Back to Journals » Cancer Management and Research » Volume 11
Total pancreatectomy compared with pancreaticoduodenectomy: a systematic review and meta-analysis
Authors Yang DJ , Xiong JJ , Liu XT , Li J , Dhanushka Layanthi Siriwardena KM , Hu WM
Received 23 November 2018
Accepted for publication 22 March 2019
Published 7 May 2019 Volume 2019:11 Pages 3899—3908
DOI https://doi.org/10.2147/CMAR.S195726
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Beicheng Sun
Du-Jiang Yang,1,* Jun-Jie Xiong,1,* Xue-Ting Liu,2 Jiao Li,3 Kanagarathna Mudiyanselage Dhanushka Layanthi Siriwardena,4 Wei-Ming Hu1
1Department of Pancreatic Surgery, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, People’s Republic of China; 2Department of Gastrointestinal Surgery, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, People’s Republic of China; 3Department of Emergency, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, People’s Republic of China; 4West China School of Medicine, Sichuan University, Chengdu 610041, Sichuan Province, People’s Republic of China
*These authors contributed equally to this work
Aim: To assess whether total pancreatectomy (TP) is as feasible, safe, and efficacious as pancreaticoduodenectomy (PD).
Materials and Methods: Major databases, including PubMed, EMBASE, Science Citation Index Expanded, Scopus and the Cochrane Library, were searched for studies comparing TP and PD between January 1943 and June 2018. The meta-analysis only included studies that were conducted after 2000. The primary outcomes were morbidity and mortality. Pooled odds ratios (ORs), weighted mean differences (WMDs) or hazard ratios (HRs) with 95 percent confidence intervals (CIs) were calculated using fixed effects or random effects models. The methodological quality of the included studies was evaluated by the Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool.
Results: In total, 45 studies were included in this systematic review, and 5 non-randomized comparative studies with 786 patients (TP: 270, PD: 516) were included in the meta-analysis. There were no differences in terms of mortality (OR: 1.44, 95% CI: 0.66–3.16; P=0.36), hospital stay (WMD: −0.60, 95% CI: −1.78–0.59; P=0.32) and rates of reoperation (OR: 1.12; 95% CI: 0.55–2.31; P=0.75) between the two groups. In addition, morbidity was not significantly different between the two groups (OR: 1.41, 95% CI: 1.01–1.97; P=0.05); however, the results showed that the TP group tended to have more complications than the PD group. Furthermore, the operation time (WMD: 29.56, 95% CI: 8.23–50.89; P=0.007) was longer in the TP group. Blood loss (WMD: 339.96, 95% CI: 117.74–562.18; P=0.003) and blood transfusion (OR: 4.86, 95% CI: 1.93–12.29; P=0.0008) were more common in the TP group than in the PD group. There were no differences in the long-term survival rates between the two groups.
Conclusion: This systematic review and meta-analysis suggested that TP may not be as feasible and safe as PD. However, TP and PD may have the same efficacy.
Keywords: total pancreatectomy, pancreaticoduodenectomy, morbidity, mortality, meta-analysis
Introduction
Surgical resection plays an essential role in patients with periampullary disease. At present, two surgical methods to treat periampullary disease, total pancreatectomy (TP) and pancreaticoduodenectomy (PD), have been reported. TP was reported for the first time in 1943.1 At that time, it was performed to avoid pancreaticojejunostomy-associated complications and to attempt to improve the long-term survival for patients with pancreatic cancer.2–5 However, some studies suggested that TP was related to increasing morbidity and mortality.6–10 Furthermore, the survival of patients with pancreatic cancer was not shown to improve after TP in several studies.11–13 It is important to note that TP also leads to permanent insufficiency of pancreatic endocrine and exocrine function, impacting long-term quality of life.14–18 Fortunately, as a result of the appearance of advanced postoperative management in recent decades, including better pancreatic enzyme formulas and insulin therapy, we are able to more effectively manage the endocrine and exocrine consequences of TP.18,19 Nevertheless, TP was not recommended as routine treatment for patients with pancreatic cancer, especially those with pancreatic ductal adenocarcinoma (PDAC).20
Currently, no randomized controlled trials (RCTs) have been reported for comparison between these two approaches. Nevertheless, a large number of retrospective comparative studies have been reported. To address this issue, we conducted the most comprehensive systematic review and meta-analysis to our knowledge to assess the quality of individual studies and to produce the most rigorous analysis evaluating whether TP can be considered to be as feasible, safe, and efficacious as PD.
Materials and methods
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) criteria were used as guidelines in the construction of the meta-analysis.21
Literature search
We performed a systematic literature search in PubMed, EMBASE, Science Citation Index Expanded, Scopus and the Cochrane Library from January 1943 to June 2018, using combinations of the following terms: “Total pancreatectomy”, “Total pancreatic resection”, “Pancreatectomy”, “Pancreaticoduodenectomy”, “Whipple”, “Pancreatic head resection”, “Duodenopancreatectomy” and “Subtotal pancreatectomy. In addition, the references of all selected articles were screened for any potential eligible studies.
Study selection
Studies were included based on the following criteria: (1) human study; (2) primary outcome was reported; (3) if studies were reported by the same institution, either the study with the larger sample size or the study with the higher quality was included; And (4) meta-analysis only include studies with a study period after 2000.
The following types of studies were excluded: abstracts, letters, editorials, expert opinions, case reports, reviews and studies without comparisons.
Data extraction
Each study was evaluated by two independent reviewers for inclusion or exclusion. Disagreements between the reviewers were resolved by consultation with a third reviewer when necessary. Data were collected by two independent researchers using standardized forms. Study characteristics, quality assessment, intraoperative and postoperative outcomes were included. Means were used for meta-analysis unless otherwise mentioned. If the mean was not reported by the author, the means or standard deviations were calculated as medians or ranges, respectively.22 The following data were extracted from each study: author, year, country, study period, study design, number of patients, age, gender, tumor size, mortality, morbidity, operation time, blood loss, blood transfusion, hospital stay, reoperation and long term survival including 3-year overall survival (3-OS) and 5-year overall survival (5-OS).
Qualitative assessment
The risk of bias in the included non-randomized studies were evaluated using a tool for assessing Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I).23
Outcomes of interest and definitions
The primary outcomes were mortality and morbidity. Mortality was defined as the number of deaths during hospitalization or within 30 days after surgery. Morbidity was defined as any complication following surgery up to the day of discharge or within 30 days of discharge.
The secondary outcomes included operation time, blood loss, blood transfusion, reoperation, hospital stay and long-term survival reported in the individual papers. Hospital stay was defined as the length of time from postsurgery to discharge from the hospital. 3-OS was defined as the number of surviving patients three years after surgery. 5-OS was defined as the number of surviving patients five years after surgery. 3-OS and 5-OS were calculated for patients with PDAC.
Statistical analysis
Meta-analysis was performed using Review Manager Version 5.3 software (The Cochrane Collaboration, Oxford, UK). For continuous and categorical variables, treatment effects were expressed as weighted mean differences (WMDs) and odds ratios (ORs) or hazard ratios (HRs) with corresponding 95% confidence intervals (CIs). For survival analysis, the data were extracted from the survival curves by referring to a method reported in a previous study, and HR was used for quantitative analysis.24 A Chi-square test was used to assess heterogeneity with a P<0.1 considered significant. I2 values were used for the evaluation of statistical heterogeneity: an I2 value of 50% or more was indicative of the presence of heterogeneity.25 A fixed effects model was initially used for all outcomes, while a random effects model was used if the test suggested rejection of the assumption of homogeneity.26 Descriptive methods were used if the data were inappropriate for meta-analysis. Sensitivity analyses were conducted to explore possible explanations for heterogeneity and to examine the influence of various exclusion criteria on the overall pooled estimate. Funnel plots were constructed to evaluate potential publication bias based on mortality and morbidity.27
Results
Results of the literature review
A flow diagram of the study is shown in Figure 1. In total, 45 studies were included in the systematic review that reported the morbidity or mortality for TP (
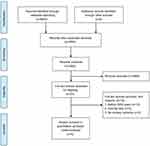 | Figure 1 Study selection flow chart according to PRISMA statement. |
A total of 23 comparative studies were included,9,18,19,30–32,34,36,39,42,43,45,46,48–50,53,58–60,62,64,65 however, two studies9,19 came from Johns Hopkins Hospital. One study was excluded because it included less relevant outcomes.9 Two studies both came from Columbia University.32,34 One study was excluded because it included less relevant outcomes.32 Moreover, one study was based on the National Surgical Quality Improvement Project database,58 and another study was based on the Nationwide Inpatient Sample database;50 both were excluded because of overlap with other studies. Two studies were excluded because of the absence of a primary outcome.49,53 Twelve studies were excluded because of the study duration before 2000.19,30,31,34,36,39,42,43,45,46,48,59 Finally, 5 studies were included in the meta-analysis.18,60,62,64,65 The study characteristics are shown in Table 1. In total, 786 patients were included, of whom 270 patients were in the TP group, and 516 patients were in the PD group. These studies came from Germany,18 Australia,60 Japan,62 Italy,64 and China.65 A total of three studies were retrospective comparative studies,60,62,65 and two studies18,64 were prospective comparative studies. Three studies used matched design.18,64,65
 | Table 1 Characteristics of the studies |
Study characteristics
The intraoperative and postoperative outcomes are shown in Table 2. The results of the meta-analysis are shown in Figures 2 and 3.
 | Table 2 Intraoperative and postoperative outcomes of the studies include in system review |
 | Figure 2 The forest plot of primary outcomes. (A) Mortality; (B) Morbidity. |
 | Figure 3 The forest plot of secondary outcomes. (A) Operation time; (B) Blood loss; (C) Blood transfusion; (D) Hospital stay; (E) Reoperation. |
Quality of studies
The methodological quality of the included studies was evaluated by ROBINS-I (Table 3). Based on ROBINS-I, three studies were graded as low-risk,18,64,65 and two studies62,64 were graded as moderate-risk.
 | Table 3 Quality of studies |
Results of meta-analysis
Primary outcome
Mortality was reported in five studies. There were no differences between the TP group and the PD group (OR: 1.44, 95%CI: 0.66–3.16; P=0.36). In total, five studies showed that there was no significant difference in morbidity (OR: 1.41, 95%CI: 1.01–1.97; P=0.05), and the results showed that the TP group tended to have more complications than the PD group.
Secondary outcomes
A longer operation time (WMD: 29.56, 95%CI: 8.23–50.89; P=0.007) and more blood loss (WMD: 339.96, 95%CI: 117.74–562.18; P=0.003) were found in the TP group than in the PD group. Blood transfusion (OR: 3.37, 95%CI: 1.25–9.12; P=0.02) in the TP group was also significantly more commom than in the PD group. However, there were no differences in hospital stay (WMD: −0.60, 95%CI: −1.78–0.59; P=0.32) and reoperation (OR: 1.12; 95%CI: 0.55–2.31; P=0.75) between the groups. There were also no differences between the two groups in terms of 3-OS (HR: 1.26, 95%CI: 0.86–1.85; P=0.24) and 5-OS (HR: 1.30, 95%CI: 0.90–1.88; P=0.16).
Sensitivity analysis
Sensitivity analysis was performed for outcomes with high heterogeneity. TP had a longer operation time in the fixed effects model (WMD: 25.39, 95%CI: 17.67–33.12; P<0.00001) and random effects model (WMD: 29.56, 95%CI: 8.23–50.89; P=0.007) than that of PD. For blood loss, the same result was found in the fixed (WMD: 280.24, 95%CI: 190.91–369.56; P<0.00001) and random (WMD: 339.96, 95%CI:00 117.74–562.18; P=0.003) effects model. There was no significant difference in blood transfusion between the fixed effects model (OR: 4.20, 95%CI: 2.46–7.18; P<0.00001) and random effects model (OR: 3.37, 95%CI: 1.25–9.12; P=0.02).
Publication bias
The funnel plots based on mortality and morbidity are shown in Figure 4. No study laid outside the limits of the 95%CI; therefore, there was no evidence of publication bias.
 | Figure 4 Funnel plot to investigate publication bias. (A) Mortality; (B) Morbidity. |
Discussion
This study summarizes the evidence to date that suggest that TP may not be as feasible and safe as PD. We found that the TP group had a longer operation time, more blood loss, and more frequent blood transfusion than the PD group. However, TP and PD may have the same efficacy because of no significant difference in overall survival.
The difference in morbidity in TP among studies from the past compared to studies in the present was vast. The lowest morbidity (17.5%) was reported by Balcom, and the highest morbidity (86.7%) was reported by Nikfarjam.48,60 Many studies suggested that more extended surgery led to higher morbidity, even in high-volume centers.13,19,60 Similar results were found in the present study. As mentioned previously, the improvement of operative techniques and postoperative management have dramatically decreased the morbidity associated with TP.62,64 Therefore, it can be inferred that morbidity was closely related to the skill of the surgeon and postoperative management. In this meta-analysis, most studies came from high-volume centers. This ensured that there were consistent skills of the surgeons and good postoperative management. Therefore, even if its mortality and morbidity have dramatically decreased in recent decades, TP remains an extended surgical procedure.
In the past, the mortality associated with TP was more than 20%.28,31,32 With the improvement of operative techniques and postoperative management, the mortality of the TP has dramatically decreased.62,64,65 However, higher mortality from TP was found in several studies.19,42,46,58 Several studies have suggested that there is no difference between the TP and PD groups.18,64 Muller reported that TP should no longer be generally avoided, because the mortality rate after elective TP was not significantly different from that associated with PD.18 Casadei also showed that postoperative mortality was similar between TP and PD.64 In this study, the mortality in TP seems to not to be different from that of PD, but for TP it never reaches the level (<2–3%) accepted for PD in high volume center. The results of the present study were consistent with the results that have been published in most of the studies for several decades.
Because TP is a more extensive operation, it is possible that the TP group had more vascular resection than the PD group.65 Therefore, longer operation times and more blood loss are not surprising. The skill and proficiency of surgeons might also be important factors for intraoperative outcomes. With more advanced technology and more skillful surgeons in the modern era, the number of patients who require blood transfusion should be less than that of previous eras.
Early studies generated a large controversy regarding the long-term survival between the two surgical procedures for patients with PDAC. The TP group with higher 5-OS than the PD group (14% vs 0%).9 In contrast, two studies reported that 5-OS was higher in the PD group than in the TP group.45,46 Nevertheless, several studies suggested that long-term survival after TP was comparable to that of PD. A large sample study reported similar 5-OS between the two groups (TP-18.5% vs PD-18.9%).19 An additional matched-pairs analysis between the TP and PD groups was carried out only for patients with PDAC, revealing similar perioperative outcomes and OS associated with the two surgical procedures.62 Furthermore, one matched study showed that similar survival of TP and PD in patients with PDAC.65 The present study also confirmed that TP and PD were similar in overall survival for patients with PDAC. A study from Satio reported worse OS and disease-free survival (DFS) in the TP group than in the PD group.62 However, using a matched analysis provided similar surgical and pathological outcomes, leading to similar OS between TP and PD matched groups.62 This suggested that long-term survival might be closely related to surgical and pathological outcomes. Therefore, TP for PDAC does not affect survival.
There are several limitations to this study: first, all included studies had non-randomized designs; second, there was significant variability in some outcomes, including the definition of morbidity and follow-up time; third, we observed some heterogeneity in certain outcome measures. This might be explained by the differences in surgical techniques, the retrospective nature of the studies, and the limited blinded outcome assessment in some of the trials. Therefore, future high-quality prospective studies are needed to confirm these results.
Conclusion
It seems reasonable to suggest that TP may not be considered to be as feasible and safe as PD. However, TP and PD may have the same efficacy. Nevertheless, there is an evident need for a well-designed study comparing TP and PD with respect to quality of life and long-term survival outcomes.
Acknowledgment
This study was supported by the Sichuan Provincial Department of Science and Technology Supporting Project (No. 2018SZ0174).
Author contributions
Du-Jiang Yang, Jun-Jie Xiong and Wei-Ming Hu designed the study. Du-Jiang Yang and Jun-Jie Xiong performed the study and wrote the paper. Du-Jiang Yang and Xue-Ting Liu assessed the studies included in this review and collected the data. Jiao Li and Kanagarathna Mudiyanselage Dhanushka Layanthi Siriwardena analysed the data. Wei-Ming Hu reviewed the manuscript. All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Rockey EW. TOTAL PANCREATECTOMY FOR CARCINOMA: CASE REPORT. Ann Surg. 1943;118(4):603.
2. Ross DE. Cancer of the pancreas: A plea for total pancreatectomy. Am J Surg. 1954;87(1):20–33.
3. Porter MR. Carcinoma of the pancreatico-duodenal area; operability and choice of procedure. Ann Surg. 1958;148(4):711–724.
4. Ihse I, Lilja P, Arnesjo B, Bengmark S. Total pancreatectomy for cancer. An appraisal of 65 cases. Ann Surg. 1977;186(6):675–680.
5. Tryka AF, Brooks JR. Histopathology in the evaluation of total pancreatectomy for ductal carcinoma. Ann Surg. 1979;190(3):373–381.
6. Gall FP, Muhe E, Gebhardt C. Results of partial and total pancreaticoduodenectomy in 117 patients with chronic pancreatitis. World J Surg. 1981;5(2):269–273.
7. Sakata N, Egawa S, Motoi F, et al. Institutional indications for islet transplantation after total pancreatectomy. J Hepatobiliary Pancreat Surg. 2008;15(5):488–492. doi:10.1007/s00534-007-1309-3
8. Kneuertz PJ, Pitt HA, Bilimoria KY, et al. Risk of morbidity and mortality following hepato-pancreato-biliary surgery. J Gastrointest Surg. 2012;16(9):1727–1735. doi:10.1007/s11605-012-1938-y
9. Schmidt CM, Glant J, Winter JM, et al. Total pancreatectomy (R0 resection) improves survival over subtotal pancreatectomy in isolated neck margin positive pancreatic adenocarcinoma. Surgery. 2007;142(4):572–580. doi:10.1016/j.surg.2007.07.016
10. Simons JP, Shah SA, Ng SC, Whalen GF, Tseng JF. National complication rates after pancreatectomy: beyond mere mortality. J Gastrointest Surg. 2009;13(10):1798–1805. doi:10.1007/s11605-009-0936-1
11. Nakao A, Ichihara T, Nonami T, et al. Clinicohistopathologic and immunohistochemical studies of intrapancreatic development of carcinoma of the head of the pancreas. Ann Surg. 1989;209(2):181. doi:10.1097/00000658-198902000-00008
12. Sarr MG, Behrns KE, van Heerden JA. Total pancreatectomy. An objective analysis of its use in pancreatic cancer. Hepato-gastroenterology. 1993;40(5):418.
13. Karpoff HM, Klimstra DS, Brennan MF, Conlon KC. Results of total pancreatectomy for adenocarcinoma of the pancreas. Arch Surg. 2001;136(1):44–47.
14. Warren KW, Poulantzas JK, Kune GA. Life after total pancreatectomy for chronic pancreatitis: clinical study of eight cases. Ann Surg. 1966;164(5):830–834.
15. Dresler CM, Fortner JG, Mcdermott K, Bajorunas DR. Metabolic consequences of (regional) total pancreatectomy. Ann Surg. 1991;214(2):131–140.
16. Billings BJ, Christein JD, Harmsen WS, et al. Quality-of-life after total pancreatectomy: is it really that bad on long-term follow-up? J Gastrointest Surg. 2005;9(8):1059–1067. doi:10.1016/j.gassur.2005.05.014
17. Gulik TMV, Nakao A, Obertop H. Extended resection for pancreatic adenocarcinoma. Oncologist. 2007;4(3):101–103.
18. Muller MW, Friess H, Kleeff J, et al. Is there still a role for total pancreatectomy? Ann Surg. 2007;246(6):966–974. doi:10.1097/SLA.0b013e31815c2ca3
19. Reddy S, Wolfgang CL, Cameron JL, et al. Total pancreatectomy for pancreatic adenocarcinoma: evaluation of morbidity and long-term survival. Ann Surg. 2009;250(2):282. doi:10.1097/SLA.0b013e3181ae9f93
20. Almond M, Roberts KJ, Hodson J, et al. Changing indications for a total pancreatectomy: perspectives over a quarter of a century. HPB. 2015;17(5):416–421. doi:10.1111/hpb.12365
21. Moher D, Liberati A, Tetzlaff J, Altman DG;
22. Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5(1):13. doi:10.1186/1471-2288-5-27
23. Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919. doi:10.1136/bmj.i4919
24.
25. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–560. doi:10.1136/bmj.327.7414.557
26. Dersimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7(3):177. doi:10.1016/0197-2456(86)90046-2
27. Egger SM, Smith GD. Investigating and dealing with publication and other biases in meta-analysis. BMJ. 2001;323(7304):101–105.
28. Wh R, Jt P, Es J, Jn K. Total pancreatectomy. Ann Surg. 1970;172(4):595. doi:10.1097/00000658-197010000-00006
29. Pliam MB, Remine WH. Further evaluation of total pancreatectomy. Arch Surg. 1975;110(5):506–512.
30. Brooks JR, Culebras JM. Cancer of the pancreas. Palliative operation, Whipple procedure or total pancreatectomy? Am J Surg. 1976;131(4):516–520.
31. Cubilla AL, Fitzgerald PJ, Fortner JG. Pancreas cancer — duct cell adenocarcinoma: survival in relation to site, size, stage and type of therapy. J Surg Oncol. 1978;10(6):465–482.
32. Cooperman AM, Herter FP, Marboe CA, Helmreich ZV, Perzin KH. Pancreatoduodenal resection and total pnacreatectomy–an institutional review. Surgery. 1981;90(4):707–712.
33. Van Heerden JA, Remine WH, Weiland LH, Mcilrath DC, Ilstrup DM. Total pancreatectomy for ductal adenocarcinoma of the pancreas. Mayo Clinic experience. Am J Surg. 1981;142(3):308–311.
34. Herter FP, Cooperman AM, Ahlborn TN, Antinori C. Surgical experience with pancreatic and periampullary cancer. Ann Surg. 1982;195(3):274–281.
35. Fortner JG. Regional pancreatectomy for cancer of the pancreas, ampulla and other related sites. Jpn J Surg. 1984;13(5):418–425.
36. Grace PA, Pitt HA, Tompkins RK, Denbesten L, Longmire W
37. Cooper MJ, Williamson RCN, Benjamin IS, et al. Total pancreatectomy for chronic pancreatitis. Br J Surg. 1987;74(10):912–915.
38. Linehan IP, Lambert MA, Brown DC, Kurtz AB, Cotton PB, Russell RCG. Total pancreatectomy for chronic pancreatitis. Gut. 1988;29(3):358–365.
39. Stone WM, Sarr MG, Nagorney DM, McIlrath DC. Chronic pancreatitis. Results of Whipple‘s resection and total pancreatectomy. Arch Surg. 1988;123(7):815–819.
40. Brooks JR, Brooks DC, Levine JD. Total pancreatectomy for ductal cell carcinoma of the pancreas. An update. Ann Surg. 1989;209(4):405. doi:10.1097/00000658-198904000-00003
41. Launois B, Franci J, Bardaxoglou E, et al. Total pancreatectomy for ductal adenocarcinoma of the pancreas with special reference to resection of the portal vein and multicentric cancer. World J Surg. 1993;17(1):122–126.
42. Baumel H, Huguier M, Manderscheid JC, Fabre JM, Houry S, Fagot H. Results of resection for cancer of the exocrine pancreas: a study from the French association of surgery. Br J Surg. 1994;81(1):102.
43. Swope TJ, Wade TP, Neuberger TJ, Virgo KS, Johnson FE. A reappraisal of total pancreatectomy for pancreatic cancer: results from US Veterans Affairs hospitals, 1987–1991. Am J Surg. 1994;168(6):582–586.
44. Fleming WR, Williamson RCN. Role of total pancreatectomy in the treatment of patients with end‐stage chronic pancreatitis. Br J Surg. 1995;82(10):1409–1412. doi:10.1002/(ISSN)1365-2168
45. Bardaxoglou E, Landen S, Maddern GJ, et al. Total pancreatectomy and subtotal duodenopancreatectomy for the management of carcinoma of the head of the pancreas: an institutional experience and evolving trends. Dig Surg. 1996;13(1):12–18. doi:10.1159/000172398
46. Ihse I, Anderson H, Andrén-Sandberg Å. Total pancreatectomy for cancer of the pancreas: is it appropriate? World J Surg. 1996;20(3):288–294.
47. Sugiyama M, Atomi Y. Pylorus-preserving total pancreatectomy for pancreatic cancer. World J Surg. 2000;24(1):66–71.
48. Jh B, Rattner DW, Warshaw AL, Chang Y, Fernandezdel CC. Ten-year experience with 733 pancreatic resections: changing indications, older patients, and decreasing length of hospitalization. Arch Surg. 2001;136(4):391. doi:10.1001/archsurg.136.4.391
49. Wagner M, Redaelli C, Lietz M, Seiler CA, Friess H, Büchler MW. Curative resection is the single most important factor determining outcome in patients with pancreatic adenocarcinoma. Br J Surg. 2004;91(5):586–594. doi:10.1002/(ISSN)1365-2168
50. McPhee JT, Hill JS, Whalen GF, et al. Perioperative mortality for pancreatectomy: a national perspective. Ann Surg. 2007;246(2):246–253. doi:10.1097/01.sla.0000259993.17350.3a
51. Kulu Y, Schmied BM, Werner J, Muselli P, Buechler MW, Schmidt J. Total pancreatectomy for pancreatic cancer: indications and operative technique. HPB. 2009;11(6):469–475. doi:10.1111/j.1477-2574.2009.00085.x
52. Murphy MM, Knaus WJ
53. Nathan H, Wolfgang CL, Edil BH, et al. Peri-operative mortality and long-term survival after total pancreatectomy for pancreatic adenocarcinoma: A population-based perspective. J Surg Oncol. 2009;99(2):87–92. doi:10.1002/jso.21189
54. Janot MS, Belyaev O, Kersting S, et al. Indications and early outcomes for total pancreatectomy at a high-volume pancreas center. HPB Surg. 2010;2010. doi:10.1155/2010/686702
55. Parsaik AK, Murad MH, Sathananthan A, et al. Metabolic and target organ outcomes after total pancreatectomy: mayo Clinic experience and meta-analysis of the literature. Clin Endocrinol (Oxf). 2010;73(6):723–731. doi:10.1111/j.1365-2265.2010.03860.x
56. Crippa S, Tamburrino D, Partelli S, et al. Total pancreatectomy: indications, different timing, and perioperative and long-term outcomes. Surgery. 2011;149(1):79–86. doi:10.1016/j.surg.2010.04.007
57. Barbier L, Jamal W, Dokmak S, et al. Impact of total pancreatectomy: short- and long-term assessment. Hpb. 2013;15(11):882–892. doi:10.1111/hpb.12054
58. Bhayani NH, Miller JL, Ortenzi G, et al. Perioperative outcomes of pancreaticoduodenectomy compared to total pancreatectomy for neoplasia. J Gastrointest Surg. 2014;18(3):549–554. doi:10.1007/s11605-013-2393-0
59. Epelboym I, Winner M, Dinorcia J, et al. Quality of life in patients after total pancreatectomy is comparable with quality of life in patients who undergo a partial pancreatic resection. J Surg Res. 2014;187(1):189–196. doi:10.1016/j.jss.2013.10.004
60. Nikfarjam M, Low N, Weinberg L, Chia PH, He H, Christophi C. Total pancreatectomy for the treatment of pancreatic neoplasms. ANZ J Surg. 2014;84(11):823–826. doi:10.1111/ans.12640
61. Hartwig W, Gluth A, Hinz U, et al. Total pancreatectomy for primary pancreatic neoplasms renaissance of an unpopular operation. Ann Surg. 2015;261(3):537–546. doi:10.1097/SLA.0000000000000791
62. Satoi S, Murakami Y, Motoi F, et al. Reappraisal of total pancreatectomy in 45 patients with pancreatic ductal adenocarcinoma in the modern era using matched-pairs analysis: multicenter study group of pancreatobiliary surgery in Japan. Pancreatology. 2015;16(4):S76–S77. doi:10.1016/j.pan.2016.06.275
63. Watanabe Y, Ohtsuka T, Matsunaga T, et al. Long-term outcomes after total pancreatectomy: special reference to survivors‘ living conditions and quality of life. World J Surg. 2015;39(5):1231–1239. doi:10.1007/s00268-015-2948-1
64. Casadei R, Ricci C, Taffurelli G, et al. Is total pancreatectomy as feasible, safe, efficacious, and cost-effective as pancreaticoduodenectomy? A single center, prospective, observational study. J Gastrointest Surg. 2016;20(9):1595–1607. doi:10.1007/s11605-016-3201-4
65. Xiong J, Wei A, Ke N, et al. A case-matched comparison study of total pancreatectomy versus pancreaticoduodenectomy for patients with pancreatic ductal adenocarcinoma. Int J Surg. 2017;48:134–141. doi:10.1016/j.ijsu.2017.10.065
66. Johnstone WC, Hoen HM, Cassera MA, et al. Total pancreatectomy for pancreatic ductal adenocarcinoma: review of the national cancer data base. HPB. 2016;18(1):21–28. doi:10.1016/j.hpb.2015.07.009
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
