Back to Journals » Journal of Pain Research » Volume 17
Tizanidine: Advances in Pharmacology & Therapeutics and Drug Formulations
Authors Zhu LL, Wang YH, Zhou Q
Received 23 January 2024
Accepted for publication 16 March 2024
Published 21 March 2024 Volume 2024:17 Pages 1257—1271
DOI https://doi.org/10.2147/JPR.S461032
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Robert B. Raffa
Ling-Ling Zhu,1,* Yan-Hong Wang,2,* Quan Zhou2
1VIP Geriatric Ward, Division of Nursing, The Second Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang Province, People’s Republic of China; 2Department of Pharmacy, The Second Affiliated Hospital, School of Medicine, Zhejiang University, Hangzhou, Zhejiang Province, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Quan Zhou, Department of Pharmacy, The Second Affiliated Hospital, School of Medicine, Zhejiang University, Jiefang Road No. 88, Shangcheng District, Hangzhou, Zhejiang Province, 310009, People’s Republic of China, Email [email protected]
Background: Skeletal muscle relaxants (SMRs) are widely used in treating musculoskeletal conditions. All SMRs, with the exception of baclofen and tizanidine, are on the list of 2023 American Geriatrics Society Beers Criteria® for potentially inappropriate medication use in older adults. In our geriatric practice, off-label use of tizanidine as preemptive analgesia drove us to find recent advances in its pharmacology and therapeutics. An update review of tizanidine was thus presented, aiming to bring the latest knowledge to clinicians and promote further research and practical exploration.
Methods: Relevant literature up to December 2023 was identified through searches of PubMed, Web of Science, and Embase.
Results: Tizanidine, a centrally acting alpha-2 adrenoceptor agonist with both antispastic and antispasmodic activity, shows efficacy in the common indications for all SMRs. From the perspective of drug safety, tizanidine has lower incidences of adverse events (injury, delirium, encephalopathy, falls, and opioid overdose) compared to baclofen, no association with risk of Alzheimer’s disease as with orphenadrine, no risk of serotonin syndrome like metaxalone when comedicated with serotonergic drugs, no significant pharmacokinetic changes in CYP2C19 poor metabolizers unlike diazepam and carisoprodol, and no physically addictive or abuse properties like carisoprodol and diazepam. From the perspective of new and potential therapeutic uses, tizanidine has additional benefits (eg, gastroprotection that can improve patient tolerance to nonsteroidal anti-inflammatory agents, anti-neuropathic pain, a key component of multimodal analgesia strategy to reduce early postoperative pain, and anti-tumor effects). New delivery systems of tizanidine are developing to improve the pharmacokinetics of oral products, including buccal patches, transdermal delivery systems, nasal spray, and in situ rectal gel.
Conclusion: Tizanidine is an SMR with unique features and may be an optimal initial choice for older adults. There would be more scientific studies, wider therapeutic applications, and new drug formulations in the future.
Keywords: musculoskeletal pain, skeletal muscle relaxants, neuropathic pain, older adults, tizanidine
Introduction
Skeletal muscle relaxants (SMRs) are commonly prescribed medications indicated for relief of muscle spasms associated with acute and painful musculoskeletal conditions. Muscle tone is often accompanied by pain symptoms, so it is crucial to integrate analgesics with SMR adjunctive therapy. Such combination use could significantly improve the effectiveness of treating spasticity and allows to reduce doses of each agent in the multimodal analgesia regimen.1
SMRs can be classified into two main categories: (1) antispasticity agents that work on the spinal cord or directly on the skeletal muscles to improve muscle hypertonicity and involuntary spasms (eg, baclofen, dantrolene), and (2) antispasmodic agents that decrease muscle spasms through alterations of central nervous system (CNS) conduction (eg, carisoprodol, cyclobenzaprine, metaxalone, methocarbamol, orphenadrine, and chlorzoxazone). The overlap between these two categories includes tizanidine and diazepam, as they have both antispastic and antispasmodic activity.2
An understanding of the pharmacology and therapeutics as well as the patient complexity helps to select an appropriate SMR for the patient. Clinicians should weigh the pros and cons of prescribing SMRs while tailoring treatments to each patient’s individual needs.
Diazepam is a long-acting benzodiazepine with sedative, hypnotic, antianxiety, and antiepileptic properties in addition to its muscle relaxant effects, and it is also a controlled substance. In addition, diazepam should be avoided in the elderly due to increased sensitivity of older adults to benzodiazepines and prominently weakened drug metabolism.1 Therefore, the use of diazepam as an SMR is limited in clinical practice, especially for older adults.
It is worth noting that all SMRs, with the exception of baclofen and tizanidine, are on the list of 2023 American Geriatrics Society (AGS) Beers Criteria® for potentially inappropriate medication use in older adults.3 Additionally, baclofen was newly added with a recommendation to avoid its use in older adults with impaired kidney function [estimated glomerular filtration rate (eGFR) <60 mL/min], given the increased risk for encephalopathy in this population.3 So tizanidine is one of the few SMRs that seem to be friendly to older adults.
Based on prescribing information, tizanidine is a centrally acting alpha-2 adrenergic receptor agonist indicated for the management of spasticity.4 In our geriatric practice, tizanidine is sometimes used as preemptive analgesia in older adults undergoing elective surgery. Such off-label use drove us to search for recent advances in the pharmacology and therapeutics of tizanidine. An update review of tizanidine is thus presented, aiming to bring the latest advances to clinicians and promote further research and practical exploration.
Methods
Search Strategy
Potentially relevant literature with title or abstract containing tizanidine from 2013 to 2023 was identified by performing searches on PubMed, Web of Science core collection, MEDLINE, and Embase.
Selection Criteria
Two reviewers (ZLL and WYH) independently retrieved the literature and screened the relevant studies. If they have a disagreement over including or excluding an article, the third reviewer (QZ) was consulted. There were 221 documents identified in PubMed. Among these, documents such as books and documents, comments, clinical conference, letters, reviews, meta-analyses, systematic reviews, case reports, and editorials were excluded (n=73). There were 233 documents from Web of Science Core Collection and MEDLINE. Among these, documents such as books, letters, meetings, reviews, case reports, editorial materials, abstract, retraction, and biography were excluded (n=69). There were 458 documents from Embase. Among these, non-article type documents were excluded (n=238). After excluding duplicated literature, 220 papers underwent further assessment. After reviewing the abstracts, documents were excluded due to reasons [the content was no close relation with therapeutic use of tizanidine or information has been described in package insert of tizanidine (n=161), non-English literature (n=27)]. Full-text articles were further assessed for eligibility. Thirty papers were finally chosen according to the inclusion/exclusion criteria (Figure 1). Valuable information was summarized by data interpretation.
 |
Figure 1 Flow chart showing selection of literature. |
Results
Brief Summary of Prescribing Information
Dosage and Administration
The recommended starting dose of tizanidine is 2 mg. Treatment can be repeated at 6 to 8 h intervals as needed. The dose of tizanidine can be gradually increased by 2–4 mg per dose, with intervals of 1–4 days between each increase, until a satisfactory reduction in muscle tone is achieved. When discontinuing tizanidine, the dose should be slowly reduced to minimize the risk of discontinuation and rebound hypertension, tachycardia, and hypertonia. Tizanidine should be used with caution in patients with renal insufficiency or any hepatic impairment.4
Drug–Drug Interactions
Coadministration of tizanidine with cytochrome P450 enzyme 1A2 (CYP1A2) inhibitors (fluvoxamine, ciprofloxacin) is contraindicated. Other CYP1A2 inhibitors should be avoided in combination with tizanidine due to potential drug interactions that may increase the risk of hypotension and sedation. Sedation can be additive when patients take tizanidine with another CNS depressant (eg, benzodiazepines, opioids, tricyclic antidepressants), so clinicians should monitor symptoms of excessive sedation.4
Adverse Drug Reactions
Adverse effects of tizanidine include somnolence, dry mouth, hypotension, bradycardia, dizziness, fatigue, weakness, hallucinations, abnormal liver function, and hepatotoxicity.4
Safety of Tizanidine versus Other SMRs
Table 1 lists a summary of the comparative safety studies of SMRs.
 |
Table 1 Summary of Comparative Safety Studies of Skeletal Muscle Relaxants |
Injury and Delirium
A retrospective cohort study showed that older adults newly initiated with baclofen had a significantly higher risk of injury and delirium compared to those who started on tizanidine for musculoskeletal pain (adjusted hazard ratio = 1.54, 3.33, respectively, P≤0.001).5
Encephalopathy
In a cohort that included 16,192 new baclofen users and 9782 new tizanidine users, oral baclofen was found to be associated with a higher 30-day risk of encephalopathy compared with tizanidine [subdistribution hazard ratio (SHR)=2.29].6
Opioid Overdose
Using four US commercial and public insurance databases, Khan et al compared the magnitude of opioid overdose risk among patients who were on long-term opioid therapy and were treated with different SMRs at the same time. In the 30-day intention-to-treat analysis, the weighted hazard ratio for opioid overdose relative to cyclobenzaprine was 2.52 for baclofen, 1.64 for carisoprodol, 1.14 for chlorzoxazone/orphenadrine, 0.46 for metaxalone, 1.00 for methocarbamol, and 1.07 for tizanidine, respectively. Concurrent use of opioids and baclofen was associated with the highest and statistically significant risk of opioid overdose (adjusted P value = 0.04).7
Fall and Fracture
Very recently, a real-world cohort study in older adults showed that the risk of fall associated with baclofen was comparable to that of cyclobenzaprine, but greater than that of tizanidine [inverse probability of treatment weighting (IPTW) incidence rate, 108.4 vs 61.9 per 1000 person-years; SHR, 1.68], whereas the risk of fracture was comparable among older users of three commonly used oral SMRs.8
Alzheimer’s Disease (AD)
A nested case-control confirmed that the use of SMRs was associated with the risk of AD. A drug-specific analysis revealed that tizanidine did not carry the risk, whereas cumulative exposure of orphenadrine (≥101 defined daily doses) was associated with the risk of AD (adjusted odds ratio 1.19).9
Hypotension
Chaugai et al compared the frequency of severe hypotensive episodes in patients receiving tizanidine and cyclobenzaprine when comedicated with strong CYP1A2 inhibitors (ciprofloxacin or fluvoxamine). The tizanidine group experienced a higher incidence of severe hypotension than the cyclobenzaprine group (2.03% vs 1.28%; odds ratio = 1.60; P = 0.029).10
New and Potential Therapeutic Uses
Table 2 summarizes the new and potential therapeutic use of tizanidine.
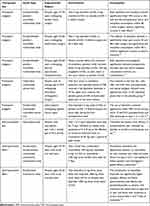 |
Table 2 Clinical Studies Demonstrating New and Potential Therapeutic Uses of Tizanidine |
Preoperative Use
A double-blinded randomized study confirmed the effects of clonidine and tizanidine on early postoperative pain in patients undergoing lumbar fusion surgery. Each group received the corresponding drug orally 1 h before surgery (group A: 4 mg tizanidine, group B: 4 mg clonidine; group C: placebo). The three groups exhibited statistically different visual analogue scale (VAS) at 2nd and 4th postoperative hours, despite comparable pain scores at 6, 12, 24 and 48 postoperative hours. The incidence of side effects (dizziness, nausea, vomiting, drowsiness, headache, and bradycardia) was comparable among the three groups.11 Another randomized clinical trial study reported the benefits of tizanidine for the management of postoperative pain after lumbar spinal fusion surgery. Patients received 4 mg oral tizanidine or placebo 1 h before surgery and 24 h after that. The tizanidine group had a significantly lower pain score at 24 and 48 h, and less morphine consumption after surgery compared with placebo (P<0.005), with comparable incidence of side effects between two groups.12
The Society for Perioperative Assessment and Quality Improvement (SPAQI) provides consensus on preoperative pain medication dosing. For example, tizanidine and baclofen could be taken preoperatively including the day of surgical procedure, whereas other SMRs (methocarbamol, orphenadrine, metaxalone, cyclobenzaprine) should be held on day of surgical procedure.19 According to expert consensus on perioperative management in enhanced recovery after spinal surgery in China and expert consensus on the implementation of enhanced recovery after surgery (ERAS) in percutaneous endoscopic interlaminar lumbar decompression/discectomy (2020), the addition of nerve repair agents, SMRs and anticonvulsants on the basis of sufficient regular use of non-steroidal anti-inflammatory drugs (NSAIDs) can improve the overall efficacy and patient satisfaction in the management of perioperative radiculalgia.20,21
Besides orthopedic surgeries, the use of oral tizanidine 4 mg and pregabaline 100 mg 90 min before laparoscopic cholecystectomy significantly relieved postoperative shoulder pain and analgesic consumption.13 Preemptive tizanidine was effective in reducing postoperative pain intensity and opioid analgesics consumption following bimaxillary orthognathic surgery.14 Patients undergoing inguinal hernia repair received tizanidine 4 mg orally or placebo pill 1 h before surgery and twice daily during the first postoperative week. Compared with placebo, the addition of tizanidine to perioperative pain treatment showed advantages in postoperative pain scores, analgesic consumption, return to daily activities, and health-related quality of life.15
Neuropathic Pain
Activation of adrenergic α2 receptors by tizanidine could induce antiallodynia in neuropathic rats in a sex-dependent manner. The antiallodynic effect of tizanidine in female neuropathic rats is due to the activation of adrenergic α2A/2B receptors, but in male neuropathic rats it is due to the activation of opioid receptors.22 Pei et al demonstrated that tizanidine has anti-nociceptive effects in spared nerve injury model of neuropathic pain by inhibiting the activation of TLR4/NF-κB p65 signaling pathway and subsequently suppressing the production of pro-inflammatory cytokines.23
The effectiveness of tizanidine in patients with neuropathic pain was assessed by Semenchuk et al. Twenty-three enrolled patients took 1 to 4 mg of tizanidine once daily for 7 days, followed by a weekly dose increase of 2 to 8 mg to reach an individual effective or maximum tolerated dose, or a maximum of 36 mg over 8 weeks. The mean average weekly pain score significantly decreased from 6.9 (baseline) to 5.2 (the end of week 8). The neuropathic pain qualities (intense, sharp, hot, dull, cold, sensitive, unpleasant, and deep pain) became significantly lower at the end of week 8 compared to baseline. Fifteen patients (68%) following tizanidine therapy had improved or much improved pain relief. Tizanidine might be an effective agent for neuropathic pain, as well as an alternative for patients poorly responsive to other medications.16 A larger, randomized placebo-controlled trial is needed.
Antitumor Effect
Osteosarcoma is the most frequent primary malignant bone tumor, affecting mainly children, adolescents, and young adults, and with a second peak of incidence in older adults. The treatment of osteosarcoma is primarily based on neo-adjuvant and adjuvant chemotherapy and surgical resection; however, it has encountered bottlenecks, especially for patients with lung metastasis and chemotherapy resistance. It is necessary to provide more effective and less toxic treatment options to high-risk patients.24
It has been found that tizanidine can inhibit the proliferation, migration, and invasion of U2 osteosarcoma cells while promoting apoptosis. The tizanidine-induced cytotoxicity within osteosarcoma cells may involve PI3K/AKT signaling pathway because the expression levels of p-AKT, p-mTOR, and p-P70S6K were reduced by tizanidine exposure. Therefore, tizanidine might be a potent therapeutic strategy for osteosarcoma treatment.25 It is worthwhile to further perform in vivo and human studies.
Data revealed that treatment of tizanidine could inhibit A549 lung cancer cell proliferation and motility by regulating Nischarin.26 Furthermore, Zhu et al observed potent therapeutic efficacy of α2-adrenergic agonists used as monotherapy in multiple tumour models. Combination of anti-programmed cell death 1 (anti-PD-1) with α2-adrenergic agonists showed a strong synergistic anti-tumour effect. The α2-adrenergic receptors involved in cancer cell proliferation, invasion, and apoptosis; therefore, α2-adrenergic agonists might substantially improve the efficacy of cancer immunotherapy.27
Gastroprotective Effect
NSAIDs are widely recommended and prescribed for the treatment of pain; however, they may exhibit gastrointestinal toxicity, which may be of particular concern when treating older patients with chronic musculoskeletal pain.
It has been found that the addition of tizanidine to naproxen or ketorolac could increase anti-inflammatory and anti-nociceptive effects of each NSAID with a lower incidence of gastric injury and spontaneous activity in rats.28 Rodriguez-Silverio et al observed the synergistic interaction between ketorolac and tizanidine in thermal hyperalgesia model and the gastroprotective activity of tizanidine against gastric damage induced by ketorolac in the rat.29
Two clinical studies have examined tizanidine against the potential gastrointestinal damage caused by NSAIDS. Pareek et al evaluated the efficacy and safety of a fixed dose combination (aceclofenac 100 mg plus tizanidine 2 mg) versus aceclofenac (100 mg) alone twice daily for 7 days in patients with acute low back pain (LBP). The aceclofenac–tizanidine combination was more effective than aceclofenac alone and had a favourable safety profile.17 A double-blind, randomized, placebo-controlled parallel group study evaluated the efficacy and tolerability in patients with LBP receiving tizanidine (4 mg three times daily) plus ibuprofen (400 mg three times daily) or placebo plus ibuprofen (400 mg three times daily) for 7 days. Compared with placebo/ibuprofen, tizanidine/ibuprofen was more effective in the treatment of moderate or severe acute LBP, while it was associated with significantly less gastrointestinal side effects. Although more patients receiving tizanidine/ibuprofen suffered drowsiness and other CNS effects, some sedation and bed rest could be beneficial for the patients with severe acute LBP.18
Hence, based on existing animal experiments and several clinical studies, tizanidine–NSAID combination therapy would be a novel approach to treat nociceptive/inflammatory conditions with improved gastric tolerability of NSAIDs.
New Delivery Systems
New delivery systems are being developed to overcome the defects of tizanidine, ie, low oral bioavailability (34–40%) due to extensive first-pass metabolism, the need for frequent administration due to short biological half-life (2.1–4.2 h) and the consequent potential for low patient compliance. Figure 2 illustrated the summary of tizanidine formulations on the market and under development.
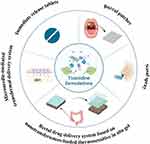 |
Figure 2 Tizanidine formulations on the market and under development. |
Buccal Patches
In vivo studies revealed that chitosan azelate-based buccal patches had about 3 times more bioavailability than the orally administered commercial product, and such patch might be an alternative to oral tizanidine to reduce the dose and dosing frequency.30
El-Mahrouk et al encapsulated tizanidine in chitosan lactate beads cross-linked with sodium tripolyphosphate, and further incorporated the beads into chitosan lactate wafer to make it easy to apply to buccal mucosa. The optimized drug delivery system has good mucosal adhesion and was non-irritating, which is beneficial to improve the pharmacokinetic characteristics of tizanidine. A randomized crossover pharmacokinetic study showed a significant increase in the relative bioavailability of tizanidine in human volunteers receiving the new formulation compared to immediate-release tablets.31
Zaman et al developed tizanidine/meloxicam loaded bilayer mucoadhesive films for buccal administration. The pharmacokinetics of buccal film significantly improved compared to the standard solution/dispersion given to the rabbits (ie, meloxicam was immediately released and permeated from immediate release layer, followed by good controlled release and permeation properties of tizanidine from sustained release films within 8 h). The mucoadhesive buccal films also had good performance (ie, uniform thickness, smooth surface, satisfactory mechanical strength).32
Transdermal Delivery System
Derma roller mediated transdermal delivery of tizanidine-loaded invasomes may be a promising strategy to increase the bioavailability of tizanidine for treating skeletal muscle spasm. After the skin was pretreated with a dermal roller, the plasma concentration of tizanidine peaked within 3 h after gel application to rats and was maintained for 24 h, whereas the peak plasma concentrations (Cmax) was not reached until 6 h after topical administration to the untreated skin.33
It is well known that transdermal drug delivery is severely limited due to the presence of a tough stratum corneum barrier, so a penetrant enhancement method is required to achieve the desired therapeutic drug concentration. Thakkar et al prepared a microneedle-mediated transdermal delivery system of tizanidine. In vivo pharmacokinetic studies showed that the relative bioavailability of microneedle patch preparation was 2.2 times higher than that of the oral marketed preparation.34 Compared to tablets currently on the market, this new formulation appears to have the potential to reduce the dose and administration frequency, thereby improving patient compliance.
Khalil et al utilized aspasomes as potential vector for transdermal delivery of tizanidine. The optimized formulation had nanoscale dispersed spherical vesicles, with good stability and ex vivo permeability, and significantly higher bioavailability in vivo than oral commercial product. It is also non-invasive and safe for the skin.35
Nasal Spray
Intranasal administration could bypass portal circulation, improve bioavailability, reduce drug absorption, and shorten the onset time of tizanidine. Vitale et al evaluated the pharmacokinetic profile of tizanidine nasal spray versus oral tablets in healthy subjects. The intranasal formulation exhibited linear pharmacokinetic characteristics and achieved Cmax in almost half of the time.36 Thakkar et al developed an intranasal cubosomal delivery system of tizanidine for CNS-specific delivery. The optimized formulation was nontoxic and targeted to the nasal mucosa.37
Rectal Delivery System
Moawad et al developed tizanidine-loaded nanotransfersomes for rectal administration. The transfersomal formulation had good permeability, with an encapsulation rate of 52.39% and a controlled release time of 8 h. The bioavailability of tizanidine was increased by about 2.18 times. The terminal elimination half-life (T1/2) was prolonged to about 10 h following in situ gel rectal administration versus oral drug solution dosing. Encapsulation of tizanidine into nanotransfersomes could achieve a dual purpose of prolonged release and increased bioavailability and thus may be considered as a promising drug delivery system for the treatment of spasticity.38
Newly Identified Drug–Drug Interactions
An open-label, multi-center, 3-period, fixed-sequence study confirmed a statistically significant drug–drug interaction between vemurafenib and tizanidine in patients with BRAFV600 mutation-positive metastatic malignancy. Oral vemurafenib 960 mg twice daily for 20 days increased plasma exposure of a single-dose tizanidine, ie, the mean Cmax and area under the concentration–time curve (AUC) of tizanidine increased approximately 2-fold and 4-fold, respectively. The underlying mechanism is that vemurafenib is a moderate inhibitor of CYP1A2, therefore caution should be exercised to concomitant use of vemurafenib and tizanidine.39
Expert Consensus on Musculoskeletal Pain Management
According to expert consensus on the management of chronic musculoskeletal pain in older adults, muscle relaxants could usually be used for the treatment of chronic LBP by relieving skeletal muscle spasm and improving blood microcirculation.40 The expert consensus on the treatment of nerve root pain in degenerative spinal conditions describes that muscle relaxants such as tizanidine can be used for those with reactive muscle spasms.41 Eperisone and tizanidine are central muscle relaxants that can be used in the treatment of a variety of chronic pain.42
According to the clinical guideline for nonspecific LBP in China, α2-adrenergic receptor agonists (such as tizanidine) not only have strong analgesic, muscle relaxant, and anti-anxiety effects, but also have a protective effect on the gastrointestinal tract, therefore the combination of this drug class with NSAIDs can effectively reduce low back pain, improve the range of motion, and offset the gastrointestinal injury caused by NSAIDs.43 According to Chinese expert consensus on diagnosis and treatment of sacroiliac joint pain, NSAIDs and/or muscle relaxants (such as tizanidine) should be given as soon as possible in patients who are less responsive to non-pharmacological treatments or whose pain is evident, whereas tramadol sustained-release tablets or strong opioid analgesics are recommended if pain interferes with sleep.44
Discussion
Alpha-2 Adrenoceptors and Pain Modulation
The alpha2 receptor has three subtypes, ie, alpha2A, alpha2B, and alpha2C adrenoceptors. Alpha2A receptors may play a role in the development of certain forms of hyperalgesia, and they are the sites of action of alpha2 agonists in reducing heat-related hyperalgesia.45 Tizanidine could display different selectivities for alpha-2 adrenoceptors subtypes, and the mean affinity values for alpha-2A, alpha-2B, and alpha-2C were 55.7, 178.0, 120.0 nmol/L, respectively.46
López-Córdoba et al described the differential in vivo contribution of spinal alpha 2A- and alpha 2C-adrenoceptors in tonic and acute evoked nociception in the rat. During acute or tonic nociceptive stimuli, presynaptic alpha 2A-adrenoceptor activation could produce anti-nociceptive stimulation at the spinal level, whereas activation of spinal alpha 2C-adrenoceptor appeared to inhibit GABAergic transmission and thus promote nociception during a tonic (inflammatory) stimulus. As for spinal alpha-2B, it did not play a role in tonic inflammatory and acute nociception.47 The net impact of non-selective alpha 2-adrenoceptor agonists is antinociception in acute and neuropathic pain conditions, suggesting the significance of alpha 2A-adrenoreceptors over alpha 2C-adrenoreceptors.
Alpha 2-Adrenoceptors and Gastroprotection
Alpha 2-adrenoceptors are widely distributed in the gastrointestinal tract, and play a crucial role in presynaptic modulation of transmitter release and gastrointestinal functions. Alpha 2-adrenoceptor agonists could inhibit gastric acid secretion, gastric emptying and motility, while alleviating gastric mucosal injury in different ulcer models. Both alpha 2B- and alpha 2C-adrenoceptor subtypes could contribute equally to the mediation of gastroprotective effect induced by alpha-2 agonists.48,49
Studies in rats have shown that tizanidine could prevent the changes of glycoproteins in the gastric mucosa and gastric juice induced by NSAIDs, reduce central vagal activity and acid secretion, and increase mucosal blood flow by regulating central alpha-2-receptors.29,50–52
Alpha 2-Adrenoceptors and Side Effects
When alpha-2 receptors are stimulated, the activity of the sympathetic nervous system decreases, thus potentially leading to a drop in blood pressure. Unlike clonidine, tizanidine had a significantly less adverse effect on blood pressure, which may be due to their differences in lipophilicity and systemic bioavailability.53,54
The sedative action of alpha-2 agonists is usually attributed to inhibition of the locus coeruleus and its ascending activating projection to the forebrain. Sedation is observed in tizanidine users, but it can be minimized by dose-titration. Patients reported improved sleep when taking tizanidine at night, suggesting a potential benefit for patients with chronic muscle pain and sleep disorders.
Alpha-2 Agonists and ERAS
The alpha-2 agonists exert supraspinal and spinal antinociceptive effects by potentiating descending pain modulatory pathways and inhibiting peripheral C fibers. Perioperative administration of alpha-2 agonists could be used as a part of multimodal analgesia to treat acute postoperative pain and reduce the consumption of opioid analgesics.55 This was associated with a reduction in anxiety, perioperative stress, inflammation, postoperative nausea and vomiting, shivering, and cognitive dysfunction. Such practice may contribute to ERAS, leading to shorter hospitalization and reduced complications and postoperative pain.
Why or How is Tizanidine Better Than Other SMRs
Beers Criteria
According to AGS 2023 updated Beers Criteria for potentially inappropriate medication use in older adults, muscle relaxants typically used to treat musculoskeletal complaints are poorly tolerated by older adults due to some defects (eg, anticholinergic adverse effects, sedation, increased risk of fractures, and questionable effectiveness at dosages tolerated by older adults). However, this criterion does not apply to baclofen and tizanidine. In addition, the AGS Beers Criteria strongly recommends that older adults avoid the use of diazepam due to elevated risk of cognitive impairment, delirium, falls, fractures, and motor vehicle accidents.3
Baclofen
Baclofen is primarily excreted by the kidneys unchanged, leaving those with reduced kidney function more susceptible to its adverse effects.56 Baclofen has higher risks of encephalopathy and opioid overdose than tizanidine,6,7 which is consistent with the characteristics of baclofen as a potent gamma-aminobutyric acid (GABA) agonist, that is, baclofen can produce toxicity through CNS suppression, clinically manifested as confusion, somnolence, or lethargy. As available evidence suggests that different SMRs have similar efficacy in treating musculoskeletal disorders or spasticity,57 prescribers may consider relatively safe agents such as cyclobenzaprine or tizanidine when concomitant opioid use is deemed necessary, or make sure that opioid antagonists are available to patients for whom baclofen is the only option.
Orphenadrine
Orphenadrine users, rather than tizanidine users, carry a higher risk of AD,9 which could be explained partly by the high anticholinergic properties of orphenadrine and the finding that anticholinergic drugs are associated with an increased risk of cognitive disorders in older adults.
Cyclobenzaprine
A drug utilization study of SMRs among commercially insured adults in the United States during the period of 2006 to 2018 showed that cyclobenzaprine was the dominant agent accounting for >50% of prescriptions, followed by methocarbamol, and tizanidine.58 In fact, cyclobenzaprine, structurally similar to the tricyclic antidepressants, can cause the expected lethargy and anticholinergic side effects. Therefore, it is recommended to avoid using cyclobenzaprine in older adults, despite that this SMR has a lower risk of encephalopathy and opioid overdose than baclofen.6,7 There is a scenario where cyclobenzaprine is safer than tizanidine in terms of hypotension when comedicated with strong CYP1A2 inhibitors.10 CYP3A4 and CYP1A2 are primarily responsible for cyclobenzaprine metabolism in human liver microsomes, whereas the primary CYP isoenzyme involved in tizanidine metabolism is CYP1A2.4,59 The contribution of CYP1A2 to the drug metabolism of tizanidine and cyclobenzaprine may determine the degree of drug interaction with strong CYP1A2 inhibitors.
Methocarbamol and Metaxalone
Methocarbamol and metaxalone are less sedated and thus can be useful for patients who cannot tolerate the sedative properties of other SMRs.60 However, they are still on the Beers criteria list as drugs to avoid in the elderly.3 Methocarbamol was associated with an increased risk of injury (odds ratio 1.42, P=0.001).61 Metaxalone showed significant dose-related inhibition of monoamine oxidase A (MAO-A) activity.62 Clinicians should understand that metaxalone may trigger potentially lethal drug interactions resulting in serotonin syndrome when prescribed with other serotonergic drugs, which are usually prescribed as components of multimodal analgesia, such as selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), and tramadol. Tizanidine is safer than metaxalone in this regard because it does not significantly interact with the other serotonergic agents mentioned above, except for the interaction with fluvoxamine that can strongly inhibit CYP1A2-mediated metabolism of tizanidine.4
Carisoprodol
Carisoprodol raises the concern of potential physical and psychological dependence, and the Drug Enforcement Administration (DEA) has listed it as a schedule IV controlled substance. Carisoprodol is metabolized by CYP2C19 to meprobamate, a sedative used in the treatment of anxiety disorders. Given the risk of CNS respiratory depression, seizures and death caused by carisoprodol overdose, and the significant pharmacokinetic change [ie, a four-fold increase in carisoprodol exposure in CYP2C19 poor metabolizers (PMs) following standard-doses compared to normal metabolizers (NMs)], carisoprodol should be used with caution in CYP2C19 PMs (approximately 3–5% of Caucasians and Africans and 15–20% of Asians), or individuals receiving concurrent CYP2C19 inhibitors or inducers.63
Diazepam
Diazepam is also classified as a Schedule IV controlled substance by DEA, with the potential for abuse. Older adults are more susceptible to adverse events than younger adults when administered with diazepam, and thus diazepam is listed as a potentially inappropriate medication in the older population based on the updated 2023 Beers criteria.3 Additionally, the Clinical Pharmacogenetics Implementation Consortium (CPIC) established a prioritization level of B/C for the development of a guideline for diazepam in the following years.64 A strong relationship between CYP2C19 and CYP2B6 phenotypes and diazepam pharmacokinetics was reported.65 The dose-corrected AUC of diazepam in CYP2C19 PMs was nearly 2-fold higher than in extensive metabolizers (EMs) or NMs. The dose-adjusted AUC of CYP2B6 PMs was 2.74 times that of EMs and 2.10 times that of NMs (P< 0.007). For CYP2C19 or CYP2B6 PMs, a 25–50% reduction in diazepam dose may be appropriate to avoid adverse reactions, dependence, and tolerability. For patients with skeletal muscle pain who are PMs of both CYP2C19 and CYP2B6 phenotypes, the dose of diazepam requires greater adjustment, or it is not recommended to use diazepam, but to switch to other alternatives such as tizanidine, which also has both antispastic and antispasmodic effects but its pharmacokinetics is not affected by the CYP2C19 and CYP2B6 genotypes.
Taken together, tizanidine may be the optimal initial choice for older adults because it shows efficacy in the common indications for all SMRs, a lower incidence of adverse events (injury, delirium, encephalopathy, fall, and opioid overdose), no association with AD risk, little possibility of being abused, pharmacokinetic characteristics independent of CYP2C19 and CYP2B6 genotypes, and the interesting additional benefits of tizanidine as an alpha2 receptor agonist (eg, gastroprotection effect that can improve patient tolerance to NSAIDS treatment and potential anti-neuropathic pain effects).
New Horizon and Further Opportunities
There are many opportunities for further research related with tizanidine. Firstly, multimodal therapy is recommended and can be achieved using concomitant administration of individual agents, fixed-dose combinations, or single agent with multiple mechanisms of action.66 The efficacy and safety of multimodal analgesic regimens including tizanidine are worthy of investigation. The pharmacological profile of the combination of tramadol-tizanidine produced antagonism in an experimental acute pain model in rats (ie, no antinociceptive effect was observed after the combination treatment as compared with individual administration).67 The underlying mechanism of this antagonistic interaction remains to be elucidated. Caution is necessary during the use of this combination in clinical practice.
Secondly, laboratory studies of new delivery systems for tizanidine should be translated into clinical trials and new drug approvals. MDM S.p.A. has completed a Phase III multicenter randomized study to assess the efficacy and safety of 0.5 mg Tizaspray® administered intranasally versus Sirdalud® 2 mg tablets in patients with acute low back pain. However, the results of this clinical trial have not been published.68
Thirdly, the efficacy and safety of different SMRs deserve further investigation. For example, tolperisone is effective in the treatment of acute painful muscle spasm and spasticity without the somnolence associated with SMRs.69 A comparative study of tizanidine versus tolperisone has not been reported.
Fourthly, it is interesting to further investigate the effect of individual patient factors on the pharmacokinetics of SMRs. Genetic polymorphisms of CYP2D6 and CYP2C19 and cigarette smoking independently and significantly affected tolperisone pharmacokinetics, and these factors together may have a greater effect (eg, non-smoking patients with the CYP2D6*10/*10 and CYP2C19 PM genotype had a 25.9-fold increase in AUC compared to smokers with the CYP2D6*wt/*wt and CYP2C19 EM genotype).70 A two-fold increase in oral baclofen clearance was associated with the single-nucleotide polymorphism of ABCC9 (rs11046232, heterozygous AT versus TT genotype).71 Smoking can reduce plasma concentrations and the effects of tizanidine. Physiologically based pharmacokinetic (PBPK)-model simulated disposition of tizanidine in various smoking sub-groups, showing that Cmax and AUC gradually decreased as the number of cigarettes smoked per day increased. For heavy smokers (>20 cigarettes per day), the dose of tizanidine may need to be higher than the average dose because the simulated Cmax and AUC are reduced by about 50% compared to nonsmokers.72 Tizanidine is primarily metabolized by CYP1A2, so it is necessary to explore the effect of CYP1A2 genetic polymorphisms on the optimal dosing of tizanidine.
Conclusion
In addition to relieving muscle spasms and chronic musculoskeletal pain, tizanidine is a particular SMR that exhibits multiple beneficial characteristics. Tizanidine is a relatively safer SMR and seems to be friendly to older adults. It can be expected that there will be more scientific studies on tizanidine as well as wider therapeutic applications. New delivery systems will also be available to better meet clinical needs.
Acknowledgments
This work was supported by the Department of Education of Zhejiang Province (grant number: Y202249568) and research projects in Zhejiang University (project code: 491020-5215F6, 491020-I4154A).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Zygmunt M, Sapa J. Muscle relaxants-the current position in the treatment of spasticity in orthopedics. Ortop Traumatol Rehabil. 2015;17:423–430. doi:10.5604/15093492.1173500
2. Witenko C, Moorman-Li R, Motycka C, et al. Considerations for the appropriate use of skeletal muscle relaxants for the management of acute low back pain. P T. 2014;39:427–435. PMID: 25050056. doi:10.1002/jcph.247
3. By the 2023 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics society 2023 updated AGS beers criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2023;71:2052–2081. doi:10.1111/jgs.18372
4. RxList [homepage on the Internet]. ZANAFLEX; 2022. Available from: https://www.rxlist.com/zanaflex-drug.htm#indications.
5. Su Zhang VR, Niu F, Lee EA, et al. Safety of baclofen versus tizanidine for older adults with musculoskeletal pain. J Am Geriatr Soc. 2023;71:2579–2584. doi:10.1111/jgs.18349
6. Hwang YJ, Chang AR, Brotman DJ, Inker LA, Grams ME, Shin JI. Baclofen and the risk of encephalopathy: a real-world, active-comparator cohort study. Mayo Clin Proc. 2023;98:676–688. doi:10.1016/j.mayocp.2022.11.004
7. Khan NF, Bykov K, Barnett ML, Glynn RJ, Vine SM, Gagne JJ. Comparative risk of opioid overdose with concomitant use of prescription opioids and skeletal muscle relaxants. Neurology. 2022;99:e1432–e1442. doi:10.1212/WNL.0000000000200904
8. Hwang YJ, Chang AR, Brotman DJ, Inker LA, Grams ME, Shin JI. Baclofen and the risk of fall and fracture in older adults: a real-world cohort study. J Am Geriatr Soc. 2024;72(1):91–101. doi:10.1111/jgs.18665
9. Rahkonen A, Taipale H, Koponen M, et al. The cumulative use of muscle relaxants and the risk of Alzheimer’s disease: a nationwide case-control study. J Alzheimers Dis. 2023;91:1283–1290. doi:10.3233/JAD-220409
10. Chaugai S, Dickson AL, Shuey MM, et al. Co-prescription of strong CYP1A2 inhibitors and the risk of tizanidine-associated hypotension: a retrospective cohort study. Clin Pharmacol Ther. 2019;105(3):703–709. doi:10.1002/cpt.1233
11. Aezi G, Shafizad M, Firouzian A, Mirani A, Hassanzadeh Kiabi F. Effects of tizanidine and clonidine on postoperative pain after lumbar fusion surgery. Interdiscip Neurosurg. 2023;31:101680. doi:10.1016/j.inat.2022.101680
12. Meskar H, Aezzi G, Kiyasari AZ, Shafizad M, Jalalian F, Alderraji M. The effect of tizanidine on postoperative pain management after lumbar fusion surgery. Exp Appl Biomed Res. 2023;24:49–56.
13. Sane S, Sayyadi S, Abbasivash R, Rezaei S, Azadfar A, Salimi S. Comparison of the effect of preoperative oral tizanidine and pregabalin on shoulder pain in laparoscopic cholecystectomy under general anesthesia. Adv Biomed Res. 2023;12:58. doi:10.4103/abr.abr_222_22
14. Dadmehr S, Shooshtari Z, Alipour M, et al. Is preemptive oral tizanidine effective on postoperative pain intensity after bimaxillary orthognathic surgery? A triple-blind randomized clinical trial. World J Plast Surg. 2022;11:37–45. doi:10.52547/wjps.11.2.37
15. Yazicioğlu D, Caparlar C, Akkaya T, Mercan U, Kulaçoğlu H. Tizanidine for the management of acute postoperative pain after inguinal hernia repair: a placebo-controlled double-blind trial. Eur J Anaesthesiol. 2016;33:215–222. doi:10.1097/EJA.0000000000000371
16. Semenchuk MR, Sherman S. Effectiveness of tizanidine in neuropathic pain: an open-label study. J Pain. 2000;1:285–292. doi:10.1054/jpai.2000.9435
17. Pareek A, Chandurkar N, Chandanwale AS, Ambade R, Gupta A, Bartakke G. Aceclofenac-tizanidine in the treatment of acute low back pain: a double-blind, double-dummy, randomized, multicentric, comparative study against aceclofenac alone. Eur Spine J. 2009;18:1836–1842. doi:10.1007/s00586-009-1019-4
18. Berry H, Hutchinson DR. Tizanidine and ibuprofen in acute low-back pain: results of a double-blind multicentre study in general practice. J Int Med Res. 1988;16:83–91. doi:10.1177/030006058801600202
19. O’Rourke MJ, Keshock MC, Boxhorn CE, et al. Preoperative management of opioid and nonopioid analgesics: Society for Perioperative Assessment and Quality Improvement (SPAQI) consensus statement. Mayo Clin Proc. 2021;96:1325–1341. doi:10.1016/j.mayocp.2020.06.045
20. Tiansheng S, Jianxiong S, Zhongjun L, et al. Expert consensus in enhanced recovery after spinal surgery in China: perioperative management. Chin J Bone Joint Surg. 2017:271–279. doi:10.3969/j.issn.2095-9958.2017.04-01
21. The Group of Minimally Invasive Spinal Surgery and Enhanced Recovery, Professional Committee of Orthopaedic Surgery and Enhanced Recovery, Association of China Rehabilitation Technology Transformation and Promotion. Expert consensus on the implementation of enhanced recovery after surgery in percutaneous endoscopic interlaminar lumbar decompression/discectomy (2020). Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2020;34:1497–1506. doi:10.7507/1002-1892.202011021
22. Rodríguez-Palma EJ, Castelo-Flores DG, Caram-Salas NL, et al. Sex-dependent antiallodynic effect of α2 adrenergic receptor agonist tizanidine in rats with experimental neuropathic pain. Eur J Pharmacol. 2022;920:174855. doi:10.1016/j.ejphar.2022.174855
23. Pei W, Zou Y, Wang W, Wei L, Zhao Y, Li L. Tizanidine exerts anti-nociceptive effects in spared nerve injury model of neuropathic pain through inhibition of TLR4/NF-κB pathway. Int J Mol Med. 2018;42:3209–3219. doi:10.3892/ijmm.2018.3878
24. Smrke A, Anderson PM, Gulia A, Gennatas S, Huang PH, Jones RL. Future directions in the treatment of osteosarcoma. Cells. 2021;10:172. doi:10.3390/cells10010172
25. Xing XW, Sun YF, Zhao J, Pan ZX, Jiang WX. Tizanidine hydrochloride exhibits a cytotoxic effect on osteosarcoma cells through the PI3K/AKT signaling pathway. J Int Med Res. 2019;47:3792–3802. doi:10.1177/0300060519850395
26. Zhao L, Zhao G, Xue Q. Tizanidine (hydrochloride) inhibits A549 lung cancer cell proliferation and motility through regulating Nischarin. Onco Targets Ther. 2020;13:291–298. doi:10.2147/OTT.S228317
27. Zhu J, Naulaerts S, Boudhan L, Martin M, Gatto L, Van den Eynde BJ. Tumour immune rejection triggered by activation of α2-adrenergic receptors. Nature. 2023;618(7965):607–615. doi:10.1038/s41586-023-06110-8
28. Patiño-Camacho SI, Déciga Campos M, Beltrán-Villalobos K, Castro-Vidal DA, Montiel-Ruiz RM, Flores-Murrieta FJ. Low doses of tizanidine synergize the anti-nociceptive and anti-inflammatory effects of ketorolac or naproxen while reducing of side effects. Eur J Pharmacol. 2017;805:51–57. doi:10.1016/j.ejphar.2017.03.021
29. Rodriguez-Silverio J, Sanchez-Mendoza ME, Arrieta-Valencia J, Rocha-Gonzalez HI, Flores-Murrieta FJ. Tizanidine increases antinociceptive effect and prevents gastric damage induced by ketorolac in the rat. Drug Dev Res. 2013;74:38–42. doi:10.1002/ddr.21054
30. Arpa MD, Nü O, Gök MK, Özgümüş S, Cevher E. Chitosan-based buccal mucoadhesive patches to enhance the systemic bioavailability of tizanidine. Int J Pharm. 2023;642:123168. doi:10.1016/j.ijpharm.2023.123168
31. El-Mahrouk GM, El-Gazayerly ON, Aboelwafa AA, Taha MS. Chitosan lactate wafer as a platform for the buccal delivery of tizanidine HCl: in vitro and in vivo performance. Int J Pharm. 2014;467:100–112. doi:10.1016/j.ijpharm.2014.03.049
32. Zaman M, Hanif M, Shaheryar ZA, Xu B. Development of tizanidine HCl-meloxicam loaded mucoadhesive buccal films: in-vitro and in-vivo evaluation. PLoS One. 2018;13:e0194410. doi:10.1371/journal.pone.0194410
33. Dsouza L, Chaudhari P, Brahmam B, Lewis SA. Derma roller mediated transdermal delivery of tizanidine invasomes for the management of skeletal muscle spasms. Eur J Pharm Sci. 2021;165:105920. doi:10.1016/j.ejps.2021.105920
34. Thakkar H, Pandya K, Patel B. Microneedle-mediated transdermal delivery of tizanidine hydrochloride. Methods Mol Biol. 2020;2059:239–258. doi:10.1007/978-1-4939-9798-5_13
35. Khalil RM, Abdelbary A, Arini SKE, Basha M, El-Hashemy HA, Farouk F. Development of tizanidine loaded aspasomes as transdermal delivery system: ex-vivo and in-vivo evaluation. J Liposome Res. 2021;31:19–29. doi:10.1080/08982104.2019.1684940
36. Vitale DC, Piazza C, Sinagra T, et al. Pharmacokinetic characterization of tizanidine nasal spray, a novel intranasal delivery method for the treatment of skeletal muscle spasm. Clin Drug Investig. 2013;33:885–891. doi:10.1007/s40261-013-0137-2
37. Thakkar H, Modi B, Patel B. Intranasal spray of cubosomal tizanidine hydrochloride for brain targeting: in vitro and in vivo characterisation. J Microencapsul. 2023;40:366–383. doi:10.1080/02652048.2023.2209651
38. Moawad FA, Ali AA, Salem HF. Nanotransfersomes-loaded thermosensitive in situ gel as a rectal delivery system of tizanidine HCl: preparation, in vitro and in vivo performance. Drug Deliv. 2017;24:252–260. doi:10.1080/10717544.2016.1245369
39. Zhang W, McIntyre C, Riehl T, et al. Effect of vemurafenib on the pharmacokinetics of a single dose of tizanidine (a CYP1A2 Substrate) in patients with BRAFV600 mutation-positive malignancies. Clin Pharmacol Drug Dev. 2020;9:651–658. doi:10.1002/cpdd.788
40. Ji Q, Yi D, Wang JY, et al. Expert consensus on management of chronic musculoskeletal pain in older adults (2019). Chin J Geriatr. 2019:500–507. doi:10.3760/cma.j.issn.0254-9026.2019.05.008
41. Chinese Association for the Study of Pain. Expert consensus on spinal degenerative nerve root pain treatment. Natl Med J China. 2019;1133–1137. doi:10.3760/cma.j.issn.0376-2491.2019.15.003
42. Kang PD, Huang ZY, Li T, et al. Expert consensus on chronic pain management in the musculoskeletal system. Chin J Bone Joint Surg. 2020:8–16. doi:10.3969/j.issn.2095-9958.2020.01.02
43. Gui R, Wang Y, Chen BH. Clinical guidelines for nonspecific low back pain in China. Chin J Spine Spinal Cord. 2022;32:258–268. doi:10.3969/j.issn.1004-406X.2022.03.09
44. Spinal pain Group Affiliated to Pain Society of Chinese Medical Association. Chinese expert consensus on diagnosis and treatment of sacroiliac joint pain. Chin J Pain Med. 2021;87–93. doi:10.3969/j.issn.1006-9852.2021.02.002
45. Smith H, Elliott J. Alpha(2) receptors and agonists in pain management. Curr Opin Anaesthesiol. 2001;14:513–518. doi:10.1097/00001503-200110000-00009
46. Piletz JE, Zhu H, Chikkala DN. Comparison of ligand binding affinities at human I1-imidazoline binding sites and the high affinity state of alpha-2 adrenoceptor subtypes. J Pharmacol Exp Ther. 1996;279:694–702.
47. López-Córdoba G, Martínez-Lorenzana G, Lozano-Cuenca J, Condés-Lara M, González-Hernández A. The differential in vivo contribution of spinal α2A- and α2C-adrenoceptors in tonic and acute evoked nociception in the rat. Front Pharmacol. 2022;13:1023611. doi:10.3389/fphar.2022.1023611
48. Gyires K, Müllner K, Rónai AZ. Functional evidence that gastroprotection can be induced by activation of central alpha(2B)-adrenoceptor subtypes in the rat. Eur J Pharmacol. 2000;396:131–135. doi:10.1016/s0014-2999(00)00231-4
49. Zádori ZS, Shujaa N, Brancati SB, Hein L, Gyires K. Both α2B- and α2C-adrenoceptor subtypes are involved in the mediation of centrally induced gastroprotection in mice. Eur J Pharmacol. 2011;669:115–120. doi:10.1016/j.ejphar.2011.07.018
50. Maeda-Hagiwara M, Watanabe H, Kanaoka R, Watanabe K. Influence of clonidine and a new related imidazoline derivative (tizanidine) on rat gastric mucosa. Pharmacology. 1986;32:109–113. doi:10.1159/000138158
51. Jain NK, Kulkarni SK, Singh A. Modulation of NSAID-induced antinociceptive and anti-inflammatory effects by alpha2-adrenoceptor agonists with gastroprotective effects. Life Sci. 2002;70:2857–2869. doi:10.1016/s0024-3205(02)01549-7
52. Emre M. The gastroprotective effects of tizanidine: an overview. Curr Therap Res. 1998;59:2–12. doi:10.1016/S0011-393X(98)85018-2
53. Kroin JS, McCarthy RJ, Penn RD, Lubenow TJ, Ivankovich AAD. Continuous intrathecal clonidine and tizanidine in conscious dogs: analgesic and hemodynamic effects. Anesth Analg. 2003;96:776–782. doi:10.1213/01.ANE.0000048087.57487.0C
54. Kawamata T, Omote K, Yamamoto H, Toriyabe M, Wada K, Namiki A. Antihyperalgesic and side effects of intrathecal clonidine and tizanidine in a rat model of neuropathic pain. Anesthesiology. 2003;98:1480–1483. doi:10.1097/00000542-200306000-00027
55. Zhang SJ, Lai G, Griffis CA, Schiltz M, Aroke EN. α2-Adrenergic receptor agonist, an attractive but underused ERAS component in improving fast-track recovery and surgical outcomes. AANA J. 2021;89:529–537.
56. Wolf E, Kothari NR, Roberts JK, Sparks MA. Baclofen toxicity in kidney disease. Am J Kidney Dis. 2018;71(2):275–280. doi:10.1053/j.ajkd.2017.07.005
57. Chou R, Peterson K, Helfand M. Comparative efficacy and safety of skeletal muscle relaxants for spasticity and musculoskeletal conditions: a systematic review. J Pain Symptom Manage. 2004;28(2):140–175. doi:10.1016/j.jpainsymman.2004.05.002
58. Li Y, Delcher C, Reisfield GM, Wei Y-J, Brown JD, Winterstein AG. Utilization patterns of skeletal muscle relaxants among commercially insured adults in the United States from 2006 to 2018. Pain Med. 2021;22(10):2153–2161. doi:10.1093/pm/pnab088
59. Wang RW, Liu L, Cheng H. Identification of human liver cytochrome P450 isoforms involved in the in vitro metabolism of cyclobenzaprine. Drug Metab Dispos. 1996;24(7):786–791.
60. See S, Ginzburg R. Choosing a skeletal muscle relaxant. Am Fam Physician. 2008;78(3):365–370.
61. Spence MM, Shin PJ, Lee EA, Gibbs NE. Risk of injury associated with skeletal muscle relaxant use in older adults. Ann Pharmacother. 2013;47(7–8):993–998. doi:10.1345/aph.1R735
62. Cherrington B, Englich U, Niruntari S, Grant W, Hodgman M. Monoamine oxidase A inhibition by toxic concentrations of metaxalone. Clin Toxicol. 2020;58(5):383–387. doi:10.1080/15563650.2019.1648815
63. RxList [homepage on the Internet]. VANADOM; 2023. Available from: https://www.rxlist.com/vanadom-drug.htm#clinpharm.
64. CPIC [hompage on the Internet]. Prioritization of CPIC guidelines. Available from: https://cpicpgx.org/prioritization-of-cpic-guidelines/.
65. Zubiaur P, Figueiredo-Tor L, Villapalos-García G, et al. Association between CYP2C19 and CYP2B6 phenotypes and the pharmacokinetics and safety of diazepam. Biomed Pharmacother. 2022;155:113747. doi:10.1016/j.biopha.2022.113747
66. Almansa C, Frampton CS, Vela JM, Whitelock S, Plata-Salamán CR. Co-crystals as a new approach to multimodal analgesia and the treatment of pain. J Pain Res. 2019;12:2679–2689. doi:10.2147/JPR.S208082
67. Beltrán-Villalobos KL, Ramírez-Marín PM, Cruz CA, Déciga-Campos M. Coadministration of tramadol and tizanidine in an experimental acute pain model in rat. Drug Dev Res. 2014;75:473–478. doi:10.1002/ddr.21229
68. ClinicalTrials.gov [hompage on the Internet]. Study to assess the efficacy and safety of tizaspray® Versus Sirdalud®, in patients with acute low back pain; 2018. Available from: https://clinicaltrials.gov/study/NCT02934061.
69. Nalamachu S, Pergolizzi J, Kaye R. Tolperisone for the treatment of acute muscle spasm of the back: results from the dose-ranging Phase 2 STAR Study (NCT03802565). J Pain Res. 2020;13:3059–3069. doi:10.2147/JPR.S278857
70. Byeon JY, Cho CK, Kang P, et al. Effects of CYP2D6 and CYP2C19 genetic polymorphisms and cigarette smoking on the pharmacokinetics of tolperisone. Arch Pharm Res. 2023;46:713–721. doi:10.1007/s12272-023-01462-1
71. McLaughlin MJ, He Y, Brunstrom-Hernandez J, et al. Pharmacogenomic variability of oral baclofen clearance and clinical response in children with cerebral palsy. PM R. 2018;10:235–243. doi:10.1016/j.pmrj.2017.08.441
72. Jogiraju VK, Heimbach T, Toderika Y, Taft DR. Physiologically based pharmacokinetic modeling of altered tizanidine systemic exposure by CYP1A2 modulation: impact of drug-drug interactions and cigarette consumption. Drug Metab Pharmacokinet. 2021;37:100375. doi:10.1016/j.dmpk.2020.100375
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
