Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 18
The Relationship Between Prognostic Nutritional Indexes and the Clinical Outcomes of Patients with Acute Exacerbation of Chronic Obstructive Pulmonary Disease
Authors Yuan FZ , Xing YL, Xie LJ, Yang DL, Shui W, Niu YY, Zhang X, Zhang CR
Received 11 January 2023
Accepted for publication 30 May 2023
Published 12 June 2023 Volume 2023:18 Pages 1155—1167
DOI https://doi.org/10.2147/COPD.S402717
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Russell
Fu-Zhen Yuan,1 Yan-Li Xing,2 Liang-Jie Xie,1 Dong-Ling Yang,1 Wei Shui,2 Yuan-Yuan Niu,2 Xin Zhang,2 Chang-Ran Zhang2
1Department of General Practice Medicine, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, 510080, People’s Republic of China; 2Department of General Medicine, East Hospital, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, 510700, People’s Republic of China
Correspondence: Chang-Ran Zhang, Department of General Medicine, East Hospital, The First Affiliated Hospital of Sun Yat-sen University, No. 183, Huangpu East Road, Huangpu District, Guangzhou, Guangdong Province, 510700, People’s Republic of China, Tel +86-13711409903, Fax +86-20-82379480, Email [email protected]
Purpose: Nutritional status is related to the clinical outcomes of patients with acute exacerbation of chronic obstructive pulmonary disease (AECOPD). The aim of this study was to investigate the association between nutritional status, measured by the prognostic nutritional index (PNI), and adverse hospitalization outcomes in patients with AECOPD.
Methods: Consecutive AECOPD patients admitted to the First Affiliated Hospital of Sun Yat-sen University between January 1, 2015 to October 31, 2021 were enrolled. We collected the clinical characteristics and laboratory data of patients. Multivariable logistic regression models were developed to assess the relationship between the baseline PNI and adverse hospitalization outcomes. A generalized additive model (GAM) was used to identify any non-linear relationship. In addition, we performed a subgroup analysis to tested the robustness of the results.
Results: A total of 385 AECOPD patients were involved in this retrospective cohort study. Based on the tertiles of PNI, patients in the lower tertiles of PNI showed more worse outcome incidence (30 [23.6%] versus 17 [13.2%] versus 8 [6.2%]; p < 0.001). Multivariable logistic regression analysis revealed that the PNI were independently associated with adverse hospitalization outcomes after adjustment for confounding factors (Odds ratio [OR] = 0.94, 95% CI: 0.91 to 0.97, P < 0.0001). After adjusting for confounders, smooth curve fitting showed a saturation effect, suggesting that the relationship between the PNI and adverse hospitalization outcomes was nonlinear. Two-piecewise linear regression model suggested that the incidence of adverse hospitalization outcomes significantly decreased with PNI level up to the inflection point (PNI = 42), and PNI was not associated with adverse hospitalization outcome after that point.
Conclusion: Decreased PNI levels at admission were determined to be associated with adverse hospitalization outcomes in patients with AECOPD. The results obtained in this study may potentially assist clinicians optimize risk evaluations and clinical management processes.
Keywords: AECOPD, nutrition, prognostic nutritional index, adverse hospitalization outcomes, non-linearity
Introduction
Chronic obstructive pulmonary disease (COPD) is characterized by chronic airway inflammation and lung parenchymal destruction, which leads to irreversible airflow limitation.1 As reported by the World Health Organization, 3.23 million people worldwide died from COPD in 2019, and more than 90% of those deaths occurred in low- and middle-income countries, including China.2 It is estimated that more than 5.4 million people will die each year from COPD and related diseases by 2060.3 Acute exacerbation of COPD (AECOPD) is the most important factor affecting the prognosis of patients with COPD and is the most common cause of hospitalization and death in COPD patients.1,4
Malnutrition often occurs in elderly hospitalized patients, and is associated with impaired respiratory function.5 It has been reported that 30 to 60% of patients hospitalized with COPD are malnourished.6,7 Among patients with COPD, malnutrition is associated with impaired lung function, increased risk of hospitalization, lowered exercise tolerance, poor quality of life, and increased risks for mortality.8–11 Moreover, malnutrition greatly increases the risks of exacerbation, prolonged lengths of stay, and increased costs.12 In addition, inflammation also plays an important role in the pathogeneses as well as the outcomes of AECOPD patients.13 Studies have shown that inflammatory biomarkers such as neutrophil-to-lymphocyte ratio,14 platelet-to-lymphocyte ratio,15 and red blood cell distribution width16 can be used to predict the prognosis of patients with AECOPD. However, since this group of biomarkers is somewhat homogeneous and not comprehensive, it is crucial to find an indicator that may reflect both the inflammation and nutritional status of patients with AECOPD.
Prognostic nutritional index (PNI), based on total lymphocyte counts and serum albumin concentrations, is a new type of parameter. Collins et al showed that lymphocyte count below 1500 is often a sign of malnutrition.8 Serum albumin is also commonly used to assess nutritional status and is closely related to the severity of malnutrition.17 Several studies have shown that COPD patients with hypoalbuminemia has been associated with a prolonged length of hospital stay during acute exacerbation, acute respiratory failure, and increased mortality.18,19 Therefore, the PNI takes into account both lymphocyte counts and serum albumin concentration levels to give a more accurate indication of the patients’ inflammation and nutritional conditions. PNI has recently been identified as a reliable and independent prognostic indicator for malignancy,20 autoimmune disease,21 and heart failure.22 However, few studies have explored the effects of PNI on prognosis of patients with AECOPD. The purpose of this study was to examine the clinical significance of PNI in patients hospitalized for AECOPD.
Methods
Study Design and Population
More detailed information regarding this study has been previously published.23 In summary, we conducted an observational study of AECOPD patients who were admitted to the First Affiliated Hospital of Sun Yat-sen University between January 1, 2015 and October 31, 2021. A total of 385 patients with AECOPD (excluding 3 patients with missing serum albumin or lymphocyte count) were included in this analysis, as shown in Figure 1. When a patient had multiple admissions to the hospital, only the first admission was recorded. The inclusion criteria were as follows: Patients were diagnosed with AECOPD at admission and at least 40 years of age or more. The exclusion criteria included other respiratory diseases (such as tuberculosis, asthma, bronchiectasis, lung cancer, interstitial lung disease), septic shock, severe cardiovascular disease (including acute left heart failure), autoimmune connective tissue diseases, and hematologic diseases (including chronic lymphocytic leukemia, multiple myeloma, and so on).
 |
Figure 1 Flowchart of the study participants. Abbreviations: AECOPD, acute exacerbation of chronic obstructive pulmonary disease; ICU, intensive care unit. |
The study was approved by the Ethics Committee of The First Affiliated Hospital of Sun Yat‐sen University (Ethics No. [2022]-179) and complied with the Declaration of Helsinki. It should be noted that due to the anonymous nature of the data, this study obtained an exemption for informed consent from the Ethics Committee.
Data Collection
All data used in this study were collected from the electronic hospital information system. The information included demographic data, comorbidities, and laboratory findings were reviewed, such as age, sex, BMI, smoking history, comorbidities (including hypertension, diabetes, arrhythmia, congestive heart failure, chronic kidney disease and coronary artery disease). In addition, we collected the age-adjusted Charlson Comorbidity Index (aCCI), which was calculated from published methodology.24 The biochemistry measurements included leukocytes, lymphocytes, neutrophils, monocytes, eosinophils (Eos), hemoglobin (Hb), platelets (PLT), red blood cells, procalcitonin (PCT), C-reactive protein (CRP), lactic dehydrogenase (LDH), N-terminal probrain natriuretic peptide (NT-proBNP), creatinine (Cr), blood urea nitrogen (BUN), albumin, serum potassium, serum sodium, serum calcium, total cholesterol (TC), triglyceride (TG), low-density lipoprotein cholesterol (LDL-c), high-density lipoprotein cholesterol (HDL-c), pH, partial pressure of carbon dioxide (PaCO2). The formula used for calculating the PNI was as follows: PNI = 10×serum albumin (g/dL)+0.005×total lymphocyte count (per mm3).25
The clinical outcomes of each patient were recorded. Poor hospitalization outcomes were considered for patients who met any one of these three criteria: Intensive care unit (ICU) admission, invasive ventilator required, or death in hospital.
Statistical Analysis
The baseline characteristics were described and compared by the tertiles of the PNI. Continuous variables are presented as mean ± SD (normal distribution) or medians and IQR (skewed distribution), while categorical variables are presented as number (%). The differences among the groups were assessed using Chi-Squared tests for the categorical variables, the One-Way Anova tests for normally distributed continuous variables. We used multiple imputations (MIs), based on five replications and a chained equation approach method in the R MI procedure,26,27 to account for missing data on the most important covariate.
Firstly, Univariate and multivariable logistic regression models were used to evaluate the relationship between the PNI and the adverse hospitalization outcomes. We evaluated the relationship of the PNI as a continuous variable with the most adverse hospitalization outcomes, and then we examined the relationship when PNI was treated as variable categorized into tertiles. In the multivariable adjusted models, sex, age, BMI, smoking status, comorbidities, leukocytes, PLT, Eos, Hb, BUN, TC, NT-proBNP, pH, PaCO2, serum potassium, serum calcium, serum sodium and required NIMV were included. The following principles were used to determine whether the potential confounders should be adjusted: Previously used or reported as relevant,28 and the potential confounders effect estimates individually changed by at least 10%.29 Secondly, potential nonlinear associations between the PNI levels and the incidence of clinical outcomes were examined with spline smoothing plot. Once non-linear relationship was found, a recursive algorithm was used to objectively calculate the inflection point. This was followed by two-piecewise linear regressions on either side of the inflection point. The optimal fitting model was determined according to the p value of logarithmic likelihood ratio test. Finally, subgroup analysis was conducted using a stratified linear regression model. Interactions were evaluated using likelihood ratio tests. Due to 18.9% of the patients missing BMI and 35% of the patients missing TC, MIs were used for the models with BMI and TC. Five imputed data sets were generated and then pooled to obtain the imputed results.27 All of the analyses were performed using Empower (R) (http://www.empowerstats.net/cn/, X&Y Solutions, Inc., Boston, MA) and R (http://www.R-project.org, The R Foundation). In this study, p value < 0.05 was considered statistically significant.
Results
Demographic Characteristics
A total of 385 patients included in the final analyses, the mean (standard deviation) age of the cohort was 74.2 (8.7) years, and 87.8% of the participants were male. The overall number of patients with adverse in-hospital outcomes was 55 (14.3%). Table 1 details the baseline demographic and biochemical characteristics which were compared according to the PNI tertiles. It can be seen that the patients with higher PNI tertiles were younger, with higher BMI, eosinophils counts, lymphocyte counts, platelet counts and hemoglobin levels than the patients with low and middle PNI tertiles (P < 0.05). It was observed that the total protein, albumin, TC, TG, and LDL-c increased with increasing tertiles of the PNI, but levels of BUN, PaCO2, PCT, CRP, LDH and NT-proBNP decreased. However, there were no significant differences observed in PNI tertiles groups in regard to aCCI, HDL-c and Cr. Moreover, individuals in the low and middle tertiles were more likely to have underlying comorbidities, including coronary artery disease, congestive heart failure, chronic kidney disease, diabetes and hypertension. The incidence of poor hospitalization outcomes significantly increased across the PNI tertiles (23.6% versus 13.2% versus 6.2% for Tertile 1 versus Tertile 2 versus Tertile 3, respectively). The results of the univariate logistic regression models for the baseline variables and adverse hospitalization outcomes are shown in Table 2. The univariate analysis indicated that DBP, coronary artery disease, lymphocytes, eosinophils, red blood cells, hemoglobin, albumin, serum calcium, TC, pH, and PNI were negatively correlated with poor hospitalization outcomes. However, platelets, D-dimer, BUN, serum potassium, length of stay, required NIMV, and PaCO2 values were positively correlated with the risks of adverse hospitalization outcomes.
 |
Table 1 Baseline Characteristics of the Cohort Study Population by Prognostic Nutritional Index Tertiles |
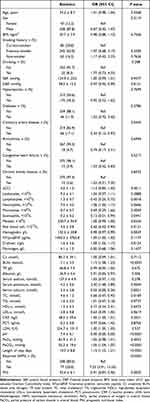 |
Table 2 Univariate Analysis for Adverse Hospitalization Outcomes |
Association Between the PNI and Adverse Hospitalization Outcomes
The results of multivariable logistic regression models examining the independent effects of PNI on adverse hospitalization outcomes are presented in Table 3. According to the crude model, the odds ratios of adverse hospitalization outcomes significantly decreased as the PNI increased and the tertiles of the PNI upgraded. There was a 9% decrease in the risks of incidence of adverse outcomes per single unit increase in the PNI (OR = 0.91; 95% CI: 0.89–0.93; P<0.0001). The OR for Tertile 3 was significantly lower than the OR for Tertile 1 (OR = 0.21; 95% CI: 0.09–0.49; P=0.0002). After accounting for sex, age, BMI, and smoking status in Model 1, PNI remained independently related to poor hospitalization outcomes, the middle tertile (OR = 0.47; 95% CI: 0.24–0.91; P=0.0253) and high tertile (OR = 0.20; 95% CI: 0.09–0.47; P=0.0002). With further adjustments for comorbidities, WBC, PLT, Eos, Hb, BUN, TC, NT-proBNP, pH, PaCO2, serum sodium, serum potassium, serum calcium and required NIMV, the risks of adverse hospitalization outcomes were significantly reduced by 6% (OR = 0.94; 95% CI: 0.91–0.97; P < 0.0001) with each 1 unit increase in the PNI. Meanwhile, compared to the lowest PNI tertile, the risks of adverse hospitalization outcomes in the middle and highest PNI tertiles were reduced by 24% (OR = 0.76, 95% CI: 0.31–1.88) and 63% (OR = 0.37, 95% CI: 0.10–1.34), respectively. In addition, considering that COPD often coexists with other diseases that may have a significant impact on disease course, we further adjusted the aCCI based on the variables adjusted in model 1 and model 2, and the results were robust (Table S1).
 |
Table 3 Unadjusted and Adjusted Logistic Regression Models of PNI and Adverse Hospitalization Outcomes |
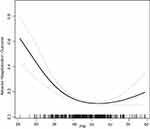
Non-Linearity Relationship of the PNI and Adverse Hospitalization Outcomes
The dose-response relationship between the PNI and the adverse hospitalization outcomes is presented in Figure 2. A nonlinear relationship (adjusted for age, sex, BMI, smoking status, comorbidities, leukocytes, PLT, Eos, Hb, BUN, TC, NT-proBNP, pH, PaCO2, serum sodium, serum potassium, serum calcium and required NIMV) was observed between the PNI and the adverse hospitalization outcomes. The incidence of poor hospitalization outcomes decreased with increases in the PNI when the PNI was less than a certain threshold. Further, two- piecewise linear regression and linear regression models were used to fit the associations between the PNI and the adverse outcomes, respectively (Table 4). The P value less than 0.001 for the log-likelihood ratio test indicated that the two-side linear regression was more appropriate for fitting the association between them. The inflection point was 42. Effect size, 95% CI and P value for the left side of the inflection point (PNI < 42) were 0.87, 0.82 to 0.92 and P < 0.0001, respectively. The results showed that when the PNI were less than 42, each unit increase in the PNI was associated with a 13% decrease in the incidence of adverse outcomes. However, the relationship was not observed on the right side of the inflection point (PNI ≥ 42) (OR = 1.05, 95% CI: 0.99–1.11, P = 0.0799). Although it was opposite to the reduced risks of adverse hospitalization outcomes on the left side of the inflection point, it lacked a statistically significant association. It appeared that a threshold effect existed for the independent association between the PNI and the adverse outcomes during hospitalization. In the two-piecewise linear regression model and GAM model, we further adjusted the aCCI based on the original model, and the results were still robust (Table S2 and Figure S1).
 |
Table 4 The Results of the Two-Piecewise Linear Regression Model |
Subgroup Analysis
We performed subgroup analysis to identify the consistency of association between the PNI and the adverse hospitalization outcomes in patients with AECOPD, considering the influence of age (< 75 versus ≥ 75 years), sex (female versus male), BMI (< 20.56 versus ≥ 20.56kg/m2), smoking history (current-smoker versus previous smoker versus non-smoker), diabetes (yes versus no), required NIMV (yes versus no), TC (< 4.6 versus ≥ 4.6mmol/L), Hb (<135 versus ≥ 135g/L), and PaCO2 (≤ 45 versus > 45mmHg) on the prognosis of AECOPD (Table 5). In the stratified analysis, the associations between those factors were similar for all strata (P > 0.05).
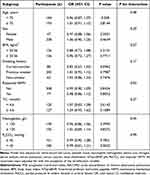 |
Table 5 Subgroup Analyses of the Association Between PNI and Adverse Hospitalization Outcomes of AECOPD Patients |
Discussion
The present study demonstrated that, based on PNI, nutrition status was independently associated with adverse hospitalization outcomes in patients with AECOPD. After adjusting for potential confounders, it was found that a positive correlation existed between the PNIs and the adverse hospitalization outcomes of the AECOPD patients. The same trend was observed when the PNI was treated as a categorical variable. Our analysis revealed non-linearity relationship between PNI and adverse hospitalization outcomes. On the left side of the inflection point, the PNI was inversely associated with adverse clinical outcomes. Therefore, according to this study’s findings, patients with AECOPD should be given nutritional assessments upon admission to hospital.
Albumin levels in serum reflect visceral protein storage, and depletion of albumin may be a sign of severe malnutrition. Research has shown that hypoalbuminemia may be caused by poor nutrition and absorption, aging, comorbid conditions, and proinflammatory cytokines that inhibit albumin production.30 Each of those factors have an accumulative impacts on hypoalbuminemia risks31 and may coexist in patients with COPD. Malnutrition exacerbates the effects of airflow obstruction, since inefficient ventilation increases the amount of energy required each day.32 The onset of malnutrition and weight loss in patients with COPD result in poor prognoses as the disease progresses. Lin et al analyzed 692 patients with AECOPD and observed that hypoproteinemia was a significant risk factor for early hospital readmission of COPD patients.33 The results revealed that hypoproteinemia more than doubled the risks of COPD early readmission (OR = 2.018, 95% CI: 1.031–3.947, p = 0.04). In addition, several studies have also demonstrated that COPD patients with hypoalbuminemia have higher mortality rates.18,34 Based on a meta-analysis, there was a significant difference between serum albumin concentrations in COPD and non-COPD patients.30 Lymphocytes participate in immune surveillance and immunoediting processes. Therefore, decreases in lymphocyte functions and counts are indications of impaired immune defense.35 Malnutrition is most commonly defined as a serum albumin level less than 3.5 g/dL or a total lymphocyte count of less than 1500 cells (per mm3).36 Prognostic nutritional index is calculated from total lymphocyte counts and serum albumin concentrations and could potentially indicate both chronic inflammation and malabsorption in AECOPD patients.
To the best of our knowledge, no previous studies have investigated the relationship between PNI and adverse hospitalization outcomes in AECOPD patients. In the present single-center retrospective study, we found that patients in the low tertile group had significantly lower BMI, leukocytes and lymphocytes counts, hemoglobin and serum albumin levels. In addition, the low tertile group also had significantly higher incidence of adverse outcomes and longer stays in hospital. Moreover, we found that after adjustment for potential confounders, the risks of adverse hospitalization outcomes was significantly reduced by 6% (OR = 0.94; 95% CI: 0.91–0.97; P < 0.0001) with each single unit increase in the PNI. Meanwhile, compared to the lowest PNI tertile, the risks of adverse hospitalization outcomes in the middle and highest PNI tertiles were reduced by 24% (OR = 0.76, 95% CI: 0.31–1.88) and 63% (OR = 0.37, 95% CI: 0.10–1.34), respectively. The results suggested that AECOPD patients with a lower PNI presented higher risks of adverse hospital outcomes. The results of subgroup analysis indicated that a stable relationship existed between the PNI and the adverse hospitalization outcomes, and no significant interaction effects were observed among the different subgroups.
Malnutrition is a common occurrence in COPD patients, resulting in poorer prognoses. However, despite its importance, malnutrition is often underappreciated in the treatments of patients with COPD. Ferreira et al revealed that nutrient supplementation resulted significant improvements in 6-minute walk test performances, respiratory muscle strength, and the health status of COPD patients with malnutrition.37 Therefore, based on the high correlation between nutrition and adverse hospitalization outcomes, this study suggested that AECOPD patients should undergo nutritional assessments and nutritional intervention at the time of hospital admission.
COPD is also often coexists with other diseases that may have a significant impact on disease course. In 1987, Charles et al proposed to use the comorbidity status of patients to predict and assess the risk of death in hospitalized patients, thus establishing the Charlson Comorbidity Index (CCI).24 The age-adjusted Charlson Comorbidity Index (aCCI), corrects the final CCI score for the age of the patient. Previous studies showed that severe AECOPD is associated with a higher prevalence of comorbidities and increased hospital readmission rate.38 In analysis of 1.6 million COPD discharges, Buhr et al found that readmission odds increased by 9% for each half standard deviation increase of CCI scores.39 However, we did not observe the association between aCCI and adverse hospitalization outcomes in our study. Moreover, In both unadjusted and adjusted analyses, aCCI seemed to have little effect on our core results (Additional file 2). We speculate that this is the result of the following possible causes. First, our sample size is still relatively small. Second, we excluded patients with tumors, myocardial infarction, leukemia, lymphoma, and autoimmune connective tissue diseases, which may affect the observation of the association between aCCI and adverse hospitalization outcomes in AECOPD patients. Therefore, a prospective cohort study with a large sample size is needed in the future to provide more reliable findings.
There are several strengths of our study. Firstly, since the PNI could be quickly calculated based on routine tests at the time of admission, clinicians may be able to identify high risk patients with AECOPD in the early stages. As a result, treatments could be modified accordingly to reduce the poor hospitalization outcomes. Secondly, this was a retrospective observational study, which was susceptible to potential confounding factors. In order to reduce residual confounding, we used strict statistical adjustment methods. In addition, we tested the robustness of the results by performing subgroup analysis in different subgroups.
However, there are also several disadvantages from the present study. Firstly, since this observational study was a retrospective design from a single medical center, the results may not be generalizable to other centers. Secondly, we excluded patients with various tumors and patients with acute myocardial infarction, leukemia, lymphoma, connective tissue disease and so on. Therefore, the findings could not be extrapolated to those patients. Finally, the data of the BMI and TC covariates were missing in 18.9% and 35% of the patients, respectively. However, multiple imputations were used to address the problem of missing data, and the results were still robust.
Conclusion
In conclusion, the PNI on admission to hospital was closely related to adverse hospitalization outcomes in AECOPD patients, showing a nonlinear relationship, and when the PNI was less than 42, the adverse hospitalization outcomes were negatively correlated with the PNI levels.
Abbreviations
COPD, chronic obstructive pulmonary disease; AECOPD, acute exacerbation of chronic obstructive pulmonary disease; PNI, prognostic nutritional index; BMI, body mass index; DBP, diastole blood pressure; SBP, systolic blood pressure; NT-proBNP, N-terminal probrain natriuretic peptide; aCCI, age-adjusted Charlson Comorbidity Index; Cr, creatinine; BUN, blood urea nitrogen; TP, total protein; TG, triglyceride; TC, total cholesterol; LDL-c, low-density lipoprotein cholesterol; HDL-c, high-density lipoprotein cholesterol; Eos, eosinophils; PLT, platelet; Lym, lymphocyte; Hb, hemoglobin; CRP, C-reactive protein; PCT, procalcitonin; LDH, lactic dehydrogenase; PaCO2, partial pressure of carbon dioxide in arterial blood; PaO2, partial pressure of oxygen in arterial blood; ICU, intensive care unit; NIMV, noninvasive mechanical ventilation; MIs, multiple imputations; OR, odd ratios; CI, confidence intervals.
Data Sharing Statement
The dataset used and analyzed during the current study can be accessed by reasonable request from the authors.
Ethics Approval and Informed Consent
This study was approved by the Ethics Review Committee of the First Affiliated Hospital of Sun Yat-sen University in April of 2022 (reference number: [2022]-179). Due to the anonymous nature of the data, an exemption for informed consent was obtained from the Investigational Ethical Review Board.
Acknowledgments
The authors would like to thank all the study participants and the First Affiliated Hospital of Sun Yat-sen University.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
The authors declare that this is a non-funded study.
Disclosure
The authors declare that they have no conflicts of interest in this work.
References
1. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for diagnosis, management, and prevention of COPD; 2022. Available from: https://goldcopd.org/.
2. World Health Organization. World Health Organization (WHO) website. Available from: https://www.who.int/zh/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd).
3. Fitzmaurice C, Abate D, Abate D, et al.; Global Burden of Disease Cancer Collaboration. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the Global Burden of Disease Study. JAMA Oncol. 2019;5(12):1749–1768. doi:10.1001/jamaoncol.2019.2996
4. Papi A, Luppi F, Franco F, Fabbri LM. Pathophysiology of exacerbations of chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2006;3(3):245–251. doi:10.1513/pats.200512-125SF
5. Grant JP. Nutrition care of patients with acute and chronic respiratory failure. Nutr Clin Pract. 1994;9(1):11–17. doi:10.1177/011542659400900111
6. Collins PF, Elia M, Stratton RJ. Nutritional support and functional capacity in chronic obstructive pulmonary disease: a systematic review and meta-analysis. Respirology. 2013;18(4):616–629. doi:10.1111/resp.12070
7. King DA, Cordova F, Scharf SM. Nutritional aspects of chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2008;5(4):519–523. doi:10.1513/pats.200707-092ET
8. Collins PF, Elia M, Kurukulaaratchy RJ, Stratton RJ. The influence of deprivation on malnutrition risk in outpatients with chronic obstructive pulmonary disease (COPD). Clin Nutr. 2018;37(1):144–148. doi:10.1016/j.clnu.2016.11.005
9. Collins PF, Stratton RJ, Kurukulaaratchy RJ, Elia M. Influence of deprivation on health care use, health care costs, and mortality in COPD. Int J Chron Obstruct Pulmon Dis. 2018;13:1289–1296. doi:10.2147/COPD.S157594
10. Gunay E, Kaymaz D, Selcuk NT, Ergun P, Sengul F, Demir N. Effect of nutritional status in individuals with chronic obstructive pulmonary disease undergoing pulmonary rehabilitation. Respirology. 2013;18(8):1217–1222. doi:10.1111/resp.12133
11. Nguyen HT, Collins PF, Pavey TG, Nguyen NV, Pham TD, Gallegos DL. Nutritional status, dietary intake, and health-related quality of life in outpatients with COPD. Int J Chron Obstruct Pulmon Dis. 2019;14:215–226. doi:10.2147/COPD.S181322
12. Hoong JM, Ferguson M, Hukins C, Collins PF. Economic and operational burden associated with malnutrition in chronic obstructive pulmonary disease. Clin Nutr. 2017;36(4):1105–1109. doi:10.1016/j.clnu.2016.07.008
13. Liu Y, Yang X, Kadasah S, Peng C. Clinical value of the prognostic nutrition index in the assessment of prognosis in critically ill patients with stroke: a retrospective analysis. Comput Math Methods Med. 2022;2022:4889920. doi:10.1155/2022/4889920
14. Emami Ardestani M, Alavi-Naeini N. Evaluation of the relationship of neutrophil-to lymphocyte ratio and platelet-to-lymphocyte ratio with in-hospital mortality in patients with acute exacerbation of chronic obstructive pulmonary disease. Clin Respir J. 2021;15(4):382–388. doi:10.1111/crj.13312
15. Kumar P, Law S, Sriram KB. Evaluation of platelet lymphocyte ratio and 90-day mortality in patients with acute exacerbation of chronic obstructive pulmonary disease. J Thorac Dis. 2017;9(6):1509–1516. doi:10.21037/jtd.2017.05.77
16. Hu GP, Zhou YM, Wu ZL, et al. Red blood cell distribution width is an independent predictor of mortality for an acute exacerbation of COPD. Int J Tuberc Lung Dis. 2019;23(7):817–823. doi:10.5588/ijtld.18.0429
17. Zhang Z, Pereira SL, Luo M, Matheson EM. Evaluation of blood biomarkers associated with risk of malnutrition in older adults: a systematic review and meta-analysis. Nutrients. 2017;9(8):829. doi:10.3390/nu9080829
18. Wang Y, Stavem K, Dahl FA, Humerfelt S, Haugen T. Factors associated with a prolonged length of stay after acute exacerbation of chronic obstructive pulmonary disease (AECOPD). Int J Chron Obstruct Pulmon Dis. 2014;9:99–105. doi:10.2147/COPD.S51467
19. Chen CW, Chen YY, Lu CL, et al. Severe hypoalbuminemia is a strong independent risk factor for acute respiratory failure in COPD: a nationwide cohort study. Int J Chron Obstruct Pulmon Dis. 2015;10:1147–1154. doi:10.2147/COPD.S85831
20. Ucar G, Ergun Y, Acikgoz Y, Uncu D. The prognostic value of the prognostic nutritional index in patients with metastatic colorectal cancer. Asia Pac J Clin Oncol. 2020;16(5). doi:10.1111/ajco.13328
21. Ye Z, Ai X, Liao Z, You C, Cheng Y. The prognostic values of neutrophil to lymphocyte ratio for outcomes in chronic obstructive pulmonary disease. Medicine. 2019;98(28):e16371. doi:10.1097/MD.0000000000016371
22. Zencirkiran Agus H, Kahraman S. Prognostic nutritional index predicts one-year outcome in heart failure with preserved ejection fraction. Acta Cardiol. 2020;75(5):450–455. doi:10.1080/00015385.2019.1661139
23. Yuan FZ, Shui W, Xing YL, Niu YY, Zhang X, Zhang CR. Association of red cell index and adverse hospitalization outcomes in chronic obstructive pulmonary disease patients with acute exacerbation: a retrospective cohort study. Int J Chron Obstruct Pulmon Dis. 2022;17:2263–2275. doi:10.2147/COPD.S373114
24. Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245–1251. doi:10.1016/0895-4356(94)90129-5
25. Mohri Y, Inoue Y, Tanaka K, Hiro J, Uchida K, Kusunoki M. Prognostic nutritional index predicts postoperative outcome in colorectal cancer. World J Surg. 2013;37(11):2688–2692. doi:10.1007/s00268-013-2156-9
26. Austin PC, White IR, Lee DS, van Buuren S. Missing data in clinical research: a tutorial on multiple imputation. Can J Cardiol. 2021;37(9):1322–1331. doi:10.1016/j.cjca.2020.11.010
27. Beesley LJ, Bondarenko I, Elliot MR, Kurian AW, Katz SJ, Taylor JM. Multiple imputation with missing data indicators. Stat Methods Med Res. 2021;30(12):2685–2700. doi:10.1177/09622802211047346
28. Peng JC, Nie F, Li YJ, Xu QY, Xing SP, Gao Y. Prognostic nutritional index as a predictor of 30-day mortality among patients admitted to intensive care unit with acute exacerbation of chronic obstructive pulmonary disease: a single-center retrospective cohort study. Med Sci Monit. 2022;28:e934687. doi:10.12659/MSM.934687
29. Romskaug R, Skovlund E, Straand J, et al. Effect of clinical geriatric assessments and collaborative medication reviews by geriatrician and family physician for improving health-related quality of life in home-dwelling older patients receiving polypharmacy: a cluster randomized clinical trial. JAMA Intern Med. 2020;180(2):181–189. doi:10.1001/jamainternmed.2019.5096
30. Zinellu E, Fois AG, Sotgiu E, et al. Serum albumin concentrations in stable chronic obstructive pulmonary disease: a systematic review and meta-analysis. J Clin Med. 2021;10(2):269. doi:10.3390/jcm10020269
31. Cabrerizo S, Cuadras D, Gomez-Busto F, Artaza-Artabe I, Marin-Ciancas F, Malafarina V. Serum albumin and health in older people: review and meta analysis. Maturitas. 2015;81(1):17–27. doi:10.1016/j.maturitas.2015.02.009
32. Kim V, Kretschman DM, Sternberg AL, DeCamp MM, Criner GJ; National Emphysema Treatment Trial Research Group. Weight gain after lung reduction surgery is related to improved lung function and ventilatory efficiency. Am J Respir Crit Care Med. 2012;186(11):1109–1116. doi:10.1164/rccm.201203-0538OC
33. Lin J, Xu Y, Wu X, et al. Risk factors associated with chronic obstructive pulmonary disease early readmission. Curr Med Res Opin. 2014;30(2):315–320. doi:10.1185/03007995.2013.858623
34. Gunen H, Hacievliyagil SS, Kosar F, et al. Factors affecting survival of hospitalised patients with COPD. Eur Respir J. 2005;26(2):234–241. doi:10.1183/09031936.05.00024804
35. Lao X, Ma L, Ma Q, et al. Hematological factors associated with immunity, inflammation, and metabolism in patients with systemic lupus erythematosus: data from a Zhuang cohort in Southwest China. J Clin Lab Anal. 2020;34(6):e23211. doi:10.1002/jcla.23211
36. Morey VM, Song YD, Whang JS, Kang YG, Kim TK. Can serum albumin level and total lymphocyte count be surrogates for malnutrition to predict wound complications after total knee arthroplasty? J Arthroplasty. 2016;31(6):1317–1321. doi:10.1016/j.arth.2015.12.004
37. Gouzi F, Maury J, Héraud N, et al. Additional effects of nutritional antioxidant supplementation on peripheral muscle during pulmonary rehabilitation in COPD patients: a randomized controlled trial. Oxid Med Cell Longev. 2019;2019:1–13. doi:10.1155/2019/5496346
38. Almagro P, Cabrera FJ, Diez J, et al. Comorbidities and short-term prognosis in patients hospitalized for acute exacerbation of COPD: the EPOC en Servicios de medicina interna (ESMI) study. Chest. 2012;142(5):1126–1133. doi:10.1378/chest.11-2413
39. Buhr RG, Jackson NJ, Kominski GF, Dubinett SM, Ong MK, Mangione CM. Comorbidity and thirty-day hospital readmission odds in chronic obstructive pulmonary disease: a comparison of the Charlson and Elixhauser comorbidity indices. BMC Health Serv Res. 2019;19(1):701. doi:10.1186/s12913-019-4549-4
 © 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2023 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.