Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
Therapeutic and scintigraphic applications of polymeric micelles: combination of chemotherapy and radiotherapy in hepatocellular carcinoma
Authors Shih Y, Peng C , Chiang P, Lin W, Luo T , Shieh M
Received 23 June 2015
Accepted for publication 2 October 2015
Published 16 December 2015 Volume 2015:10(1) Pages 7443—7454
DOI https://doi.org/10.2147/IJN.S91008
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Lei Yang
Ying-Hsia Shih,1,2 Cheng-Liang Peng,2 Ping-Fang Chiang,1,2 Wuu-Jyh Lin,2 Tsai-Yueh Luo,2,3 Ming-Jium Shieh1,4
1Institute of Biomedical Engineering, National Taiwan University, Taipei, Taiwan; 2Isotope Application Division, Institute of Nuclear Energy Research, Taoyuan, Taiwan; 3Institute of Radiological Science, Central University, Taichung, Taiwan; 4Department of Oncology, National Taiwan University Hospital, College of Medicine, National Taiwan University, Taipei, Taiwan
Abstract: This study evaluated a multifunctional micelle simultaneously loaded with doxorubicin (Dox) and labeled with radionuclide rhenium-188 (188Re) as a combined radiotherapy and chemotherapy treatment for hepatocellular carcinoma. We investigated the single photon emission computed tomography, biodistribution, antitumor efficacy, and pathology of 188Re-Dox micelles in a murine orthotopic luciferase-transfected BNL tumor cells hepatocellular carcinoma model. The single photon emission computed tomography and computed tomography images showed high radioactivity in the liver and tumor, which was in agreement with the biodistribution measured by γ-counting. In vivo bioluminescence images showed the smallest size tumor (P<0.05) in mice treated with the combined micelles throughout the experimental period. In addition, the combined 188Re-Dox micelles group had significantly longer survival compared with the control, 188ReO4 alone (P<0.005), and Dox micelles alone (P<0.01) groups. Pathohistological analysis revealed that tumors treated with 188Re-Dox micelles had more necrotic features and decreased cell proliferation. Therefore, 188Re-Dox micelles may enable combined radiotherapy and chemotherapy to maximize the effectiveness of treatment for hepatocellular carcinoma.
Keywords: 188Re-Dox micelles, radiotherapeutic, chemotherapeutic, hepatocellular carcinoma
Introduction
Hepatocellular carcinoma (HCC) is one of the most common cancer that causes death and has poor prognosis, resulting in patient survival rates in the range of 3%–5%.1 However, between 20% and 35% of patients are eligible for this invasive procedure due to the delayed diagnosis of HCC in patients with advanced cancer.2 Current treatment modalities for HCC include surgical tumor removal or transcatheter arterial embolization, chemotherapy, and external radiotherapy.
The chemotherapeutic drug doxorubicin (Dox) is commonly used to treat HCC. However, clinical studies have shown its serious side effects, such as dose-dependent cardiotoxicity and hepatotoxicity.3 Previous researches recently have revealed that nano-sized carrier enhance the accumulation of Dox in the tumor site and decrease the side effect.4,5
Intra-arterial administration of therapeutic radiopharmaceuticals and chemotherapeutic agents can deliver a tumoricidal dose with less non-tumorous hepatic tissue damage.6,7 However, transcatheter arterial embolization of yttrium-90 (90Y)-micospheres requires accurate skill and the use of specialized equipment, and the skeletal accumulation of 90Y could result in bone marrow depression.8
Rhenium-188 (188Re) is now considered as a better isotope for in vivo studies and clinical applications in both the imaging and therapeutic treatment of HCC.9,10 Since 188Re (t1/2=16.9 hours) decays by 15% gamma emission (Eγ=155 keV) and by 85% beta emission (Eβ−max=2.12 meV), its incorporation into polymeric micelles, which composed of amphiphilic block copolymers for drug delivery,11–13 could have both diagnostic and therapeutic applications.14–19 The high β-energy emission (2.1 meV) of 188Re provides the optimal therapeutic dose and deep tissue penetration for cancer therapy as previously studies described.17,20–22 By the enhanced permeability and retention (EPR) effect, nano-sized carrier could passive targeting23 and efficiently accumulate in tumors.24–27
In this study, we evaluated a nano-sized drug delivery system based on methoxy-poly (ethylene glycol)-block-poly(ε-caprolactone) (mPEG-b-PCL) micelles28 containing radiopharmaceuticals – 188Re and chemotherapeutic agents – Dox for combined radio/chemotherapy. The multifunctional micelles loaded with Dox and labeled with 188Re were developed to synergistically enhance cancer therapy. By using a murine model with luciferase-transfected BNL tumor cells (BNL/Luc) HCC, both the therapeutic efficacy and mechanism of this delivery system were evaluated.
Materials and methods
Preparation of 188Re-Dox micelles
The amphiphilic block copolymers, including mPEG-b-PCL and Fmoc-NH-PEG-b-PCL, were synthesized by the ring-opening polymerization of ε-caprolactone, and deprotected by 20% piperidine in dimethylformamide.15,29 Conjugation of diethylene triamine pentaacetic acid (DTPA) dianhydride with the amino group of N2H-PEG-b-PCL to synthesis the DTPA-PEG-b-PCL as described previously.17,30 The molecular weights of copolymers were characterized by 1H nuclear magnetic resonance (NMR, Bruker Avance-500 MHz FT-NMR; Bruker Corporation, Billerica, MA, USA) and gel permeation chromatography (Waters 510 pump/410 differential refractometer).
The doxorubicin-loaded DTPA micelles (Dox micelles) were prepared by the solvent evaporation method.29,31,32 The concentration of Dox was quantified by determining the absorbance at 480 nm with a UV–visible spectrophotometer (BioMate 3S, Thermo Electron Corporation, Hudson, NH, USA). The drug encapsulation efficiency is the amount of drug encapsulated divided by the amount of drug added multiplied by 100%. The mean diameters of the micelles were characterized with a Delsa™ Nano Particle Analyzer (Beckman Coulter, Fullerton, CA, USA). The morphology of the micelles was observed by H-7650 transmission electron microscopy (TEM, Hitachi Ltd., Tokyo, Japan).
The morphology of the micelles was observed by H-7650 TEM (Hitachi Ltd). The radiolabeling yields of the 188Re-DTPA-micelles were determined by a radio-thin layer chromatography imaging scanner (AR2000, Bioscan, Washington, DC, USA). The 188Re-Dox micelles were prepared by reacting a mixture of Dox micelles, 188Re-perrhenate (188ReO4, ~370 MBq), and stannous chloride for 1 hour at 37°C.27,28,33,34
Measurement of in vitro release
The in vitro release profiles of 188Re and Dox from the 188Re-Dox micelles were studied as described previously.15 A 2 mL volume of 188Re-Dox micelles were placed in a dialysis bag (molecular weight cutoff 3.5 kDa), and the dialysis bag was incubated in 50 mL of phosphate-buffered saline (PBS) (pH ~7.4) with gently stirring at 37°C, and the release medium was collected at predetermined time intervals. The release of Dox was quantified using high-performance liquid chromatography (HPLC), using a Waters Symmetry C18 reversed-phase column (150 mm ×3.9 mm, 5 mm) and a Waters HPLC instrument (Waters HPLC Alliance 2695 with a Waters 2487 dual λ absorbance detector; Waters, Milford, MA, USA). The mobile phase consisted of a 0.1% trifluoroacetic acid solution (pH=3.0) and acetonitrile at a flow rate of 1 mL/min. Dox was detected using a λ absorbance detector with a wavelength of 480 nm. The released 188Re from the 188Re-Dox micelles was quantified using a gamma-counter (Cobra series Model 5003, Packard Instrument Company, Inc., Meriden, CT, USA). All results are the mean ± standard deviation.
Establishment of the murine hepatocellular carcinoma model
Orthotopic murine liver tumor was generated in Balb/c mice using the BNL/Luc cell line. Female Balb/c athymic (nut/nut) nude mice 5–6 weeks of age were purchased from the National Laboratory Animal Center (Taipei, Taiwan). The in vivo experimental protocols were approved by the College of Medicine and the College of Public Health Institutional Animal Care and Use Committee (National Taiwan University; IACUC, number 20130041). All experiments involving the BNL/Luc cell line were performed in accordance with the rules of the Biosafety Level 2 Laboratory. The BNL/Luc cells were cultured in a humidified 5% CO2 incubator at 37°C in Dulbecco’s Modified Eagle’s Medium (Gibco BRL, Gaithersburg, MD, USA), containing 10% fetal bovine serum (Gibco BRL) and 1% antibiotics (antibiotic–antimycotic; Gibco BRL). The BNL/Luc tumors were established by orthotopic injection of 105 cells into one capsule of hepatic lobes. After 7 days post-incubation, the mice were monitored by bioluminescence imaging using IVIS imaging system (Xenogen, Alameda, CA, USA) to check for tumor growth. Mice were anesthetized and intraperitoneally injected with D-luciferin (Xenogen) for bioluminescence imaging.
Micro-SPECT/CT and biodistribution of 188Re-micelles
Micro-single photon emission computed tomography (SPECT)/computed tomography (CT) was also performed to evaluate the distribution of the 188Re-Dox micelles in the mice bearing orthotopic BNL/Luc tumors. SPECT and CT images were obtained from micro-SPECT/CT scanner system (XSPECT, Gamma Medica, Northridge, CA, USA). Mice bearing BNL/Luc tumors were received the 188Re-Dox micelles (equivalent to 22.2 MBq of 188Re) when that tumors volume reached 150–200 mm3. After intravenous injection 1, 4, 24, and 48 hours, the SPECT images merged with CT images were obtained from the animal in exactly the same position as described previously.17
The tumor-bearing mice (n=3 per time point) were sacrificed 1, 4, 8, 24, and 48 hours after injection of 188Re-Dox micelles, and then the blood, heart, liver, spleen, lung, kidney, smooth muscle, small intestine, urinary bladder, bone, brain, and tumors were collected, and the radioactivity of those organs was measured by a gamma counter. Distribution data were shown as the percentage of injected dose per gram of organs (ID%/g). Each column represents the mean ± standard deviation.
The biodistribution of doxorubicin was studied by ex vivo fluorescence images using an IVIS imaging system. After 188Re-Dox micelles injected 48 hours, the mice were sacrificed and obtained the heart, liver, spleen, lung, kidney, intestine, and tumors. Aforementioned organs were isolated to estimate the organs distribution of doxorubicin.
After the biodistribution studies, tumors and livers of mice were frozen, and embedded in optimal cutting temperature compound (Tissue Tek, Sakura, Torrance, CA, USA) for the frozen sections. The frozen sections were placed in contact with a BASMS 2040 imaging plate (Fujifilm, Tokyo, Japan) for the autoradiography, and the images were analyzed by an FLA-5100 reader (Fujifilm) and Multi Gauge V3.0 software (Fujifilm).
To identify the cluster of differentiation 31 (CD-31) expression and the distribution of doxorubicin in the tumor tissues (n=3) by immunofluorescence staining, the BNL/Luc tumor-bearing mice were intravenously administered with 188Re-Dox micelles at 10 mg/kg of doxorubicin. The tumor tissues were collected at 24 and 48 hours after injection, frozen, optimal cutting temperature embedded, and sectioned. The sections were washed continuous three times with PBS, then fixed in acetone for 15 minutes. The sections slide was farther blocked with 5% normal goat serum for 10 minutes at room temperature. The rat-anti-mouse CD-31 monoclonal antibody (rat anti-mouse, diluted 1:100, BioLegend, San Diego, CA, USA) were overlaid all sections with at 4°C overnight. The fluorescein isothiocyanate-labeled goat-anti-rat IgG (diluted 1:400, Sigma, St Louis, MO, USA) was added on slide and incubated at room temperature for 1 hour. The CD-31 staining slides were observed under a fluorescence microscope (Olympus Corporation, Tokyo, Japan).
Anti-tumor efficacy of 188Re-Dox micelles
The mice implanted with BNL/Luc tumor were assigned to four groups as follows: control-PBS (n=4), 188ReO4 (n=4), Dox micelles (n=5), and 188Re-Dox micelles (n=5). The 188Re-Dox micelles were administered via the tail vein with 10 mg/kg of doxorubicin and 22.2 MBq of 188Re on days 0, 6, and 12. Body weight changes were measured during observation days. The mice death dates were recorded for the survival curve by the Kaplan–Meier method. Survival evaluate of all groups was analyzed by log-rank test, and when <0.05, P-value was considered significant difference. The tumor growth inhibition (TGI) percentage of mice was calculated by the relative tumors volume at the mice sacrificed time points.
The therapeutic responses were monitored by bioluminescence imaging during observation days. The BNL/Luc tumor of mice was imaged and measured by IVIS system by Living Imaging software (Xenogen) on days 0, 6, 13, 19, and 26.
The 188Re-Dox micelles were administered via the tail vein with 10 mg/kg of doxorubicin and 22.2 MBq of 188Re. The tumor tissues (n=3) were collected at 24, 36, 48 hours after injection. For the hematoxylin and eosin (H&E) staining and immunohistochemical studies, all of the tumor tissues were formalin-fixed and paraffin-embedded. The necrosis in tumor tissue was detected by nicotinamide adenine dinucleotide phosphate (NADPH)-diaphorase staining.35 The section slides were reacted with NADPH-diaphorase reaction solution (10 mL of 1 M Trizma solution) containing 80 mg NADPH and 5 mg nitroblue tetrazolium for 30 minutes at 37°C. The section slides were also analyzed by proliferating cell nuclear antigen (PCNA) immunohistochemical staining for the proliferation study as described previously.18 The liver and spleen tissues (n=3) were collected at 24, 48 hours, and 7 days after the injection of the 188Re-Dox micelles for H&E staining.
Statistical analysis
All results are the mean ± standard deviation from ten distinct regions of each of the three tumors per experimental group.
Results
Characterization of the 188Re-Dox micelles
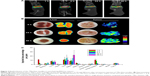
We synthesized mPEG-b-PCL and DTPA-PEG-b-PCL as described previously (Figure 1A).17 The molecular weights (Mn,NMR) and degree of polymerization (DPPCL) of mPEG-b-PCL and DTPA-PEG-b-PCL copolymers were 15,400 Da (DPPCL: 91.1) and 14,000 Da (DPPCL: 78.9), respectively, as determined by 1H NMR spectroscopy. The conjugation efficiency of DTPA dianhydride to NH2-PEG-PCL was 76.8% as described previously.17 The Dox micelles were observed by TEM (Figure 1B), exhibited spherical shape with sizes consistent with those observed by DLS (Figure 1C). The size of Dox micelles using various D/P ratios for Dox loading ranged from 107.5 to 194.0 nm (Table 1 and Figure 1C). The radiochemical purities of the 188Re-Dox micelles with D/P ratios of 1:5, 1:10, and 1:20 were 89.4%, 94.1%, and 90.1%, respectively (Figure 1D). The Dox micelles with a D/P ratio of 1:5 or 1:20 were larger in particle sizes with lower encapsulation efficiencies and radiochemical purities than those with a D/P ratio of 1:10 (Table 1).
The in vitro release profiles of 188Re (Figure 2A) and Dox (Figure 2B) from the 188Re-Dox micelles in PBS solution at pH 5.0 and 7.4 were assessed. A two-phase release profile was detected with a relatively fast release in the first phase after 8 hours, followed by a sustained and slower release over a prolonged period of time.
Micro-SPECT/CT imaging and biodistribution of the 188Re-Dox micelles in mice bearing BNL/Luc tumors
The biodistributions of the 188Re-Dox micelles were evaluated in mice bearing BNL/Luc tumors. The micro-SPECT/CT imaging discovered that the radioactivity accumulated in both the liver and tumor at 1, 4, 24, and 48 hours after the injection of the 188Re-Dox micelles (Figure 3A). To confirm the biodistributions of the 188Re-Dox micelles from the micro-SPECT/CT images, autoradiography was performed. The autoradiography studies clearly showed significant accumulation of radioactivity from the 188Re-Dox micelles in the liver beginning 4 hours after injection (Figure 3B). However, the tumor accumulation of radioactivity also increased with time after the injection.
The highest accumulation had occurred in the liver, followed by in the tumor, spleen, lung, and kidney at 1 hour after the injection of the 188Re-Dox micelles (Figure 3C). The radioactivity in the blood and kidney decreased quickly over the time course, with the urinary bladder displaying increased radioactivity.
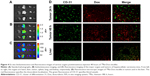
The ex vivo biodistribution of Dox was evaluated in mice bearing BNL/Luc tumors. Images obtained by the IVIS imaging system revealed the accumulation of Dox in the heart, liver, tumor, spleen, lung, and kidney at 48 hours after the injection of the 188Re-Dox micelles. As shown in Figure 4A–C, higher bioluminescence and fluorescence from Dox occurred in the tumor compared with other major organs.
The distribution of Dox correlation with the blood vessels between liver and tumor was evaluated; tissue sections were stained with a CD-31 immunostaining (Figure 4D). This analysis shown that the 188Re-Dox micelles extravagated from the blood vessels and that doxorubicin delivery was more accumulation in tumors compared with in normal liver.
Anti-tumor effect of the 188Re-Dox micelles
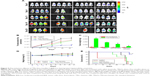
We investigated the antitumor efficacy and toxicity of the 188Re-Dox micelles on BNL/Luc tumor-bearing mice (Figure 5). The therapeutic responses were monitored by bioluminescence imaging after various treatments during the evaluation period. Four groups of Balb/c mice bearing orthotopic BNL/Luc tumors were treated with an intravenous injection of combined 188Re-Dox micelles, with 188ReO4 alone, with Dox-micelles alone, or with PBS. Anti-tumor effect of the 188Re-Dox micelles was shown a series of typical optical images of luminescence-expressing tumors after the various treatments (Figure 5A). Mice in the PBS-control and in the 188ReO4 alone group shown rapid progression of the tumor burden.
The antitumor efficacy of the 188Re-Dox micelles was compared with that of the 188ReO4 and the Dox-micelles. As shown in Figure 5B, the tumor burden was measured and quantified based on the luminescence intensity, as performed in Figure 5A. The average photon flux was measured as a function of time after the treatments. The 188Re-Dox micelle group gave the lowest photon collection, which suggests the best antitumor efficacy on days 13, 19, and 26 (compared with the control groups of 188ReO4 alone and of Dox micelle alone). As expected, the mice that received the combined 188Re-Dox micelle treatment exhibited the lowest tumor burden, as shown in Figure 5C. Reduced tumor volumes were measured at the sacrificed time points (day 31) after the combined treatment with the 188Re-Dox micelles compared with the control single treatment groups (P<0.05). After the treatment with the 188Re-Dox micelles, a TGI percentage of 84.8% was detected compared with the control. TGI percentage of 42.0% and 60.7% was detected for the treatments of 188ReO4 alone and Dox-micelles alone, respectively.
Decrease in body weight was recorded to monitor treatment-induced toxicity (Figure 5D). The control groups treated with PBS, 188ReO4 (22.2 MBq of 188Re) alone, or Dox-micelles (10 mg/kg of Dox) alone gradually increased in mean body weight by 122%, 106%, and 113%, respectively, from days 19 to 26. Two of the four mice died after the second dose of 188ReO4 on day 6. Mice treated with the 188Re-Dox micelles (10 mg/kg of doxorubicin and 22.2 MBq of 188Re) had lost a maximum of approximately 5% of their weight on day 23.
We also compared the long-term survival of the mice bearing BNL/Luc tumors in the PBS (n=4), 188ReO4 alone (n=4), Dox micelles alone (n=5), and 188Re-Dox micelle (n=5), respectively. Figure 5E shows all mice in the control, 188ReO4 alone, and Dox micelles alone of single treatment groups had died owing to aggressive tumor growth by day 31. The average survival times were 25.7, 16.5, and 24.4 days for the control, 188ReO4 alone, and Dox micelle alone groups, respectively. There was no significant difference among the average survival times of these three groups (P>0.05). On the contrary, the survival rates of the hepatoma mice treated with the 188Re-Dox micelles were 80% on day 31.
To further examine the therapy effect of the 188Re-Dox micelle in vivo, the tumors were further analyzed by immunohistochemical staining (Figure 6A). The H&E staining showed different tumor tissue morphologies in different treatment groups. The image of 188Re-Dox micelles treat group was found more necrotic tissue in the tumors, compared with control and other groups. In contrast, less necrosis was observed in tumors treated with 188ReO4 alone or control. The PCNA immunostaining revealed that the cell proliferation was significantly decreased in the tumors treated with the 188Re-Dox micelles (~19.6%±6.8%) compared with the tumors treated with 188ReO4 alone (~50.7%±8.7%; P<0.05), with Dox micelles alone (~50.7%±8.7%; P<0.05), or with PBS alone (control mice) (~68.0%±9.6%; P<0.05) (Figure 6B). The histological analysis demonstrated that 188Re-Dox micelles could not cause significant toxicity in normal liver and spleen as shown in Figure 6C.
Discussion
The Dox micelles with a D/P ratio of 1:10 had the highest encapsulation efficiency of 98.2% and a radiochemical purity of 94.1% (Figure 1 and Table 1). It had reported that nanoparticles with a size arranging from 10 to 100 nm are more efficient as drug delivery systems and exhibit high cellular uptake and tumor accumulation,25–27 whereas the reticuloendothelial system filters away nanoparticles with sizes larger than 200 nm.36,37 The Dox micelle with a D/P ratio of 1/10 was employed as the drug carrier for this study.
Results of in vitro release profiles of 188Re and Dox indicate that the drug release from the 188Re-Dox micelles would occur under acidic or neutral conditions, which is under physiological conditions (Figure 2).
The 188Re-Dox micelles could maintain the 188Re and micelles in the liver and tumor area until 48 hours. There was no significant accumulation of radioactivity found in any other organs through SPECT imaging (Figure 3). The biodistributions of the 188Re-Dox micelles were also determined by γ-counting. The results suggested that the 188Re-Dox micelles were distributed to most of the major organs and the tumor. Biodistribution studies of the 188Re-Dox micelles results (Figure 3) implied that the radioactivity of the 188Re-Dox micelles was eliminated by the urinary system. The tumor-to-muscle ratio of the 188Re-Dox micelles increased from 92.97±98.15 at 1 hour after the injection to 229.88±23.10 at 48 hours after the injection. This result could be explained by the EPR effect, where the 188Re-Dox micelles could passively target to tumors site. The greater accumulation of Dox in the BNL/Luc tumor could propose an improved antitumor efficacy with low toxicity caused by nonspecific targeting.38,39
Although the results of autoradiography clearly shown significant accumulation of the 188Re-Dox micelles in the liver (Figure 3), the fluorescence of doxorubicin in the liver was low. The fluorescence images were obtained from the tissue sections shown similar biodistribution of doxorubicin as that acquired by IVIS imaging (Figure 4). The tumor displayed a higher accumulation of Dox than the liver tissue at 24 and 48 hours following the 188Re-Dox micelle delivery. This could be explained by the absence of doxorubicin release from the 188Re-Dox micelles in the liver. Consequently, the 188Re-Dox micelles would probably not induce significant hepatotoxicity. On the contrary, there was significant fluorescence of doxorubicin found in the tumors. The result suggesting that after the uptake of the 188Re-Dox micelles by cancer cells, Dox was released as previously described.40
Comparatively, mice that received the 188Re-Dox micelles or Dox micelles alone shown significantly lower tumor burdens on day 6 and on day 13 than did those that received PBS or 188ReO4 alone (Figure 5). Treatment with the 188Re-Dox micelles delayed the appearance and decreased the intensity of luminescence by up to days 26; however, the expression of luminescence did not decrease in the other groups. This suggests that the 188Re-Dox micelles were the inhibitoriest toward tumor growth compared with the other single treatments.
Although we observed body weight loss in mice receiving the 188Re-Dox micelles, all mice administered with triple doses of the 188Re-Dox micelles had survived on day 26, indicating that the combination of radiotherapy and chemotherapy mediated by the 188Re-Dox micelles did not cause obvious toxicity (Figure 5).
In pathology studies (Figure 6), results of H&E staining indicated that more necrosis was seen in tumors treated with the 188Re-Dox micelles, such as coagulation, vacuolation, and decreased nuclear staining in tumor tissues as described previously.18 This was in good agreement with the loss of NADPH-diaphorase activity.35 The tumor tissue sections of all experiment groups were stained with NADPH-diaphorase staining for the assessment of tissue viability. Necrotic tissue in tumor sections showed loss of NADPH-diaphorase activity.35 As shown in Figure 6A, the tumor treated with 188Re-Dox micelles had more necrotic features than that treated with 188ReO4 alone or Dox micelles alone. PCNA staining shown high expression in tumors treated with 188ReO4, Dox micelles, and PBS alone, but very low in tumors treated with the 188Re-Dox micelles (Figure 6B). According to the results of immunohistochemical staining, the 188Re-Dox micelles cause cell death through necrosis. These imply that the treatment with the 188Re-Dox micelles could greatly increase tumor necrosis and reduce the tumor proliferation but not cause significant toxicity in normal liver and spleen (Figure 6C).
Conclusion
In summary, we had prepared and characterized multifunctional 188Re-Dox micelles simultaneously loaded with doxorubicin and labeled with 188Re for cancer radiotherapy combined with chemotherapy. The components in the 188Re-Dox micelles acted synergistically against tumor growth, demonstrating better performance than either component (188ReO4 or Dox micelles) individually in mice bearing orthotopic liver tumors. Therefore, the 188Re-Dox micelles would be a synergistic combinatorial treatment modality with both radiotherapy and chemotherapy for orthotopic HCC in mice model.
Author contributions
Ying-Hsia Shih designed the experiments, wrote the manuscript, and performed the experiments. Cheng-Liang Peng contributed to experimental suggestions and performed the experiments. Ping-Fang Chiang performed the experiments. Wuu-Jyh Lin contributed to experimental suggestions. Tsai-Yueh Luo contributed to experimental design and suggestions, and manuscript revision. Ming-Jium Shieh contributed to experimental suggestions and manuscript revision. All authors contributed toward data analysis, drafting and critically revising the paper, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Acknowledgments
This research was funded by the National Science Council of the Republic of China (Taiwan) (NSC 102-2320-B-002-038-MY3) and the Ministry of Health and Welfare, Taiwan (MOHW103-TDU-N-211-133006).
Disclosure
The authors report no conflicts of interest in this work.
References
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. Ca-a Cancer J Clin. 2005;55(2):74–108. | ||
Cha C, DeMatteo RP, Blumgart LH. Surgery and ablative therapy for hepatocellular carcinoma. J Clin Gastroenterol. 2002;35(5):S130–S137. | ||
Park S, Yoon J, Bae S, et al. Therapeutic use of H2O2-responsive anti-oxidant polymer nanoparticles for doxorubicin-induced cardiomyopathy. Biomaterials. 2014;35(22):5944–5953. | ||
Zheng L, Gou M, Zhou S, et al. Antitumor activity of monomethoxy poly(ethylene glycol)-poly (epsilon-caprolactone) micelle-encapsulated doxorubicin against mouse melanoma. Oncol Rep. 2011;25(6):1557–1564. | ||
Lv S, Tang Z, Li M, et al. Co-delivery of doxorubicin and paclitaxel by PEG-polypeptide nanovehicle for the treatment of non-small cell lung cancer. Biomaterials. 2014;35(23):6118–6129. | ||
Gray B, Van Hazel G, Hope M, et al. Randomised trial of SIR-spheres ((R)) plus chemotherapy vs chemotherapy alone for treating patients with liver metastases from primary large bowel cancer. Ann Oncol. 2001;12(12):1711–1720. | ||
Gulec SA, Mesoloras G, Dezarn WA, McNeillie P, Kennedy AS. Safety and efficacy of Y-90 microsphere treatment in patients with primary and metastatic liver cancer: the tumor selectivity of the treatment as a function of tumor to liver flow ratio. J Transl Med. 2007;5:15. | ||
Wang S-J, Lin W-Y, Lui W-Y, Chen M-N, Tsai Z-T, Ting G. Hepatic artery injection of yttrium-90-lipiodol: biodistribution in rats with hepatoma. J Nucl Med. 1996;37(2):332–335. | ||
Hamoudeh M, Kamleh MA, Diab R, Fessi H. Radionuclides delivery systems for nuclear imaging and radiotherapy of cancer. Adv Drug Deliv Rev. 2008;60(12):1329–1346. | ||
Luo T-Y, Tang IC, Wu Y-L, et al. Evaluating the potential of 188Re-SOCTA–trastuzumab as a new radioimmunoagent for breast cancer treatment. Nucl Med Biol. 2009;36(1):81–88. | ||
Gref R, Minamitake Y, Peracchia MT, Trubetskoy V, Torchilin V, Langer R. Biodegradable long-circulating polymeric nanospheres. Science. 1994;263(5153):1600–1603. | ||
Kwon GS, Okano T. Polymeric micelles as new drug carriers. Adv Drug Deliv Rev. 1996;21(2):107–116. | ||
Savic R, Luo LB, Eisenberg A, Maysinger D. Micellar nanocontainers distribute to defined cytoplasmic organelles. Science. 2003;300(5619):615–618. | ||
Shahin M, Ahmed S, Kaur K, Lavasanifar A. Decoration of polymeric micelles with cancer-specific peptide ligands for active targeting of paclitaxel. Biomaterials. 2011;32(22):5123–5133. | ||
Peng CL, Lai PS, Lin FH, Wu SYH, Shieh MJ. Dual chemotherapy and photodynamic therapy in an HT-29 human colon cancer xenograft model using SN-38-loaded chlorin-core star block copolymer micelles. Biomaterials. 2009;30(21):3614–3625. | ||
Matsumura Y. Preclinical and clinical studies of NK012, an SN-38-incorporating polymeric micelles, which is designed based on EPR effect. Adv Drug Deliv Rev Deliv Rev. 2011;63(3):184–192. | ||
Peng C-L, Shih Y-H, Lee P-C, Hsieh TM-H, Luo T-Y, Shieh M-J. Multimodal image-guided photothermal therapy mediated by Re-188-labeled micelles containing a cyanine-type photosensitizer. ACS Nano. 2011;5(7):5594–5607. | ||
Peng C-L, Shih Y-H, Liang K-S, et al. Development of in situ forming thermosensitive hydrogel for radiotherapy combined with chemotherapy in a mouse model of hepatocellular carcinoma. Mol Pharm. 2013;10(5):1854–1864. | ||
Shieh MJ, Peng CL, Chiang WL, et al. Reduced skin photosensitivity with meta-tetra(hydroxyphenyl)chlorin-loaded micelles based on a poly(2-ethyl-2-oxazoline)-b-poly(d,l-lactide) diblock copolymer in vivo. Mol Pharm. 2010;7(4):1244–1253. | ||
Tang IC, Luo T-Y, Liu S-W, et al. Synthesis and application of 188Re-MN-16ET/lipiodol in a hepatocellular carcinoma animal model. Nucl Med Biol. 2011;38(7):1043–1052. | ||
Lin W-Y, Luo T-Y, Tsai S-C, Kao C-H, Tang IC, Huang P-W. A comparison of Re-188-MN-16ET-lipiodol and transcatheter arterial chemoembolization in the treatment of hepatoma: an animal study. Nucl Med Biol. 2013;40(3):437–441. | ||
Huang P-W, Tsai S-C, Luo T-Y, Kao C-H, Lin W-Y. Therapeutic efficacy of 188Re-MN-16ET lipiodol in an animal model of hepatocellular carcinoma. Ann Nucl Med. 2013;27(6):532–537. | ||
Bertrand N, Wu J, Xu X, Kamaly N, Farokhzad OC. Cancer nanotechnology: the impact of passive and active targeting in the era of modern cancer biology. Adv Drug Deliv Rev. 2014;66:2–25. | ||
Wu J, Akaike T, Maeda H. Modulation of enhanced vascular permeability in tumors by a bradykinin antagonist, a cyclooxygenase inhibitor, and a nitric oxide scavenger. Cancer Res. 1998;58(1):159–165. | ||
Ferrari M. Nanogeometry: beyond drug delivery. Nat Nano. 2008;3(3):131–132. | ||
Popović Z, Liu W, Chauhan VP, et al. A nanoparticle size series for in vivo fluorescence imaging. Angew Chem Int Ed. 2010;49(46):8649–8652. | ||
Cabral H, Matsumoto Y, Mizuno K, et al. Accumulation of sub-100 nm polymeric micelles in poorly permeable tumours depends on size. Nat Nano. 2011;6(12):815–823. | ||
Wu J, Chu C-C. Block copolymer of poly(ester amide) and polyesters: synthesis, characterization, and in vitro cellular response. Acta Biomater. 2012;8(12):4314–4323. | ||
Peng CL, Shieh MJ, Tsai MH, Chang CC, Lai PS. Self-assembled star-shaped chlorin-core poly(c-caprolactone)-poly(ethylene glycol) diblock copolymer micelles for dual chemo-photodynamic therapies. Biomaterials. 2008;29(26):3599–3608. | ||
Fonge H, Lee H, Reilly RM, Allen C. Multifunctional block copolymer micelles for the delivery of 111In to EGFR-positive breast cancer cells for targeted auger electron radiotherapy. Mol Pharm. 2009;7(1):177–186. | ||
Cheng CC, Huang CF, Ho AS, et al. Novel targeted nuclear imaging agent for gastric cancer diagnosis: glucose-regulated protein 78 binding peptide-guided In-111-labeled polymeric micelles. Int J Nanomed. 2013;8:1385–1391. | ||
Cheng CC, Lu N, Peng CL, et al. Targeting to overexpressed glucose-regulated protein 78 in gastric cancer discovered by 2D DIGE improves the diagnostic and therapeutic efficacy of micelles-mediated system. Proteomics. 2012;12(15–16):2584–2597. | ||
Chen Y, Xiong Q-F, Yang X-Q, He L, Huang Z-W. Evaluation of 188Re-DTPA-deoxyglucose as a potential cancer radiopharmaceutical. Am J Roentgenol. 2010;194(3):761–765. | ||
Hsieh B-T, Hsieh J-F, Tsai S-C, et al. Rhenium-188-labeled DTPA: a new radiopharmaceutical for intravascular radiation therapy. Nucl Med Biol. 1999;26(8):967–972. | ||
Zuchini R, Huang CH, Tsai HW, et al. Electromagnetic thermoablation to treat thrombocytopenia in cirrhotic and hypersplenic rats. J Gastroenterol Hepatol. 2010;25(9):1578–1586. | ||
Litzinger DC, Buiting AMJ, Vanrooijen N, Huang L. Effect of liposome size on the circulation time and intraorgan distribution of amphipathic poly(ethylene glycol)-containing liposomes. Biochim Biophys Acta Biomembr. 1994;1190(1):99–107. | ||
Moghimi SM, Hunter AC, Murray JC. Long-circulating and target-specific nanoparticles: theory to practice. Pharmacol Rev. 2001;53(2):283–318. | ||
Poste G, Kirsh R. Site-specific (targeted) drug delivery in cancer therapy. Nat Biotechnol. 1983;1(10):869–878. | ||
Fisher JW, Sarkar S, Buchanan CF, et al. Photothermal response of human and murine cancer cells to multiwalled carbon nanotubes after laser irradiation. Cancer Res. 2010;70(23):9855–9864. | ||
Lu P-L, Chen Y-C, Ou T-W, et al. Multifunctional hollow nanoparticles based on graft-diblock copolymers for doxorubicin delivery. Biomaterials. 2011;32(8):2213–2221. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.