Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 15
The Protective Effect of HBO1 on Cigarette Smoke Extract-Induced Apoptosis in Airway Epithelial Cells
Authors Chen L, Luo L, Kang N, He X, Li T, Chen Y
Received 14 October 2019
Accepted for publication 10 December 2019
Published 6 January 2020 Volume 2020:15 Pages 15—24
DOI https://doi.org/10.2147/COPD.S234634
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Chunxue Bai
Lin Chen,1–3,* Lijuan Luo,1–3,* Naixin Kang,1–3 Xue He,1–3 Tiao Li,1–3 Yan Chen1–3
1Department of Pulmonary and Critical Care Medicine, The Second Xiangya Hospital, Central South University, Changsha 410011, Hunan, People’s Republic of China; 2Research Unit of Respiratory Disease, Central South University, Changsha 410011, Hunan, People’s Republic of China; 3Diagnosis and Treatment Center of Respiratory Disease, Central South University, Changsha 410011, Hunan, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Yan Chen
Department of Pulmonary and Critical Care Medicine, The Second Xiangya Hospital, Central South University, 139 Renmin Middle Road, Changsha 410011, Hunan, People’s Republic of China
Tel +86 731 85295148
Email [email protected]
Purpose: Epigenetic modification is one of most important mechanisms underlying the pathogenesis of chronic obstructive pulmonary disease (COPD). The purpose of this study was to determine whether histone acetyltransferase binding to ORC1 (HBO1) can protect against cigarette smoke (CS)-induced cell apoptosis and sustain normal histone acetylation in COPD.
Methods: Human lung tissue samples were obtained from patients who underwent lung resection. The emphysema mouse model and HBO1 overexpressing mice were each established by intraperitoneal injection with cigarette smoke extract (CSE) or intratracheal lentiviral vectors instillation. TUNEL (terminal deoxynucleotidyl transferase dUTP nick end labeling) assays were used to assess apoptotic ratio in mice. The apoptosis of human bronchial epithelial cells (HBECs) was assayed by flow cytometry. HBO1, B-cell lymphoma-2 (BCL-2), and H3K14ac protein expression were detected by Western blotting. HBO1 mRNA expression was measured by quantitative real-time polymerase chain reaction.
Results: Protein expression of HBO1 was decreased significantly in lung tissue from COPD patients and CSE-treated emphysema mouse models. Overexpression of HBO1 attenuated CSE-induced emphysematous changes, as well as apoptosis in the lungs of COPD mice. In vitro, the HBO1 protein degraded in a time- and dose-dependent course with CSE treatment. With flow cytometry, we proved that HBO1 could reverse the apoptosis of HBECs induced by CSE. Furthermore, HBO1 overexpression promoted the expression of anti-apoptotic BCL-2 protein and enhanced H3K14 acetylation in airway epithelial cells.
Conclusion: These findings demonstrate that the key histone modulator HBO1 plays a protective role in COPD pathogenesis that may shed light on potential therapeutic targets to inhibit the progress of COPD.
Keywords: chronic obstructive pulmonary disease, apoptosis, HBO1, emphysema
Introduction
Chronic obstructive pulmonary disease (COPD) is a heterogeneous disease characterized by persistent respiratory symptoms and irreversible airflow limitation. It has become the fourth leading cause of death worldwide,1 and in the coming decades, further increases in COPD morbidity and mortality are expected.2 Epidemiological data has proved that cigarette smoking is the most common risk factor for COPD development and progression throughout the world. The airway epithelium is the first line of defense against noxious particles or gases entering the lungs. Increasing evidence has demonstrated that airway epithelial cell dysfunction plays a critical role in COPD pathogenesis.3,4
Epigenetic alternations including DNA methylation, post-modifications of histone modification, and non-coding RNAs are increasingly studied in the context of COPD. In eukaryotic cells, histone acetylation is related to chromatin structure and gene expression.5 Histone acetyltransferases (HATs) and deacetylases (HDACs) are responsible for the acetylation status of a given chromatin locus.6 HATs can provide transcriptional machinery access to the DNA template by catalyzing the acetylation of histones. Conversely, HDACs are responsible for the removal of acetyl groups from the lysine residues of histones, resulting in transcriptional repression.6 HBO1 (histone acetyltransferase binding to ORC1) is a member of the HAT family that possesses a highly conserved MYST domain.7 The MYST domain contains an acetyl-CoA binding motif and a zinc finger and is responsible for the catalytic activity of HBO1.8 HBO1 acts on histones H3 and H4 and engages in a variety of biological processes including DNA replication and transcriptional regulation.9 During the G1 phase of the cell cycle, HBO1 can interact with Cdt1 and be recruited to perform the replication functions required for loading the minichromosome maintenance (MCM) complex onto origin DNA and the assembly of pre-replicative complexes (pre-RCs).10 During the S phase, HBO1 is required for the acetylation of H3K14, which is necessary for CDC45 recruitment and activation of DNA replication.11 Deleting HBO1 from embryonic cells causes a reduction of global H3K14 acetylation and decreases the expression of genes that regulate embryonic development.12 Conversely, HBO1 can lead to excessive cell replication and proliferation in tumors.13–15
In the present study, we assessed the involvement of HBO1 in COPD pathogenesis.
Materials And Methods
Subjects
Peripheral lung tissue specimens were obtained from patients who underwent resection for lung cancers. The tissue specimens were resected at least 5 cm away from the cancerous tissue. We included 32 subjects: nonsmokers (n=15), and smokers with COPD (n=5) and without COPD (n=12). The diagnosis of COPD was followed by the guidelines of the Global Initiative for Chronic Obstructive Pulmonary Disease (GOLD) (http://www.goldcopd.com). The patients with COPD had airflow limitation (forced expiratory volume in 1 s/forced vital capacity [FEV1/FVC] <0.7). Subject age, sex, smoking history, and lung function (FEV1pred and FEV1/FVC) are listed in Table 1. Exclusion criteria consisted of lung diseases other than COPD and lung tumors, other systemic diseases, pulmonary infection and antibiotic exposure up to 4 weeks before surgery, use of inhaled or oral glucocorticoids up to 3 months before surgery, a history of chemotherapy or radiotherapy, and a history of occupational exposure. The study was approved by the Clinical Trial and Ethics Committee of the Second Xiangya Hospital of Central South University. Written informed consent was obtained from all human subjects before their enrollment into the study, which was conducted in accordance with the Declaration of Helsinki.
 |
Table 1 Clinical Characteristics of the Study Subjects |
Animals
The emphysema mouse model was built according to the protocol of previous study.16 with slight modifications. Six-week-old, specific pathogen-free, BALB/c mice (21–23 g each) (Slyke Jingda, Hunan, China) were injected intraperitoneally with cigarette smoke extract (CSE) or phosphate-buffered saline (PBS) at days 0, 11, and 22 (0.3 mL per injection). Lentivirus (109 ifu/mL) (GeneChem, Shanghai, China) or PBS (0.1 mL) was injected intratracheally at day 14. The animals were sacrificed on day 28. All animal care and experimental protocols were approved by the Animal Care and Use Committee of Hunan Provincial People’s Hospital and were conducted in accordance with the committee’s animal care and use guidelines.
CSE Preparation
Cigarettes (Furong, Changde Cigarette Company, Hunan, China) were combusted using a modified syringe-driven apparatus (products of 5 cigarettes were collected in 10 mL PBS for animal experiments, and products of 1 cigarette were collected in 20 mL Dulbecco’s minimum essential media [DMEM] for cell experiments). Each cigarette contained 12 mg tar, 1.1 mg nicotine, and 14 mg carbon monoxide. The smoke was bubbled through DMEM/PBS then filtered through a 0.2-μm pore-size filter. The 100% CSE sample was titrated to a pH of 7.2–7.4 and diluted with DMEM/PBS to obtain the required concentration. CSE was freshly prepared for every experiment.
Cell Lines and Culture
HBECs were purchased from the Chinese Academy of Sciences (Shanghai, China) and cultured in DMEM (Hyclone, Logan, UT, USA) supplemented with 10% fetal bovine serum and 50 U/mL penicillin and streptomycin (Gibco,Thermo Fisher Scientific, Waltham, MA, USA) at 37°C in a 5% CO2 culture chamber. Starvation for 24 h was performed before exposure to CSE, siRNA, and/or lentivirus.
Small-Interfering RNA Preparation and Transfection
HBECs were cultured in a 12-well plate for 24 h to 40–60% confluence in complete medium without antibiotics. Then, non-targeting negative control siRNA or HBO1 siRNA (RiboBio, Guangzhou, China) were transfected at 50 nM for 48 hours following the manufacturer’s instructions.
Lentivirus HBO1 Preparation and Transfection
HBO1 and the vector lentivirus were transfected into HBECs following the manufacturer’s instructions. Lentivirus was transfected into the HBECs at a multiplicity of infection of 10. Cells positive for green fluorescent protein were considered infected.
Isolation of RNA and Real-Time Polymerase Chain Reaction (PCR)
The total RNA was isolated from HBECs using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) and then treated with DNase I (Invitrogen) according to the manufacturer’s instructions. Total RNA was then reverse-transcribed using the RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific). Subsequently, real-time PCR was performed using a StepOne Plus real-time PCR system (Life Technologies, Carlsbad, CA, USA). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as an internal control. The sequences of all primers used were:
HBO1-F: 5′-AAAAGTTGGCTCCCCAGAAC-3′
HBO1-R: 5′-GCCGTCTCCTGACTGATTTC-3′
GAPDH-F: 5′-TCAAGAAGGTGGTGAAGCAG-3′
GAPDH-R: 5′-CGTCAAAGGTGGAGGAGTG-3′
Western Blot Analysis
The total isolated soluble proteins were measured using bicinchoninic acid assay kits, separated by polyacrylamide gel electrophoresis, transferred onto polyvinylidene fluoride membranes, then incubated with antibodies to HBO1 (ab190908, Abcam, Cambridge, UK), BCL-2 (ab196495, Abcam), and H3K14ac (#7627s, Cell Signaling Technology, Danvers, MA, USA). After washing, membranes were incubated with goat anti-mouse and goat anti-rabbit IgG horseradish peroxidase-conjugated secondary antibodies (Proteintech Group, Inc., Rosemont, IL, USA). The bound complexes were detected using enhanced chemiluminescence (Santa Cruz Biotechnology, Dallas, TX, USA). The data were normalized to β-actin or H3 levels.
Lung Tissue Morphometry
Lung tissue samples were fixed in 4% formaldehyde, cut into 3.5-mm-thick sections, and stained with hematoxylin and eosin (HE). Morphometry was quantified by the values of mean linear intercept (MLI) and destructive index (DI) at a magnification of 100× as previously described.16
Lung Immunohistochemistry (IHC)
Sections were incubated with anti-HBO1 (ab190908, Abcam) to detect HBO1 expression. Scoring was performed according to the method of Chiappara et al17 with slight modification. The results were blindly evaluated by two investigators. HBO1 expression was classified according to the staining intensity: no staining (0), weak staining (1), moderate staining (2), or strong staining (3). The staining areas were scored as 0 (0%), 1 (1–10%), 2 (11–50%), and 3 (51–100%). The staining index was calculated as the product of the proportion of positive cells and the staining intensity score. High-power fields of bronchial tissues were randomly sampled, and calculations were performed on five images per section. The average score of five images from each tissue section was used for statistical analyses.
TUNEL Analysis
Terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) assays were performed with an in situ Apoptosis Detection Kit (Hoffman-La Roche Ltd., Basel, Switzerland). We randomly selected 5/9 areas to measure the apoptotic index (AI). Fields containing non-parenchymal structures such as large airways or vessels were excluded.
Flow Cytometry (FCM) Analysis
HBECs were harvested from 6-well plates and washed with PBS twice, centrifuged (800 rpm, 5 min), then mixed with 500 µL binding buffer. After resuspension, 10 µL propidium iodide (PI) and Annexin V-fluorescein isothiocyanate (FITC) were added, following the manufacturer’s instructions (KGA108, KeyGen, Nanjing, China). Finally, the HBECs were examined with the flow cytometer (A00-1-1102, Beckman, USA).
Statistical Analysis
The data are presented as mean ± SEM. Statistical significance was calculated using one-way analysis of variance (ANOVA) or Kruskal–Wallis tests.
Results
HBO1 Protein Decreased Significantly in COPD Airway Epithelium
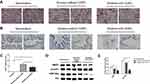
The study subjects’ baseline characteristics are presented in Table 1. Age, FEV1pred, and FEV1/FVC did not differ between groups. The numbers of pack-years between smoking groups were not significantly different. The values of FEV1pred and FEV1/FVC in the smokers with COPD group was significantly decreased compared to the nonsmokers and smokers without COPD groups.
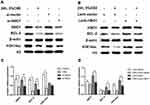
HE staining was performed to detect the degree of emphysema in peripheral lung tissue specimens. The results showed that the degree of emphysema in smokers with and without COPD was much more severe than in nonsmokers (Figure 1A). By Western blot, HBO1 protein levels were significantly downregulated in smokers with and without COPD compared to nonsmokers (Figure 1D and E). Next, IHC analysis demonstrated that HBO1 was distributed in the airway epithelium, and its abundance was significantly downregulated in smokers compared with nonsmokers. (Figure 1B and C). These results indicated that HBO1 protein levels were significantly decreased in the airway epithelium of COPD patients.
HBO1 Attenuates Cell Apoptosis and Emphysema in the Mouse Model
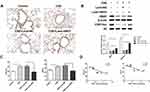
In mice exposed to CSE, HBO1 protein levels detected by Western blot and IHC were significantly decreased (Figure 2A and B). The MLI and DI values were significantly increased in the CSE group compared with the control group, and the values were decreased in Lenti-HBO1 groups compared with Lenti-NC groups (Figure 2C). Furthermore, HBO1 protein levels negatively correlated with the MLI and DI (Figure 2D). We performed TUNEL assays to clarify whether decreased HBO1 was associated with apoptosis in the CSE-exposed mouse model. The results showed significantly fewer TUNEL-positive cells in the control and Lenti-HBO1 groups compared to the CSE and Lenti-NC groups, respectively (Figure 3A and B). These results indicate that HBO1 could attenuate CSE-induced cell apoptosis and emphysema in mouse models.
HBO1 Prevents CSE-Induced Apoptosis in HBECs
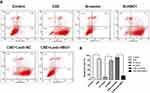
We sought to determine whether HBO1 could prevent the apoptosis of HBECs exposed to CSE. FCM analysis with Annexin V-FITC/PI staining showed HBEC apoptosis was significantly upregulated in the CSE and Si-HBO1 groups when compared with the control and Si-vector groups, respectively. HBO1 overexpression in the Lenti-HBO1 group reduced the apoptosis of HBECs exposed to CSE compared with the Lenti-NC group (Figure 4). These results illustrate that HBO1 can prevent CSE-induced apoptosis in HBECs.
CSE Decreases HBO1 Protein Expression in HBECs
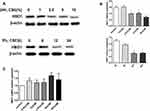
Next, we sought to elucidate the possible mechanism by HBO1 prevents CSE-induced apoptosis in HBECs. We detected HBO1 expression in HBECs exposed to CSE. Dose- and time-dependence were observed for HBEC expression of HBO1 protein (Figure 5A and B). However, mRNA levels did not differ in HBECs exposed to different concentrations of CSE (Figure 5C). These results indicate that CSE decreases HBO1 protein levels but not mRNA levels.
HBO1 Is Required for H3K14 Acetylation
To detect the possible mechanism by which HBO1 prevents CSE-induced apoptosis in HBECs, we measured the expression of H3K14ac in lung specimens and HBECs. H3K14ac protein was significantly decreased in the human specimens (Figure 1D), lung tissues of mice (Figure 2B), and HBECs (Figure 6). The protein levels of BCL-2 and H3K14ac were significantly decreased in the Si-HBO1 group compared with the Si-vector group. HBO1 overexpression in the Lenti-HBO1 group led to upregulated H3K14ac and BCL-2 protein levels (Figure 6). These results indicate that HBO1 is required for H3K14 acetylation and BCL-2 protein expression in HBECs exposed to CSE.
Discussion
Our results showed that HBO1 protein levels were significantly downregulated in the airway epithelium of patients with COPD, and HBO1 overexpression can attenuate CSE-induced cell apoptosis and emphysema in mice. In vitro, HBO1 can prevent the apoptosis of CSE-exposed HBECs. Moreover, HBO1 is required for H3K14 acetylation and BCL-2 protein expression. These findings illustrate that HBO1 is required for H3K14 acetylation and the survival of HBECs exposed to CSE.
Histone acetylation has been well documented in the pathogenesis of COPD.18 The levels of H4 acetylation in smokers and H3 acetylation in ex-smokers were significantly increased compared with nonsmokers.19 Significant decreases in HDAC activity and protein levels, attributed to histone hyperacetylation, were also reported in COPD.20 The hyperacetylation of histones is associated with pro-inflammatory cytokine expression, which is an important mechanism for the chronic inflammation in COPD. An imbalance of HATs and HDACs may affect the acetylation status of H3/H4. However, HAT activity was not significantly different in patients with or without COPD.20
HBO1 protein levels in CSE-exposed HBECs were significantly decreased, but mRNA levels were not significantly different. It seems that the effect of CSE on HBO1 expression is at the level of its stability, rather than its transcription. As a possible mechanism, HBO1 is phosphorylated by MeK1 and then targeted by the ubiquitin E3 ligase subunit FBXW15 for recognition, site-specific ubiquitination, and degradation.21
HBO1 is a member of the HAT family and exerts acetyltransferase activity towards H3 and H4 chromatin substrates by forming HBO1 complexes, including ING4/5, and scaffold proteins (JADE or BRPF).11,22,23 In our study, HBO1 was required for H3K14 acetylation in vivo and in vitro. The acetylation of H3K14 is associated with multiple physiological processes including cell proliferation,12,21,24,25 embryonic development,12 and fetal liver erythropoiesis.22 The H3 and H4 lysine residues acetylated by HBO1 were determined by the scaffold proteins, including JADE1/2/3 and BRPF1/2/3, but how HBO1 complexes modulate the acetylation of lysine 14 of H3 is still unknown.
Apoptosis of epithelial and endothelial cells is one of the most important mechanisms in COPD development.26 We observed a clear effect of HBO1 on the regulation of apoptosis in CSE-exposed HBECs, which may be a reasonable explanation for the role of HBO1 in COPD development. Kueh and colleagues demonstrated that the development of HBO1-deficient embryos was arrested at the 10-somite stage.12 The decrease in HBO1 or H3K14ac protein levels could be a useful biomarker of CSE-induced apoptosis in HBECs.
HBO1 is involved in multiple molecular processes including DNA replication origin binding and DNA-binding transcriptional factor activity.27,28 HBO1 acetylates both H3 and H4, which is important in DNA replication licensing and the completion of DNA replication. HBO1 interacts with Cdt1, associating MCM2-7 with chromatin to trigger DNA replication during the G1 phase; in the S phase, DNA replication is terminated by Cdt1 degradation and geminin recruitment.27 Cell cycle arrest in the G1 phase was reported in A549 cells with HBO1 silencing.21 The abnormalities of DNA replication and cell cycle arrest caused by HBO1 degradation may explain the apoptosis of HBECs exposed to CSE. However, the connection between H3K14 acetylation and BCL-2 expression is still unknown, and the precise molecular mechanism requires further research. Understanding the mechanism of HBO1 in modulating CSE-exposed HBEC apoptosis will help clarify the pathogenesis of COPD.
Although HBO1 plays a critical role in the pathogenesis of COPD, especially in airway epithelial cell apoptosis, the specific molecular mechanism of HBO1 on regulating histone acetylation and apoptosis needs to be further explored.
Conclusions
In summary, HBO1 protein levels were downregulated significantly in the airway epithelium of patients with COPD. HBO1 can attenuate CSE-induced cell apoptosis and emphysema in mice. In vitro, HBO1 can prevent the apoptosis of HBECs exposed to CSE. The results might be related with the level of H3K14 acetylation. These findings demonstrate that HBO1 plays a protective role in COPD pathogenesis, and enhancing its expression may be a new therapy to prevent COPD.
Acknowledgments
The authors would like to thank all the participants involved in this study.
Author Contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that there are no competing interests in this work.
References
1. Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2095–2128. doi:10.1016/S0140-6736(12)61728-0
2. Wang C, Xu J, Yang L, et al. Prevalence and risk factors of chronic obstructive pulmonary disease in China (the China Pulmonary Health [CPH] study): a national cross-sectional study. Lancet. 2018;391(10131):1706–1717. doi:10.1016/S0140-6736(18)30841-9
3. Hiemstra PS, McCray PB
4. Brody JS, Steiling K. Interaction of cigarette exposure and airway epithelial cell gene expression. Annu Rev Physiol. 2011;73:437–456. doi:10.1146/annurev-physiol-012110-142219
5. Shen Y, Wei W, Zhou DX. Histone acetylation enzymes coordinate metabolism and gene expression. Trends Plant Sci. 2015;20(10):614–621. doi:10.1016/j.tplants.2015.07.005
6. Shahbazian MD, Grunstein M. Functions of site-specific histone acetylation and deacetylation. Annu Rev Biochem. 2007;76:75–100. doi:10.1146/annurev.biochem.76.052705.162114
7. Avvakumov N, Lalonde ME, Saksouk N, et al. Conserved molecular interactions within the HBO1 acetyltransferase complexes regulate cell proliferation. Mol Cell Biol. 2012;32(3):689–703. doi:10.1128/MCB.06455-11
8. Tao Y, Zhong C, Zhu J, Xu S, Ding J. Structural and mechanistic insights into regulation of HBO1 histone acetyltransferase activity by BRPF2. Nucleic Acids Res. 2017;45(10):5707–5719. doi:10.1093/nar/gkx142
9. Doyon Y, Cayrou C, Ullah M, et al. ING tumor suppressor proteins are critical regulators of chromatin acetylation required for genome expression and perpetuation. Mol Cell. 2006;21(1):51–64. doi:10.1016/j.molcel.2005.12.007
10. Iizuka M, Stillman B. Histone acetyltransferase HBO1 interacts with the ORC1 subunit of the human initiator protein. J Biol Chem. 1999;274(33):23027–23034. doi:10.1074/jbc.274.33.23027
11. Feng Y, Vlassis A, Roques C, et al. BRPF3-HBO1 regulates replication origin activation and histone H3K14 acetylation. EMBO J. 2016;35(2):176–192. doi:10.15252/embj.201591293
12. Kueh A, Dixon M, Voss A, Thomas T. HBO1 is required for H3K14 acetylation and normal transcriptional activity during embryonic development. Mol Cell Biol. 2011;31(4):845–860. doi:10.1128/MCB.00159-10
13. Wang Y, Chen S, Tian W, et al. High-expression HBO1 predicts poor prognosis in gastric cancer. Am J Clin Pathol. 2019;152(4):517–526. doi:10.1093/ajcp/aqz065
14. Li R, Zhou R, Zhang J. Function of PM2.5 in the pathogenesis of lung cancer and chronic airway inflammatory diseases. Oncol Lett. 2018;15(5):7506–7514. doi:10.3892/ol.2018.8355
15. Quintela M, Sieglaff DH, Gazze AS, et al. HBO1 directs histone H4 specific acetylation, potentiating mechano-transduction pathways and membrane elasticity in ovarian cancer cells. Nanomedicine. 2019;17:254–265. doi:10.1016/j.nano.2019.01.017
16. Zhang Y, Cao J, Chen Y, et al. Intraperitoneal injection of cigarette smoke extract induced emphysema, and injury of cardiac and skeletal muscles in BALB/C mice. Exp Lung Res. 2013;39(1):18–31. doi:10.3109/01902148.2012.745910
17. Chiappara G, Gjomarkaj M, Virzì A, et al. The role of p21 Waf1/Cip1 in large airway epithelium in smokers with and without COPD. Biochim Biophys Acta. 2013;1832(10):1473–1481. doi:10.1016/j.bbadis.2013.04.022
18. Sundar IK, Yao H, Rahman I. Oxidative stress and chromatin remodeling in chronic obstructive pulmonary disease and smoking-related diseases. Antioxid Redox Signal. 2013;18(15):1956–1971. doi:10.1089/ars.2012.4863
19. Szulakowski P, Crowther AJ, Jiménez LA, et al. The effect of smoking on the transcriptional regulation of lung inflammation in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2006;174(1):41–50. doi:10.1164/rccm.200505-725OC
20. Ito K, Ito M, Elliott WM, et al. Decreased histone deacetylase activity in chronic obstructive pulmonary disease. N Engl J Med. 2005;352(19):1967–1976. doi:10.1056/NEJMoa041892
21. Zou C, Chen Y, Smith RM, et al. SCF(Fbxw15) mediates histone acetyltransferase binding to origin recognition complex (HBO1) ubiquitin-proteasomal degradation to regulate cell proliferation. J Biol Chem. 2013;288(9):6306–6316. doi:10.1074/jbc.M112.426882
22. Mishima Y, Miyagi S, Saraya A, et al. The Hbo1-Brd1/Brpf2 complex is responsible for global acetylation of H3K14 and required for fetal liver erythropoiesis. Blood. 2011;118(9):2443–2453. doi:10.1182/blood-2011-01-331892
23. Lalonde ME, Cheng X, Côté J. Histone target selection within chromatin: an exemplary case of teamwork. Genes Dev. 2014;28(10):1029–1041. doi:10.1101/gad.236331.113
24. Newman DM, Voss AK, Thomas T, Allan RS. Essential role for the histone acetyltransferase KAT7 in T cell development, fitness, and survival. J Leukoc Biol. 2017;101(4):887–892. doi:10.1189/jlb.1MA0816-338R
25. Mishima Y, Wang C, Miyagi S, et al. Histone acetylation mediated by Brd1 is crucial for Cd8 gene activation during early thymocyte development. Nat Commun. 2014;5:5872. doi:10.1038/ncomms6872
26. Yoshida T, Tuder RM. Pathobiology of cigarette smoke-induced chronic obstructive pulmonary disease. Physiol Rev. 2007;87(3):1047–1082. doi:10.1152/physrev.00048.2006
27. Miotto B, Struhl K. JNK1 phosphorylation of Cdt1 inhibits recruitment of HBO1 histone acetylase and blocks replication licensing in response to stress. Mol Cell. 2011;44(1):62–71. doi:10.1016/j.molcel.2011.06.021
28. Gaudet P, Livstone MS, Lewis SE, Thomas PD. Phylogenetic-based propagation of functional annotations within the gene ontology consortium. Brief Bioinform. 2011;12(5):449–462. doi:10.1093/bib/bbr042
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.