Back to Journals » Infection and Drug Resistance » Volume 13
Therapeutic Drug Monitoring and Nephrotoxicity of Teicoplanin Therapy in Chinese Children: A Retrospective Study
Authors Sun D, Zhang T, Mi J, Dong Y, Liu Y, Zhang Y, Zhang D, Wang T, Cheng H, Dong Y
Received 23 July 2020
Accepted for publication 19 September 2020
Published 12 November 2020 Volume 2020:13 Pages 4105—4113
DOI https://doi.org/10.2147/IDR.S272982
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Suresh Antony
Dan Sun,1,* Tao Zhang,1,* Jie Mi,2 Yuzhu Dong,3 Yang Liu,1 Ying Zhang,1 Di Zhang,1 Taotao Wang,1 Hua Cheng,2 Yalin Dong1
1Department of Pharmacy, The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, Shaanxi 710061, People’s Republic of China; 2Department of Pharmacy, Xi’an Children’s Hospital, Xi’an, Shaanxi 710003, People’s Republic of China; 3Department of Pharmacy, The Third Affiliated Hospital of Chongqing Medical University, Chongqing 401120, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Yalin Dong
Department of Pharmacy, The First Affiliated Hospital of Xi’an Jiaotong University, Xi’an, Shaanxi 710061, People’s Republic of China
Tel/ Fax +86-29-85323241
Email [email protected]
Hua Cheng
Department of Pharmacy, Xi’an Children’s Hospital, Xi’an, Shaanxi 710003, People’s Republic of China
Tel/ Fax +86-29-87692090
Email [email protected]
Purpose: This study aims to 1) describe the distribution characteristics of teicoplanin trough concentration (Cmin) and explore the related influencing factors and 2) evaluate the nephrotoxicity of teicoplanin in children.
Patients and Methods: A cohort of children who were treated with teicoplanin intravenously were included in this retrospective study. Regression analysis was performed to explore the factors associated with the fluctuations of teicoplanin Cmin and the development of nephrotoxicity. Classification and regression tree analysis was used to identify the population at high risk for teicoplanin nephrotoxicity.
Results: A total of 269 plasma samples from 186 children were collected. Underexposure (Cmin < 10 mg/L) was documented in 52.7% of cases. The Cmin/dose after administering the loading dose was strongly associated with age (P = 0.008), weight (P = 0.039), and serum creatinine (P = 0.022). The Cmin/dose after administering the maintenance dose was strongly associated with gender (P = 0.014) and serum creatinine (P = 0.006). Cmin (P = 0.012) and the concomitant treatment with amphotericin B (P = 0.001) were the independent risk factors for teicoplanin-related nephrotoxicity. Children who were concomitantly treated by amphotericin B with teicoplanin Cmin > 9.81 mg/L or patients with teicoplanin Cmin > 21.94 mg/L were at high risk for nephrotoxicity.
Conclusion: The fluctuations of teicoplanin Cmin could be affected by age, weight, gender, and serum creatinine. Cmin and concomitant treatment with amphotericin B were the independent risk factors for nephrotoxicity. We suggested that the therapeutic drug monitoring of teicoplanin should be performed in children.
Keywords: teicoplanin, trough concentration, children, nephrotoxicity, factor
Introduction
Gram-positive bacterial infections remain a significant health concern in pediatric patients.1,2 Teicoplanin is a glycopeptide antibiotic exhibiting high antibacterial activities against the majority of gram-positive bacteria. It is widely used for the treatment of skin and soft tissue infections, bone and joint infections, pneumonia, urinary tract infections and infective endocarditis in pediatrics worldwide,3 as the superior tissue penetration, slower rate of elimination and fewer adverse reactions compared with vancomycin.4
Previous studies showed that teicoplanin trough concentration (Cmin) was significantly associated with improved clinical response, and a Cmin of > 60 mg/L would increase the likelihood of nephrotoxicity.5–7 Generally, it is recommended that, for most gram-positive infections, teicoplanin Cmin should be maintained at least 10 mg/L (measured by high performance liquid chromatography, HPLC) or 15 mg/L (measured by fluorescence polarization immunoassay, FPIA) in clinical practice.8,9 In addition, large inter-individual and intra-individual variation of teicoplanin Cmin have been observed in both adult and pediatric patients,10–16 and it has been reported that drug underexposure was documented in more than half of the cases.3 Weight, serum creatinine and other clinical factors were found to be significantly correlated with the Cmin of teicoplanin.3,17–19
Clinical data focused on therapeutic drug monitoring (TDM) of teicoplanin in pediatric patients, especially in children, is very limited. And factors influencing teicoplanin Cmin have not been fully elucidated. Yamada et al evaluated the Cmin of teicoplanin after the recommended loading dose in 36 children and found that age and renal function were significantly associated with teicoplanin Cmin.17 Another large-scale retrospective TDM study (280 pediatric patients) conducted by Strenger et al indicated that age and gender contributed to the variation of teicoplanin concentration.19 However, more researches are needed to further explore the influencing factors of teicoplanin Cmin due to the limited data.
Besides, compared to vancomycin, teicoplanin lead to a lower risk of nephrotoxicity.20,21 However, nephrotoxicity is associated with greater hospital costs and longer length of stay in non-critically ill children.22 To date, only Yamada et al reported that the incidence of nephrotoxicity in children was 2.3%, and no significant difference in renal impairment was found between the groups with Cmin > 20 mg/L and Cmin < 20 mg/L (measured by FPIA).17 However, the risk factors of teicoplanin-related nephrotoxicity have not been fully explored in children.
In this regard, this study aimed to conduct a retrospective study to 1) describe the distribution characteristics of teicoplanin Cmin and explore the related influencing factors associated with Cmin; 2) analyze and evaluate the incidence of teicoplanin-related nephrotoxicity and its risk factors in children.
Patients and Methods
Study Population
In this retrospective study, we included a cohort of children who were treated with teicoplanin intravenously at the First Affiliated Hospital of Xi’an Jiaotong University and Xi’an Children’s Hospital between March 2017 and December 2018. Inclusion criteria were: 1) patients aged from 2 months to 18 years old; 2) patients who were clinically diagnosed or suspected with gram-positive bacterial infections, such as respiratory infections, bloodstream infections, bone and joint infections, skin and soft tissue infections and so on, and then were treated with teicoplanin (for children aged 2 months to 12 years, loading dose: one single dose of 10 mg/kg body weight administered intravenously every 12 hours, repeated 3 times; maintenance dose: one single dose of 6–10 mg/kg body weight administered intravenously once a day. For children aged >12 years, loading dose: 6 mg/kg body weight every 12 hours for 3 intravenous or intramuscular administrations; maintenance dose: 6 mg/kg body weight intravenous or intramuscular once a day). The specific prescription of each patient was determined by physicians; 3) patients who underwent TDM of teicoplanin. Exclusion criteria were: 1) patients who underwent continuous renal replacement therapy or peritoneal dialysis; 2) patients who received teicoplanin for less than 48 hours. This study protocol was approved by the Ethics Committee of the two hospitals.
Data Collection
The patient’s clinical data were extracted through the hospital’s electronic medical record system and the nursing system. The main information was as follows: 1) demographic data, such as gender, age, and weight; 2) underlying diseases and diagnosis of infection; 3) dosage regimen of teicoplanin and the concomitant medication (defined as the other drugs were given at or almost at the same day with teicoplanin before performing TDM for more than one day); 4) laboratory test results, such as creatinine, transaminase (aspartate aminotransferase and alanine transaminase) and platelet count; 5) adverse reactions information, such as nephrotoxicity.
Blood Sample Collection and Concentration Measurement
At least 3 days after the first dose of teicoplanin, plasma samples were taken from patients 30 minutes before the next dose. A validated HPLC method established by our research group was used to quantify the plasma concentrations of teicoplanin. The linear range was 5.63–125.00 mg/L (r = 0.9995). The minimum limit of quantitation was 5.63 mg/L. The average relative standard deviations of intra-day and inter-day were 3.2% and 6.8%, respectively, and the mean relative error of intra-day and inter-day were 0.12% and 0.10%, respectively.23
Definitions
Teicoplanin Cmin were divided into two groups: Cmin in the first group were the plasma concentrations measured on day 3 or 4 after the first dose of teicoplanin. Cmin in another group were the plasma concentrations measured at steady state (the teicoplanin dosage was not changed for ≥ 72 h).24 Underexposure was defined as the Cmin of teicoplanin was less than 10 mg/L.
To explore factors affecting the fluctuation of teicoplanin Cmin, we performed an analysis to investigate the relationship between different factors and the value of Cmin/Dose (C/D). C/D were divided into two groups: one was C3/4min/DCLD (the ratio between Cmin on day 3 or day 4 after teicoplanin administration and the cumulative loading dose), and the other was CSSmin/DMD (the ratio between the teicoplanin Cmin at steady state and the maintenance dose). Nephrotoxicity was defined as a serum creatinine (SCr) increase at least 1.5 times that of the baseline.25
Statistical Analysis
Statistical analyses were performed using SPSS 19.0. The mean ± standard deviation (SD) or median (interquartile range, IQR) was used to describe continuous variables depending on the normality of distribution, whereas percent or count was used to describe categorical variables. Univariate and multivariate linear regression analysis was applied to determine potential factors affecting the teicoplanin C/D. Univariate and multivariate logistic regression analyses were applied to explore the potential risk factors for nephrotoxicity. Variables with P < 0.1 in the univariate analysis were combined in the multivariate analysis. Logistic regression and classification and regression tree (CART) analysis were performed to identify the patient population with a greater or less likelihood of developing nephrotoxicity. In this study, variables with P < 0.1 in a multivariate logistic regression analysis were included in the CART analysis.
Results
Baseline Patient Characteristics
One hundred and eighty-six children were included in this study. Patients were typically male (56.5%), with a median age of 3.82 years (IQR 1.53–6.27 years). The median weight was 15.3 kg (IQR 11.0–21.0 kg). The main reason for teicoplanin therapy was respiratory infections (58.1%) and malignancy was the most common comorbid condition (46.2%). Only 25.8% of patients were proven to be infected with gram-positive bacterial. Median albumin and SCr were 35.0 g/L and 26 µmol/L, respectively (Table 1).
 |
Table 1 Patients’ Characteristics |
Distribution of Teicoplanin Trough Concentrations
The distribution of teicoplanin Cmin was shown in Figure 1. A total of 269 teicoplanin Cmin were included in this study. The median of Cmin was 9.49 mg/L (IQR 5.97–15.35 mg/L) and 52.7% of Cmin is less than 10 mg/L. Among the 269 teicoplanin Cmin, 56 Cmin were obtained on day 3 or 4 after the initial teicoplanin administration, the median was 8.43 mg/L (IQR 6.41–15.02 mg/L) (Figure 1, panel A). The median of the remaining 213 teicoplanin Cmin obtained at steady state was 9.77 mg/L (IQR 5.89–15.60 mg/L) (Figure 1, panel B). Compared with the steady state Cmin, those obtained on day 3 or day 4 were more likely to be < 10 mg/L.
Factors Influencing Teicoplanin C/D
Factors Influencing Teicoplanin C3/4min/DCLD
As shown in Table 2, gender, age, weight, concomitant use of carbapenems, and SCr were allowed to be included in the multivariate analyses due to those factors had the P value < 0.1. Finally, it was found that teicoplanin C3/4min/DCLD was significantly affected by age (P = 0.008), weight (P = 0.039), and SCr (P = 0.022).
 |
Table 2 Univariate and Multivariate Analyses of the Variables Tested for Potential Association with C3/4min/DCLD |
Factors Influencing Teicoplanin CSSmin/DMD
Accordingly, Table 3 shows the factors that influencing the teicoplanin CSSmin/DMD. Although univariate analyses indicated that gender, weight, and SCr had the P value < 0.1, no significant correlation was found between weight and teicoplanin CSSmin/DMD in the multivariate analyses. Gender and SCr were found to be independently associated with teicoplanin CSSmin/DMD (P = 0.014, P = 0.006, respectively).
 |
Table 3 Univariate and Multivariate Analyses of the Variables Tested for Potential Association with CSSmin/DMD |
Nephrotoxicity Analysis
One hundred and eighty-two children were eligible for nephrotoxicity analysis. Nine of one hundred and eighty-two (4.9%) patients occurred nephrotoxicity. As shown in Table 4, both univariable and multivariable logistic analysis indicated that teicoplanin Cmin and the concomitant use of amphotericin B were the risk factors for the incidence of nephrotoxicity related to teicoplanin (P = 0.012, P = 0.001, respectively). Further CART model showed that children with concomitant treatment with amphotericin B had teicoplanin Cmin > 9.81 mg/L or patients with teicoplanin Cmin > 21.94 mg/L were at high risk for nephrotoxicity (Figure 2). Inversely, patients who were concomitantly treated by amphotericin B with teicoplanin Cmin < 9.81 mg/L or those with teicoplanin Cmin < 21.94 mg/L were at low risk of nephrotoxicity.
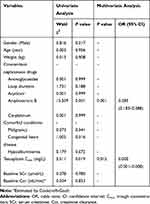 |
Table 4 Univariable and Multivariable Analysis of Risk Factors for Nephrotoxicity in Patients Receiving Teicoplanin |
Discussion
In this retrospective study, we described the distribution characteristics of teicoplanin Cmin and investigated the potential factors affecting the concentration of teicoplanin based on 269 TDM data from 186 patients. In addition, we explored the risk factors related to the nephrotoxicity of teicoplanin and identified patients with high-risk of nephrotoxicity.
Teicoplanin Cmin of 9.49 mg/L (IQR 5.97, 15.35 mg/L) was observed in this study. When Cmin > 10 mg/L was defined as the therapeutic target of teicoplanin, more than half of the teicoplanin Cmin (52.7%) did not achieve the therapeutic target, which was similar to previous studies.16,26 Zhao et al reported the teicoplanin Cmin was 11.8 mg/L (IQR 3.0, 49.6 mg/L), and 48% of children had sub-therapeutic steady-state Cmin.16 Yamada et al reported that the median Cmin of teicoplanin was 16.3 (3.6, 25.9) mg/L in children (measured by FPIA), and 46.2% of patients did not achieve a trough value ≥ 15 mg/L was in the recommended dose regimen group.17 Given those findings, it seems that teicoplanin Cmin existed inadequate exposure in most patients. We suggest that the dose increase of teicoplanin and the exploration of Cmin-related influencing factors should be conducted in children to achieve better clinical responses.
In this study, age, weight, and SCr were found to be the factors that exerted a significant effect on teicoplanin C3/4min/DCLD. Our results were partially similar with a previous study with a relatively small sample size (36 children) which reported that teicoplanin Cmin would be influenced by age, SCr and estimated glomerular filtration when giving a fixed-dose.17 Since teicoplanin is mainly excreted by the kidneys,27 impaired renal function usually causes a decrease of teicoplanin clearance.28 In addition, drug clearance and volume of distribution in children also changed with the increase of age and weight, further affecting the drug exposure. Strenger et al found that compared with school-age children, young children had significantly lower initial teicoplanin Cmin.19 Besides, several studies have also indicated that weight could significantly influence the pharmacokinetics of teicoplanin in children.3,16,29 Therefore, it is necessary to consider age, weight and serum creatinine to achieve the therapeutic target of teicoplanin in children as soon as possible.
For the CSSmin/DMD, in addition to SCr, gender was also found to be significantly related to teicoplanin CSSmin/DMD. Similar to the recent results concluded from Cai et al, which demonstrated that there were gender-related differences in teicoplanin Cmin.29 Another study found that adolescent girls had significantly higher initial and follow-up teicoplanin Cmin than adolescent boys, and the authors suggested that gender-related differences in protein binding, tissue distribution or renal excretion of teicoplanin may explain this difference.19 More prospective studies are expected in the future to explore the causes of this phenomenon.
In the field of TDM, studies rarely focused on the nephrotoxicity of teicoplanin in children. The incidence of nephrotoxicity was 4.9% according to the current study. A retrospective study found that the overall incidence of nephrotoxicity in children was 2.3%.17 We found that teicoplanin Cmin and concomitant treatment with amphotericin B were independent risk factors for nephrotoxicity of teicoplanin in children. Moreover, there was an increased risk of nephrotoxicity when the Cmin of teicoplanin exceed 21.94 mg/L in children or the Cmin exceed 9.81 mg/L in children with concomitant use of amphotericin B. To our knowledge, this is the first study to report the risk factors of teicoplanin-related nephrotoxicity and the high-risk population in children. Previous studies reported that a Cmin of > 60 mg/L increased the likelihood of nephrotoxicity of teicoplanin.6 Similarly, Wilson reported that adult patients with teicoplanin Cmin > 60 mg/L had a higher incidence of nephrotoxicity compared to patients with teicoplanin Cmin of 20–40 mg/L.30 However, another study drew a contradictory conclusion that there was no significant correlation between the Cmin and the occurrence of nephrotoxicity in adults.5 The most common adverse reaction of amphotericin B is nephrotoxicity. A previous study showed that amphotericin B combined with vancomycin was associated with impaired renal function.31 In this study, 4 of 9 patients developing nephrotoxicity were received concomitant treatment with amphotericin B. The nephrotoxicity threshold of teicoplanin was decreased from 21.94 mg/L to 9.81 mg/L when combining amphotericin B and teicoplanin, it may be caused by the synergy between the two drugs. This result suggested that the concomitant use of amphotericin B could aggravate the nephrotoxicity possibility of teicoplanin. Therefore, more attention should be paid to the children who received concomitant treatment with amphotericin B and teicoplanin in the future, even when the Cmin does not reach the therapeutic target.
There are several limitations in our study. Firstly, this study was a retrospective study, neither the number of TDM samples nor the timing of TDM for each patient was consistent. Secondly, the relationship between drug exposure and clinical efficacy was not explored because there are few microbiologically documented gram-positive bacterial infections in this study. Larger-scale, multicenter and prospective studies are expected to further validate the results of teicoplanin TDM in children.
Conclusion
This is a retrospective study of teicoplanin in a larger sample size of children patients. The current dose regimen of teicoplanin may result in inadequate drug exposure in children. Age, weight, gender and SCr are important factors that can significantly influence the fluctuation of teicoplanin Cmin. Teicoplanin Cmin and concomitant treatment with amphotericin B were independent risk factors for nephrotoxicity. It is recommended to perform TDM in children to ensure the efficacy and safety of teicoplanin.
Ethics Approval and Informed Consent
This study was approved by the Ethics Committee of the First Affiliated Hospital of Xi’an Jiaotong University and the Ethics Committee of Xi’an Children’s Hospital, respectively. Waiving of informed consent was given due to the retrospective, non-interventional study design. All patient data were collected anonymously. This study was performed in accordance with the Declaration of Helsinki. Patients were ensured about the confidentiality of their information.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was supported by the Special Fund for Drug Development and Rational Application of Chinese Pharmacological Society (No. CTDM (XM-2017-01-001-14)) and the Key Research and Development Program in Shaanxi Province of China (No. 2019ZDLSF01-05) for supporting this study.
Disclosure
Dan Sun and Tao Zhang are co-first authors for this study. The authors declare that they have no conflicts of interest.
References
1. Venkatesh MP, Placencia F, Weisman LE. Coagulase-negative staphylococcal infections in the neonate and child: an update. Semin Pediatr Infect Dis. 2006;17(3):120–127. doi:10.1053/j.spid.2006.06.005
2. Maldonado ME, Acuña M, Álvarez AM, et al. Microorganismos aislados de hemocultivos en niños con cáncer y neutropenia febril de alto riesgo en cinco hospitales de Santiago, Chile, período 2012-2015. Revista Chilena de Infectología. 2018;35(2):140–146. doi:10.4067/s0716-10182018000200140
3. Ramos-Martin V, Paulus S, Siner S, et al. Population pharmacokinetics of teicoplanin in children. Antimicrob Agents Chemother. 2014;58(11):6920–6927. doi:10.1128/AAC.03685-14
4. Spencer CM, Bryson HM. Teicoplanin. A pharmacoeconomic evaluation of its use in the treatment of gram-positive infections. Pharmacoeconomics. 1995;7(4):357–374. doi:10.2165/00019053-199507040-00009
5. Zhou LJ, Gao YQ, Cao W, et al. Retrospective analysis of relationships among the dose regimen, trough concentration, efficacy, and safety of teicoplanin in Chinese patients with moderate–severe Gram-positive infections. Infect Drug Resist. 2018;11:29–36. doi:10.2147/IDR.S146961
6. Abdul-Aziz MH, Alffenaar JC, Bassetti M, et al. Antimicrobial therapeutic drug monitoring in critically ill adult patients: a position paper. Intensive Care Med. 2020;46:1127–1153.
7. Wang TT, Li N, Hu SS, et al. Factors on trough teicoplanin levels, associations between levels, efficacy and safety in patients with gram-positive infections. Int J Clin Pharmacol Ther. 2015;53(05):356–362. doi:10.5414/CP202247
8. Targocid EMC 200mg powder for solution for injection/infusion or oral solution. Available from: https://www.medicines.org.uk/emc/product/2926/smpc.
9. Yuan DX. Teicoplanin for infusion. Available from: http://drugs.dxy.cn/drug/82481/detail.htm#4.
10. Kim S-H, Kang C-I, Huh K, et al. Evaluating the optimal dose of teicoplanin with therapeutic drug monitoring: not too high for adverse event, not too low for treatment efficacy. Eur J Clin Microbiol Infect Dis. 2019;38(11):2113–2120. doi:10.1007/s10096-019-03652-6
11. Barco S, Mesini A, Barbagallo L, et al. A liquid chromatography-tandem mass spectrometry platform for the routine therapeutic drug monitoring of 14 antibiotics: application to critically ill pediatric patients. J Pharm Biomed Anal. 2020;186:113273. doi:10.1016/j.jpba.2020.113273
12. Yoshida T, Yoshida S, Okada H, et al. Risk factors for decreased teicoplanin trough concentrations during initial dosing in critically ill patients. Pharmazie. 2019;74:120–124.
13. Kato H, Hamada Y, Hagihara M, et al. Retrospective study of teicoplanin loading regimen that rapidly achieves target 15–30 μg/mL serum trough concentration. J Infect Chemother. 2016;22(5):308–313. doi:10.1016/j.jiac.2016.01.019
14. Byrne CJ, Egan S, Fennell JP, et al. Teicoplanin use in adult patients with haematological malignancy: exploring relationships between dose, trough concentrations, efficacy and nephrotoxicity. Int J Antimicrob Agents. 2015;46(4):406–412. doi:10.1016/j.ijantimicag.2015.05.019
15. Ramos-Martin V, Neely MN, McGowan P, et al. Population pharmacokinetics and pharmacodynamics of teicoplanin in neonates: making better use of C-reactive protein to deliver individualized therapy. J Antimicrob Chemother. 2016;71(11):3168–3178. doi:10.1093/jac/dkw295
16. Zhao W, Zhang D, Storme T, et al. Population pharmacokinetics and dosing optimization of teicoplanin in children with malignant haematological disease. Br J Clin Pharmacol. 2015;80(5):1197–1207. doi:10.1111/bcp.12710
17. Yamada T, Kubota T, Yonezawa M, et al. Evaluation of teicoplanin trough values after the recommended loading dose in children with associated safety analysis. Pediatr Infect Dis J. 2017;36(4):398–400. doi:10.1097/INF.0000000000001456
18. Yamada T, Kubota T, Nakamura M, et al. Evaluation of teicoplanin concentrations and safety analysis in neonates. Int J Antimicrob Agents. 2014;44(5):458–462. doi:10.1016/j.ijantimicag.2014.07.005
19. Strenger V, Hofer N, Rödl S, et al. Age- and gender-related differences in teicoplanin levels in paediatric patients. J Antimicrob Chemother. 2013;68:2318–2323.
20. Cavalcanti AB, Goncalves AR, Almeida CS, et al. Teicoplanin versus vancomycin for proven or suspected infection. Cochrane Database Syst Rev. 2010;6:CD007022.
21. Kato‐Hayashi H, Niwa T, Ohata K, et al. Comparative efficacy and safety of vancomycin versus teicoplanin in febrile neutropenic patients receiving hematopoietic stem cell transplantation. J Clin Pharm Ther. 2019;44(6):888–894. doi:10.1111/jcpt.13011
22. Moffett BS, Goldstein SL. Acute Kidney Injury and Increasing Nephrotoxic-Medication Exposure in Noncritically-Ill Children. Clin J Am Soc Nephrol. 2011;6(4):856–863. doi:10.2215/CJN.08110910
23. Hu S, Pang C, Dong H, et al. Determination of teicoplanin serum concentration in critically ill patients. Chine J Hospital Pharmacy. 2011;31:753–755.
24. Sánchez A, López-Herce J, Cueto E, et al. Teicoplanin pharmacokinetics in critically ill paediatric patients. J Antimicrob Chemother. 1999;44(3):407–409. doi:10.1093/jac/44.3.407
25. Kellum JA, Bellomo R, Ronco C. Definition and classification of acute kidney injury. Nephron Clin Pract. 2008;109(4):c182–187. doi:10.1159/000142926
26. Jung J, Lee K, Oh J, et al. Therapeutic drug monitoring of teicoplanin using an LC–MS/MS method: analysis of 421 measurements in a naturalistic clinical setting. J Pharm Biomed Anal. 2019;167:161–165. doi:10.1016/j.jpba.2019.02.001
27. Bernareggi A, Borghi A, Borgonovi M, et al. Teicoplanin metabolism in humans.. Antimicrob Agents Chemother. 1992;36(8):1744. doi:10.1128/AAC.36.8.1744
28. Gao LL, Xu H, Ye Q, et al. Population pharmacokinetics and dosage optimization of teicoplanin in children with different renal functions. Front Pharmacol. 2020;11:552. doi:10.3389/fphar.2020.00552
29. Cai J, Liu XF, Ni HQ. Analysis of monitoring results and influencing factors of blood drug concentration of different dosages of teicoplanin. Pharmacy Today. 2020;30:565–568.
30. Wilson AP. Comparative safety of teicoplanin and vancomycin. Int J Antimicrob Agents. 1998;10(2):143–152. doi:10.1016/S0924-8579(98)00025-9
31. Feiten HDS, Okumura LM, Martinbiancho JK, et al. Vancomycin-associated nephrotoxicity and risk factors in critically ill children without preexisting renal injury. Pediatr Infect Dis J. 2019;38(9):934–938. doi:10.1097/INF.0000000000002391
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


