Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 13
The outcome and the influencing factors of the age of onset in post-mortem of chronic bronchitis patients: a retrospective study
Authors Zhu L, Ni Z, Luo X, Zhang Z, Wang S, Meng Z, Gu X, Wang X
Received 15 November 2017
Accepted for publication 15 December 2017
Published 21 February 2018 Volume 2018:13 Pages 645—652
DOI https://doi.org/10.2147/COPD.S157084
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Chunxue Bai
Linyun Zhu,1 Zhenhua Ni,2 Xuming Luo,1 Zhuhua Zhang,1 Shiqiang Wang,1 Ziyu Meng,1 Xiandong Gu,1 Xiongbiao Wang1
1Department of Respiratory Medicine, Putuo Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai, 2Central Laboratory, Putuo Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai, China
Purpose: Chronic bronchitis is thought to occur in elderly patients, and smoking seems to be an important risk factor. The outcomes related to the age of onset in patients with chronic bronchitis are still unclear.
Patients and methods: A retrospective study was conducted on deceased patients whose diagnosis included bronchitis from 2010 to 2016. Patients were separated into two groups according to the age of onset (Group I, age ≤50 years old; Group II, age >50 years old). Information regarding disease course, smoking history, death age, number of admissions per year, Hugh Jones Index, and self-reported comorbidities of the patients was recorded.
Results: The courses of chronic cough and sputum were 33.38±7.73 years and 14.44±8.60 years in Group I and Group II, respectively (p<0.05). The death ages of Group I and Group II were 77.65±7.87 years and 84.69±6.67 years, respectively (p<0.05). There was a significant negative correlation between the number of hospital admissions per year and the age of onset. The age of onset was negatively associated with daily smoking count (r=-0.210) and total smoking count (r=-0.146). In Group I, there were fewer cases of coronary heart disease (OR =0.41 [0.24–0.71]), neurological diseases (OR =0.48 [0.24–0.97]), and total comorbidities (OR =0.67 [0.54–0.85]) than in Group II.
Conclusion: Patients with early onset chronic bronchitis had a longer history, younger death age, poorer health status, and lower incidence of comorbidities.
Keywords: chronic airway disease, comorbidity, Hugh Jones Index, smoking, hospital admission, disease course
Introduction
Chronic bronchitis is often thought to be an age-related disease that occurs after years of cigarette smoking. It is characterized by chronic coughing, chest tightness, and dyspnea. Chronic bronchitis occurs not only in older individuals but also in younger people. The age of onset is determined by the interaction of gene polymorphisms and environmental factors. Smoking status,1 location, occupational dust exposure, type of house,2 radiation,3 allergic history, childhood asthma, parental bronchitis symptoms,4 and biomass fuels5 are all related to the incidence of chronic bronchitis. Chronic bronchitis also shows a moderate familial aggregation, particularly in women.6 The T5-TG12 haplotype of the cystic fibrosis transmembrane receptor (CFTR) gene,7 CFTR dysfunction due to smoking,8 and aberrant expression of epigenetic markers may cause a higher incidence of chronic bronchitis.9 Although chronic bronchitis is associated with impaired quality of life, hospital admissions, and increased mortality,10 it has not garnered much attention since the introduction of the term chronic obstructive pulmonary disease. Previous studies have shown that chronic bronchitis notably increases the risk of continuous airflow limitation and all-cause mortality in subjects <50 years old, but not among subjects ≥50 years old.11 Few studies have focused on the clinical features, related factors, and outcomes according to the age of onset in chronic bronchitis. Our study aims to determine the relationship between the age of onset and severity of chronic bronchitis, the influencing factors of the age of onset on comorbidities, and the cause of death in patients with chronic bronchitis.
Patients and methods
Participants
We conducted a retrospective study by reviewing the medical records of deceased patients admitted to the Shanghai Putuo District Central Hospital and whose diagnosis included chronic bronchitis (ICD-10: J42) from 2010 to 2016. Data were acquired from the hospital database. This study enrolled 558 patients whose baseline characteristics, disease history, and self-reported comorbidities were recorded.
Patients with chronic bronchitis were defined as having a sputum-producing cough daily for at least 3 months per year for two consecutive years. Smoking patients are defined as having a smoking history, including current and former smokers. Smoking history included smoking duration, daily smoking count, and total smoking count. The number of hospital admissions per year was considered to be an average admission of the past year.
The age of onset was defined as the first occurrence of recurrent cough and sputum, which was reported by the patients in their medical records as a retrospective indicator. Patients were separated into two groups according to the age of onset. The age of Group I was ≤50 years, while the participants in Group II were >50 years old. The course of disease was detailed through retrospective data and defined as the occurrence of this disease until the patients’ death. The clinical dyspnea index was evaluated using the Hugh Jones Index. The self-reported comorbidities included coronary heart disease (ischemic heart disease, myocardial infarction, and coronary artery disease), diabetes, heart failure, hypertension, malignant tumor, mental disease (depression, mania, dementia), neurological disease, and renal failure (renal function lower than chronic kidney disease [CKD] stage III), all of which were referred from the Charlson Comorbidity Index.
Statistical analysis
All data are expressed as the mean ± SD for continuous variables and as frequencies and percentages for categorical variables. Continuous data were compared using the Student’s t-test or the Wilcoxon’s test, while categorical data were compared using the chi-squared and Fisher’s exact tests. Spearman’s rank correlation techniques were used to analyze the relationships between several continuous variables. The survival analysis used the Cox proportional hazard model. A p-value <0.05 was considered statistically significant for single comparisons. All the reported p-values were two sided. Data were analyzed using the SPSS 22.00 software package (SPSS Inc, Chicago, IL, USA).
Ethics approval and consent to participate
The study protocol was approved by the institutional review board of Putuo Hospital, Shanghai University of Traditional Chinese Medicine. The patients’ approval or informed consent was not required for a retrospective review of their records. The patient data used in our study did not include any identifying information.
Results
Five hundred fifty-eight deceased patients who met all of the criteria were included in this study. There were 405 males (72.6%) and 153 females (27.4%). Patients were divided into two groups according to the age of onset. Group I (the age of onset was ≤50 years) included 63 patients, whereas Group II (the age of onset was >50 years) included 495 patients.
Longer course of disease, earlier age of death, and poorer dyspnea status in early onset chronic bronchitis patients
The average course of chronic cough and sputum was 16.71±10.66 years. The age of onset was 67.32±12.35 years, and the age of death was 83.89 years. The course of chronic cough and sputum was notably longer in Group I than in Group II. The Hugh Jones Index showed significant differences between these two groups. The age of onset was significantly negatively correlated with the number of hospital admissions per year (Table 1).
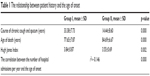  | Table 1 The relationship between patient history and the age of onset |
The correlation between smoking history and the age of onset
There were 233 patients with a history of smoking, of which only 35 patients had not quit smoking. There were more patients with a smoking history in Group I than in Group II (Table 2). There were no differences between Group I and Group II in terms of daily smoking counts, smoking duration, and total smoking counts. Furthermore, the age of onset was negatively associated with daily smoking counts (r=−0.210, p=0.001) and total smoking counts (r=−0.146, p=0.027), but there was no correlation between the duration of smoking and the age of onset (r=−0.116, p=0.078, Figure 1).
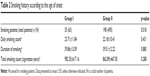  | Table 2 Smoking history according to the age of onset |
The incidence of self-reported comorbidities according to the age of onset in patients with chronic bronchitis
There were eight comorbidities considered, including coronary heart disease (ischemic heart disease, myocardial infarction, and coronary artery disease), diabetes, heart failure, hypertension, malignant tumor, mental disease (depression, mania, dementia), neurological disease, and renal failure (renal function lower than CKD stage III). In Group I (early onset chronic bronchitis patients), there were fewer cases of coronary heart diseases (OR =0.41 [0.24–0.71]), neurological diseases (OR =0.48 [0.24–0.97]), and total complications (OR =0.67 [0.54–0.85]) than in Group II (late-onset chronic bronchitis patients). However, the incidences of diabetes, heart failure, hypertension, malignant tumor, mental disease (including depression, mania, and dementia), and renal failure (renal function lower than CKD stage III) were similar in the two groups (Figure 2).
  | Figure 2 The relationship between the incidence of self-comorbidities and the age of onset. |
The relationship between cause of death and the age of onset
There was no difference in the cause of death in the two groups. Most patients died from chronic respiratory disease (37.6%) and cardiovascular disease (28.9%). Other cause of death included neurological disease, digestion disease, malignant tumor, and so on (Figure 3).
  | Figure 3 The relationship between cause of death and the age of onset. |
The relationship between the survival curves of the two groups and the age of onset
The mean ages of death were 77.65±7.87 years and 84.69±6.67 years (p<0.05) in Group I and Group II, respectively (Figure 4).
  | Figure 4 The mortality between the two groups as a function of the age of onset. |
Discussion
Chronic bronchitis often occurs in elderly individuals. An investigation of community-dwelling individuals aged 40–80 years found that the incidence of chronic bronchitis was 12.7%,12 and other studies observed similar results.13,14 Though smoking is the most important risk factor, low socioeconomic class and urban living,10,15 gender, asthma history,16 changes in living habits,14 odor or the musty smell of mildew/mold in the house, exposure to 1,1,1-trichloro-2,2-bis(4-chlorophenyl)ethane (DDT),49 body mass index,17 and air pollution18 play roles in the development of chronic bronchitis. Studies have demonstrated that aging (especially >50 years old) is also a high risk factor for chronic bronchitis.14,19 As a result of persistent mucus hypersecretion, longer courses of productive cough are related to quicker reductions in the lung function.20 Early onset chronic bronchitis has a higher risk to develop into irreversible airflow limitation and all-cause mortality.11
Though chronic bronchitis is related to aging, our study found that the early onset patients had longer productive cough courses, in which the age of death was much lower than the other patients. Lindberg et al showed that productive cough has a significantly higher risk for death even after adjustment for common risk factors.21 With the progress of disease, chronic bronchitis has an increased risk for frequent exacerbation.22,23 Not only did patients with chronic bronchitis have a higher frequency of exacerbation, but also had longer stays in the hospital.23 We found that the age of onset is negatively correlated with the number of hospital admissions, which was similar to these results. The earlier the onset, the more frequently these patients presented with exacerbation.
In the early onset group, patients showed higher Hugh Jones Index values indicating a poorer health status in our study. Respiratory symptoms are common in chronic bronchitis or COPD. Previous studies have shown that cough is associated with the health-related quality of life,24 that is, poor quality of life, and breathing problems of chronic bronchitis that limit daily activities.25 Patients with chronic bronchitis had a greater incidence of chronic dyspnea and activity restriction,26 and chronic bronchitis significantly lowered exercise capacity in COPD patients.27 There may be a reasonable explanation for the occurrence of early onset chronic bronchitis with longer courses of disease and higher activity restrictions. On the other hand, Riesco et al discovered that active smoking was significantly associated with higher grades of dyspnea.28 Long-term smoking negatively affects the health of patients.29 The early onset subjects showed higher rates of smoking, which may lead to increase levels of dyspnea shown as higher Hugh Jones Index scores.
Smoking has been proven to be a definitive risk factor for the incidence of chronic bronchitis. Liu et al conducted a prospective study to assess the duration of smoking and airway symptoms. They found that smoking duration showed a linear relationship with symptoms including frequent productive cough, frequent shortness of breath, and shortness of breath affecting physical activity.29 In our study, though duration of smoking was not related to the age of onset, daily smoking count and total smoking count are negatively associated with the age of onset. Using the time to first cigarette (TTFC) after waking parameter as an indicator of nicotine dependence, current smokers who were found to have shorter TTFC times had an increased risk of chronic bronchitis.30 While quitting or reducing smoking might lead to fewer chronic productive cough symptoms,31 other studies have shown that the smokers who quit because of illness had a significantly higher prevalence of chronic respiratory disease.32 Smoking-related chronic mucus hypersecretion usually resolves following smoking cessation, but the duration of smoking correlated with poorer airway disease activity that reflected the underlying course.20 There were strong associations between smoking history (including current smokers and ex-smokers) and high incidence of chronic respiratory diseases.32 In our study, the daily smoking count and total smoking count were greater with earlier onset, which could perhaps explain why these subjects had a younger age of onset. We found that early onset patients had more active smoking histories and a higher smoking rate than the other group. Not only did smoking history has an impact on the incidence of chronic bronchitis, but also the amount of cigarettes smoked contributed to this disease.
Subjects with physician-diagnosed COPD were more likely have coexisting arthritis, depression, osteoporosis, cancer, coronary heart disease, congestive heart failure, and stroke.33 Our study found that heart failure, coronary heart disease, and hypertension are the most common complications. Furthermore, in the early onset group, the incidences of coronary heart disease and neurological disease were lower than in the other group. We speculate that as patients grow older, these age-related diseases become more common.
Renal failure is an often neglected problem with regard to chronic bronchitis. A previously conducted meta-analysis demonstrated that COPD was found to be associated with a significantly increased prevalence of CKD (OR =2.20).34 We found that the incidence of chronic renal failure was similar in both groups, so this disease may not be associated with the age of onset.
Chronic bronchitis increased the risk of ischemic events in all age groups. This observation reached significance for patients >60 years of age, especially over the previous 2 months.35 The early onset age group presented with a lower incidence of neurological disease likely because these diseases are associated with aging.
Diabetes mellitus (DM) is becoming more common. Previous research has shown that the incidence of diabetes is associated with accumulated smoking exposure, impaired spirometry pattern pulmonary function, reduced 6-minute walking distance without the influence of body mass index, high blood pressure, and high cholesterol.36 Patients with chronic bronchitis have an increased risk of type 2 diabetes, which is independent of smoking. In addition, chronic bronchitis has a genetic correlation to diabetes.37 However, there is a decreased incidence of inflammatory diseases such as chronic bronchitis in diabetic patients; thus, type 2 diabetes might reduce the risk of these diseases.38 We found that there is no difference between the two groups in terms of the incidence of diabetes. Both aging and chronic inflammation might interact in diabetes as a comorbidity of chronic bronchitis. Although DM was not associated with reduced quality of life and poorer pulmonary function, untreated DM was related to reduced quality of life and worse pulmonary function.39
Lung cancers are more common in chronic bronchitis. Several studies have found that COPD increases the risk of lung cancer,40 and that these lung cancers might be more aggressive.41 Other malignant tumors were found in our study, including colorectal cancer or leukemia. Some are associated with a history of smoking,42 while some are not well known.
In our study, there were no differences in the incidence of mental illness between the two groups, but it is worth noting that both groups of chronic bronchitis patients presented with mental illness. Mental illnesses, especially depression, gradually increase43 and become a threat to health in elderly individuals. Both depression and chronic respiratory disease lead to cognitive impairments at the early stages of chronic airway damage and progress with worsening conditions.44 Individuals with mental illness have a significantly increased incidence of chronic physical health disorders compared with people without mental illness.45 Patients with chronic bronchitis presented with worse mental well-being than those without chronic bronchitis.46 Therefore, clinicians and researchers should pay more attention to the mental state of patients with chronic bronchitis.
We found that most patients died from chronic respiratory diseases, which was similar to other studies.47 There was no difference between the two groups and no differences in all-cause of death in the patients. Instant airway inflammation and longer durations of smoking may account for the earlier death of patients. Mekov et al discovered that the risk factors for increased mortality were age, FEV1 values, severe exacerbation in the previous year, and reduced quality of life.48 Chronic bronchitis increased all-cause mortality and mortality from respiratory causes, cardiovascular diseases, and cancer.23 In our study, early onset patients had more frequent exacerbation and poorer health status, which could cause death at younger ages.
Our retrospective study on chronic bronchitis focused on the case histories of deceased patients. Many of these subjects were in critical condition with a long course of disease, which may have introduced some bias into the dataset. Until now, little research focusing on the patient’s history prior to death has been conducted, and as a result, this study could lay the foundation for future investigations that center on early onset patients.
Conclusion
We found that patients with early onset chronic bronchitis had a longer history, younger death age, more smokers, poorer health status, and lower incidence of comorbidities. Further, we need a large, prospective cohort study focusing on the age of onset in chronic bronchitis.
Acknowledgments
LZ has received grants for scientific work from the Foundation of the budget project of Shanghai University of Traditional Chinese Medicine in China. XW has received grants for scientific work from the Foundation of the Putuo key clinical specialist construction Programs in China and the Foundation of Department of Respiratory Medicine Development Fund of Putuo District in China.
Disclosure
The authors report no conflicts of interest in this work.
References
Pärna K, Põld M, Ringmets I. Physicians’ views on the role of smoking in smoking-related diseases: findings from cross-sectional studies from 1982–2014 in Estonia. TobInduc Dis. 2017;15:31. | ||
Mahesh PA, Jayaraj BS, Chaya SK, et al. Variation in the prevalence of chronic bronchitis among smokers: a cross-sectional study. Int J Tuberc Lung Dis. 2014;18(7):862–869. | ||
Azizova TV, Zhuntova GV, Haylock R, et al. Chronic bronchitis incidence in the extended cohort of Mayak workers first employed during 1948–1982. Occup Environ Med. 2017;74(2):105–113. | ||
Dharmage SC, Perret JL, Burgess JA, et al. Current asthma contributes as much as smoking to chronic bronchitis in middle age: a prospective population-based study. Int J Chron Obstruct Pulmon Dis. 2016;11:1911–1920. | ||
Sehgal M, Rizwan SA, Krishnan A. Disease burden due to biomass cooking-fuel-related household air pollution among women in India. Glob Health Action. 2014;7(1):25326. | ||
Meteran H, Backer V, Kyvik KO, Skytthe A, Thomsen SF. Heredity of chronic bronchitis: a registry-based twin study. Respir Med. 2014;108(9):1321–1326. | ||
Wang P, Naruse S, Yin H, et al. The susceptibility of T5-TG12 of the CFTR gene in chronic bronchitis occurrence in a Chinese population in Jiangsu province, China. J Biomed Res. 2012;26(6):410–417. | ||
Wu DD, Song J, Bartel S, Krauss-Etschmann S, Rots MG, Hylkema MN. The potential for targeted rewriting of epigenetic marks in COPD as a new therapeutic approach. Pharmacol Ther. Epub2017 Aug 19. | ||
Raju SV, Tate JH, Peacock SK, et al. Impact of heterozygote CFTR mutations in COPD patients with chronic bronchitis. Respir Res. 2014;15:18. | ||
Axelsson M, Ekerljung L, Eriksson J, et al. Chronic bronchitis in West Sweden – a matter of smoking and social class. Eur Clin Respir J. 2016;3:30319. | ||
Guerra S, Sherrill DL, Venker C, Ceccato CM, Halonen M, Martinez FD. Chronic bronchitis before age 50 years predicts incident airflow limitation and mortality risk. Thorax. 2009;64(10):894–900. | ||
Marcus BS, McAvay G, Gill TM, Vaz FCA. Respiratory symptoms, spirometric respiratory impairment, and respiratory disease in middle-aged and older persons. J Am Geriatr Soc. 2015;63(2):251–257. | ||
Mwangi J, Kulane A, Van Hoi L. Chronic diseases among the elderly in a rural Vietnam: prevalence, associated socio-demographic factors and healthcare expenditures. Int J Equity Health. 2015;14:134. | ||
Landis SH, Muellerova H, Mannino DM, et al. Continuing to Confront COPD International Patient Survey: methods, COPD prevalence, and disease burden in 2012–2013. Int J Chron Obstruct Pulmon Dis. 2014;9:597–611. | ||
Miele CH, Jaganath D, Miranda JJ, et al. Urbanization and daily exposure to biomass fuel smoke both contribute to chronic bronchitis risk in a population with low prevalence of daily tobacco smoking. COPD. 2016;13(2):186–195. | ||
Holm M, Torén K, Andersson E. Incidence of chronic bronchitis: a prospective study in a large general population. Int J Tuberc Lung Dis. 2014;18(7):870–875. | ||
Pahwa P, Karunanayake CP, Rennie DC, et al. Prevalence and associated risk factors of chronic bronchitis in First Nations people. BMC Pulm Med. 2017;17(1):95. | ||
Maji KJ, Arora M, Dikshit AK. Burden of disease attributed to ambient PM2.5 and PM10 exposure in 190 cities in China. Environ Sci Pollut Res Int. 2017;24(12):11559–11572. | ||
Tutar N, Yeşilkaya S, Memetoğlu ME, Özel D, Boşnak E. The prevalence of chronic bronchitis in adults living in the center of Gumushane. TuberkToraks. 2013;61(3):209–215. | ||
Allinson JP, Hardy R, Donaldson GC, Shaheen SO, Kuh D, Wedzicha JA. The presence of chronic mucus hypersecretion across adult life in relation to chronic obstructive pulmonary disease development. Am J Respir Crit Care Med. 2016;193(6):662–672. | ||
Lindberg A, Sawalha S, Hedman L, Larsson LG, Lundbäck B, Rönmark E. Subjects with COPD and productive cough have an increased risk for exacerbations and death. Respir Med. 2015;109(1):88–95. | ||
Lahousse L, LJM S, Joos GF, Franco OH, Stricker BH, Brusselle GG. Epidemiology and impact of chronic bronchitis in chronic obstructive pulmonary disease. Eur Respir J. 2017;50(2):1602470. | ||
Pelkonen MK, Notkola IK, Laatikainen TK, Jousilahti P. Chronic bronchitis in relation to hospitalization and mortality over three decades. Respir Med. 2017;123:87–93. | ||
Deslee G, Burgel PR, Escamilla R, et al. Impact of current cough on health-related quality of life in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2016;11:2091–2097. | ||
Accordini S, Corsico AG, Calciano L, et al. The impact of asthma, chronic bronchitis and allergic rhinitis on all-cause hospitalizations and limitations in daily activities: a population-based observational study. BMC Pulm Med. 2015;15:10. | ||
Elbehairy AF, Raghavan N, Cheng S, et al. Physiologic characterization of the chronic bronchitis phenotype in GOLD grade IB COPD. Chest. 2015;147(5):1235–1245. | ||
Zhang W, Lu H, Peng L, et al. Chronic bronchitis leads to accelerated hyperinflation in COPD patients during exercise. Respirology. 2015;20(4):618–625. | ||
Riesco JA, Alcázar B, Trigueros JA, Campuzano A, Pérez J, Lorenzo JL. Active smoking and COPD phenotype: distribution and impact on prognostic factors. Int J Chron Obstruct Pulmon Dis. 2017;12: 1989–1999. | ||
Liu Y, Pleasants RA, Croft JB, et al. Smoking duration, respiratory symptoms, and COPD in adults aged ≥45 years with a smoking history. Int J Chron Obstruct Pulmon Dis. 2015;10:1409–1416. | ||
Guertin KA, Gu F, Wacholder S, et al. Time to first morning cigarette and risk of chronic obstructive pulmonary disease: smokers in the PLCO cancer screening trial. PLoS One. 2015;10(5):e0125973. | ||
Kainu A, Pallasaho P, Pietinalho A. No change in prevalence of symptoms of COPD between 1996 and 2006 in Finnish adults – a report from the FinEsS Helsinki Study. Eur Clin Respir J. 2016;3:31780. | ||
Kurmi OP, Li L, Wang J, et al. COPD and its association with smoking in the Mainland China: a cross-sectional analysis of 0.5 million men and women from ten diverse areas. Int J Chron Obstruct Pulmon Dis. 2015;10:655–665. | ||
Schnell K, Weiss CO, Lee T, et al. The prevalence of clinically-relevant comorbid conditions in patients with physician-diagnosed COPD: a cross-sectional study using data from NHANES 1999–2008. BMC Pulm Med. 2012;12:26. | ||
Gaddam S, Gunukula SK, Lohr JW, Arora P. Prevalence of chronic kidney disease in patients with chronic obstructive pulmonary disease: a systematic review and meta-analysis. BMC Pulm Med. 2016;16(1):158. | ||
Piñol-Ripoll G, de la Puerta I, Santos S, Purroy F, Mostacero E. Chronic bronchitis and acute infections as new risk factors for ischemic stroke and the lack of protection offered by the influenza vaccination. Cerebrovasc Dis. 2008;26(4):339–347. | ||
Kinney GL, Baker EH, Klein OL, et al. Pulmonary predictors of incident diabetes in smokers. Chronic Obstr Pulm Dis. 2016;3(4):739–747. | ||
Meteran H, Backer V, Kyvik KO, Skytthe A, Thomsen SF. Comorbidity between chronic obstructive pulmonary disease and type 2 diabetes: a nation-wide cohort twin study. Respir Med. 2015;109(8): 1026–1030. | ||
Zheng Y, Zhang G, Chen Z, Zeng Q. Relationship between type 2 diabetes and inflammation diseases: cohort study in Chinese Adults. Iran J Public Health. 2015;44(8):1045–1052. | ||
Mekov EV, Slavova YG, Genova MP, et al. Diabetes mellitus type 2 in hospitalized COPD patients: impact on quality of life and lung function. Folia Med (Plovdiv). 2016;58(1):36–41. | ||
Gonzalez J, Marín M, Sánchez-Salcedo P, Zulueta JJ. Lung cancer screening in patients with chronic obstructive pulmonary disease. Ann Transl Med. 2016;4(8):160. | ||
Sanchez-Salcedo P, Zulueta JJ. Lung cancer in chronic obstructive pulmonary disease patients, it is not just the cigarette smoke. Curr Opin Pulm Med. 2016;22(4):344–349. | ||
Bailie L, Loughrey MB, Coleman HG. Lifestyle risk factors for serrated colorectal polyps: a systematic review and meta-analysis. Gastroenterology. 2017;152(1):92–104. | ||
Kim MT, Kim KB, Han HR, Huh B, Nguyen T, Lee HB. Prevalence and predictors of depression in Korean American elderly: findings from the memory and aging study of Koreans (MASK). Am J Geriatr Psychiatry. 2015;23(7):671–683. | ||
Dal Negro RW, Bonadiman L, Tognella S, Bricolo FP, Turco P. Extent and prevalence of cognitive dysfunction in chronic obstructive pulmonary disease, chronic non-obstructive bronchitis, and in asymptomatic smokers, compared to normal reference values. Int J Chron Obstruct Pulmon Dis. 2014;9:675–683. | ||
Scott D, Burke K, Williams S, Happell B, Canoy D, Ronan K. Increased prevalence of chronic physical health disorders in Australians with diagnosed mental illness. Aust N Z J Public Health. 2012;36(5): 483–486. | ||
Meek PM, Petersen H, Washko GR, et al. Chronic bronchitis is associated with worse symptoms and quality of life than chronic airflow obstruction. Chest. 2015;148(2):408–416. | ||
Pesce G. Mortality rates for chronic lower respiratory diseases in Italy from 1979 to 2010: an age-period-cohort analysis. ERJ Open Res. 2016;2(1):00093–2015. | ||
Mekov E, Slavova Y, Tsakova A, et al. One-year mortality after severe COPD exacerbation in Bulgaria. Peer J. 2016;4:e2788. | ||
Ye M, Beach J, Martin JW, Senthilselvan A. Association between lung function in adults and plasma DDT and DDE levels: results from the Canadian Health Measures Survey. Environ Health Perspect, 2015; 123(5):422–427. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

