(5):625–632. | ||
Fernandez WG, Yard EE, Comstock RD. Epidemiology of lower extremity injuries among U.S. high school athletes. Acad Emerg Med. 2007;14(7):641–645. | ||
Nichols JF, Rauh MJ, Barrack MT, Barkai HS, Pernick Y. Disordered eating and menstrual irregularity in high school athletes in lean-build and nonlean-build sports. Int J Sport Nutr Exerc Metab. 2007;17(4):364–377. | ||
Nichols JF, Rauh MJ, Lawson MJ, Ji M, Barkai HS. Prevalence of the female athlete triad syndrome among high school athletes. Arch Pediatr Adolesc Med. 2006;160(2):137–142. | ||
Thein-Nissenbaum JM, Rauh MJ, Carr KE, Loud KJ, McGuine TA. Associations between disordered eating, menstrual dysfunction, and musculoskeletal injury among high school athletes. J Orthop Sports Phys Ther. 2011;41(2):60–69. | ||
Hoch AZ, Pajewski NM, Moraski L, et al. Prevalence of the female athlete triad in high school athletes and sedentary students. Clin J Sport Med. 2009;19(5):421–428. | ||
Tabler J, Utz RL. The influence of adolescent eating disorders or disordered eating behaviors on socioeconomic achievement in early adulthood. Int J Eat Disord. 2015;48(6):622–632. | ||
Herpertz-Dahlmann B, Dempfle A, Konrad K, Klasen F, Ravens-Sieberer U, BELLA study group. Eating disorder symptoms do not just disappear: the implications of adolescent eating-disordered behaviour for body weight and mental health in young adulthood. Eur Child Adolesc Psychiatry. 2015;24(6):675–684. | ||
Joy E, Kussman A, Nattiv A. 2016 update on eating disorders in athletes: a comprehensive narrative review with a focus on clinical assessment and management. Br J Sports Med. 2016;50(3):154–162. | ||
Smith AR, Fink EL, Anestis MD, et al. Exercise caution: over-exercise is associated with suicidality among individuals with disordered eating. Psychiatry Res. 2013;206(2–3):246–255. | ||
Barrack MT, Rauh MJ, Nichols JF. Prevalence of and traits associated with low BMD among female adolescent runners. Med Sci Sports Exerc. 2008;40(12):2015–2021. | ||
World Health Organization multicenter study on menstrual and ovulatory patterns in adolescent girls. II. Longitudinal study of menstrual patterns in the early postmenarcheal period, duration of bleeding episodes and menstrual cycles. World Health Organization Task Force on Adolescent Reproductive Health. J Adolesc Health Care. 1986;7(4):236–244. | ||
Harber VJ, Petersen SR, Chilibeck PD. Thyroid hormone concentrations and muscle metabolism in amenorrheic and eumenorrheic athletes. Can J Appl Physiol. 1998;23(3):293–306. | ||
Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO Study Group. World Health Organ Tech Rep Ser. 1994;843:1–129. | ||
Bianchi ML, Baim S, Bishop NJ, et al. Official positions of the International Society for Clinical Densitometry (ISCD) on DXA evaluation in children and adolescents. Pediatr Nephrol. 2010;25(1):37–47. | ||
Khan KM, Liu-Ambrose T, Sran MM, Ashe MC, Donaldson MG, Wark JD. New criteria for female athlete triad syndrome? As osteoporosis is rare, should osteopenia be among the criteria for defining the female athlete triad syndrome? Br J Sports Med. 2002;36(1):10–13. | ||
Ackerman KE, Nazem T, Chapko D, et al. Bone microarchitecture is impaired in adolescent amenorrheic athletes compared with eumenorrheic athletes and nonathletic controls. J Clin Endocrinol Metab. 2011;96(10):3123–3133. | ||
Ackerman KE, Putman M, Guereca G, et al. Cortical microstructure and estimated bone strength in young amenorrheic athletes, eumenorrheic athletes and non-athletes. Bone. 2012;51(4):680–687. | ||
Heaney RP, Abrams S, Dawson-Hughes B, et al. Peak bone mass. Osteoporos Int. 2000;11(12):985–1009. | ||
Julián-Almárcegui C, Gómez-Cabello A, Huybrechts I, et al. Combined effects of interaction between physical activity and nutrition on bone health in children and adolescents: a systematic review. Nutr Rev. 2015;73(3):127–139. | ||
Rosen DS; American Academy of Pediatrics Committee on Adolescence. Identification and management of eating disorders in children and adolescents. Pediatrics. 2010;126(6):1240–1253. | ||
Melin A, Tornberg AB, Skouby S, et al. The LEAF questionnaire: a screening tool for the identification of female athletes at risk for the female athlete triad. Br J Sports Med. 2014;48(7):540–545. | ||
Ode JJ, Pivarnik JM, Reeves MJ, Knous JL. Body mass index as a predictor of percent fat in college athletes and nonathletes. Med Sci Sports Exerc. 2007;39(3):403–409. | ||
Klein DA, Poth MA. Amenorrhea: an approach to diagnosis and management. Am Fam Physician. 2013;87(11):781–788. | ||
Gray SH. Menstrual disorders. Pediatr Rev. 2013;34(1):6–17; quiz 17–18. | ||
Crabtree NJ, Arabi A, Bachrach LK, et al; International Society for Clinical Densitometry. Dual-energy X-ray absorptiometry interpretation and reporting in children and adolescents: the revised 2013 ISCD Pediatric Official Positions. J Clin Densitom. 2014;17(2):225–242. | ||
Warr BJ, Woolf K. The female athlete triad: patients do best with a team approach to care. JAAPA. 2011;24(4):50–55. | ||
Nikolaidis PT, Ziv G, Arnon M, Lidor R. Physical characteristics and physiological attributes of female volleyball players--the need for individual data. J Strength Cond Res. 2012;26(9):2547–2557. | ||
Joy EA, Wilson C, Varechok S. The multidisciplinary team approach to the outpatient treatment of disordered eating. Cur Sports Med Rep. 2003;2(6):331–336. | ||
Temme KE, Hoch AZ. Recognition and rehabilitation of the female athlete triad/tetrad: a multidisciplinary approach. Cur Sports Med Rep. 2013;12(3):190–199. | ||
Zach KN, Smith Machin AL, Hoch AZ. Advances in management of the female athlete triad and eating disorders. Clin Sports Med. 2011;30(3):551–573. | ||
Dold M, Aigner M, Klabunde M, Treasure J, Kasper S. Second-generation antipsychotic drugs in anorexia nervosa: a meta-analysis of randomized controlled trials. Psychother Psychosom. 2015;84(2):110–116. | ||
Flament MF, Bissada H, Spettigue W. Evidence-based pharmacotherapy of eating disorders. Int J Neuropsychopharmacol. 2012;15(2):189–207. | ||
Bacaltchuk J, Hay P. Antidepressants versus placebo for people with bulimia nervosa. Cochrane Database Syst Rev. 2003;(4):CD003391. | ||
Nikolaidis PT. Weight status and physical fitness in female soccer players: is there an optimal BMI? Sport Sci Health. 2014;10(1):41–48. | ||
Arends JC, Cheung MY, Barrack MT, Nattiv A. Restoration of menses with nonpharmacologic therapy in college athletes with menstrual disturbances: a 5-year retrospective study. Int J Sport Nutr Exerc Metab. 2012;22(2):98–108. | ||
Bergström I, Crisby M, Engström AM, et al. Women with anorexia nervosa should not be treated with estrogen or birth control pills in a bone-sparing effect. Acta Obstet Gynecol Scand. 2013;92(8):877–880. | ||
Warren MP, Brooks-Gunn J, Fox RP, et al. Persistent osteopenia in ballet dancers with amenorrhea and delayed menarche despite hormone therapy: a longitudinal study. Fertil Steril. 2003;80(2):398–404. | ||
Fredericson M, Kent K. Normalization of bone density in a previously amenorrheic runner with osteoporosis. Med Sci Sports Exerc. 2005;37(9):1481–1486. | ||
Miller KK, Lee EE, Lawson EA, et al. Determinants of skeletal loss and recovery in anorexia nervosa. J Clin Endocrinol Metab. 2006;91(8):2931–2937. | ||
Misra M. Long-term skeletal effects of eating disorders with onset in adolescence. Ann N Y Acad Sci. 2008;1135:212–218. | ||
Viapiana O, Gatti D, Dalle Grave R, et al. Marked increases in bone mineral density and biochemical markers of bone turnover in patients with anorexia nervosa gaining weight. Bone. 2007;40(4):1073–1077. | ||
Zanker CL, Cooke CB, Truscott JG, Oldroyd B, Jacobs HS. Annual changes of bone density over 12 years in an amenorrheic athlete. Med Sci Sports Exerc. 2004;36(1):137–142. | ||
Bolton JG, Patel S, Lacey JH, White S. A prospective study of changes in bone turnover and bone density associated with regaining weight in women with anorexia nervosa. Osteoporos Int. 2005;16(12):1955–1962. | ||
Compston JE, McConachie C, Stott C, et al. Changes in bone mineral density, body composition and biochemical markers of bone turnover during weight gain in adolescents with severe anorexia nervosa: a 1-year prospective study. Osteoporos Int. 2006;17(1):77–84. | ||
Bailey CA, Brooke-Wavell K. Exercise for optimising peak bone mass in women. Proc Nutr Soc. 2008;67(1):9–18. | ||
Wallace BA, Cumming RG. Systematic review of randomized trials of the effect of exercise on bone mass in pre- and postmenopausal women. Calcif Tissue Int. 2000;67(1):10–18. | ||
American College of Sports Medicine, Chodzko-Zajko WJ, Proctor DN, et al. American College of Sports Medicine position stand. Exercise and physical activity for older adults. Med Sci Sports Exerc. 2009;41(7):1510–1530. | ||
Bell DR, Blackburn JT, Ondrak KS, et al. The effects of oral contraceptive use on muscle stiffness across the menstrual cycle. Clin J Sport Med. 2011;21(6):467–473. | ||
Cobb KL, Bachrach LK, Sowers M, et al. The effect of oral contraceptives on bone mass and stress fractures in female runners. Med Sci Sports Exerc. 2007;39(9):1464–1473. | ||
Lopez LM, Grimes DA, Schulz KF, Curtis KM, Chen M. Steroidal contraceptives: effect on bone fractures in women. Cochrane Database Syst Rev. 2014;(6):CD006033. | ||
Ross AC, Taylor CL, Yaktine AL, Del Valle HB, editors. Dietary Reference Intakes for Calcium and Vitamin D. Washington, DC: National Academies Press; 2011. | ||
Shimasaki Y, Nagao M, Miyamori T, et al. Evaluating the risk of a fifth metatarsal stress fracture by measuring the serum 25-hydroxyvitamin D levels. Foot Ankle Int. 2016;37(3):307–311. | ||
Lappe J, Cullen D, Haynatzki G, Recker R, Ahlf R, Thompson K. Calcium and vitamin D supplementation decreases incidence of stress fractures in female navy recruits. J Bone Miner Res. 2008;23(5):741–749. | ||
Arvinen-Barrow M, Massey WV, Hemmings B. Role of sport medicine professionals in addressing psychosocial aspects of sport-injury rehabilitation: professional athletes’ views. J Athl Train. 2014;49(6):764–772. | ||
American Academy of Pediatrics. Committee on Sports Medicine and Fitness. Medical concerns in the female athlete. Pediatrics. 2000;106(3):610–613. | ||
Official position of the International Society for Clinical Densitometry. 2005. Available from: http://www.iscd.org/official-positions/2013-iscd-official-positions-pediatric/. Accessed May 26, 2016. | ||
NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy. Osteoporosis prevention, diagnosis, and therapy. JAMA. 2001;285(6):785–795. | ||
Covassin T, Beidler E, Ostrowski J, Wallace J. Psychosocial aspects of rehabilitation in sports. Clin Sports Med. 2015;34(2):199–212. | ||
Walker N, Thatcher J, Lavallee D. Psychological responses to injury in competitive sport: a critical review. J R Soc Promot Health. 2007;127(4):174–180. | ||
Yang J, Schaefer JT, Zhang N, Covassin T, Ding K, Heiden E. Social support from the athletic trainer and symptoms of depression and anxiety at return to play. J Athl Train. 2014;49(6):773–779. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

