Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
Total Antioxidant Status in Stable Chronic Obstructive Pulmonary Disease
Authors Hlavati M , Tomić S, Buljan K, Buljanović V , Feldi I, Butković-Soldo S
Received 28 May 2020
Accepted for publication 7 September 2020
Published 6 October 2020 Volume 2020:15 Pages 2411—2419
DOI https://doi.org/10.2147/COPD.S264944
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Marina Hlavati,1,2 Svetlana Tomić,2 Krunoslav Buljan,2 Vikica Buljanović,3 Ivan Feldi,4 Silva Butković-Soldo2
1Department of Diagnostic and Therapeutical Procedures, General Hospital Našice, Našice 31500, Croatia; 2Faculty of Medicine Osijek, University Josip Juraj Strossmayer Osijek, Osijek 31000, Croatia; 3Department for Biochemical Analysis, General Hospital Našice, Našice 31500, Croatia; 4Department of Internal Medicine, General Hospital Našice, Našice 31500, Croatia
Correspondence: Marina Hlavati Department of Diagnostic and Therapeutical Procedures
General Hospital Našice, Bana Jelačića 10, Našice 31500, Croatia
Tel +385915810485
Fax +38531613826
Email [email protected]
Objective: This study evaluates the total antioxidant status (TAS) in plasma of stable chronic obstructive pulmonary disease (COPD) patients. Earlier studies of their relationship showed inconsistent findings.
Patients and Methods: We compared TAS between 90 COPD patients and 30 age- and sex-matched controls (mean age 67 ± 7.9, 87 males and 33 females) according to airway obstruction severity, gender, smoking status (current/ former/ non-smoker), smoking-dose, the number of exacerbations in the previous year, nutritional status and hypercapnia.
Results: There were no differences in pack-years between COPD and controls, neither in COPD groups. The median time from the last exacerbation was 5 months (interquartile range 3− 8.3). TAS was significant higher in COPD than controls (1.68 [1.55− 1.80] versus 1.59 [1.54− 1.68], respectively; P = 0.03). TAS was significantly higher in COPD men than women (1.7 [1.6− 1.8] versus 1.57 [1.5− 1.7], respectively; P = 0.001). In COPD groups, there were no significant differences between the severity of airway obstruction and TAS. We found significant positive correlation between pack-years and TAS in all participants (Rho = 0.429, P = 0.004) and COPD patients (Rho = 0.359, P = 0.02), but not in controls. TAS was a significant predictor of COPD (β = 3.26; P = 0.04; OR = 26.01; 95% CI: 1.20 to 570.8). We failed to find significant differences between TAS and smoking status, frequency of exacerbations in the previous year, nutritional status and hypercapnia.
Conclusion: TAS was a significant predictor of COPD. TAS was a significantly higher in stable COPD than controls, higher in COPD men than women, but there was no significant correlation between TAS and the airway obstruction severity. Our results suggest that it could be appropriate to include the time from the last exacerbation in the oxidant–antioxidant balance analysis of COPD patients.
Keywords: chronic obstructive pulmonary disease, antioxidants, airway obstruction severity, exacerbation, smoking
Introduction
The pathogenesis of chronic obstructive pulmonary disease (COPD) is in strong causal link with increased oxidative burden. The harmful oxidants and defensive intracellular and extracellular antioxidant systems are in gentle balance.1 Antioxidants and antioxidant enzymes have an essential function against the oxidative burden in smokers and COPD lungs.2 Oxidant–antioxidant imbalance has a significant role in different stages of COPD severity.3 It occurs when the resident antioxidants are insufficient or cannot increase enough to counteract an increased oxidative burden. The target of treating oxidative stress should be either to reduce oxidant formation or to increase antioxidants.4
Cigarette smoking is the most common identified risk factor for COPD.5 Despite that, only a minority of smokers develop COPD symptoms that are clinically significant.6 Approximately 13% of smokers have COPD and the risk for disease increased with the number of cigarettes consumed.7 Smoking cessation has the greatest impact on disease course and it is a key to prevent future COPD exacerbations.8,9 But even in former smokers that have signs of persistent chronic airflow limitation, there are signs of a low-grade systemic inflammation. It shows that, once COPD develops, smoking cessation may not stop the disease from progressing.10
Since it is difficult to measure each antioxidant in different biological samples, various methods have been promoted to determine the total antioxidant status (TAS). Some of them can define TAS in plasma of COPD patients.1 Reports on the relationship between the oxidant–antioxidant imbalance and pulmonary function in COPD patients showed inconsistent results.11 In stable COPD, there were reports of no significant differences of TAS,12,13 as well as a significant decrease of TAS compared to controls.11,14 In a study without the characteristics of disease activity, findings suggested a lower TAS in patients compared to healthy controls, confirming a presence of systematically oxidant–antioxidant imbalance in COPD patients.15 Some authors found a significant reduction in TAS only in exacerbation of COPD.16,17 Earlier reports that compared TAS and the airway obstruction severity in COPD patients, were found no significant differences.11,18,19
In the present study, we have analyzed the predictive value of TAS for COPD and compared the plasma TAS values in stable COPD patients according to their clinical data and severity of the disease with healthy controls.
Patients and Methods
Study Subject
This was a single-center, cross-sectional observational study, taken in general hospital, between March and August 2018. The participants were above 18 years, both sexes, with a diagnosis of COPD and healthy volunteers. COPD patients were defined as stable based on having no exacerbation for the previous 2 months. The data about previous exacerbations were collected retrospectively. None of them use systemic corticosteroid medication during the study.
Exclusion criteria were: active malignant disease, febrile state, nonsteroidal anti-inflammatory and antioxidant therapy (vitamins A, C and E). COPD patients with arterial hypertension, hyperlipidemia, ischemic heart disease and diabetes mellitus were included if they were under good therapy control.
The sample size was calculated to be 120, with an effect size of 0.35, level of significance 0.05 and power 0.9 (G*power software, version 3.1.9.2, by Franz Faul, University Kiel, Germany).
The study was approved by the ethics committee of General Hospital Našice (No. 01-497/3-2017) and Faculty of Medicine Osijek, University Josip Juraj Strossmayer Osijek, Croatia (No. 2158-61-07-17-209). All data were anonymized and the study was conducted in accordance with the amended Declaration of Helsinki. Informed consent was obtained from all individual participants included in the study. All participants signed an informed consent form before entering the study.
Study Design and Variables
According to international established Global Initiative for Chronic Obstructive Lung Disease (GOLD) criteria, COPD diagnosis was confirmed by post-bronchodilator forced expiratory volume in the one second (FEV1) to forced vital capacity (FVC) ratio (FEV1/FVC) of <0.70 in patients with appropriate symptoms. Spirometry findings of airflow limitation severity were based on FEV1 and divided COPD in mild, moderate and severe/very severe group (GOLD classification).20 According to number of exacerbations in the previous year, they were divided into non-exacerbators (0), infrequent exacerbators (1) and frequent exacerbators (≥2).21 A fourth group was healthy volunteers (FEV1/FVC >70% and FEV1 >80% of predicted value). The participants were demanded to refrain from caffeine, alcohol and increased physical activity at least 12 hours before entering the study.
Height and weight as theanthropometric measurements were determined, body mass index (BMI) calculated and expressed as weight (kg)/height (m2). Smoking status was recorded as current, former and non-smoker. We defined former smokers as those who had quit smoking at least one year prior to participating because the others are most likely to relapse.22 Pack-years were calculated using the formula: number of cigarettes per day x number of smoking years/20.11 We defined light smoking as <20 and heavy smoking as ≥20 pack-years.23 Spirometry (Spiroscoutᴿ Ganshorn, Germany) was carried out by specialized technician according to American Thoracic Society/European Respiratory Society standardization to all participants.24
Biochemical Analysis
The capillary and venous blood was taken for biochemical analysis. An acid-base status was analyzed in COPD patients (Simens RAPIDLab 348EX, Germany) and complete blood count (Sysmex XN-1000 SA-01, Sysmex Europe GmbH), C-reactive protein (CRP) and TAS (analyzer Beckman Coulter DXC 700 AU, USA) in all. For TAS analysis, separated serum samples were frozen on – 20°C, collected up to 14 days, collected ones were defrosted and analyzed in one day by colorimetric assay. The procedure was repeated until we reached the target number of participants. Blood samples for other parameters were analyzed instantly after taking.
The plasma antioxidant status was measured using a commercially available TAS kit (reagents Randox Laboratories Ltd, United Kingdom). The TAS assay is based on incubation of 2,2’-azino-di-3-ethylbenzthiazoline sulphonate (ABTS) with peroxidase (metmyoglobin) and hydrogen peroxide (H2O2) to produce the radical cation ABTS+. This has a relatively stable blue-green color, which is measured at 600 nm. Antioxidants in the added sample cause suppression of this colour production to a degree which is proportional to their concentration. The absorbance was read after exactly 3 minutes. Reference range is set at 1.30 − 1.77 and results are expressed as mmol/L.25
Statistical Methods
Categorical data are represented by absolute and relative frequencies. Numerical data are described by the arithmetic mean and the standard deviation or the median and the limits of the interquartile range. Variations of categorical variables were tested by χ2 test. The normality of the distribution of numeric variables was tested by the Shapiro–Wilk test. The differences between the normal distribution of numerical variables between the two independent groups were tested by the Student t test, and in the case of a deviation from the normal distribution by Mann–Whitney U-test. The differences between the normal distribution of numeric variables between groups considering the weight of COPD were tested for deviation from the normal distribution by Kruskal–Wallis test (post hoc Conover test). The correlation of numeric variables was evaluated by Spearman’s coefficient of correlation ρ (rho). The estimate of the predictive value was tested by logistic regression analysis. All P values are two-sided. The level of significance is set to Alpha = 0.05. For statistical analysis, statistical software MedCalc Statistical Software version 18.11.3 (MedCalc Software bvba, Ostend, Belgium; http://www.medcalc.org; 2019) and SPSS (IBM Corp. Released 2015. IBM SPSS Statistics for Windows, Version 23.0, Armonk, NY: IBM Corp.) were used.
Results
We included 120 participants, 90 COPD patients and 30 healthy volunteers ─ controls, mean age 67 ± 7.9, 87 males and 33 females. Current COPD smokers had more heavy smokers and control group light ones, but according to pack-years there were no differences. As expected, lung function was significantly impaired in COPD patients versus controls. TAS was a significantly higher in COPD than controls (Table 1).
 |
Table 1 Baseline Characteristics of COPD Patients and Controls |
Among current COPD smokers, according to airflow obstruction severity, there was a significant rate of heavy smokers in severe/very severe group and light smokers in mild COPD group, but according to pack-years there were no differences. The severe/very severe COPD had a lower BMI and more infrequent and frequent exacerbators than other COPD groups. In COPD groups, there were no significant differences between the severity of airway obstruction and the TAS values (Table 2).
 |
Table 2 Baseline Characteristics of COPD Patients, According to Airflow Obstruction Severity |
For all COPD patients, according to smoking status, pack-years, the frequency of exacerbations in the previous year and the findings of hypercapnia in blood samples, we failed to find significant differences in TAS values (Table 3).
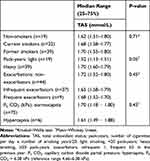 |
Table 3 TAS Value in COPD, According to Smoking Status, Pack-Years, Exacerbations in the Previous Year and Hypercapnia |
For all participants, with Spearman’s rank correlation coefficient Rho, there was no significant correlation between TAS and the parameters of lung functions. For all participants, control and COPD group, with Spearman’s rank correlation coefficient Rho there was no significant correlation between BMI and pack-years, neither between BMI and TAS (data not displayed).
We found a significant positive correlation between pack-years and TAS in all participants and COPD patients, but not in controls (Figure 1A–C).
According to gender, in the controls, there were no significant differences between TAS values in men and women (1.62 [1.5–1.7] versus 1.56 [1.5–1.6], respectively; P = 0.34). In COPD patients, TAS was significantly higher in men than women 1.7 [1.6–1.8] versus (1.57 [1.5–1.7], respectively; P = 0.001, Mann–Whitney U-test).
Logistic regression analysis showed that TAS was a significant predictor of COPD (β = 3.26; P = 0.04; OR = 26.01, 95% CI: 1.20 to 570.8).
Discussion
The aim of this study was to test the predictive value of the antioxidative defense parameter − TAS for COPD and the value of TAS in plasma of COPD patients with stable disease state, according to airway obstruction severity, gender, smoking status, smoking-dose, the number of exacerbations in the previous year, nutritional status and hypercapnia. In our study, TAS was a significant predictor of COPD. TAS was a significantly higher in COPD than controls, higher in COPD men than women, but there was no significant correlation between TAS and the airway obstruction severity.
In an addition to the pulmonary impairment, COPD is associated with skeletal muscle wasting and muscle dysfunction. In a study with stable COPD patients, the biopsy of peripheral skeletal muscle in rest was performed and biochemical analysis of antioxidant parameters in muscle and oxidants in blood. The results of this study were the overall antioxidant capacity, included TAS, in muscle bioptate of COPD patients compared to healthy controls, but without any changes in oxidant parameters of blood. Probably the most influential way by which the antioxidant defense system can be triggered is exposed to oxidants. Although in study the analysis of oxidant parameters in muscle was not performed, they concluded that it is likely that the observed increased antioxidant capacity is a compensatory adaptation to elevated oxidants in skeletal muscle of patients with COPD.26
Our COPD participants were in a stable state of disease. In the previous study, the COPD patients with an acute exacerbation of disease had decreased antioxidant capacity in plasma at the beginning of treatment, for at least 48 hours, with a rise at the end of an exacerbation when they were considered to be clinically stable enough for discharge. However, the antioxidant capacity values had not returned to normal levels 5–10 days after admission.16 Previous studies that detected decreased TAS values defined stable COPD as those with 1 month14,19 or 2 months11 of period after last exacerbation. They did not state neither took in account precise time from the last exacerbation like we did in our study. Our median time of five months from last exacerbation and higher values of TAS compared to controls could suggest a possible involvement of time needed for recovery of antioxidant defense system. The gentle oxidant–antioxidant balance and their dynamic relationship anyhow include deliberation of time in which the analyzed biological sample was taken. It could be appropriate to include the time from the last exacerbation in the oxidant–antioxidant balance analysis of COPD patients. It should be considered in future prospective studies and possible interventions aimed to establish such balance. Since we did not determine the level of oxidants in serum of our participants, we could not know if the increased level of antioxidant is a compensatory adaptation to oxidative stress.
Our study confirmed earlier reports data that were no significant correlation between TAS and the airway obstruction severity.11,18,19
In COPD patients, TAS was significantly higher in men. All women in our study were in post-menopausal period, thereby eliminated the effect of estrogen on oxidative stress which could occur in different age groups. Estrogen has helpful vasoactive and antioxidative characteristics and its decline in postmenopausal period is associated with increased oxidative stress.27 Determine hormonal status was beyond the scope of this study, so we could not know whether this is the reason for the observed gender difference in TAS values.
In our COPD group, 10% were frequent exacerbators, and in a previous study, the reported rate was 13.6%.21 The frequency of exacerbations did not correlate with TAS values. Our severe to very severe COPD patients were underweight and moderate ones were obese. Underweight individuals without COPD disease showed decreased antioxidant status that was improved after re-nutrition.28 In our study, there was no correlation between nutritional status and TAS in COPD patients and controls. Obesity is correlated with systemic inflammation and accompanied by high-oxidative stress.29 It may stimulate TAS and the high values of antioxidant potential may be interpreted by compensatory response to aggravated oxidative stress in individuals with higher content of adipose tissue.30 The earlier study that analyzed differences between obese and non-obese COPD smokers found higher value of antioxidative marker in the blood of obese ones.31 The recent study that analyzed muscle fat infiltration in COPD patients, according to nutritional status found that muscle fat infiltration is associated with increase inflammation/oxidative stress and reduction of TAS, independent of BMI. Their underweight subgroup had elevated risk of exacerbations.32 It would be useful to prospectively monitor frequent exacerbators and COPD patients, according to nutritional status, the dynamics of TAS values over time and other clinical features of their disease phenotype.
In our study, we failed to find a correlation between TAS and hypercapnia, possibly because of the small sample size of that subgroup. Hypercapnia is frequently observed in COPD patients and, on a molecular level, participate in mitochondrial dysfunction. It represents as increased mitochondrial production of reactive oxygen species and causes oxidant–antioxidant imbalance.33
We excluded the intake of dietary antioxidants; accordingly, the potential confusion impact of nutrition and lifestyle routine could not be part of explanations of our conclusions. Earlier study with the male COPD patients who had smoked ≥20 pack-years exhibited an advantageous association between dietary antioxidant vitamin intake and FEV1.34 There is increasing interest in strategies based on nutritional or pharmacological stimulation of endogenous antioxidative defense genes. The Nuclear Erythroid-Related Factor 2 (Nrf2) is the leading antioxidative and cell-protective transcriptive nuclear factor regulated by those genes.35,36 Recently, the first study that analyzed expression of the Nrf2 gene in the samples of alveolar macrophages, nasal and bronchial epithelial and mononuclear cells of the periphery blood in current and former COPD smokers was performed. The expression of the Nrf2 genes was significantly higher only in bronchial epithelium of COPD smokers. There were no differences in TAS and inflammation markers in serum, plasma, bronchoalveolar lavage and exhaled breath condensate. The number of participants was not big enough to correlate between TAS and smoking-dose in the current smokers group. The conclusion was that the recognition of differences in expression of the Nrf2 gene between different biological systems could help that targeting of specific cells could have therapeutic potency of Nrf2 activators as it was already confirmed in animal and in vitro studies.37 Such analysis far exceeds our technical capabilities, but it would certainly be useful to include genetic analysis in addition to the clinical manifestations of COPD in order to better understand the severity of disease, conduct therapeutic decisions, evaluate response to target interventions and provide prognostic information.
According to available data, except increased oxidants, inflammation and acute exacerbation, TAS in COPD patients could depend on gender, hormonal status in women, smoking status, smoking-dose, exacerbation number in the previous year, possibly the time from the last exacerbation, nutritional status, chronic hypercapnia, antioxidant supplementation, but also genotype and type of biological sample that was analyzed. The compensatory mechanisms of dynamic interaction between oxidant and antioxidant and the need for retention the balance, additional complicate the need for incorporating mentioned risk factors and their combination in definitive conclusions of finding interdependence.
Limitation and Strengths
The limitation of our study is the measurement of TAS in plasma, which might not reflect the local levels in the lungs. the other biological samples might be more appropriate for estimate oxidant–antioxidant imbalance in COPD patients. We performed only one measurement of TAS at a random time point which might ignore any variation of them over time. The concentrations of antioxidants that were measured at a single point may not represent the usual concentration of the study participants.38
TAS is the only biomarker that we performed because of technical and financial reasons. The combination of biomarkers increases the predictive value compared with single biomarker and clinical variables. On the other side, the literature data propose that a history of previous exacerbations is so firm correlate with future exacerbations that biomarkers may not contribute supplementary relevant information.39
The technical disadvantage of our study was the inability to freeze blood samples at − 80°C. That is why we were unable simultaneous defreezing all sample collection and their analysis in one act. We found higher values of TAS in the heavy COPD smoker group than in light one, but without significance; a significant positive correlation between pack-years and TAS in all participants and COPD patients, but not controls; and also TAS as predictor of COPD, but with a wide range of 95% confidence interval. A larger sample size would give bigger statistical power to detect significant associations and increase confidence in our findings.
Our conclusions are based upon results from a cross-sectional study and some of the aims could have been better addressed in a prospective study. Because the above limitations, the results could be potentially biased.
The strength of our study was a comparison of COPD patients with the well-matched control group. We exclude the intake of dietary antioxidants from the study and the potential impact of physical activity on antioxidant capacity. In COPD patients, during exercise, a significant increase in levels of blood markers of oxidative stress was observed that could be related to the disturbances of oxidant–antioxidant balance.12
Conclusions
The plasma TAS values in COPD patients depend on various factors. In our study, TAS was a significant predictor of COPD. TAS was a significantly higher in stable COPD than controls, higher in COPD men than women, but there was no significant correlation between TAS and the airway obstruction severity. We failed to find significant differences between TAS in stable COPD and smoking status, frequency of exacerbations in the previous year, nutritional status and hypercapnia. The literature data on TAS value dynamics and our results suggest that it could be appropriate to include the time from the last exacerbation in the oxidant–antioxidant balance analysis of COPD patients. However, prospective studies are needed for further evaluation.
Abbreviations
COPD, chronic obstructive pulmonary disease; TAS, total antioxidant status; GOLD, Global Initiative for Chronic Obstructive Lung Disease; FEV1, forced expiratory volume in the one second; FEV1/FVC ratio, forced expiratory volume in the one second to forced vital capacity ratio; BMI, body mass index; CRP, C-reactive protein; Nrf2, Nuclear Erythroid-Related Factor 2.
Data Sharing Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Acknowledgments
We gratefully acknowledge the time and effort of our research participants.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This study was not funded.
Disclosure
The authors declare no conflicts of interest in this work.
References
1. Zinellu E, Zinellu A, Fois AG, Carru C, Pirina P. Circulating biomarkers of oxidative stress in chronic obstructive pulmonary disease: a systematic review. Respir Res. 2016;17(1):1–11. doi:10.1186/s12931-016-0471-z
2. Rahman I, Kinnula VL. Strategies to decrease ongoing oxidant burden in chronic obstructive pulmonary disease. Expert Rev Clin Pharmacol. 2012;5(3):293–309. doi:10.1586/ecp.12.16
3. Singh S, Verma SK, Kumar S, et al. Evaluation of oxidative stress and antioxidant status in chronic obstructive pulmonary disease. Scand J Immunol. 2017;85(2):130–137. doi:10.1111/sji.12498
4. Rahman IMW. Antioxidant pharmacological therapies for COPD. Curr Opin Pharmacol. 2013;12(3):256–265. doi:10.1016/j.coph.2012.01.015.ANTIOXIDANT
5. Eisner MD, Anthonisen N, Coultas D, et al. An official American Thoracic Society public policy statement: novel risk factors and the global burden of chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2010;182(5):693–718. doi:10.1164/rccm.200811-1757ST
6. Li Y, Cho MH, Zhou X. What do polymorphisms tell us about the mechanisms of COPD? Clin Sci. 2017;131(24):2847–2863. doi:10.1042/cs20160718
7. Zhong N, Wang C, Yao W, et al. Prevalence of chronic obstructive pulmonary disease in China: a large, population-based survey. Am J Respir Crit Care Med. 2007;176(8):753–760. doi:10.1164/rccm.200612-1749OC
8. Bai JW, Chen XX, Liu S, Yu L, Xu JF. Smoking cessation affects the natural history of COPD. Int J COPD. 2017;12:3323–3328. doi:10.2147/COPD.S150243
9. Viniol C, Vogelmeier CF. Exacerbations of COPD. Eur Respir Rev. 2018;27:170103. doi:10.1183/16000617.0103-2017
10. Gan WQ, Man SFP, Senthilselvan A, Sin DD. Association between chronic obstructive pulmonary disease and systemic inflammation: a systematic review and a meta-analysis. Thorax. 2004;59(7):574–580. doi:10.1136/thx.2003.019588
11. Ben Anes A, Fetoui H, Bchir S, et al. Increased oxidative stress and altered levels of nitric oxide and peroxynitrite in tunisian patients with chronic obstructive pulmonary disease: correlation with disease severity and airflow obstruction. Biol Trace Elem Res. 2014;161(1):20–31. doi:10.1007/s12011-014-0087-4
12. Koechlin C, Couillard A, Cristol JP, et al. Does systemic inflammation trigger local exercise-induced oxidative stress in COPD? Eur Respir J. 2004;23(4):538–544. doi:10.1183/09031936.04.00069004
13. Can U, Yerlikaya FH, Yosunkaya S. Role of oxidative stress and serum lipid levels in stable chronic obstructive pulmonary disease. J Chin Med Assoc. 2015;78(12):702–708. doi:10.1016/j.jcma.2015.08.004
14. Aydemir Y, Aydemir Ö, Şengül A, et al. Comparison of oxidant/antioxidant balance in COPD and non-COPD smokers. Heart Lung. 2019;48(6):566–569. doi:10.1016/j.hrtlng.2019.07.005
15. Lakhdar R, Denden S, Mouhamed MH, et al. Correlation of EPHX1, GSTP1, GSTM1, and GSTT1 genetic polymorphisms with antioxidative stress markers in chronic obstructive pulmonary disease. Exp Lung Res. 2011;37(4):195–204. doi:10.3109/01902148.2010.535093
16. Rahman I, Skwarska E, MacNee W. Attenuation of oxidant/antioxidant imbalance during treatment of exacerbations of chronic obstructive pulmonary disease. Thorax. 1997;52(6):565–568. doi:10.1136/thx.52.6.565
17. Stanojkovic I, Kotur-Stevuljevic J, Milenkovic B, et al. Pulmonary function, oxidative stress and inflammatory markers in severe COPD exacerbation. Respir Med. 2011;105(Suppl.1):31–37. doi:10.1016/S0954-6111(11)70008-7
18. Ekin S, Arısoy A, Gunbatar H, et al. The relationships among the levels of oxidative and antioxidative parameters, FEV1 and prolidase activity in COPD. Redox Rep. 2017;22(2):74–77. doi:10.1080/13510002.2016.1139293
19. Tavilani H, Nadi E, Karimi J, Goodarzi MT. Oxidative stress in COPD patients, smokers and non-smokers subjects. Respir Care. 2012;57(12):2090–2094. doi:10.4187/respcare.01809
20. Mirza S, Clay RD, Koslow MA, Scanlon PD. COPD guidelines: a review of the 2018 GOLD report. Mayo Clin Proc. 2018;93(10):1488–1502. doi:10.1016/j.mayocp.2018.05.026
21. Beeh KM, Glaab T, Stowasser S, et al. Characterisation of exacerbation risk and exacerbator phenotypes in the POET-COPD trial. Respir Res. 2013;14:1–8. doi:10.1186/1465-9921-14-116
22. Sulsky SI, Fuller WG, Van Landingham C, Ogden MW, Swauger JE, Curtin GM. Evaluating the association between menthol cigarette use and the likelihood of being a former versus current smoker. Regul Toxicol Pharmacol. 2014;70(1):231–241. doi:10.1016/j.yrtph.2014.07.009
23. Guo E, Liu P, Qian Z, et al. Number of cigarettes smoked per day, smoking index, and intracranial aneurysm rupture: a case–control study. Front Neurol. 2018;9:1–7. doi:10.3389/fneur.2018.00380
24. Miller MR, Hankinson J, Brusasco V, et al. Standardisation of spirometry. Eur Respir J. 2005;26(2):319–338. doi:10.1183/09031936.05.00034805
25. Rice-Evans C, Miller NJ. Total antioxidant status in plasma and body fluids. Methods Enzymol. 1994;234:279–293. doi:10.1016/0076-6879(94)34095-1
26. Gosker HR, Bast A, Haenen GRMM, et al. Altered antioxidant status in peripheral skeletal muscle of patients with COPD. Respir Med. 2005;99(1):118–125. doi:10.1016/j.rmed.2004.05.018
27. Signorelli SS, Neri S, Sciacchitano S, et al. Behaviour of some indicators of oxidative stress in postmenopausal and fertile women. Maturitas. 2006;53(1):77–82. doi:10.1016/j.maturitas.2005.03.001
28. Oliveras-López MJ, Ruiz-Prieto I, Bolaños-Ríos P, De la Cerda F, Martín F, Jáuregui-Lobera I. Antioxidant activity and nutritional status in anorexia nervosa: effects of weight recovery. Nutrients. 2015;7(4):2193–2208. doi:10.3390/nu7042193
29. Leo F, Rossodivita AN, Segni CD, et al. Frailty of obese children: evaluation of plasma antioxidant capacity in pediatric obesity. Exp Clin Endocrinol Diabetes. 2016;124(8):481–486. doi:10.1055/s-0042-105280
30. Keaney JF, Larson MG, Vasan RS, et al. Obesity and systemic oxidative stress: clinical correlates of oxidative stress in the Framingham study. Arterioscler Thromb Vasc Biol. 2003;23(3):434–439. doi:10.1161/01.ATV.0000058402.34138.11
31. Ben MS, Sfaxi I, Tabka Z, Ben SH, Rouatbi S. Oxidative stress and lung function profiles of male smokers free from COPD compared to those with COPD: a case-control study. Libyan J Med. 2014;9(1):1–13. doi:10.3402/ljm.v9.23873
32. Vivodtzev I, Moncharmont L, Tamisier R, et al. Quadriceps muscle fat infiltration is associated with cardiometabolic risk in COPD. Clin Physiol Funct Imaging. 2018;38(5):788–797. doi:10.1111/cpf.12481
33. Meyer A, Zoll J, Charles AL, et al. Skeletal muscle mitochondrial dysfunction during chronic obstructive pulmonary disease: central actor and therapeutic target. Exp Physiol. 2013;98(6):1063–1078. doi:10.1113/expphysiol.2012.069468
34. Hong JY, Lee CY, Lee MG, Kim YS. Effects of dietary antioxidant vitamins on lung functions according to gender and smoking status in Korea: a population-based cross-sectional study. BMJ Open. 2018;8(4):1–9. doi:10.1136/bmjopen-2017-020656
35. Bewley MA, Budd RC, Ryan E, et al. Opsonic phagocytosis in chronic obstructive pulmonary disease is enhanced by Nrf2 agonists. Am J Respir Crit Care Med. 2018;198(6):739–750. doi:10.1164/rccm.201705-0903oc
36. Mizumura K, Maruoka S, Shimizu T, Gon Y. Role of Nrf2 in the pathogenesis of respiratory diseases. Respir Investig. 2019;1–8. doi:10.1016/j.resinv.2019.10.003
37. Sidhaye VK, Holbrook JT, Burke A, et al. Compartmentalization of anti-oxidant and anti-inflammatory gene expression in current and former smokers with COPD. Respir Res. 2019;20(1):1–9. doi:10.1186/s12931-019-1164-1
38. Ford ES, Li C, Cunningham TJ, Croft JB. Associations between antioxidants and all-cause mortality among US adults with obstructive lung function. Br J Nutr. 2014;112(10):1662–1673. doi:10.1017/S0007114514002669
39. Zemans RL, Jacobson S, Keene J, et al. Multiple biomarkers predict disease severity, progression and mortality in COPD. Respir Res. 2017;18(1):1–10. doi:10.1186/s12931-017-0597-7
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

