Back to Journals » Journal of Blood Medicine » Volume 15
The Montana Interfacility Blood Network: A Novel Lifesaving “Hand-off” for the Optimal Care of Rural Patients
Authors Riha GM, Johnson A, Arnold S , Englehart MS, Thompson SJ
Received 5 December 2023
Accepted for publication 1 March 2024
Published 20 March 2024 Volume 2024:15 Pages 141—146
DOI https://doi.org/10.2147/JBM.S442134
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Martin H Bluth
Gordon M Riha,1 Alyssa Johnson,2 Sadie Arnold,3 Michael S Englehart,1 Simon J Thompson4
1Trauma & General Surgery, Billings Clinic, Billings, MT, USA; 2EMS & Trauma Systems Montana, Chronic Disease Prevention and Health Promotion Bureau, Montana Department of Public Health and Human Services, Helena, MT, USA; 3Laboratory Services, Billings Clinic, Billings, MT, USA; 4Collaborative Science & Innovation, Billings Clinic, Billings, MT, USA
Correspondence: Simon J Thompson, Collaborative Science & Innovation, Billings Clinic, Billings, MT, USA, Tel +1 503 267 7314, Fax +1 406 435-1586, Email [email protected]
Purpose: The state of Montana encompasses and defines rural health care as it is known in the United States (US) today. This vast area is punctuated by pockets of health care availability with varying access to blood products for transfusion. Furthermore, timely transport is frequently challenged by weather that may limit air transportation options, resulting in multiple hours in ground transport to definitive care.
Patients and Methods: The Montana State Trauma Care Committee (MT-STCC) developed the Montana Interfacility Blood Network (MT-IBN) to ensure blood availability in geographically distanced cases where patients may otherwise not survive. The index case that led to the formal development of the MT-IBN is described, followed by a second case illustrating the IBN process.
Results: This process and development manuscript details the innovative efforts of MT-STCC to develop this fledgling idea unique to rural US health care. We review guidelines that have been developed to define broad aspects of the MT-IBN including the reason to share resources, proper packaging, paperwork necessary for transfer, and how to provide resources directly to the patient. Finally, we describe implementation within the state.
Conclusion: The MT-IBN was developed by MT-STCC to facilitate the hand-off of lifesaving blood to patients being transported by ground to definitive care in Montana without having to stop at an intermediary facility. This has already led to lives saved in areas that are limited in blood availability due to rurality.
Keywords: rural health care, blood transfusion, health services accessibility, trauma
Introduction
Background
Massive hemorrhage remains a major cause of preventable death in trauma.1 Of these deaths, 33–56% occur during the prehospital period.2 The key components of treatment are immediate hemorrhage control3 and blood product transfusion to restore volume and manage coagulopathy.4 The European guidelines on management of major bleeding and coagulopathy following major trauma5 suggest that bleeding trauma patients should be rapidly transported to major trauma centers to institute appropriate treatment as soon as possible. Furthermore, the importance of immediate access to blood products for hemorrhagic shock is well known in the trauma literature. Hemorrhage is responsible for 30–40% of trauma mortality.2
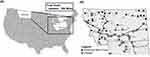
The state of Montana (Figure 1A) encompasses over 147,000 square miles with approximately 7 people per square mile,6 which truly defines rural health care as it is known in the United States today. This vast area is punctuated by pockets of health care availability with varying access to blood products for transfusion and surgical care. Furthermore, timely transport to definitive care is challenged by weather that may limit air transportation options, resulting in multiple hours in ground transport. Herein, we report the index case prompting the development of a novel interfacility blood network, and a second case illustrating the implementation and outcome of the new process.
Index Case #1
A 30-year-old female presented to a rural health care center in Lame Deer, MT with severe abdominal pain and a syncopal episode. The patient developed hypotension with transient response to fluid resuscitation, with no blood product availability in this associated clinic or region. Due to the frigid weather, ongoing snowfall, and low aviation ceiling, helicopter and fixed wing transport were not possible. The patient was transported by ground 104 miles to the closest integrated tertiary care and Level II trauma center, in Billings, MT. Enroute, the patient required bag mask ventilation due to altered mental status 60 miles from Billings and then went into cardiac arrest 7 miles from Billings requiring CPR. Upon arrival at the tertiary facility, after a transfer time of 3 hours and 18 minutes, the patient underwent massive transfusion protocol and immediate surgical intervention where she was noted to have a ruptured ectopic pregnancy with evacuation of 5 liters of intraperitoneal blood. Post-operatively, the patient was extubated and made a full physiologic and neurologic recovery.
Out of necessity and for the optimal care of patients, the Montana State Trauma Care Committee (MT-STCC) developed the Montana Interfacility Blood Network (MT-IBN). We hypothesize that the MT-IBN will ultimately save lives in a state dominated by vast distances between health care facilities.
Materials and Methods
All massive transfusions were peer reviewed at an ACS verified level II trauma center (tertiary trauma facility (regional care facility)) by a Blood Utilization Committee, a multi-disciplinary monthly meeting formed in August 2015 and led by a physician chair. In review of the Index Case #1, it was noted that ground transport passed two facilities that had blood availability on the way to definitive care. Further discussion remarked on the published availability of a state “microbrew map” and thus raised the question of why a state blood map was not available.
Blood banking specialists developed a preliminary map detailing locations of packed red blood cells (PRBCs) throughout the states of Montana and northern Wyoming (Figure 1B). The preliminary idea of a blood sharing network was then discussed at the MT-STCC on May 19, 2019. A follow-up discussion occurred at the August 14, 2019, MT-STCC meeting, and a blood subcommittee was then created on November 13, 2019. This blood subcommittee met twice through 2019 and 2020 in which the blood map was reviewed, and a blood sharing procedure draft was developed.
Step 1 - Determine Interest
To gauge potential interest across the state and the potential for provider buy-in, a questionnaire was initially distributed via the state trauma forums to all trauma coordinators at hospitals affiliated with the Montana Trauma system in January 2020; this consisted of 41 state trauma designated hospitals. Subsequently, the results were compiled and reported at the February 12, 2020, MT-STCC meeting (Supplemental Materials 1).
Step 2 - Guideline Development
A multidisciplinary team set forth to develop guidelines for a blood sharing network utilizing the blood product availability map. Five broad categories (Figure 2) were determined to be necessary for patient safety, blood banking regulatory concerns, and statewide implementation, including a blood product availability map.7
 |
Figure 2 Blood Product Sharing Guidelines Developed for Statewide Implementation. Note: Supplementary Materials can be accessed here: https://www.dovepress.com/get_supplementary_file.php?f=442134.docx. |
1. Reason to share resource identified
2. Blood resources must be available
3. Proper packaging
4. Paperwork for transfer
5. Resupply and billing
Step 3 - Stakeholder Review
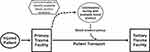
The MT-IBN concept (Figure 3) was formally presented at the Montana statewide American Society for Clinical Laboratory Science (ASCLS) and the regional Rimrock Trauma Conference in April 2021. The developers further discussed the concept at various Trauma Education Assessment & Management (TEAM) courses, the Montana equivalent of the American College of Surgeons’ Rural Trauma Team Development Courses,8 throughout 2021. An update was provided to the Montana Department of Public Health and Human Services with a Clinical Laboratory Improvement Amendment (CLIA) update in December 2021 that was sent to laboratory providers statewide. Discussions then continued with a presentation at the Montana Public Health Association conference in April 2022 and included meetings with key leaders of the Montana Hospital Association (MHA). Following this, the MT-IBN concept underwent final review and approval by MT-STCC in April 2022.
Step 4 – Implementation
Administrative policy and guidelines for personnel were distributed statewide along with education for Emergency Medical Services (EMS) and trauma personnel as discussed further in the Results.
Written informed consent was obtained from the patients for publication of their cases.
Results
The questionnaire was sent to all hospitals designated by the Montana Trauma system and included trauma coordinators, Trauma Medical Directors, and trauma staff (n=112), with the aim to obtain a respondent from each facility. A total of 40 individuals from 41 state-designated facilities responded to the questionnaire. Aproximately 92.5% responded that the ambulance should be able to pick up blood products for administration to a patient during transport to a higher level of care (Supplemental Materials 1). Furthermore, 75% of respondents believed that such a process was either probably or definitely needed. Aproximately 48.7% responded that sharing of blood products for patients “passing by” and not seen at the facility was allowed, but without policy or process. However, many responded that gaining approval for such process at their facility would be somewhat difficult (57.5%), very difficult (12.5%), or extremely difficult (5%).
Following approval of the MT-IBN at STCC, packets were compiled containing the following: a cover letter, a quick start guide containing the five broad guidelines discussed above, Laboratory Guide for Blood Product Sharing (Supplemental Materials 2), packing guidelines (Supplemental Materials 3 and 4), and transfer documents (Supplemental Materials 5 and 6). Rollout included respective education of EMS and trauma personnel at the Rocky Mountain Rural Trauma pre-conference symposium and of hospital providers at the MHA in September 2021.
Case #2
A patient with severe gastrointestinal (GI) hemorrhage presented to a rural critical access hospital (CAH) in rural Montana where workup revealed a hemoglobin of 5.8 gm/dl. This facility had only 1 unit of O negative blood available. The patient required ground transport from the primary CAH to definitive care in Billings (82.8 miles away). Midway between the rural facility and Billings on the Interstate Highway is another Critical Access Hospital, 41.3 miles from the primary CAH. The provider at the primary CAH called the nurse supervisor of the midway CAH at 0200 to discuss the situation and confirm if they could provide intermediary lifesaving blood. Laboratory personnel at the midway CAH were called in, readied a unit of O negative blood, and arranged for the local law enforcement officer to take the blood to the on/off ramp to the highway. The patient did not need to be registered, observed, or unnecessarily delayed at the midway CAH. Instead, the ambulance met the local law enforcement officer at the highway on/off ramp, and the ambulance crew transferred and transfused the required blood on the way to the Billings facility. The patient had a favorable outcome.
Discussion
Two recent randomized controlled trials, Prehospital Air Medical Plasma (PAMPer)9 and the Control of Major Bleeding After Trauma Trial (COMBAT),10 provided opposing views on the implementation of prehospital plasma transfusion. However, a post hoc analysis combining the two trials suggested that there was a benefit when prehospital transfer times are longer than 20 minutes.11 Additionally, distance from a trauma center and time to treatment are well-documented sources of disparity in rural care. A 2019 study12 demonstrated that states with poor trauma center access had relatively higher pre-hospital death rates. These states with a high pre-hospital death burden had a lower proportion of the population with access to a Level I/II trauma center within 1 hour of injury.12 This is further supported by other studies which have demonstrated that rural residents are 14% more likely to die from traumatic injury compared to non-rural residents.13
Patient care in Montana is influenced by inherent rural disparities and geographic isolation (Figure 1A). Despite this land mass, the 2020 US census demonstrated an average population density of 7.4 people per square mile compared to the average US population of 94 per square mile. The Montana state trauma system was comprised of 41 state-designated trauma facilities (4 ACS-verified Level II, 3 ACS-verified Level III, 11 community trauma hospitals, and 23 trauma receiving facilities). Timely access to a trauma center is not possible in many areas of the state at the current time.
The majority (75%) of respondents to the questionnaire believed that the MT-IBN blood handoff was necessary for optimal patient outcomes but that it would be difficult gaining administrative approval at their individual facility. MT-STCC collaboration with numerous state societies in conjunction with MT-STCC oversight of state trauma destination protocols helped alleviate individual facility administrative difficulties and garner widespread adoption. This led to a profound impact on rural health care delivery with very preliminary evidence of improved survival in the state as seen in Case #2.
During initial MT-IBN discussions, the top concern from providers was to avoid a transfusion-based reaction. A decision was made early in development to begin implementation with PRBCs only and exclude fresh frozen plasma or platelets. Only type O blood was given, with type O negative for females less than 50 years of age; whilst type O positive blood was available for males (18 years of age and older) and females 50 years of age and older. Transfusion of emergent release PRBCs before completion of routine blood bank testing carries a low risk of non-ABO alloantibody mediated hemolytic transfusion reaction rate of 0.4%.14 Other studies have demonstrated a 3% rate of alloimmunization due to emergent release transfusion, a 0.3% rate of incompatible, and a 0.02% rate of delayed hemolytic transfusion reaction.15
A secondary benefit of the MT-IBN includes enhanced care of non-trauma patients such as gynecologic/obstetric bleeding diathesis and GI hemorrhage. The development of the network ensures blood availability in cases whereby patients may otherwise not survive. The uniqueness of the MT-IBN stems from the fact that blood sharing may occur between dozens of unaffiliated hospital systems and small critical access facilities without the need for the patient to stop for care at the intermediary facility. The transport of patients in the MT-IBN does not represent an Emergency Medicine Treatment and Active Labor Act (EMTALA) violation, as patients are stopping at an intermediary facility for a resource instead of an evaluation.16
The closest published and known use of prehospital transfusion such as the MT-IBN concept is through the Southwest Texas Regional Advisory Council (STRAC). STRAC is a collaboration with South Texas Blood and Tissue Center, UT Health San Antonio, University Health Systems, and the US Army Institute of Surgical Research/San Antonio Military Medical Center. STRAC has developed a network of cold stored whole blood products which can be delivered and transfused in the prehospital setting on helicopters and by emergency medical services. They utilized a “Brothers in Arms” system which relies on a broad network of O positive males for donation.17,18 The rurality of Montana would not make this process feasible, and thus the MT-IBN is better suited for this region of the country.
Limitations
The development of this project had limitations. Federal laws dictate that without the appropriate licensing, blood cannot cross state lines. Even though some tertiary centers in Montana have affiliates in Wyoming, Idaho, and the western Dakotas, blood cannot cross over state lines in transport on the way to definitive care. There are current ongoing regulatory discussions to reconcile this issue. Furthermore, participation in MT-IBN is not mandated, and thus, some facilities may opt out of participation if asked to act as an intermediary/donating facility.
Conclusion
The MT-IBN was developed by the MT-STCC to facilitate the hand-off of lifesaving blood to patients being transported by ground to definitive care in Montana. This unique, unparalleled network has already led to lives saved in a state with limited blood availability due to rurality.
Disclosure
All authors have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work. All the authors declare that there is no conflict of interest in this work.
References
1. Curry N, Hopewell S, Doree C, Hyde C, Brohi K, Stanworth S. The acute management of trauma hemorrhage: a systematic review of randomized controlled trials. Crit Care. 2011;15. doi:10.1186/cc10096
2. Kauvar DS, Lefering R, Wade CE. Impact of hemorrhage on trauma outcome: an overview of epidemiology, clinical presentations, and therapeutic considerations. J Trauma. 2006;60(6 Suppl):S3–11. doi:10.1097/01.ta.0000199961.02677.19
3. Holcomb JB. Methods for improved hemorrhage control. Crit Care. 2004;8(Suppl 2):S57–60. doi:10.1186/cc2407
4. Ledgerwood AM, Blaisdell W. Coagulation challenges after severe injury with hemorrhagic shock. J Trauma Acute Care Surg. 2012;72(6):1714–1718. doi:10.1097/TA.0b013e318245225c
5. Spahn DR, Bouillon B, Cerny V, et al. The European guideline on management of major bleeding and coagulopathy following trauma: fifth edition. Critical Care. 2019;23(1):98. doi:10.1186/s13054-019-2347-3
6. McCotter EL, Peterson AL, Savell TC, Orr LC, Besel JM, Thompson SJ. Bringing trauma education to the frontier: overcoming distance barriers utilizing a virtual platform. J Trau Nurs. 2023;30(4):235–241. doi:10.1097/jtn.0000000000000734
7. Montana ETS EMS and Hospital Resources. Montana Department of Public Health and Human Services; 2023. Available from: https://mtdphhs.maps.arcgis.com/apps/instant/interactivelegend/index.html?appid=3500d5f8dd20401ebe8a4fd86ab502d3.
8. Bauman ZM, Loftus J, Hodson A, et al. Rural trauma team development course instills confidence in critical access hospitals. World J Surg. 2020;44(5):1478–1484. doi:10.1007/s00268-019-05359-3
9. Sperry JL, Guyette FX, Brown JB, et al. Prehospital plasma during air medical transport in trauma patients at risk for hemorrhagic shock. N Engl J Med. 2018;379(4):315–326. doi:10.1056/NEJMoa1802345
10. Chapman MP, Moore EE, Chin TL, et al. Combat: initial experience with a randomized clinical trial of plasma-based resuscitation in the field for traumatic hemorrhagic shock. Shock. 2015;44(1):63–70. doi:10.1097/shk.0000000000000376
11. Pusateri AE, Moore EE, Moore HB, et al. Association of prehospital plasma transfusion with survival in trauma patients with hemorrhagic shock when transport times are longer than 20 minutes: a post hoc analysis of the PAMPer and COMBAT clinical trials. JAMA Surgery. 2020;155(2):e195085–e195085. doi:10.1001/jamasurg.2019.5085
12. Hashmi ZG, Jarman MP, Uribe-Leitz T, et al. Access delayed is access denied: relationship between access to trauma center care and pre-hospital death. J Am Coll Surg. 2019;228(1):9–20. doi:10.1016/j.jamcollsurg.2018.09.015
13. Jarman MP, Castillo RC, Carlini AR, Kodadek LM, Haider AH. Rural risk: geographic disparities in trauma mortality. Surgery. 2016;160(6):1551–1559. doi:10.1016/j.surg.2016.06.020
14. Goodell PP, Uhl L, Mohammed M, Powers AA. Risk of hemolytic transfusion reactions following emergency-release RBC transfusion. Am J Clin Pathol. 2010;134(2):202–206. doi:10.1309/ajcp9ofjn7fltxdb
15. Mulay SB, Jaben EA, Johnson P, Badjie K, Stubbs JR. Risks and adverse outcomes associated with emergency-release red blood cell transfusion. Transfusion. 2013;53(7):1416–1420. doi:10.1111/j.1537-2995.2012.03922.x
16. Warby R, Leslie SW, Borger J. EMTALA And Transfers. In: StatPearls. StatPearls Publishing LLC; 2023.
17. Braverman MA, Smith A, Shahan CP, et al. From battlefront to homefront: creation of a civilian walking blood bank. Transfusion. 2020;60:S167–s172. doi:10.1111/trf.15694
18. Schaefer R, Long T, Wampler D, et al. Operationalizing the deployment of low-titer o-positive whole blood within a regional trauma system. Mil Med. 2021;186(Suppl 1):391–399. doi:10.1093/milmed/usaa283
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.