Back to Journals » The Application of Clinical Genetics » Volume 11
The role of PAX9 promoter gene polymorphisms in causing hypodontia: a study in the Jordanian population
Authors Abu-Siniyeh A , Khabour OF , Owais AI
Received 9 August 2018
Accepted for publication 31 October 2018
Published 21 November 2018 Volume 2018:11 Pages 145—149
DOI https://doi.org/10.2147/TACG.S183212
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Martin Maurer
Ahmed Abu-Siniyeh,1 Omar F Khabour,1 Arwa I Owais2
1Department of Medical Laboratory Sciences, Faculty of Applied Medical Sciences, Jordan University of Science and Technology, Irbid, Jordan; 2Department of Applied Dental Sciences, Faculty of Applied Medical Sciences, Jordan University of Science and Technology, Irbid, Jordan
Background: The congenital absence of one or few teeth, hypodontia, is considered one of the utmost dental ageneses in human beings. Several genes have been shown to be involved in the development of hypodontia such as paired box gene 9 (PAX9). The expression of PAX9 is controlled by several polymorphic elements in the promoter region of the gene on 14q13.3 locus. The aim of this study was to find any association between PAX9 c.-912T>C (rs2073247) and c.-1031G>A (rs2073244) promoter polymorphisms and the development of hypodontia among the Jordanian population.
Methods: Genotyping of the polymorphisms in 72 unrelated subjects with hypodontia was performed using PCR-restriction fragment length polymorphism (RFLP) technique and compared with that of 72 normal healthy unrelated control individuals.
Results: The hypodontia group had a significantly higher -1031GG genotype (P<0.01) and a significantly lower -912TC genotype (P<0.01) compared with the control group. The results suggest that the transcriptional activity of PAX9 gene is affected by polymorphisms in the promoter region of this gene and is associated with hypodontia phenotype.
Conclusion: The rs2073247) and rs2073244 promoter polymorphisms of PAX9 might play a role in the development of hypodontia in the Jordanian population.
Keywords: PAX9, hypodontia, Jordan, polymorphism, promoter
Introduction
Hypodontia, the congenital absence of less than six permanent teeth, is one of the most prevalent craniofacial congenital malformations in human beings,1 with an overall incidence of 2.0%–10.0%, depending on the population studied excluding the absence of third molars.2 Hypodontia is common for the lower second premolars and the upper lateral incisors.3,4 Hypodontia can occur as a result of both environmental and genetic factors. For example, trauma of the dental region, chemotherapy, or radiotherapy has been shown to be associated with hypodontia. Genetic factors such as variations in MSX1 and paired box gene 9 (PAX9) have also been shown to be related to non-syndromic hypodontia.5,6
The PAX9 gene is a member of the paired box (PAX) family of transcriptional factors. The gene is localized on the long arm of chromosome 14 (14q21) and has been shown to play an important role in dental development especially in the bud stage.3,7,8 Mutations in PAX9 gene have been shown to cause familial tooth agenesis.9–12 Moreover, several polymorphisms in PAX9 gene have been shown to increase the risk factor for the development of hypodontia. The c.-1031G>A and c.-912T>C single-nucleotide polymorphisms (SNPs) in PAX9 gene have been shown to be associated with hypodontia in the Caucasian population.13 Similarly, Bianchi et al14 showed that the C allele of c.-915G>C SNP of PAX9 gene is related to hypodontia. Therefore, polymorphic variants of PAX9 gene in addition to mutations might be a risk factor for tooth agenesis.
This study aimed to investigate the association between PAX9 c.-1031G>A and c.-912T>C promoter polymorphisms and the development of hypodontia among the Jordanian population.
Subjects and methods
In this study, 72 unrelated healthy subjects diagnosed with non-syndromic hypodontia and a similar number of healthy controls of unrelated individuals with the same mean age of 20 years were enrolled.
Sample collection was done in the Dental Teaching Center, Jordan University of Science and Technology (JUST), Jordan. Approval from the institutional review board (IRB) at the university was obtained. Both the IRB at JUST committees utilized the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments. The procedures and possible discomforts, risks, and benefits were explained to the patients involved, and their written informed consents were obtained.
The selection criteria were as follows: healthy, unrelated individuals with agenesis of one to five permanent teeth, excluding third molars, and the lack of any other craniofacial malformations. Control subjects were all healthy, unrelated, and had normal primary and permanent dentition and did not show any craniofacial abnormalities. The diagnosis was confirmed by the same dentist after obtaining thorough history and making orthopantomographs for all subjects.
The DNA for the genotyping of examined SNPs was isolated from the blood collected in EDTA sterile tubes using DNA extraction kit obtained from Promega Corporation (Fitchburg, WI, USA) according to the manufacturer’s instructions. DNA concentration and quality were assessed spectrophotometrically, and DNA was stored at –20°C until used.
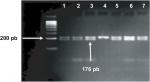
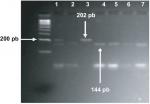
Polymorphisms of PAX9 c.-1031G>A and c.-912T>C were typed by restriction fragment length polymorphism (RFLP) according to the criteria specified by Peres et al13 using HaeIII and MseI restriction enzymes (Fermentas, Glen Burnie, MD, USA) respectively. For both polymorphisms, the used primers were as follows: forward: 5’-AGC CTG AAT CCT GTG TGC AC-3’ and reverse: 5’-CTA ATC TAA AGT GTA CCG TAT GC-3’. Amplification was performed in PCR tubes containing 100 ng of DNA, 1 µM of each primer and ready-to-use PCR master mix obtained from Promega Corporation. An Eppendorf thermocycler (Eppendorf, Hamburg, Germany) were used, and the cycling conditions were as follows: 95°C for 5 minutes and 35 cycles at 95°C for 60 seconds, 58°C for 30 seconds, and 72°C for 60 seconds, followed by a final extension of 5 minutes at 72°C. The amplified fragment (202 bp) was detected using 2% agarose gel electrophoresis prior to restriction digestion, which was carried out for each polymorphism separately in 20 µL final volume reaction using 10 µL of PCR product and 1 unit of HaeIII for c.-1031G>A or 2.0 units of MseI for c.-912T>C. The reactions were incubated at 37°C for 5 hours. Amplified fragment for -1031G and -912T alleles stayed uncut, whereas the -1031A allele cut with HaeIII to give 176 bp and 26 bp fragments (Figure 1), and -912C allele cut with MseI to give 144 bp and 58 bp fragments (Figure 2).
Hardy–Weinberg equation was used to test whether the examined SNPs were in equilibrium. The SPSS (version 21; IBM Corporation, Armonk, NY, USA) program was used for all statistical evaluations. P-value of <0.05 was considered significant.
Results
Male-to-female ratio was 1:1.18 in the hypodontia group and 1:1.25 in the control group (P>0.05; Table 1). About 16.7% of hypodontia subjects had three or more missing teeth, while the percentage of subjects with one and two missing teeth was 83.3% (Table 1). The most prevalent missing teeth in the sample were incisors and the premolars (Table 1).
  | Table 1 Aspects of population studied |
Table 2 summarizes the genotype distributions and allele frequencies of c.-1031G>A PAX9 SNPs in different groups. Significant differences in the distribution of genotypes of the c.-1031G>A SNPs were detected (Fisher’s exact test, P<0.001) with the GG homozygous genotype being more frequent among hypodontia subjects. Accordingly, the distribution of G allele to A allele was different between hypodontia subjects and controls (chi-squared test, P<0.001). Therefore, the presence of the -1031 GG homozygous genotype increases the risk of hypodontia in the studied population.
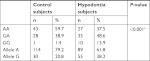  | Table 2 Genotype and allele frequencies of PAX c.-1031G>A polymorphism in the Jordanian population Notes: aFisher’s exact test. bChi-squared test. |
The distribution of genotypes and the frequencies of alleles at PAX9 c.-912T>C promoter SNPs in the two groups are summarized in Table 3. Similar to c.-1031G>A SNPs, the distribution of PAX9 c.-912T>C SNP genotypes (Fisher’s exact test, P<0.001) and T to C allele (chi-squared test, P<0.001) was significantly different between the hypodontia group and the control group. Among the hypodontia group, only one subject (<1% of the sample) was CT heterozygous compared with 24 subjects (33% of the sample) in the control group. Therefore, the presence of the -912TC heterozygous genotype might be associated with low risk of hypodontia in the Jordanian population.
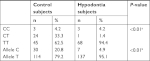  | Table 3 Genotype and allele frequencies of PAX9 c.-912T>C polymorphism in the Jordanian population Notes: aFisher’s exact test. bChi-squared test. |
Discussion
In this study, we found that PAX9 c.-1031G>A and c.-912T>C gene promoter polymorphisms were strongly associated with the development of non-syndromic hypodontia in the Jordanian population. The results showed that the presence of the -1031 GG and -912TC genotypes might be associated with hypodontia in the Jordanian population.
The role of PAX9 in tooth development is well established. PAX9 is a member of gene family that encodes for transcriptional factors which is important during embryogenesis.15,16 PAX9 plays a significant role during odontogenesis in the progressive and reciprocal signal transduction pathways that normally occur in epithelial–mesenchymal cells.6 The gene product of PAX9 appears early in the mesenchyme before ectodermal thickening and reaches its higher expression level during the initiation of bud and cap stages.17 In the dental mesenchyme, PAX9 protein interacts with MSX1 homeoprotein to regulate BMP4, which has an implication in downstream signaling cascade that is essential for enamel knot formation.16,18,19 Therefore, proper regulation of PAX9 signaling pathway is essential for odontogenic potential of mesenchyme and normal tooth development.
The results of this study agree with the finding in the Caucasian population (12). About 13.8% of hypodontia patients had GG genotype at -1031 position, while it was present in one subject from the control group (1.4%). Similar finding was reported in the Caucasian population in Brazil where the GG genotype at -1031 (9.8%) in hypodontia subjects compared with 2.9% in controls.13 Regarding the c.-912T>C polymorphism, the frequency of the TT genotype was 94.4% in the hypodontia group compared with 62.5% in the control group. In the Caucasian population, Peres et al13 showed that TT genotype was strongly associated with hypodontia. In their study, TT genotype was 12.7% in hypodontia subjects compared with 2.8% in controls. However, in the present study, the frequency of the T/C genotype was higher in the control group (33.3%) compared with the hypodontia subjects (1.4%) which indicated that the T/C genotype was associated with low risk of hypodontia. In contrast, Peres et al13 reported similar frequencies of the T/C genotype in hypodontia individuals and the control group. On the other hand, a study from Turkey showed that AC haplotype of c.-1031G>A and c.-912T>C SNPs carries less risk in having hypodontia.20 The discrepancy between the different studies could be due to variations in the genetic background and in the frequency of the T/C alleles in the different populations. Despite such disparity, results of the current study and those of the study by Peres et al,13 provide strong evidence for the involvement of the PAX9 c.-912T>C promoter polymorphism in causing hypodontia phenotype in the two populations. In addition, the association between gene polymorphisms and hypodontia might have population specificity being affected by certain environmental factors or/and population genetic background.
Several mutations and polymorphisms in PAX9 gene were reported to cause hypodontia in other populations (Table 4). For example, the deletion of the PAX9 gene in American and Polish families was described to cause autosomal dominant agenesis which involves several primary and permanent molars.10,21 In the American population, frame shift mutations in exon 2 of PAX9 were reported in individuals suffer from hypodontia.9,22 In addition, missense mutations in PAX9 gene were also reported to cause hypodontia in several populations.4,9,11,16,21,23–27 Furthermore, nonsense mutations in PAX9 that produce truncated proteins were identified to cause familial hypodontia and oligodontia phenotypes in subjects from Denmark, Finland, Spain, and USA.22,28–30 In the Caucasian population in Brazil, the G-915C SNP in the promoter region of PAX9 gene was found to be associated with hypodontia.14 Mitsui et al31 reported two novel mutations (c.73_75delATC and c.146C>T) in the paired domain of PAX9 in patients with non-syndromic tooth agenesis from Japan. In a study from Saudi Arabia, a new mutation p.N40I in PAX9 was detected in two patients with non-syndromic tooth agenesis.32 A new initiation codon mutation (changes the ATG initiation codon to AGG) related to oligodontia was identified in the Chinese population.33 Thus, the abovementioned literature indicates a significant role for PAX9 in tooth development in human beings.
  | Table 4 Mutations and polymorphisms in PAX9 gene that has been shown to cause tooth agenesis |
Conclusion
Findings of this study indicated that c.-1031G>A and c.-912T>C promoter polymorphisms in PAX9 gene might be associated with hypodontia in the Jordanian population. The analysis of genes that involved in the development of hypodontia is important for understanding the genetic basis of hypodontia and might help in the management of this condition.
Acknowledgments
The study was funded by Deanship of Research, JUST. The authors thank Mr Ahmad Abu-Wardeh for his technical help in the study.
Disclosure
The authors report no conflicts of interest in this work.
References
Arte S, Nieminen P, Pirinen S, Thesleff I, Peltonen L. Gene defect in hypodontia: exclusion of EGF, EGFR, and FGF-3 as candidate genes. J Dent Res. 1996;75(6):1346–1352. | ||
Breeze J, Dover MS, Williams RW. Contemporary surgical management of hypodontia. Br J Oral Maxillofac Surg. 2017;55(5):454–460. | ||
Hu JC, Simmer JP. Developmental biology and genetics of dental malformations. Orthod Craniofac Res. 2007;10(2):45–52. | ||
Matalova E, Fleischmannova J, Sharpe PT, Tucker AS. Tooth agenesis: from molecular genetics to molecular dentistry. J Dent Res. 2008;87(7):617–623. | ||
Cobourne MT. Familial human hypodontia—is it all in the genes? Br Dent J. 2007;203(4):203–208. | ||
Thesleff I. Genetic basis of tooth development and dental defects. Acta Odontol Scand. 2000;58(5):191–194. | ||
Jernvall J, Thesleff I. Reiterative signaling and patterning during mammalian tooth morphogenesis. Mech Dev. 2000;92(1):19–29. | ||
Underhill DA. Genetic and biochemical diversity in the Pax gene family. Biochem Cell Biol. 2000;78(5):629–638. | ||
das P, Hai M, Elcock C, et al. Novel missense mutations and a 288-bp exonic insertion in PAX9 in families with autosomal dominant hypodontia. Am J Med Genet A. 2003;118A(1):35–42. | ||
das P, Stockton DW, Bauer C, et al. Haploinsufficiency of PAX9 is associated with autosomal dominant hypodontia. Hum Genet. 2002;110(4):371–376. | ||
Mensah JK, Ogawa T, Kapadia H, Cavender AC, D’Souza RN. Functional analysis of a mutation in PAX9 associated with familial tooth agenesis in humans. J Biol Chem. 2004;279(7):5924–5933. | ||
Stockton DW, das P, Goldenberg M, D’Souza RN, Patel PI. Mutation of PAX9 is associated with oligodontia. Nat Genet. 2000;24(1):18–19. | ||
Peres RC, Scarel-Caminaga RM, do Espírito Santo AR, Line SR. Association between PAX-9 promoter polymorphisms and hypodontia in humans. Arch Oral Biol. 2005;50(10):861–871. | ||
Bianchi FJ, de Oliveira TF, Saito CB, Peres RC, Line SR. Association between polymorphism in the promoter region (G/C-915) of PAX9 gene and third molar agenesis. J Appl Oral Sci. 2007;15(5):382–386. | ||
Dahl E, Koseki H, Balling R. Pax genes and organogenesis. Bioessays. 1997;19(9):755–765. | ||
Kapadia H, Mues G, D’Souza R. Genes affecting tooth morphogenesis. Orthod Craniofac Res. 2007;10(4):237–244. | ||
Peters H, Neubüser A, Balling R. Pax genes and organogenesis: Pax9 meets tooth development. Eur J Oral Sci. 1998;106 (Suppl 1):38–43. | ||
Ogawa T, Kapadia H, Feng JQ, Raghow R, Peters H, D’Souza RN. Functional consequences of interactions between Pax9 and Msx1 genes in normal and abnormal tooth development. J Biol Chem. 2006;281(27):18363–18369. | ||
Ogawa T, Kapadia H, Wang B, D’Souza RN. Studies on Pax9-Msx1 protein interactions. Arch Oral Biol. 2005;50(2):141–145. | ||
Isman E, Nergiz S, Acar H, Sari Z. PAX9 polymorphisms and susceptibility with sporadic tooth agenesis in Turkish populations: a case-control study. BMC Genomics. 2013;14:733. | ||
Mostowska A, Biedziak B, Trzeciak WH. A novel mutation in PAX9 causes familial form of molar oligodontia. Eur J Hum Genet. 2006;14(2):173–179. | ||
Frazier-Bowers SA, Scott MR, Cavender A, Mensah J, D’Souza RN. Mutational analysis of families affected with molar oligodontia. Connect Tissue Res. 2002;43(2-3):296–300. | ||
Jumlongras D, Lin JY, Chapra A, et al. A novel missense mutation in the paired domain of PAX9 causes non-syndromic oligodontia. Hum Genet. 2004;114(3):242–249. | ||
Klein ML, Nieminen P, Lammi L, Niebuhr E, Kreiborg S. Novel mutation of the initiation codon of PAX9 causes oligodontia. J Dent Res. 2005;84(1):43–47. | ||
Kobielak A, Kobielak K, Wiśniewski AS, Mostowska A, Biedziak B, Trzeciak WH. The novel polymorphic variants within the paired box of the PAX9 gene are associated with selective tooth agenesis. Folia Histochem Cytobiol. 2001;39(2):111–112. | ||
Lammi L, Halonen K, Pirinen S, Thesleff I, Arte S, Nieminen P. A missense mutation in PAX9 in a family with distinct phenotype of oligodontia. Eur J Hum Genet. 2003;11(11):866–871. | ||
Zhao J, Hu Q, Chen Y, Luo S, Bao L, Xu Y. A novel missense mutation in the paired domain of human PAX9 causes oligodontia. Am J Med Genet A. 2007;143A(21):2592–2597. | ||
Hansen L, Kreiborg S, Jarlov H, Niebuhr E, Eiberg H. A novel nonsense mutation in PAX9 is associated with marked variability in number of missing teeth. Eur J Oral Sci. 2007;115(4):330–333. | ||
Nieminen P, Arte S, Tanner D, et al. Identification of a nonsense mutation in the PAX9 gene in molar oligodontia. Eur J Hum Genet. 2001;9(10):743–746. | ||
Tallón-Walton V, Manzanares-Céspedes MC, Arte S, et al. Identification of a novel mutation in the PAX9 gene in a family affected by oligodontia and other dental anomalies. Eur J Oral Sci. 2007;115(6):427–432. | ||
Mitsui SN, Yasue A, Masuda K, et al. Novel PAX9 mutations cause non-syndromic tooth agenesis. J Dent Res. 2014;93(3):245–249. | ||
Shahid M, Balto HA, Al-Hammad N, et al. Mutations in MSX1, PAX9 and MMP20 genes in Saudi Arabian patients with tooth agenesis. Eur J Med Genet. 2016;59(8):377–385. | ||
Liang J, Qin C, Yue H, He H, Bian Z. A novel initiation codon mutation of PAX9 in a family with oligodontia. Arch Oral Biol. 2016;61:144–148. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.