Back to Journals » Drug Design, Development and Therapy » Volume 8
Time and dose relationships between schisandrin B- and schisandrae fructus oil-induced hepatotoxicity and the associated elevations in hepatic and serum triglyceride levels in mice
Authors Zhang Y, Pan S, Zhou S, Wang X, Sun N, Zhu P, Chu Z, Yu Z, Ko K
Received 10 May 2014
Accepted for publication 10 June 2014
Published 19 September 2014 Volume 2014:8 Pages 1429—1439
DOI https://doi.org/10.2147/DDDT.S67518
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Video abstracted presented by Yi Zhang.
Views: 445
Yi Zhang,1 Si-Yuan Pan,1 Shu-Feng Zhou,2 Xiao-Yan Wang,1 Nan Sun,1 Pei-Li Zhu,1 Zhu-Sheng Chu,1 Zhi-Ling Yu,3 Kam-Ming Ko4
1Department of Pharmacology, School of Chinese Materia Medica, Beijing University of Chinese Medicine, Beijing, People's Republic of China; 2Department of Pharmaceutical Sciences, College of Pharmacy, University of South Florida, Tampa, FL, USA; 3School of Chinese Medicine, Hong Kong Baptist University, 4Division of Life Science, Hong Kong University of Science and Technology, Hong Kong, People's Republic of China
Background: Schisandrin B (Sch B), a dibenzocyclooctadiene compound, is isolated from schisandrae fructus (SF). This study was conducted to compare the time- and dose-response between Sch B- and SF oil (SFO)-induced changes in hepatic and serum parameters in mice.
Methods: Institute of Cancer Research (ICR) mice were given a single oral dose of Sch B (0.125–2 g/kg) or SFO (0.3–5 g/kg). Serum alanine aminotransferase (ALT) activity, hepatic malondialdehyde, and triglyceride (TG) levels were measured at increasing time intervals within 6–120 hours postdosing.
Results: Serum ALT activity was elevated by 60%, with maximum effect (Emax) =45.77 U/L and affinity (KD) =1.25 g/kg at 48–96 hours following Sch B, but not SFO, treatment. Sch B and SFO treatments increased hepatic malondialdehyde level by 70% (Emax =2.30 nmol/mg protein and KD =0.41 g/kg) and 22% (Emax =1.42 nmol/mg protein and KD =2.56 g/kg) at 72 hours postdosing, respectively. Hepatic index was increased by 16%–60% (Emax =11.01, KD =0.68 g/kg) and 8%–32% (Emax =9.88, KD =4.47 g/kg) at 12–120 hours and 24–120 hours after the administration of Sch B and SFO, respectively. Hepatic TG level was increased by 40%–158% and 35%–85%, respectively, at 12–96 hours and 6–48 hours after Sch B and SFO treatment, respectively. The values of Emax and KD for Sch B/SFO-induced increase in hepatic TG were estimated to be 22.94/15.02 µmol/g and 0.78/3.03 g/kg, respectively. Both Sch B and SFO increased serum TG (up to 427% and 123%, respectively), with the values of Emax =5.50/4.60 mmol/L and KD =0.43/2.84 g/kg, respectively.
Conclusion: The findings indicated that Sch B/SFO-induced increases in serum/hepatic parameters occurred in a time-dependent manner, with the time of onset being serum TG level < hepatic TG level < hepatic index < serum ALT activity. However, the time of recovery of these parameters to normal values varied as follow: serum TG level < hepatic TG level and liver injury < hepatic index. The Emax and affinity of Sch B on tissue/enzyme/receptor were larger than those of SFO.
Keywords: pharmacodynamics, hepatomegaly, alanine aminotransaminase, affinity, maximum effect
Introduction
Pharmacokinetics (PK) and pharmacodynamics (PD) are integral parts of drug development and the rational use of drug in clinical situations,1 which is exemplified by the clinical potential for combined pharmacokinetically- and pharmacodynamically-guided dosing (PK/PD dosing) of chemotherapy agents in the treatment of cancer patients.2,3 PK is defined as the study of the changes of concentrations of drugs and/or their metabolite(s) with time in blood and tissues as well as the time course of drug absorption, distribution, metabolism, and excretion.4–6 PD refers to the study of the time course and intensity of therapeutic and adverse effects of a drug, which are related to the receptor binding, postreceptor events, and chemical interactions. In this regard, the approach of PK/PD modeling is adopted to investigate the dose-response relationship for some drugs in vivo.7–9 However, for other drugs, a simpler relationship between the concentration and effect is modeled mathematically to conceptualize receptor occupancy and drug response in an idealized in vitro system.10–12
Owing to the belief that naturally-occurring herbs or compounds are much safer than chemical synthetic drugs, herbal medicines/products have become more and more popular over the world. However, as to how the pharmacological basis for the efficacy and safety of herbal medicines can be established remains challenging. Therefore, the investigation of PK and PD of herbal medicinal products has been an area of intensive research.13,14 It is well known that PD analysis of drug-receptor or drug-enzyme interaction can be conveniently performed in vitro, but PD parameters can also be obtained in vivo.15,16 The dose-response relationship can be described by Clark’s equation, which is mathematically derived from the mass-action law.17 The maximum effect (Emax) and affinity (KD) can also be determined by using Scott’s plot method in vivo.
Schisandrae fructus (SF) (“Wu-Wei-Zi” in Chinese), which has been widely used for thousands of years in the People’s Republic of China, is found in Northwestern China, Korea, and in the Russian Far East.18 Schisandrin B (Sch B) (Figure 1) is an active dibenzocyclooctadiene derivative isolated from SF. Previous studies in our laboratory have shown that Sch B treatment significantly increased serum and hepatic triglycerides (TG), as well as hepatic index in mice.16,19,20 In the present study, the correlation and values of Emax/KD of Sch B and SF oil (SFO) on serum/hepatic TG levels and hepatic index/injury were investigated in mice.
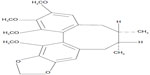  | Figure 1 The chemical structure of schisandrin B. |
Materials and methods
Herbal material and extraction procedure
SF was purchased from the Anguo Chinese herbs market in Hebei province, People’s Republic of China, and authenticated as the fruit of Schisandra chinensis (Turcz.) Baillon (“Bei-Wu-Wei-Zi” in Chinese) by Professor Chun-Sheng Liu in the Beijing University of Chinese Medicine. The SF seeds were removed, washed, and dried at room temperature. The seeds were then pulverized using an industrial blender. For the preparation of the SFO extract, 500 g of powdered SF seeds was extracted twice with five volumes of petroleum ether (60°C–80°C) under reflux. The pooled extract was filtered by filter paper and concentrated under reduced pressure by rotaevaporation to obtain SFO, with a yield of 20% (w/w) (ie, 5 g of SF seeds for every 1 g of extract), and stored at 4°C until use.
Chemicals and reagents
Sch B was purified from the petroleum ether extract of dried SF by silica gel column chromatography as previously described.21 The purity of Sch B, as determined by high performance liquid chromatography analysis, was higher than 95%. Betis extra virgin olive oil was purchased from a local market. Assay kit for TG (certificate number 135991) was purchased from Zhongsheng Beikong Biotechnology Science Inc. (Beijing, People’s Republic of China). The assay kit for alanine aminotransferase (ALT) was obtained from Beijing Leadman Biochemistry Co, Ltd (Beijing, People’s Republic of China). Malondialdehyde (MDA) (certificate number 20130325) and Coomassie brilliant blue G250 (certificate number 20130325) were purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, People’s Republic of China).
Animal treatment
Male Institute of Cancer Research (ICR) mice (Grade II, certificate number 118, SCXK [jing] 2006-0009), weighing 18–20 g, were purchased from the Vital River Laboratory Animal Technology Co., Ltd. (Beijing, People’s Republic of China). They were maintained at 20°C–21°C, with a relative humidity of 50%–55% and allowed free access to water and food. Animals were housed ten in each cage, and ten mice were assigned to each group. Experiments were performed when the animals had attained a body weight of 25–28 g. All experimental procedures were approved by the University Committee on Research Practice in Beijing University of Chinese Medicine.
Experimental design
Design one
In this study, the time response of Sch B and SFO on serum/hepatic TG, hepatic index, hepatic MDA production, and hepatic injury was estimated. Mice were treated with either oral Sch B (1 g/kg) or SFO (2.5 g/kg) suspended in olive oil. The doses of Sch B and SFO were chosen with reference to our previous studies and median lethal dose value. Control animals were orally administered the vehicle (ie, olive oil, 5 mL/kg) only. After 6, 12, 24, 48, 72, 96, and 120 hours postdosing, mice were sacrificed under light ether anesthesia. Blood samples were collected from the orbital vein, and liver tissue samples were also obtained and subjected to biochemical analysis.
Design two
This study was designed to estimate the dose response of Sch B and SFO on the above mentioned parameters. Mice were orally administered with either Sch B at increasing doses of 0.125, 0.25, 0.5, 1, and 2 g/kg or SFO 0.3, 0.6, 1.25, 2.5, and 5 g/kg, suspended in olive oil. Control (ie, non-Sch B- or non-SFO-treated) animals were given the vehicle. Mice were sacrificed at the time point of Emax for each parameter: serum and hepatic TG level, serum ALT activity, hepatic index, and hepatic MDA level.
Biochemical analysis
Serum samples were prepared by centrifuging whole blood for 8 minutes at 2,000× g and stored at −70°C until used for biochemical analysis. Liver tissue samples were homogenized in nine volumes of saline using two 10-second bursts of a tissue disintegrator at 13,500 rpm, and the homogenate was then centrifuged at 2,000× g for 15 minutes to obtain the supernatants. Hepatic supernatant (30 μL) and serum (10 μL) were used to determine TG levels using the GPO-PAP (glycerol-3-phosphate oxidase and phenol + aminophenazone) method. Serum ALT activity was measured by automatic Biochemistry Analyzer (Synchron CX4 PRO; Beckman Coulter, Inc., Brea, CA, USA). Aliquots of 100 μL of hepatic supernatant were used to measure hepatic MDA and protein level using thiobarbiturate acid and G-250 dye method, respectively. Assays were performed using assay kits according to the manufacturer’s instructions.
Measurement of hepatic index
The liver was excised and weighed. Hepatic index was estimated from the ratio of liver weight to body weight (liver weight/body weight ×100).
Statistical analysis
All values are expressed as means ± standard error of the mean. Data were analyzed by one-way analysis of variance (ANOVA) using SPSS (version 16.0) statistical analysis program, and then differences among means were analyzed using Dunnett’s multiple comparisons test or post hoc analysis. Differences were considered significant at P<0.05. The parameters of Emax and KD of Sch B and SFO were obtained using the Scott’s plot method. KD is the affinity constant for the ligand-receptor interaction. A low- and high-affinity interaction is characterized by a high and low KD value, respectively. Correlation test was performed using the Pearson product-moment correlation coefficient method.
Results
Time- and dose-response relationship of Sch B- and SFO-induced changes in serum ALT activity
In this study, serum ALT activity was determined as an indirect measure of liver function in Sch B or SFO-treated mice. While SFO treatment (2.5 g/kg) produced no detectable change in serum ALT activity, Sch B (1 g/kg) caused increases in ALT activity by 49%, 68%, and 27% (P<0.01) at 48, 72, and 96 hours postdosing, respectively, with the maximal effect observed at 72 hours (Figure 2A) and the values for Emax being estimated at 45.77 U/L and KD 1.25 g/kg (Figure 2B).
Time- and dose-response relationship of Sch B- and SFO-induced changes in hepatic MDA production
MDA, which is the byproduct of oxidation of phospholipids, was an indirect measure of lipid peroxidation. In addition to assessing liver function, the effects of Sch B and SFO treatment on hepatic MDA contents were examined. Sch B and SFO treatment increased hepatic MDA levels by 70% (P<0.01) and 22% (P<0.05) at 72 hours postdosing, respectively (Figure 3A). Sch B (0.125–2 g/kg) and SFO (0.3–5 g/kg) dose-dependently elevated hepatic MDA levels by 16%–94% and 1%–20% at 72 hours posttreatment, respectively, with estimated values for Emax of 2.30 and 1.42 nmol/mg protein, respectively, and KD of 0.41 and 2.56 g/kg, respectively (Figure 3B).
Time- and dose-response relationship of Sch B- and SFO-induced changes in hepatic index
Figure 4 shows that Sch B and SFO time- and dose-dependently increased the hepatic index. Sch B increased the hepatic index by 18%–60% (P<0.01) from 12–120 hours posttreatment. SFO increased the hepatic index (17%–31%; P<0.01) from 24–120 hours postdosing. The maximal effects of Sch B and SFO on hepatic index were observed at 48 hours postdosing (Figure 4A). Sch B (0.125–2 g/kg) and SFO (0.3–5 g/kg) dose-dependently increased the hepatic index by 16%–60% and 8%–32% at 48 hours posttreatment, respectively, with values for Emax of 11.01 and 9.88, respectively, and KD of 0.68 and 4.47 g/kg, respectively, being estimated (Figure 4B).
Time- and dose-response relationship of Sch B- and SFO-induced changes in hepatic TG level
Figure 5 shows that treatment with Sch B and SFO increased hepatic TG contents in a time- and dose-dependent manner. Sch B (1 g/kg) increased hepatic TG by 76%, 114%, 158%, 112%, and 40% (P<0.01) at 12, 24, 48, 72, and 96 hours posttreatment, respectively. Hepatic TG contents were increased by 85%, 82%, 60%, and 35% at 6, 12, 24, and 48 hours, respectively, after the SFO (2.5 g/kg) treatment. The maximal effects of Sch B- and SFO-induced elevation in hepatic TG level were observed at 48 and 6 hours posttreatment, respectively (Figure 5A). Sch B and SFO gradually increased hepatic TG levels by 65%–209% and 33%–90% (P<0.01) at 48 and 6 hours posttreatment, respectively, with the values for Emax being estimated at 22.94 and 15.02 μmol/g, respectively, and for KD at 0.78 and 3.03 g/kg, respectively (Figure 5B).
Time- and dose-response relationship of Sch B- and SFO-induced changes in serum TG level
Both Sch B and SFO treatment increased serum TG levels in a time- and dose-dependent manner. Serum TG levels were elevated by 69%, 262%, 427%, 289%, and 100% (P<0.01) at 6, 12, 24, 48, and 72 hours, respectively, after dosing with Sch B at 1 g/kg when compared with the control group. However, SFO at dose of 2.5 g/kg increased serum TG levels by 89% and 123% (P<0.01) at 12 and 24 hours postdosing, respectively. The maximal effect of Sch B- and SFO-induced hypertriglyceridemia was observed at 24 hours posttreatment (Figure 6A). Sch B and SFO dose-dependently increased serum TG levels by approximately 268% (P<0.01) and 154% (P<0.01) at 24 hours posttreatment, with values for Emax being 5.50 and 4.60 mmol/L and KD being 0.43 and 2.84 g/kg, respectively (Figure 6B).
Effect of Sch B and SFO on body weight
The body weight of mice was suppressed on the first two days following the Sch B and SFO treatment. Sch B (1 g/kg) significantly lowered the body weight by 202% and 62% at 24 and 48 hours postdosing, respectively, when compared with the control group. SFO treatment (2.5 g/kg) caused body weight loss by 245% and 46% at 24 and 48 hours posttreatment, respectively (Figure 7).
Correlation analysis of Sch B and SFO on serum/hepatic TG, hepatomegaly, and serum ALT activity
Correlation analysis measures the relationship between two variables. In the present study, it was found that Sch B and SFO treatment caused increases in serum/hepatic TG levels, hepatomegaly, and liver damage as indicated by abnormal serum ALT activity. Therefore, the relationship between serum and hepatic TG, hepatic TG, and hepatomegaly as well as liver size and serum ALT activity were analyzed in order to explore their correlation, which was expressed as the value of Pearson’s r. The data of correlation analysis were collected from the corresponding dose-effect response. Results showed that there was a high positive correlation between the serum and hepatic TG change after Sch B and SFO treatment (r=0.507, P=0.000; r=0.598, P=0.000, respectively), followed by hepatic TG and hepatomegaly (r=0.309, P=0.019; r=0.439, P=0.001, respectively). Hepatic index and serum ALT activity also showed a positive correlation in Sch B-treated mice (r=0.354, P=0.013), but not in SFO-treated mice (r=0.061, P=0.672) (Figure 8A–F).
Time course of Sch B-/SFO-induced changes in serum/hepatic TG level, hepatic index, and serum ALT activity
The percent changes with respect to control values for serum/hepatic TG level, hepatic index, and serum ALT activity were analyzed at increasing time intervals within 6–120 hours postdosing with Sch B or SFO in mice. Results indicated that the time of onset/recovery of changes in serum TG level, hepatic TG level, hepatic index, and serum ALT activity was 6/72, 12/96, 12/120, and 48/96 hours and 12/24, 6/48, and 24/120 hours after Sch B and SFO treatment, respectively. The maximal effects of Sch B-/SFO-induced changes in serum TG, hepatic TG, hepatic index, serum ALT activity were observed at 24/24, 48/6, 48/48, and 72 hours postdosing, respectively. There was no detectable change of serum ALT activity in SFO-treated mice (Figure 9).
Discussion
In the present study, Sch B and SFO treatment increased serum ALT activity, hepatic index, and serum/hepatic TG levels in mice. It is generally known that TG biosynthesis mainly involves endogenous and exogenous pathways. The exogenous pathway of TG biosynthesis permits efficient absorption of dietary lipids to form chylomicrons in the intestines. The endogenous pathway of TG biosynthesis is derived predominantly from the esterification of long chain fatty acid (FA) in the liver, and TG is then transported to the circulation by very low density of lipoprotein (VLDL). Hypertriglyceridemia results primarily from increases in VLDL and chylomicrons in the blood, which have been attributed to the over production of VLDL in the liver and disorder of intestinal TG-rich lipoprotein production.22 As for FA, it is degraded through mitochondrial β-oxidation;23 therefore, the inhibition of β-oxidation will lead to the production of TG from FA. It has been reported that Sch B treatment could inhibit β-oxidation and increase plasma FA levels.24 In addition, it has been shown that SFO contains FAs (octadecadienoic acid [93.3%] and octadecenoic acid [4.0%]) and other compounds such as Sch B (2.2%) as major chemical components.25–27 Hence, it is reasonable to speculate that FA and Sch B in SFO mainly contribute to the hypertriglyceridemia caused by SFO. Our results showed that Sch B and SFO treatment increased serum and hepatic TG levels in a time- (6−96 hours postdosing) and dose-dependent manner. Scott’s plot analysis showed that the potency of Sch B-induced elevation in both serum and hepatic TG levels is higher than that of SFO. Furthermore, serum TG level was about 1.2 fold higher than that of hepatic TG after Sch B treatment. The lower KD (high affinity) values of hypertriglyceridemia caused by Sch B indicated that the TG-elevating action of Sch B may be mediated by enzyme(s) and/or receptor(s).
Sch B-/SFO-induced hepatomegaly in association with hepatic steatosis possibly involves the promotion of lipogenesis and retardation of lipolysis, which, when acting in concert, result in the accumulation of lipids in the liver.28 However, the possibility of drug-induced proliferation of liver cells and/or changes in cellular composition(s) cannot be excluded. The deposition of excessive hepatic lipids may trigger liver cell injury and lipid peroxidation, with the resultant increase in oxidative stress. The increased oxidative stress in turn induces the expression of inflammatory cytokines, which have been considered to be causally involved in the development of tissue damage, fibrosis, and inflammation in hepatic steatosis.29
Sch B and SFO used in the current study were found to increase the hepatic index in a time- and dose-dependent manner. Sch B was found to be 1.3 fold more effective in increasing hepatic index than SFO, as evidenced by the Emax values. While Sch B has been shown to protect against cell injury caused by various toxicants via antioxidant mechanisms,30,31 results obtained from the present study suggested that Sch B at a dose of 1 g/kg might result in hepatotoxicity, as indicated by increases in serum ALT activity and hepatic MDA level. Sch B-induced increase in serum ALT activity might be related to the drug-induced hepatomegaly, hepatic steatosis, and/or its direct toxic effect on liver cells at such a high dosage. Histological examination of hepatic tissue sections (hematoxylin and eosin, original magnification ×20) showed that hepatic lipid deposits appeared as small vacuoles within the cytoplasm of liver cells in drug-treated mice, but there were no significant changes in the inflammatory status (data not shown). This observation indicated that the extent of Sch B- and SFO-induced liver damage was not beyond the threshold for triggering histopathological changes.
Although SF, a traditional herbal medicine, has been used as tonic and adaptogen in the People’s Republic of China and Russia, since the 1970s, experimental and clinical investigations have demonstrated the hepatoprotective effect of SF and/or its related compounds such as Sch B, bifendate, and bicyclol (see review32) in rodents and the symptom-relieving effect in patients with viral hepatitis. However, in the present study, Sch B and SFO treatment were found to cause increases in serum and hepatic TG levels, hepatic index, and serum ALT activity in mice. Sch B- and SFO-induced hepatomegaly showed a more protracted time course (from 12–120 hours postdosing), followed by hypertriglyceridemia (from 6–96 hours) and liver injury (from 48–96 hours) in Sch B-treated mice. Therefore, caution should be exercised when a high dose of an active compound, which is otherwise nontoxic at low doses of administration, is recommended for human use. For example, it has been shown that SF and Sch B possess antioxidant and liver protective effects. But our results indicated that Sch B and SFO at high doses caused increases in oxidative stress and liver injury. In this connection, Smejkal et al33 and Chyau et al34 found that Schisandra chinensis lignans and Sch B at high concentrations could kill BY-2 and HepG2 cells in vitro. However, the biochemical mechanism underlying the cytotoxic effect is largely unknown. Therefore, future studies are warranted to investigate the mechanism(s) involved in the Sch B and SFO-induced changes in liver functions particularly the TG metabolism.
In conclusion, the results obtained from the present study showed that serum ALT activity was elevated at 24–96 hours after Sch B, but not SFO, treatment. Sch B- and SFO-induced hepatomegaly and elevation in hepatic MDA content were observed at 12–120 and 72 hours postdosing, respectively. Sch B/SFO treatment increased serum/hepatic TG levels (up to 427%) in a time/dose-dependent manner, with the time course of changes spanning from 6–96/12–24 hours and 12–96/6–48 hours postdosing, respectively. Analysis of PD parameters indicated that Sch B induced hypertriglyceridemia, hepatomegaly, and liver damage in a more effective manner than SFO. Moreover, the affinity of Sch B on tissues was higher than that of SFO. Correlation analysis indicated that the degree of Sch B/SFO-induced hepatic TG accumulation was positively correlated with the extents of hepatomegaly and liver injury in mice.
Acknowledgments
This work was supported by a grant from the National Natural Science Foundation of China (grant number 31071989).
Disclosure
The authors report no conflicts of interest in this work.
References
Fosdick A, Zheng J, Pflanz S, et al. Pharmacokinetic and pharmacodynamic properties of GS-9620, a novel Toll-like receptor 7 agonist, demonstrate interferon-stimulated gene induction without detectable serum interferon at low oral doses. J Pharmacol Exp Ther. 2014;348(1):96–105. | |
Arpon DR, Gandhi MK, Martin JH. A new frontier in haematology – combining pharmacokinetic with pharmacodynamic factors to improve choice and dose of drug. Br J Clin Pharmacol. 2014;78(2):274–281. | |
Saif MW, Choma A, Salamone SJ, Chu E. Pharmacokinetically guided dose adjustment of 5-fluorouracil: a rational approach to improving therapeutic outcomes. J Natl Cancer Inst. 2009;101(22):1543–1552. | |
Rezende VM, Rivellis A, Novaes MM, de Alencar Fisher Chamone D, Bendit I. Quantification of imatinib in human serum: validation of a high-performance liquid chromatography-mass spectrometry method for therapeutic drug monitoring and pharmacokinetic assays. Drug Des Devel Ther. 2013;7:699–710. | |
Yoon S, Lee H, Kim TE, et al. Comparative steady-state pharmacokinetic study of an extended-release formulation of itopride and its immediate-release reference formulation in healthy volunteers. Drug Des Devel Ther. 2014;8:123–128. | |
Golor G, Hu K, Ruffin M, et al. A first-in-man study to evaluate the safety, tolerability, and pharmacokinetics of pasireotide (SOM230), a multireceptor-targeted somatostatin analog, in healthy volunteers. Drug Des Devel Ther. 2012;6:71–79. | |
Salphati L, Wong H, Belvin M, et al. Pharmacokinetic-pharmacodynamic modeling of tumor growth inhibition and biomarker modulation by the novel phosphatidylinositol 3-kinase inhibitor GDC-0941. Drug Metab Dispos. 2010;38(9):1436–1442. | |
Betts AM, Clark TH, Yang J, et al. The application of target information and preclinical pharmacokinetic/pharmacodynamic modeling in predicting clinical doses of a Dickkopf-1 antibody for osteoporosis. J Pharmacol Exp Ther. 2010;333(1):2–13. | |
Romero E, Vélez de Mendizabal N, Cendrós JM, et al. Pharmacokinetic/pharmacodynamic model of the testosterone effects of triptorelin administered in sustained release formulations in patients with prostate cancer. J Pharmacol Exp Ther. 2012;342(3):788–798. | |
Ahmed AA, Marki A, Gaspar R, et al. β(2)-Adrenergic activity of 6-methoxykaempferol-3-O-glucoside on rat uterus: in vitro and in silico studies. Eur J Pharmacol. 2011;667(1–3):348–354. | |
Alvarez-Jaimes L, Sutton SW, Nepomuceno D, et al. In vitro pharmacological characterization of RXFP3 allosterism: an example of probe dependency. PLoS One. 2012;7(2):e30792. | |
Corboz MR, Rivelli MA, Shah H, et al. Role of α2-adrenoceptors in electrical field stimulation-induced contraction of pig nasal mucosa and pharmacologic characterization of a novel α2C-adrenoceptor agonist. Am J Rhinol Allergy. 2013;27(2):84–90. | |
Zhang YZ, An R, Yuan J, Wang Y, Gu QQ, Wang XH. [Analysis and comparison of intestinal absorption of components of Gegenqinlian decoction in different combinations based on pharmacokinetic parameters]. Yao Xue Xue Bao. 2013;48(10):1611–1617. Chinese. | |
Bhattaram VA, Graefe U, Kohlert C, Veit M, Derendorf H. Pharmacokinetics and bioavailability of herbal medicinal products. Phytomedicine. 2002;9(Suppl 3):1–33. | |
Pan SY, Han YF. Comparison of the inhibitory efficacy of four belladonna drugs on gastrointestinal movement and cognitive function in food-deprived mice. Pharmacology. 2004;72(3):177–183. | |
Pan SY, Dong H, Guo BF, et al. Effective kinetics of schisandrin B on serum/hepatic triglyceride and total cholesterol levels in mice with and without the influence of fenofibrate. Naunyn Schmiedebergs Arch Pharmacol. 2011;383(6):585–591. | |
Chou TC. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol Rev. 2006;58(3):621–681. | |
Panossian A, Wikman G. Pharmacology of Schisandra chinensis Bail.: an overview of Russian research and uses in medicine. J Ethnopharmacol. 2008;118(2):183–212. | |
Pan SY, Dong H, Han YF, Li WY, Zhao XY, Ko KM. A novel experimental model of acute hypertriglyceridemia induced by schisandrin B. Eur J Pharmacol. 2006;537(1–3):200–204. | |
Pan SY, Jia ZH, Zhang Y, et al. Novel mouse model of combined hyperlipidemia associated with steatosis and liver injury by a single-dose intragastric administration of schisandrin B/cholesterol/bile salts mixture. J Pharmacol Sci. 2013;123(2):110–119. | |
Ip SP, Poon MK, Wu SS, et al. Effect of schisandrin B on hepatic glutathione antioxidant system in mice: protection against carbon tetrachloride toxicity. Planta Med. 1995;61(5):398–401. | |
Trevaskis NL, Charman WN, Porter CJ. Acute hypertriglyceridemia promotes intestinal lymphatic lipid and drug transport: a positive feedback mechanism in lipid and drug absorption. Mol Pharm. 2011;8(4):1132–1139. | |
Bartlett K, Eaton S. Mitochondrial beta-oxidation. Eur J Biochem. 2004;271(3):462–469. | |
Kwan HY, Hu YM, Chan CL, et al. Lipidomics identification of metabolic biomarkers in chemically induced hypertriglyceridemic mice. J Proteome Res. 2013;12(3):1387–1398. | |
An LP, Wang YP, Liu XM, et al. [Effect of Schisandrae fructus oil on type 2 diabetic rats induced by streptozotocin]. Chin Tradit Herbal Drugs. 2012;43(3):552–556. Chinese. | |
Hou DY, Hui RH, Li TC, Diao QP. [Determination of fatty acids and octadecadienoic acid in Schisandra chinensis]. J Anshan Normal Univ. 2011;(2):26–29. Chinese. | |
Zhang T, Bo J, Xu W, Geng, N. [Analysis of fatty acids composition in the oil of schisandra chinensis by GC-MS]. J Cent Univ Nat (Nat Sci Edition). 2012;2(21):29–31. Chinese. | |
Buang Y. The mechanism of hepatomegaly associated with hepatic steatosis. J Pharm Res. 2011;4(12):4404–4407. | |
Day CP, James OF. Steatohepatitis: a tale of two “hits”? Gastroenterology. 1998;114(4):842–845. | |
Lam PY, Leong PK, Chen N, Ko KM. Schisandrin B enhances the glutathione redox cycling and protects against oxidant injury in different types of cultured cells. Biofactors. 2011;37(6):439–446. | |
Chang CY, Chen YL, Yang SC, et al. Effect of schisandrin B and sesamin mixture on CCl(4)-induced hepatic oxidative stress in rats. Phytother Res. 2009;23(2):251–256. | |
Pan SY, Zhou SF, Gao SH, et al. New perspectives on how to discover drugs from herbal medicines: CAM’s outstanding contribution to modern therapeutics. Evid Based Complement Alternat Med. 2013;2013: 627375. | |
Smejkal K, Slapetová T, Krmencík P, et al. Evaluation of cytotoxic activity of Schisandra chinensis lignans. Planta Med. 2010;76(15):1672–1677. | |
Chyau CC, Ker YB, Chang CH, et al. Schisandra chinensis peptidoglycan-assisted transmembrane transport of lignans uniquely altered the pharmacokinetic and pharmacodynamic mechanisms in human HepG2 cell model. PLoS One. 2014;9(1):e85165. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.








