Back to Journals » OncoTargets and Therapy » Volume 11
Thymosin α1 suppresses migration and invasion of PD-L1 high-expressing non-small-cell lung cancer cells via inhibition of STAT3–MMP2 signaling
Authors Bo C, Wu Q, Zhao H, Li X, Zhou Q
Received 22 June 2018
Accepted for publication 15 September 2018
Published 23 October 2018 Volume 2018:11 Pages 7255—7270
DOI https://doi.org/10.2147/OTT.S177943
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Leo Jen-Liang Su
Cong Bo,1,* Qiang Wu,1,* Hai Zhao,2 Xuebing Li,3 Qinghua Zhou1,3
1Lung Cancer Center, West China Hospital, Sichuan University, Chengdu, China; 2Department of Critical Care Medicine, West China Hospital, Sichuan University, Chengdu, China; 3Tianjin Key Laboratory of Lung Cancer Metastasis and Tumor Microenvironment, Tianjin Lung Cancer Institute, Tianjin Medical University General Hospital, Tianjin, China
*These authors contributed equally to this work
Background: Thymosin α1 (Tα1) is one of the most commonly used immunomodulators for metastatic non-small-cell lung cancer (NSCLC) patients in many countries. Despite the identification of the direct suppression on cancer cell proliferation, little is known about its effect on metastasis and metastasis-related signaling such as matrix metalloproteinases (MMPs) and programmed cell death ligand 1 (PD-L1).
Materials and methods: NSCLC cells with distinguishing PD-L1 expression levels were treated with Tα1. siRNAs were used to knockdown PD-L1. Cell migration and invasion abilities were evaluated by wound-healing and transwell assays. The xenograft model by BALB/c nude mice was constructed to test the inhibitory effect of Tα1 on metastasis in vivo. The expression levels of metastasis-related signaling pathways and key molecules were assessed by Western blot (WB) and quantitative reverse transcriptase PCR (qRT-PCR).
Results: Tα1 significantly suppressed cell migration and invasion in PD-L1 high-expressing H1299, NL9980, and L9981 cells but not in PD-L1 low-expressing A549 or SPC-A-1 cells. This difference was demonstrated by mouse model in vivo as well. Knocking down of PD-L1 significantly impaired the inhibition of cell migration and invasion caused by Tα1 treating in PD-L1 high-expressing cells. Besides, Tα1 inhibited the activation and translocation of STAT3 and the expression of MMP2 in PD-L1 high-expressing NSCLC cells. Moreover, the treatment of STAT3 activator colivelin could partly reverse the Tα1-induced MMP2 suppression and the migration phenotype.
Conclusion: Tα1 significantly suppresses migration and invasion in PD-L1 high-expressing NSCLC cells compared with PD-L1 low-expressing NSCLC cells in vitro and in vivo, through the downregulation of STAT3–MMP2 signaling. These different responses to Tα1, together with the depiction of Tα1-induced signaling changes, suggest a potential benefit of Tα1 for PD-L1-positive NSCLC patients, enlightening the combination of Tα1 with target therapy or immune checkpoint inhibitors.
Keywords: matrix metalloproteinase 2, non-small-cell lung cancer, programmed cell death ligand 1, STAT3, thymosin α1
Introduction
Lung cancer is the leading cause of both new cancer diagnoses and cancer-related deaths worldwide.1 Non-small-cell lung cancer (NSCLC) comprises 85%–90% of all lung cancers.2 Despite the increasing therapeutic means such as targeted drugs and stereotactic body radiation therapy, the 5-year overall survival rate of NSCLC remains less than 15%.3 This is largely because of the strong invasive and metastatic potentials of NSCLC. Recent data show that approximately 90% of NSCLC patients die due to distant metastases rather than primary tumor;4,5 hence, there is an urgent need to restrain tumor metastasis in NSCLC.
Thymosin α1 (Tα1), found in highest concentrations in the thymus, is a 28-amino-acid peptide first described and characterized by Goldstein et al.6,7 In 1966, Goldstein et al purified a calf thymus extract termed thymosin fraction V (TFV), which can be divided into α-thymosin, β-thymosin, and γ-thymosin according to their isoelectric points.7,8 Tα1 is the most abundant α-thymosin in TFV and has an immunomodulatory activity 10–1,000 times as potent as TFV.9 The immunomodulatory effects of Tα1 include enhancing dendritic cell and antibody responses, modulating cytokine and chemokine production, and blocking steroid-induced apoptosis of thymocytes, but it is centered primarily on the augmentation of T-lymphocyte function.10,11 T lymphocytes play a pivotal role in tumor immune surveillance and pathogen clearance; hence, Tα1 is now in clinical trials worldwide for the treatment of several types of malignancies including NSCLC.10,12 In China, Tα1 has been approved and the oncologists often use it as a complementary treatment during chemo- or radiotherapy, the improved quality of life after which has been observed.13
Interestingly, besides immunomodulatory effect, Tα1 has shown to act directly on the tumor cells. Moody et al14 first described the antigrowth effect of Tα1 on NCI-H1299 NSCLC cell line in vitro and in vivo. Subsequent studies further revealed the antiproliferative and apoptosis-inducing effects on lung cancer, breast cancer, and leukemia cells.9,15,16 However, little is known about the effect on metastasis. Furthermore, when it comes to the impact of Tα1 on the pathway of tumor cells, all the aforementioned studies only outlined the relationship with the PI3K/AKT pathway.15
In the present study, we investigated the migration and invasion of NSCLC cells treated with Tα1, as well as the related signaling pathway changes in NSCLC cells. First, we observed the distinguishing reductions in migration and invasion in the NSCLC cells with different PD-L1 expression levels: PD-L1 high-expressing NSCLC cells (H1299, NL9980, and L9981) showed a significant suppression of metastasis after Tα1 treatment, while PD-L1 low-expressing cells (A549 and SPC-A-1) did not. The phenotypic changes after knocking down of PD-L1 verified the relationship between different responses and PD-L1 levels in NSCLC. Additionally, STAT3–matrix metalloproteinase 2 (MMP2) pathway was found to be associated with the metastatic changes in PD-L1 high-expressing NSCLC cells, which was further confirmed by the treatment of STAT3 activator. Our study depicted a more comprehensive molecular diagram of NSCLC cells treated with Tα1, enlightening the combination of Tα1 and targeted therapy in the future trials.
Materials and methods
Agents and antibodies
Tα1 was generously provided by Dr Qinghua Zhou (West China Hospital, Sichuan University, Chengdu, China). Tα1 powder was dissolved in the standard solvent mannitol and used for cell treatment immediately. Colivelin (# sc-361153) was purchased from Santa Cruz Biotechnology Inc. (Dallas, TX, USA), and LY294002 (# S1105) was purchased from Selleck Chemicals (Houston, TX, USA). Colivelin and LY294002 were dissolved in dimethyl sulfoxide (DMSO), stored in 1.5 mL sterile plastic tubes at −20°C, thawed, and used on the day of the experiment.
The antibodies used in this study include PD-L1 (# 13684; Cell Signaling Technology, Boston, MA, USA), phosphorylated-STAT3 (Tyr705) (p-STAT3; # 9145; Cell Signaling Technology), STAT3 (# 9139; Cell Signaling Technology), MMP2 (# 4022; Cell Signaling Technology), MMP9 (# 2270; Cell Signaling Technology), β-actin (AF0003; Beyotime Institute of Biotechnology, Shanghai, China), and PCNA (# 13110; Cell Signaling Technology).
Cell cultures
Human bronchial epithelial Beas-2B cells and human NSCLC cells (95C, 95D, A549, NCI-H1299, SPC-A-1, NCI-H460, and YTMLC-90) were purchased from the Cell Bank of the Chinese Academy of Science (Shanghai, China), while the sub-cell lines, high-metastatic L9981 and low-metastatic NL9980, were isolated and established from a human lung large cell carcinoma cell line by Dr Qinghua Zhou.17 Luciferase-expressing L9981 (L9981-luc) and A549 (A549-luc) cells were established by Tianjin Lung Cancer Institute (Tianjin, China). All the cells were cultured at 37°C in the Roswell Park Memorial Institute (RPMI)-1640 medium (Thermo Fisher Scientific, Waltham, MA, USA) supplemented with 10% FBS (Thermo Fisher Scientific), 100 U/mL of penicillin, and 100 μg/mL of streptomycin in a humidified atmosphere of 5% CO2.
Wound-healing assay
The NSCLC cells were seeded into six-well plates and cultured to 90% confluence. A sterile 1 mL pipette tip (Corning Incorporated, Corning, NY, USA) was used to scratch straight lines in the cell layer to create wounds. Then, the cells were washed with PBS twice and cultured in the RPMI-1640 medium with Tα1 of different concentrations. Wounds were observed at different time points within the scrape lines, and representative points were marked and photographed at five individual fields. The widths of wound gaps were measured using the ImageJ software.
Migration and invasion assays
A transwell apparatus with 8 μm filter inserts (# 3422; Corning Incorporated) was used to measure cell migration ability. The apparatus with 50 μL of matrigel coated at the bottom of the upper chamber was used to measure invasion ability. Six hundred microliters of medium with 10% FBS was added into the lower chamber, and 5×104 cells after treatment were added into the upper chamber without FBS. After an incubation of 18 h (migration assay) or 24 h (invasion assay) at 37°C, the inner side of the chamber was wiped with a cotton swab to remove the cells and the outer side was fixed by isopropanol for 20 min and then stained by 0.05% crystal violet for 20 min. The stained cells of each chamber were photographed in five randomly selected fields and subjected to cell counting. The average cell number in the five fields was recorded, and each experiment was repeated at least three times.
Cell counting kit-8 (CCK-8) assay
A CCK-8 (CK04-3000T; Dojindo Laboratories Co., Ltd, Kumamoto, Japan) was used to assess cell viability. The cells (7,000 cells/well) were seeded in 96-well plates. After 24 h of incubation, the cells were further treated with Tα1 for 12, 24, 36, or 48 h. Cell viability was detected by adding 10 μL of CCK-8 dissolved in 90 μL of RPMI-1640 medium, and the absorbance at 450 nm was recorded using a multi-label counter after 2 h incubation.
Colony formation assay
Briefly, 500 cells were seeded in 12-well plates. After 24 h of incubation, the cells were treated with 45, 90, or 180 μM Tα1 for 12–16 days. The cells were fixed with 4% formaldehyde for 20 min at room temperature (RT), and the colonies were stained with 0.5% crystal violet and manually counted. Colonies with at least 100 cells were considered to represent viable cells.
In vivo assay
All animal experiments were approved by the ethics committee of Tianjin Medical University. Immunodeficient BALB/c mice (aged 5–7 weeks, female) were purchased from the Institute of Zoology (Chinese Academy of Sciences, Beijing, China) and housed with sterilized cages under 12 h light/dark cycle, 18°C–22°C, and 50%–60% relative humidity. Food and drinking water were provided ad libitum. After 1 week adaptation, each mouse was inoculated with 0.1 mL of tumor cell suspension (9×106 A549-luc or 1×107 L9981-luc) at the right posterior flank and in vivo imaged using an IVIS200 (Xenogen; Caliper Life Sciences, Hopkinton, MA, USA). These data were identified at 0 weeks. When tumors became visible (50–80 mm3), the mice were subcutaneously injected with Tα1 dissolved in mannitol (20 μg/g body weight, once a day), while the control mice were injected with mannitol only. Luciferase activity was measured every 7 days and analyzed by the Image-Pro Plus software (Media Cybernetics, Bethesda, MD, USA).
Protein extraction and Western blotting
Cells after treatment were washed with ice-cold PBS for three times. RIPA lysis buffer (P0013C; Beyotime Institute of Biotechnology) with 1% phenylmethanesulfonyl fluoride solution (PMSF) was used for cell lysis and total protein collection. For nuclear and cytoplasmic protein extraction, Nuclear and Cytoplasmic Protein Extraction Kit (P0027; Beyotime Institute of Biotechnology) was used and the protocol is as follows. First, cells in 12-well plates were scraped off and collected by centrifugation. Each cell pellet was dissolved in 100 μL cytoplasmic protein extraction agent A with 1% of PMSF by 5 min vortex and 15 min incubation on ice. Next, 5 μL of cytoplasmic protein extraction agent B was added, followed by another 5 min vortex and 1 min incubation on ice. Then, the sample was centrifuged at 16,000× g for 5 min at 4°C, and the supernatant containing cytoplasmic protein was frozen immediately for further analysis. The pellet was re-suspended in 25 μL of nuclear protein extraction agent with 1% of PMSF, followed by 30 s vortex and 30 min incubation on ice. Finally, the supernatant containing the nuclear protein was obtained after another centrifugation at 16,000× g for 10 min at 4°C. Protein lysates were boiled at 100°C for 10 min, separated with 10% SDS-PAGE, and then transferred into the nitrocellulose (NC) membrane (EMD Millipore, Billerica, MA, USA). Membrane was blocked by skim milk dissolved in 5% Tris-buffered saline with Tween-20 buffer (TBST) at RT for 1 h. After the incubation with primary antibodies overnight at 4°C, the blots were then incubated with corresponding goat antirabbit (A0208, 1:2,500; Beyotime Institute of Biotechnology) or goat antimouse (A0216, 1:2,500; Beyotime Institute of Biotechnology) IgG horseradish peroxidase-conjugated secondary antibody for 1 h at RT. After that, the membrane was washed with TBST five times for 7 min at each time. The signals were visualized with electrochemiluminescence substrates (EMD Millipore).
Quantitative reverse transcriptase PCR (qRT-PCR)
The total RNA of NSCLC cells was extracted using the TRIzol reagent (Thermo Fisher Scientific) and reversely transcribed to single-strand complementary DNA with the Reverse Transcriptase M-MLV (RNase H-) (Takara, Shiga, Japan). Primers of PD-L1, MMP2, MMP9, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were synthesized by the Beijing Genomics Institute (Beijing, China). Reactions were amplified in a 7900 Fast RT-PCR System (Thermo Fisher Scientific) and carried out under the following conditions: initial denaturation at 95°C for 10 min, 35 cycles of denaturation at 95°C for 20 s, annealing at 60°C for 10 s, and polymerization at 72°C for 30 s. The cycle threshold (CT) values of the target genes were identified, and mRNA levels were calculated as a ratio of normalized GAPDH level according to the 2−ΔΔCT method. Gene-specific primers are listed in Table 1.
  | Table 1 Primer sequences for real-time PCR analysis |
siRNA interference and transfections
PD-L1-siRNA was purchased from GenePharma (Shanghai, China). The targeting sequences are shown in Table 2. H1299, NL9980, L9981, A549, and SPC-A-1 cells were cultured overnight to a confluence of 50%. Transfections without serum were conducted with 80 nM PD-L1 siRNAs or negative control siRNAs using the Lipofectamine 2000 reagent (Thermo Fisher Scientific) for 6 h. After culturing with serum for additional 42 h, the cells were trypsinized and divided evenly for Western blot (WB), qRT-PCR, and Tα1 treatment experiments.
  | Table 2 Target sequences of PD-L1 siRNA |
Statistical analysis
Data in bar graphs are expressed as the mean ± SD from at least three independent experiments. Statistical analysis was performed using the Student’s unpaired t-test or one-way ANOVA. All statistical analyses were performed with the GraphPad Prism 6 (GraphPad Software, Inc., La Jolla, CA, USA). A P-value of ≤0.05 was considered statistically significant.
Results
Tα1 suppresses migration and invasion in PD-L1 high-expressing NSCLC cells but not in PD-L1 low-expressing NSCLC cells
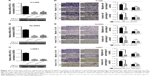
First, the study revealed the distinguishing levels of PD-L1 transcription and expression in different NSCLC cell lines. Compared with the normal bronchial epithelial cells (Beas-2B), H1299, NL9980, and L9981 presented high PD-L1 levels, while A549 and SPC-A-1 presented a lower expression (Figure 1). These five NSCLC cell lines were selected for the following experiments. In wound-healing assay, the migration abilities of H1299, NL9980, and L9981 cells were attenuated by Tα1 in both time- and dose-dependent manners (Figure 2A, P<0.001). Transwell assay further showed the significant suppression of migration and invasion after 48 h treatment with 90 and 180 μM Tα1 in H1299, NL9980, and L9981 (Figures 2B and 3A–C, P<0.001). However, as for PD-L1 low-expressing cells, A549 and SPC-A-1, the migration and invasion reductions were not significant in the quantitative assessment of transwell assay (Figures 2B and 3D and E, P>0.05). Additionally, though CCK-8 and colony formation assays revealed a significant suppression of proliferation by Tα1 in both PD-L1 high-expressing NSCLC cells (H1299, NL9980, and L9981) and PD-L1 low-expressing NSCLC cell (A549) as reported,18 there was still a tendency that the suppression was stronger in PD-L1 high-expressing cells (Figure S1). In summary, NSCLC cells with high PD-L1 expression have a distinct response of migration and invasion to Tα1.
Tα1 attenuates metastases of PD-L1 high-expressing NSCLC but not low-expressing NSCLC in nude mice
PD-L1 high-expressing cell L9981-luc was inoculated at the right posterior flank of eight nude mice. Four mice received Tα1 treatment, while the other four mice were administrated with the solvent. The same treatment was performed in the other two groups of nude mice inoculated with PD-L1 low-expressing cell A549-luc. Among those inoculated with L9981-luc, significantly fewer metastatic lesions appeared in the Tα1 treatment group compared with the control group. Inconsistently, mice inoculated with A549-luc displayed similar amount of metastatic lesions between control and Tα1 treatment groups and there was no significant difference in the fluorescence intensity between the metastatic lesions of the two groups as well (Figure 3F).
Knocking down of PD-L1 impairs the inhibitory effects of cell migration and invasion caused by Tα1 treating in PD-L1 high-expressing NSCLC cells
To verify the relationship between different responses and PD-L1 levels, PD-L1 was knocked down by siRNAs in H1299, NL9980, and L9981 cells (Figure 4A–C). PD-L1-silenced cells with negative control cells were treated with or without 180 μM Tα1 for 48 h. Similar to A549 and SPC-A-1 cells, the migration and invasion suppression of PD-L1-silenced cells could not be observed (Figure 4D–F, P>0.05), while the negative control cells remained a significant suppression (Figure 4D–F, P<0.001).
Tα1 inhibits the activation of STAT3 and suppresses the expression of MMP2 in PD-L1 high-expressing NSCLC cells
To explore the mechanism whereby Tα1 suppressed the migration and invasion in PD-L1 high-expressing cells, H1299, NL9980, and L9981 cells were treated with 180 μM Tα1 for 48 h. Western blotting and qRT-PCR showed that the mRNA and protein levels of MMP2 were decreased significantly (Figure 5A and B), along with the inhibited activation (Figure 5A, C, and D) and the reduced translocation (Figure 5E) of STAT3. Subsequently, STAT3 activator colivelin was applied combining with or without Tα1. Colivelin-induced activation of STAT3 was accompanied by the elevated MMP2 expression (Figure 5F). Moreover, the MMP2 suppression caused by Tα1 exposure could partly be reversed by colivelin (Figure 5F), and similar results were observed in the phenotype change in migration (Figure 5G). Overall, in PD-L1 high-expressing NSCLC cells, the STAT3–MMP2 pathway is the target, whereby Tα1 suppresses migration and invasion.
PD-L1 low-expressing NSCLC cells present a significantly lower expression of MMP2, which could not be suppressed by Tα1
To investigate why Tα1 could not suppress the migration and invasion of PD-L1 low-expressing NSCLC cells significantly, A549 and SPC-A-1 were exposed to 180 μM Tα1 for 48 h. However, the mRNA and protein levels of MMP2 (Figure 5A and B), as well as the activation of STAT3 (Figure 5A), were not inhibited by Tα1. Besides, the expression of STAT3, MMP2, and PD-L1 was assessed in H1299, L9981, A549, and SPC-A-1 cells with or without knocking down of PD-L1. A significantly lower expression of p-STAT3 and MMP2 was positively correlated with the lower PD-L1 expression in A549 and SPC-A-1 (Figure 5H). Moreover, knocking down of PD-L1 led to the decreased protein levels of p-STAT3 and MMP2 (Figure 5H). Similar correlation was demonstrated in mRNA levels between MMP2 and PD-L1 (Figure 5I). LY294002 has been reported to downregulate the PD-L1 expression in various NSCLC cells,19,20 and the PD-L1 protein in L9981 and A549 could be downregulated by LY294002 effectively, which was also accompanied by the decreased protein level of MMP2 (Figure 5J). Overall, both intrinsic and induced low expression of PD-L1 are associated with the low expression of p-STAT3 and MMP2, indicating that Tα1 only effects on STAT3–MMP2 pathway with high levels of molecule expression.
Discussion
Tα1 is one of the most commonly used immunomodulators for metastatic NSCLC patients in many countries.13,21 In this study, we investigated the direct effect of Tα1 on cell metastasis of NSCLC in vitro and in vivo and explored the signaling changes associated. We found that Tα1 could significantly suppress the migration and invasion in PD-L1 high-expressing cells. On the contrary, it did not function as metastatic suppressor in either PD-L1 low-expressing cells or PD-L1-silenced NSCLC cells. Furthermore, this suppressive effect in PD-L1 high-expressing NSCLC cells was demonstrated to be associated with the downregulation of STAT3–MMP2 signaling. Schematic model proposed in Figure 6 summarized the regulating process.
PD-L1 is a coinhibitory molecule expressed mainly on macrophages, dendritic cells, activated T and B cells, and tumor cells.22 The PD-L1 expressing on cancer cells, when binding to its receptor programmed cell death 1 (PD-1) expressing on activated T cells, can impair the anticancer function of T cells.22,23 High expression of PD-L1 in tumor tissues has been reported as a poor prognostic biomarker in NSCLC patients,24–26 and quite a few evidence showed a positive association between PD-L1 expression and metastatic ability of NSCLC cells.27–30 Taking epithelial–mesenchymal transition (EMT) for example, data in Figure S2 revealed a protein profile facilitating EMT in PD-L1 high-expressing NSCLC cell lines, while Asgarova et al28 showed a reversible upregulation of PD-L1 during cytokine-driven EMT in NSCLC cells. In the present study, the lower expression of PD-L1 not only meant a significantly weak inhibitory effect of Tα1 on metastatic phenotypes and MMP2 expression but also signified a consistent lower protein expression on the STAT3–MMP2 pathway. These results suggest the association between PD-L1 and the metastasis-associated molecule MMP2 in NSCLC cells. Indeed, recent study showed that PD-L1 blockade decreased both the mRNA and protein levels of MMP2 in tumor tissues,31 which is consistent with our results. In contrast, the positive correlation between PD-L1 and MMP2 may also explain, in turn, why Tα1 could not suppress the MMP2 expression and the metastatic phenotypes in PD-L1 low-expressing cells. One most probable explanation is that the protein level of MMP2 in PD-L1 low-expressing cells is too low to be suppressed significantly by Tα1. Another possibility is that PD-L1 is exactly the intermediary molecule between Tα1 and STAT3–MMP2 pathway. Therefore, adequate amounts of PD-L1 in PD-L1 high-expressing cells can effectively respond to Tα1, though it was not decreased by Tα1.
Discontinuously expressed in normal tissues, MMPs are often highly expressed on malignant cells, degrading extracellular matrix protein and ultimately invading other tissues.32,33 MMP2 is one of the key enzymes in the activation of other MMPs,34 and the overexpression of which contributes to the metastasis and angiogenesis of lung cancer.35,36 Several immunomodulators, including triticumoside, mushroom-derived polysaccharide, and timosaponin AIII, have been found to suppress MMP2 expression, thereby inhibiting the invasion and migration of NSCLC cells.37–39 As for Tα1, little is known about its effect on MMPs. We investigated the antimetastasis effect of Tα1, observing a significant downregulation of MMP2, but not MMP9, on both mRNA and protein levels. To explore whether the simultaneously observed inactivation of STAT3 had influenced the transcription of MMP2, H1299, NL9980, and L9981 were co-treated with STAT3 activator colivelin and Tα1. The Tα1-induced suppression of both MMP2 protein and cell migration phenotype was partly reversed by STAT3 activator colivelin, suggesting an association between the inhibited STAT3 and the decreased MMP2. Several studies have showed that STAT3–MMPs’ regulation plays important roles in the antimetastasis effect of many biological extracts such as silibinin and plumbagin.40,41 Our results support these findings and further define the MMP2, rather than MMP9, as the target of STAT3 in the Tα1 treatment of NSCLC cells.
The involved molecules of this study enlighten the following three combination modes for advanced NSCLC: 1) Tα1 plus STAT3 inhibitors, a novel target therapy on the way from trial to clinic; 2) Tα1 plus pembrolizumab, an immune checkpoint (PD-1/PD-L1) inhibitor for the first- and subsequent-line treatment of NSCLC; and 3) Tα1 plus bisphosphonate, a classic inhibitor of MMPs for the clinical management of bone metastases. Among them, the combination of Tα1 with pembrolizumab is promising. A PD-L1 expression level of ≥50% is an indicator for first-line pembrolizumab therapy in advanced NSCLC patients,42 while the antimetastasis role of Tα1 is specifically effective in PD-L1 high-expressing NSCLC cells. Furthermore, as a safe immunopotentiator, Tα1 may augment anticancer efficiency of the immune checkpoint inhibitor in vivo. The combination of the two drugs is expected in animal experiments and future clinical trials.
Conclusion
Tα1 can significantly suppress cell migration and invasion in PD-L1 high-expressing cells rather than in low-expressing NSCLC cells in vitro and in vivo. This suppression was associated with the inhibition of STAT3–MMP2 signaling. These results not only depict a more comprehensive molecular diagram of NSCLC cells treated with Tα1 but also enlighten the combination of Tα1 and immune checkpoint inhibitors in the future trials.
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (No 81572288 to Qinghua Zhou and No 81302002 to Xuebing Li), the Key Project of International Cooperation of Science and Technology Innovation Between Governments, the National Key Research and Development Plan of China (No 2016YEE0103400 to Qinghua Zhou), the Tianjin Natural Science Foundation (No 14JCQNJC12300 to Xuebing Li), and the “New Century” Talent Training Project of Tianjin Medical University General Hospital (2014 to Xuebing Li). Thymosin α1 used in this study is produced by SciClone Pharmaceuticals, Inc. (NASDAQ:SCLN). The authors thank the members of Tianjin Lung Cancer Institute for helpful technical support.
Disclosure
The authors report no conflicts of interest in this work.
References
GLOBOCAN [webpage on the Internet]. Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012; 2012. Available from: http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx. Accessed September 28, 2018. | ||
Reck M, Popat S, Reinmuth N, et al. Metastatic non-small-cell lung cancer (NSCLC): ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2014;25(Suppl 3):iii27–iii39. | ||
Bai X, Meng L, Sun H, Li Z, Zhang X, Hua S. MicroRNA-196b inhibits cell growth and metastasis of lung cancer cells by targeting Runx2. Cell Physiol Biochem. 2017;43(2):757–767. | ||
Dela Cruz CS, Tanoue LT, Matthay RA. Lung cancer: epidemiology, etiology, and prevention. Clin Chest Med. 2011;32(4):605–644. | ||
Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66(2):115–132. | ||
Pierluigi B, D’Angelo C, Fallarino F, et al. Thymosin alpha1: the regulator of regulators? Ann N Y Acad Sci. 2010;1194:1–5. | ||
Goldstein AL, Slater FD, White A, Preparation WA. Preparation, assay, and partial purification of a thymic lymphocytopoietic factor (thymosin). Proc Natl Acad Sci U S A. 1966;56(3):1010–1017. | ||
Hooper JA, Mcdaniel MC, Thurman GB, Cohen GH, Schulof RS, Goldstein AL. Purification and properties of bovine thymosin. Ann N Y Acad Sci. 1975;249:125–144. | ||
Fan YZ, Chang H, Yu Y, Liu J, Wang R. Thymosin alpha1 suppresses proliferation and induces apoptosis in human leukemia cell lines. Peptides. 2006;27(9):2165–2173. | ||
Li J, Liu CH, Wang FS. Thymosin alpha 1: biological activities, applications and genetic engineering production. Peptides. 2010;31(11):2151–2158. | ||
Moretti S, Oikonomou V, Garaci E, Romani L. Thymosin α1: burying secrets in the thymus. Expert Opin Biol Ther. 2015;15(Suppl 1):51–58. | ||
Jiang J, Wang X, Tian J, Li L, Lin Q. Thymosin plus cisplatin with vinorelbine or gemcitabine for non-small cell lung cancer: a systematic review and meta-analysis of randomized controlled trials. Thorac Cancer. 2011;2(4):213–220. | ||
Cheepsattayakorn A, Cheepsattayakorn R. Lung cancer chemotherapy, new treatment and related patents. Recent Pat Anticancer Drug Discov. 2014;9(3):372–381. | ||
Moody TW, Fagarasan M, Zia F, Cesnjaj M, Goldstein AL. Thymosin alpha 1 down-regulates the growth of human non-small cell lung cancer cells in vitro and in vivo. Cancer Res. 1993;53(21):5214–5218. | ||
Guo Y, Chang H, Li J, et al. Thymosin alpha 1 suppresses proliferation and induces apoptosis in breast cancer cells through PTEN-mediated inhibition of PI3K/Akt/mTOR signaling pathway. Apoptosis. 2015;20(8):1109–1121. | ||
Moody TW. Thymosin alpha1 as a chemopreventive agent in lung and breast cancer. Ann N Y Acad Sci. 2007;1112:297–304. | ||
Zhou Q, Wang Y, Che G, et al. Establishment and their biological characteristics of clonal cell subpopulations (NL9980 and L9981) from a human lung large cell carcinoma cell line (WCQH-9801). Zhongguo Fei Ai Za Zhi. 2003;6(6):464–468. | ||
D L, X X. Effect of thymosin a1 on the apoptosis of lung cancer cell A549. Chin J Clin Pharmacol. 2016;32(19):1776–1779. | ||
Abdelhamed S, Ogura K, Yokoyama S, Saiki I, Hayakawa Y. AKT-STAT3 pathway as a downstream target of EGFR signaling to regulate PD-L1 expression on NSCLC cells. J Cancer. 2016;7(12):1579–1586. | ||
Ota K, Azuma K, Kawahara A, et al. Induction of PD-L1 expression by the EML4-ALK oncoprotein and downstream signaling pathways in non-small cell lung cancer. Clin Cancer Res. 2015;21(17):4014–4021. | ||
Garaci E, Pica F, Matteucci C. Historical review on thymosin α1 in oncology: preclinical and clinical experiences. Expert Opin Biol Ther. 2015;15(Suppl 1):31–39. | ||
Lin K, Cheng J, Yang T, Li Y, Zhu B. EGFR-TKI down-regulates PD-L1 in EGFR mutant NSCLC through inhibiting NF-κB. Biochem Biophys Res Commun. 2015;463(1–2):95–101. | ||
Soria JC, Marabelle A, Brahmer JR, Gettinger S. Immune checkpoint modulation for non-small cell lung cancer. Clin Cancer Res. 2015;21(10):2256–2262. | ||
Mu CY, Huang JA, Chen Y, Chen C, Zhang XG. High expression of PD-L1 in lung cancer may contribute to poor prognosis and tumor cells immune escape through suppressing tumor infiltrating dendritic cells maturation. Med Oncol. 2011;28(3):682–688. | ||
Okuma Y, Hishima T, Kashima J, Homma S. High PD-L1 expression indicates poor prognosis of HIV-infected patients with non-small cell lung cancer. Cancer Immunol Immunother. 2018;67(3):495–505. | ||
Takamori S, Toyokawa G, Okamoto I, et al. Clinical significance of PD-L1 expression in brain metastases from non-small cell lung cancer. Anticancer Res. 2018;38(1):553–557. | ||
Chen L, Gibbons DL, Goswami S, et al. Metastasis is regulated via microRNA-200/ZEB1 axis control of tumour cell PD-L1 expression and intratumoral immunosuppression. Nat Commun. 2014;5:5241. | ||
Asgarova A, Asgarov K, Godet Y, et al. PD-L1 expression is regulated by both DNA methylation and NF-kB during EMT signaling in non-small cell lung carcinoma. Oncoimmunology. 2018;7(5):e1423170. | ||
Kim S, Koh J, Kim MY, et al. PD-L1 expression is associated with epithelial-to-mesenchymal transition in adenocarcinoma of the lung. Hum Pathol. 2016;58:7–14. | ||
Zhang J, Gao J, Li Y, et al. Circulating PD-L1 in NSCLC patients and the correlation between the level of PD-L1 expression and the clinical characteristics. Thorac Cancer. 2015;6(4):534–538. | ||
Zhao L, Li C, Liu F, et al. A blockade of PD-L1 produced antitumor and antimetastatic effects in an orthotopic mouse pancreatic cancer model via the PI3K/Akt/mTOR signaling pathway. Onco Targets Ther. 2017;10:2115–2126. | ||
Lee MM, Chen YY, Liu PY, Hsu S, Sheu MJ. Pipoxolan inhibits CL1-5 lung cancer cells migration and invasion through inhibition of MMP-9 and MMP-2. Chem Biol Interact. 2015;236:19–30. | ||
Chambers AF, Matrisian LM. Changing views of the role of matrix metalloproteinases in metastasis. J Natl Cancer Inst. 1997;89(17):1260–1270. | ||
Kleiner DE, Stetler-Stevenson WG. Matrix metalloproteinases and metastasis. Cancer Chemother Pharmacol. 1999;43(Suppl):S42–S51. | ||
Tokuraku M, Sato H, Murakami S, Okada Y, Watanabe Y, Seiki M. Activation of the precursor of gelatinase A/72 kDa type IV collagenase/MMP-2 in lung carcinomas correlates with the expression of membrane-type matrix metalloproteinase (MT-MMP) and with lymph node metastasis. Int J Cancer. 1995;64(5):355–359. | ||
Rojiani MV, Alidina J, Esposito N, Rojiani AM. Expression of MMP-2 correlates with increased angiogenesis in CNS metastasis of lung carcinoma. Int J Clin Exp Pathol. 2010;3(8):775–781. | ||
Poudel B, Ki HH, Luyen BT, Lee YM, Kim YH, Kim DK. Triticumoside induces apoptosis via caspase-dependent mitochondrial pathway and inhibits migration through downregulation of MMP2/9 in human lung cancer cells. Acta Biochim Biophys Sin. 2016;48(2):153–160. | ||
Jung O, Lee J, Lee YJ, et al. Timosaponin AIII inhibits migration and invasion of A549 human non-small-cell lung cancer cells via attenuations of MMP-2 and MMP-9 by inhibitions of ERK1/2, Src/FAK and β-catenin signaling pathways. Bioorg Med Chem Lett. 2016;26(16):3963–3967. | ||
Lee KR, Lee JS, Song JE, Ha SJ, Hong EK. Inonotus obliquus-derived polysaccharide inhibits the migration and invasion of human non-small cell lung carcinoma cells via suppression of MMP-2 and MMP-9. Int J Oncol. 2014;45(6):2533–2540. | ||
Byun HJ, Darvin P, Kang DY, et al. Silibinin downregulates MMP2 expression via Jak2/STAT3 pathway and inhibits the migration and invasive potential in MDA-MB-231 cells. Oncol Rep. 2017;37(6):3270–3278. | ||
Yan W, Tu B, Liu YY, et al. Suppressive effects of plumbagin on invasion and migration of breast cancer cells via the inhibition of STAT3 signaling and down-regulation of inflammatory cytokine expressions. Bone Res. 2013;1(4):362–370. | ||
Ettinger DS, Wood DE, Aisner DL, et al. Non-small cell lung cancer, version 5.2017, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2017;15(4):504–535. |
Supplementary materials
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.