Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 10 » Issue 1
The inspiratory capacity/total lung capacity ratio as a predictor of survival in an emphysematous phenotype of chronic obstructive pulmonary disease
Authors French A, Balfe D, Mirocha J, Falk J, Mosenifar Z
Received 31 October 2014
Accepted for publication 10 January 2015
Published 13 July 2015 Volume 2015:10(1) Pages 1305—1312
DOI https://doi.org/10.2147/COPD.S76739
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Russell
Aimee French, David Balfe, James M Mirocha, Jeremy A Falk, Zab Mosenifar
Cedars-Sinai Medical Center, Division of Pulmonary and Critical Care Medicine, Los Angeles, CA, USA
Background: Forced expiratory volume in 1 second (FEV1) grades severity of COPD and predicts survival. We hypothesize that the inspiratory capacity/total lung capacity (IC/TLC) ratio, a sensitive measure of static lung hyperinflation, may have a significant association with survival in an emphysematous phenotype of COPD.
Objectives: To access the association between IC/TLC and survival in an emphysematous phenotype of COPD.
Methods: We performed a retrospective analysis of a large pulmonary function (PF) database with 39,050 entries, from April 1978 to October 2009. Emphysematous COPD was defined as reduced FEV1/forced vital capacity (FVC), increased TLC, and reduced diffusing capacity of the lungs for carbon monoxide (DLCO; beyond 95% confidence intervals [CIs]). We evaluated the association between survival in emphysematous COPD patients and the IC/TLC ratio evaluated both as dichotomous (≤25% vs >25%) and continuous predictors. Five hundred and ninety-six patients had reported death dates.
Results: Univariate analysis revealed that IC/TLC ≤25% was a significant predictor of death (hazard ratio [HR]: 2.39, P<0.0001). Median survivals were respectively 4.3 (95% CI: 3.8–4.9) and 11.9 years (95% CI: 10.3–13.2). Multivariable analysis revealed age (HR: 1.19, 95% CI: 1.14–1.24), female sex (HR: 0.69, 95% CI: 0.60–0.83), and IC/TLC ≤25% (HR: 1.69, 95% CI: 1.34–2.13) were related to the risk of death. Univariate analysis showed that continuous IC/TLC was associated with death, with an HR of 1.66 (95% CI: 1.52–1.81) for a 10% decrease in IC/TLC.
Conclusion: Adjusting for age and sex, IC/TLC ≤25% is related to increased risk of death, and IC/TLC as a continuum, is a significant predictor of mortality in emphysematous COPD patients.
Keywords: emphysema, pulmonary function testing, mortality
Introduction
COPD represents an important public health challenge that is both treatable and preventable.1 Currently, COPD is the fourth leading cause of death worldwide and is expected to be the third leading cause of death by 2020.2 Despite efforts from the medical community, the Centers for Disease Control (CDC) reports that smoking rates in the US have only slightly declined since 1997 from 24.7% to 20.8%.3 With COPD prevalence increasing, accurate assessments of COPD comorbidities and mortality are needed.4–25 COPD results from the interplay between genetic susceptibility and exposure to environmental stimuli.26
In 1977, Fletcher and Peto described the natural history of COPD, including its relationship with smoking and decline in forced expiratory volume in 1 second (FEV1).27 Since that time, COPD has been characterized as a poorly reversible airflow limitation, most often defined by FEV1.8 Historically, the reduction in FEV1 has been used to define the severity of COPD and frequently cited as an important predictor of mortality.24 Additionally, FEV1 is frequently targeted as a clinical endpoint in COPD clinical trials.28–34 Other clinical measurements such as inspiratory capacity (IC), 6-minute walk test (6MWT), the BODE index (body mass index, airflow obstruction, dyspnea, and exercise), and dyspnea questionnaires may have stronger associations with mortality than FEV1.7,12–16,19,24,35–39
Several publications have focused on the use of the IC/total lung capacity (TLC) ratio, a measure of static lung hyperinflation, which has been shown to be strongly associated with exercise-associated dynamic hyperinflation as well as exercise tolerance.4,8,38,40 Additionally, studies evaluating the use of resting IC have also demonstrated a strong association between IC and functional exercise limitation in COPD patients.41,42 In 2004, Casanova et al evaluated the role of the IC/TLC ratio, in conjunction with the BODE index and FEV1.7 Their cohort consisted of 689 patients (95% male) with 183 deaths (178 males and five females) with a median follow-up less than 3 years; their results suggested that when compared to FEV1 and the BODE index, an IC/TLC ratio of ≤25% offered the best combined sensitivity and specificity for predicting all-cause mortality in COPD patients.7 We hypothesize that the IC/TLC ratio is associated with risk of death, when used to evaluate a large cohort over an extended period of follow-up and could be a useful clinical tool in assessing patients with an emphysematous phenotype of COPD.
Methods
We performed a retrospective analysis of a large pulmonary function (PF) database, consisting of 39,050 entries, from our institution, which encompasses a broad patient population of inpatients and outpatients from April 1978 to October 2009. Cedars-Sinai Medical Center Institutional Review Board approval (Pro-00012916) was obtained for the study. We evaluated the IC/TLC ratio and its association with survival. All available studies were evaluated, which consisted of 39,050 entries. We defined COPD patients with an emphysematous phenotype as those with a reduced FEV1/FVC ratio, increased TLC, and reduced diffusing capacity of the lungs for carbon monoxide (DLCO; all beyond 95% confidence intervals) using established normal values.42–44 If the same patient had multiple PF studies, only the first recorded PF was included in the study analysis. Lastly, patients were excluded if they were under the age of 18 years. Using the Social Security National Death Index (SSNDI), we established absolute dates of death. If the subject did not have a reported date of death in the SSNDI, they were assumed to be alive 8 weeks prior to the date that the SSNDI was queried. Univariate analysis was completed to evaluate the relationship between IC/TLC ratio and survival. Additionally, disease severity was graded using the FEV1 as defined by the Global Initiative for Chronic Obstructive Lung Disease (GOLD) criteria.46 Body mass index (BMI) was treated as ordered categories (BMI: <20 low, ≥20–25 normal, ≥25 overweight/obese). As done previously in the NETT trial, DLCO was treated as a binary variable, with a cut-off of <22%.47 Finally, multivariable analysis assessing the associations of age, sex, FEV1, DLCO, IC, and BMI in conjunction with the IC/TLC ratio was performed. All studies were performed on equipment manufactured by the SensorMedics Corporation. IC measurements were made during the enhanced spirometry section with deep inspiration occurring after a period of stable tidal breathing; all tests were performed according to the American Thoracic Society (ATS) criteria. Single-breath DLCO measurements were made according to ATS criteria with a 10-second breath-hold; correction for hemoglobin was not performed. TLC was measured using plethysmography as per ATS criteria. Forced expiratory spirometry was obtained pre- and post-spirometry; plethysmography was obtained post-bronchodilator.
Numerical variables were summarized by mean and standard deviation or median and interquartile range (IQR), as appropriate, depending on the normality of the distribution. Group differences on numerical variables were assessed by Student’s t-tests (normality was satisfied) or the Wilcoxon rank-sum test (normality was violated). Categorical variables were summarized by frequency and percentage and compared across groups by the chi-square test or Fisher’s exact test, as appropriate. Survival was estimated by the Kaplan–Meier method. Group differences in survival were assessed by the log-rank test. Survival differences across ordinal explanatory variables were assessed by trend tests. Factors associated with the risk of death were assessed by multivariable Cox proportional hazards models. The proportional hazards assumption was assessed by the supremum test and by analysis of Schoenfeld residuals. Hazard ratios (HRs) and their associated 95% CIs were reported where possible. Two-sided P-values were reported for all comparisons. Statistical calculations were made using SAS software (v9.1.3; SAS Institute Inc., Cary, NC, USA).
Results
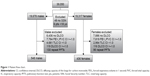
Our database consisted of 39,050 PF tests; 14,322 were excluded because they had no DLCO recorded. The remaining 24,728 PFs that were excluded, either did not meet criteria for obstruction or were repeat PF tests. Nine-hundred and eighty-four patient PFs meeting criteria were analyzed (Figure 1). The study cohort consisted of 436 females and 548 males. Five hundred and ninety-six subjects had reported dates of death.
Baseline characteristics are reported in Table 1. As displayed in Table 1, there were notable differences between the ‘dead’ and ‘alive’ groups. ‘Alive’ patients were younger, had a higher BMI, a higher FEV1/FVC ratio, and higher predicted FEV1 with a larger percentage being defined as having mild COPD. In regards to lung volumes, ‘alive’ patients had a slightly lower TLC, higher DLCO, higher IC, and had better IC/TLC ratios than patients in the ‘dead’ group.
The potential predictors of all-cause mortality are shown in Tables 2 and 3.
Univariate analysis revealed that IC/TLC, % DLCO, sex, age, BMI, IC, and FEV1 were all significantly associated with survival (Table 2; Figures 2, 3, and 4). Using an IC/TLC ≤25% compared to IC/TLC >25%, there was a significant decrease in survival (HR: 2.39, 95% CI: 2.03–2.82, P<0.0001). The median survival was 4.3 years for IC/TLC ≤25% (95% CI: 3.8–4.9 years) versus 11.9 years for IC/TLC >25% (95% CI: 10.3–13.2 years). Five-year survival for IC/TLC ≤25% versus IC/TLC >25% was 43.9% (95% CI: 38.7%–49.0%) versus 73.4% (95% CI: 69.6%–76.9%), respectively. Additionally, a DLCO <22% versus a DLCO ≥22% was associated with an increased risk of death (HR: 2.08, 95% CI: 1.68–2.57, P<0.0001). The median survival for a DLCO <22% was 3.77 years (95% CI: 2.94–4.60 years). Females had reduced risk of death (HR: 0.78, 95% CI: 0.66–0.92, P=0.003). Median survival for females was 8.7 years (95% CI: 7.7–10.1 years) versus 7.0 years for males (95% CI: 5.7–8.2 years). A 5-year increase in age revealed a 23% increased risk of death (HR: 1.23, 95% CI: 1.18–1.28, P<0.0001). Compared to a normal BMI (≥20 to <25, the reference level), patients with a BMI <20 were at greater risk of death (HR: 1.22, 95% CI: 0.99–1.51, P=0.068). However, a BMI ≥25 appeared to be protective (HR: 0.81, 95% CI: 0.67–0.97, P=0.022). An IC ≥80% was associated with a reduced risk of death (HR: 0.48, 95% CI: 0.41–0.57, P<0.0001). Lastly, FEV1 was evaluated using moderate COPD disease, as defined by GOLD criteria, as the reference group: patients with mild COPD were less likely to die (HR: 0.48, 95% CI: 0.35–0.65, P<0.001); however, both severe (HR: 1.38, 95% CI: 1.11–1.70, P=0.003) and very severe COPD (HR: 1.81, 95% CI: 1.46–2.23, P<0.0001) patients were at increased risk of death.
  | Figure 3 Patient survival by IC/TLC. |
  | Figure 4 Patient survival by sex. |
In addition, a multivariable analysis was preformed to assess the associations of age, sex, FEV1, % DLCO, IC, and BMI in conjunction with the IC/TLC ratio. Analysis revealed that age (5-year increase in age), sex, IC/TLC ≤25%, FEV1, BMI <20, and DLCO <22% remained statistically significant factors associated with the risk of death (Table 3). Patients with a DLCO <22% had an increased risk of death when compared to patients with a DLCO ≥22% (HR: 1.28, 95% CI: 1.01–1.61, P=0.043) (Figure 5). Compared to a normal BMI (≥20 to <25, the reference level), patients with a BMI <20 were at greater risk of death (HR: 1.22, 95% CI: 0.97–1.53, P=0.089). In contrast, a BMI ≥25 appeared to be protective (HR: 0.81, 95% CI: 0.67–0.97, P=0.022) (Figure 6). Lastly, FEV1 was evaluated using moderate COPD disease, as defined by GOLD criteria, as the reference group. Patients with mild COPD were less likely to die (HR: 0.66, 95% CI: 0.48–0.90, P=0.009). Additionally, neither severe (HR: 1.01, 95% CI: 0.80–1.27, P=0.95) nor very severe COPD (HR: 1.06, 95% CI: 0.79–1.41, P=0.71) patients were at increased risk of death, compared to the moderate group in the multivariate analysis. In summary, increasing age, IC/TLC ≤25%, DLCO <22%, and a BMI <20 were associated with increased risk of death. Female sex and a higher BMI were protective.
  | Figure 5 Patient survival by DLCO. |
  | Figure 6 Survival by BMI level. |
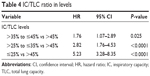
Lastly, we completed a univariate analysis of the IC/TLC ratio as a continuous predictor of the risk of death. Over the entire range of IC/TLC (8%–63%), a 10% decrease in IC/TLC was associated with a 66% increase in the risk of death (HR: 1.66, 95% CI: 1.52–1.81, P<0.0001). For clinical applicability, we categorized the ratio into four levels: IC/TLC >45% (the reference level), >35% to ≤45%, >25% to ≤35%, and ≤25% (Table 4; Figure 7). Compared to patients with IC/TLC >45%, patients with IC/TLC >35% to ≤45% were at 76% increased risk of death (HR: 1.76, 95% CI: 1.07–2.89); patients with IC/TLC >25% to ≤35% were at 182% increased risk of death (HR: 2.82, 95% CI: 1.76–4.53); and patients with IC/TLC ≤25% were at 423% increased risk of death (HR: 5.23, 95% CI: 3.28–8.35). Median survivals (95% CI) were 4.3 (3.8–4.9) years for patients with IC/TLC ≤25%, 8.9 (7.7–10.5) years for patients with IC/TLC >25% to ≤35%, and 14.1 (11.9–18.7) years for patients with IC/TLC >35% to ≤45%. Median survival was not yet reached in patients with IC/TLC >45% (30-year survival estimate 52.3%).
  | Table 4 IC/TLC ratio in levels |
  | Figure 7 Patient survived by IC/TLC level. |
Discussion
Using an IC/TLC ratio of ≤25%, a representation of static lung hyperinflation, we were able to demonstrate that the IC/TLC ratio correlated with risk of death in patients with an emphysematous phenotype of COPD. Patients with an IC/TLC ratio ≤25% had a median survival of 4.3 years versus 11.9 years for those patients with an IC/TLC >25%. The use of the IC/TLC ratio, as a single predictor variable, in patients with COPD, will allow clinicians to better evaluate and risk-stratify patients with an emphysematous phenotype of COPD.
In the 35 years since Fletcher and Peto described the natural history of COPD, our understanding about the disease process has been an ongoing development.27 Although initial studies supported FEV1 as the best predictor of COPD mortality,47 more recent literature has revealed that a variety of factors are more accurate predictors of mortality than FEV1.7,12–16,19,24,34–39 Interest has been shown in the role of the vitamin D pathway in COPD, with vitamin D deficiency correlating with the risk of COPD.26
In addition to IC/TLC and FEV1, we also examined the relationship of IC, age, BMI, DLCO, and sex (Tables 1 and 3) and their associations with the risk of death. Specifically, our multivariable analysis demonstrated that IC alone was not a reliable predictor of mortality, contrary to what has been previously reported in a milder COPD population.24 We were able to demonstrate that age and BMI were associated with risk of death but did not demonstrate as clear an association as the IC/TLC. In regards to sex, Kaplan–Meier analysis revealed that females had a survival advantage when compared directly to males with COPD. Ringbaek et al previously reported that when compared to standardized mortality rates expected in the general population, it appeared that females with COPD actually have more years of life lost than their male counterparts.21 Furthermore, when examining the DLCO, we found a significant association with risk of death. However, our cohort had only 126 patients with a DLCO ≤22%.
Our study had a number of strengths. To our knowledge, it is the first study to evaluate mortality over 34 years of reported data in an emphysematous phenotype of COPD. Additionally, our cohort consisted of 44.3% (436) females, and to date, is one of the largest cohorts of females used to evaluate COPD mortality. We used an easily identifiable IC/TLC cut-off of 25%, which has previously been validated.7
The study does have limitations. It is a PF test-based study, assumptions about the patients’ clinical phenotype were made on the basis of PF data, and no smoking history or radiological data were available. We did not evaluate or compare the BODE index or 6MWT. On the other hand, we did evaluate other well-defined variables including age, BMI, and FEV1 in the analysis. We did not have PaO2 values, therefore could not evaluate/correlate the degree of hypoxemia and its role in survival. The study inclusion criteria required a DLCO for the patient to be included in the analysis. In doing so, we eliminated patients who may have obstructive disease secondary to asthma, but undoubtedly removed many patients with COPD. We did not evaluate specific cause of death as previous studies have done, which prevented us from excluding patients that died from nonrespiratory events.
Our study demonstrates that the IC/TLC ratio ≤25% is strongly related to risk of death in an emphysematous phenotype of COPD patients. When compared to FEV1 and other previously accepted variables, IC/TLC was a better predictor of survival in both univariate and multivariate analysis. Specifically, we found FEV1 to be weakly associated with survival. When using patients with moderate severity as the reference group, those patients with mild disease were noted to have improved survival. Patients with severe and very severe disease did not have a statistically significant difference in mortality when compared to those individuals with moderately severe disease.
We believe that the reduced IC/TLC ratio, reflective of static lung hyperinflation, plays an important role in patients with an emphysematous phenotype of COPD. As hyperinflation progresses, patients experience increasing dyspnea and reduced exercise capacity. Dynamic lung hyperinflation is associated with increasing end-expiratory lung volumes, which is associated with airflow limitation and loss of elastic recoil. In COPD, emphysematous changes in the lung are secondary to destruction of the elastic tissue in the lung parenchyma. As the lung parenchyma loses it elasticity, lung recoil is reduced, resulting in a compensatory increase in functional reserve capacity (FRC); as the FRC increases, the IC is reduced.49 Therefore, in patients with COPD, the relationship between IC and TLC gradually declines as static lung hyperinflation increases. We found that the IC/TLC ratio strongly correlates with risk of death. In addition to risk of death, static lung hyperinflation has been reported to correlate with exercise capacity and cardiac performance in patients with COPD, thus adding to the clinical use of the IC/TLC ratio.
Previous studies of COPD patients often refer to the FEV1 and its rate of decline to define treatment success or failure. Current treatment guidelines are based on the GOLD-defined FEV1, an endpoint that we found to have little correlation with survival. We believe that the IC/TLC ratio may be a more relevant variable for such analysis, possible arguing for further studies.
In conclusion, a significant number of COPD patients have PF tests that include measurement of lung volumes. The IC/TLC ratio, like the BODE index, has demonstrated a correlation with COPD survival, is easy to obtain, and should be considered in the assessment of patients with an emphysematous phenotype of COPD.
Disclosure
The authors report no conflicts of interest in this work.
References
Centers for Disease Control and Prevention (CDC). Deaths from chronic obstructive pulmonary disease United States, 2000–2005. MMWR Morb Mortal Wkly Rep. 2008;57(45):1229–1232. | ||
Xu JQ, Kochanek KD, Murphy SL, Tejeda-Vera B. Deaths: Final Data for 2007. National Vital Statistics Reports. Vol 58, no 19. Hyattsville, MD: National Health Center for Health Statistics; 2010. Available from: http://www.cdc.gov/nchs/data/nvsr/nvsr58/nvsr58_19.pdf. Accessed January 20, 2015. | ||
Centers for Disease Control and Prevention (CDC). Cigarette smoking among adults – United States, 2006. MMWR Morb Mortal Wkly Rep. 2007;56(44):1157–1161. | ||
Albuquerque AL, Nery LE, Villaça DS, et al. Inspiratory fraction and exercise impairment in COPD patients with GOLD stages II–III. Eur Respir J. 2006;28(5):939–944. | ||
Bourdin A, Burgel PR, Chanez P, Garcia G, Perez T, Roche N. Recent advances in COPD: pathophysiology, respiratory physiology and clinical aspects, including comorbidities. Eur Respir J. 2009;18(114): 198–212. | ||
Calverly PM, Anderson JA, Celli B, et al; TORCH investigators. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. N Engl J Med. 2007;356:775–789. | ||
Casanova C, Cote C, Marin JM, et al. Distance and oxygen desaturation during the 6-min walk test as predictors of long-term mortality in patients with COPD. Chest. 2008;134(4):746–752. | ||
Casanova C, Cote C, de Torres JP, et al. Inspiratory-to-total lung capacity ratio predicts mortality in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2005;171(6):591–597. | ||
Soler-Cataluña JJ, Martínez-García MA, Román Sánchez P, Salcedo E, Navarro M, Ochando R. Severe acute exacerbations and mortality in patients with chronic obstructive pulmonary disease. Thorax. 2005;60(11):925–931. | ||
Celli BR, Vestbo J, Jenkins CR, et al; Investigators of the TORCH Study. Sex differences in mortality and clinical expressions of patients with chronic obstructive pulmonary disease. The TORCH experience. Am J Respir Crit Care Med. 2011;183(3):317–322. | ||
Celli BR, Decramer M, Kesten S, Liu D, Mehra S, Tashkin DP; UPLIFT Study Investigators. Mortality in the 4-year trial of tiotropium (UPLIFT) in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2009;180(10):948–955. | ||
Celli, Bartolome R. Predictors of mortality in COPD. Respir Med. 2010;104:773–779. | ||
Cote CG, Pinto-Plata V, Kasprzyk K, Dordelly LJ, Celli BR. The 6-min walk distance, peak oxygen uptake, and mortality in COPD. Chest. 2007;132(6):1778–1785. | ||
Fan VS, Curtis JR, Tu SP, McDonell MB, Fihn SD; Ambulatory Care Quality Improvement Project Investigators. Using quality of life to predict hospitalization and mortality in patients with obstructive lung diseases. Chest. 2002;122(2):429–436. | ||
Martinez FJ, Foster G, Curtis JL, et al; NETT Research Group. Predictors of mortality in patients with emphysema and severe airflow obstruction. Am J Respir Crit Care Med. 2006;173(12):1326–1334. | ||
Nishimura K, Izumi T, Tsukino M, Oga T. Dyspnea is a better predictor of 5-year survival than airway obstruction in patients with COPD. Chest. 2002;121(5):1434–1440. | ||
Oga T, Nishimura K, Tsukina M, et al. Health status measured with the CRQ does not predict mortality in COPD. Eur Respir J. 2002;20(5):1147–1151. | ||
Oga T, Nishimura K, Tsukino M, Sato S, Hajiro T. Analysis of the factors related to mortality in chronic obstructive pulmonary disease: role of exercise capacity and health status. Am J Respir Crit Care Med. 2003;167(4):544–549. | ||
Pinto-Plato VM, Cote C, Cabral H, Taylor J, Celli BR. The 6-min walk distance: change over time and value as a predictor of survival in severe COPD. Eur Respir J. 2004;23(1):28–33. | ||
Sabino PG, Silva BM, Brunetto AF. Nutritional status is related to fat-free mass, exercise capacity and inspiratory strength in severe chronic obstructive pulmonary disease patients. Clinics (Sao Paulo). 2010;65(6):599–605. | ||
Ringbaek T, Seersholm N, Viskum K. Standardised mortality rates in females and males with COPD and asthma. Eur Respir J. 2005;25(5):891–895. | ||
Sin DD, Anthonisen NR, Soriano JB, Agusti AG. Mortality in COPD: role of comorbidities. Eur Respir J. 2006;28(6):1245–1257. | ||
Sin DD, Wu L, Anderson JA, et al. Inhaled corticosteroids and mortality in chronic obstructive pulmonary disease. Thorax. 2005;60(12): 992–997. | ||
Tantucci C, Donati P, Nicosia F, et al. Inspiratory capacity predicts mortality in patients with chronic obstructive pulmonary disease. Respir Med. 2008;102(4):613–619. | ||
de Torres JP, Cote CG, López MV, et al. Sex differences in mortality in patients with COPD. Eur Respir J. 2009;33(3):528–535. | ||
Decramer M, Janssens W, Miravitlles M. Chronic obstructive pulmonary disease. Lancet. 2012;379(9823):1341–1351. | ||
Fletcher C, Peto R. The natural history of chronic airflow obstruction. BMJ. 1977;1(6077):1645–1648. | ||
Jenkins CR, Jones PW, Calverley PM, et al. Efficacy of salmeterol/fluticasone by GOLD stage of chronic obstructive pulmonary disease: analysis from the randomised, placebo-controlled TORCH study. Respir Res. 2009;10:59. | ||
Celli BR, Thomas NE, Anderson JA, et al. Effects of pharmacotherapy on rate of decline of lung function in chronic obstructive pulmonary disease: results from the TORCH study. Am J Respir Crit Care Med. 2008;178(4):332–338. | ||
Decramer M, Celli BR, Kesten S, Lystig T, Mehra S, Tashkin DP; UPLIFT investigators. Effect of triotropium on outcomes in patients with moderate chronic obstructive pulmonary disease (UPLIFT): a prespecified subgroup analysis of a randomised controlled trial. Lancet. 2009;374(9696):1171–1178. | ||
Gan WQ, Man SF, Senthilselvan A, Sin DD. Association between chronic obstructive pulmonary disease and systemic inflammation: a systemic review and a meta-anaylsis. Thorax. 2004;59(7):574–580. | ||
Miravitlles M, Anzueto A. Insights into interventions in managing COPD patients: lessons from the TORCH and UPLIFT studies. Int J Chron Obstruct Pulmon Dis. 2009;4:185–201. | ||
Soriano JB, Sin DD, Zhang X, et al. A pooled analysis of FEV1 decline in COPD patients randomized to inhaled corticosteroids or placebo. Chest. 2007;131(3):682–689. | ||
Celli BR, Cote CG, Marin JM, et al. The body-mass index, airflow obstruction, dyspnea, and exercise capacity index in chronic obstructive pulmonary disease. N Engl J Med. 2004;350(10):1005–1012. | ||
Glaab T, Vogelmeier C, Buhl R. Outcome measures in chronic obstructive pulmonary disease (COPD): strengths and limitations. Respir Res. 2010;11:79. | ||
Freitas CG, Pereira CA, Viegas CA. Inspiratory capacity, exercise limitations, markers of severity, and prognostic factors in chronic obstructive pulmonary disease. J Bras Pneumol. 2007;33(4):389–396. | ||
Landbo C, Prescott E, Lange P, Vestbo J, Almdal TP. Prognostic value of nutritional status in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1999;160(6):1856–1861. | ||
Marin JM, Carrizo SJ, Gascon M, Sanchez A, Gallego B, Celli BR. Inspiratory capacity, dynamic hyperinflation, breathlessness, and exercise performance during the 6-minute-walk test in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2001;163(6):1395–1399. | ||
Puhan MA, Garcia-Aymerich J, Frey M, et al. Expansion of the prognostic assessment of patients with chronic obstructive pulmonary disease: the updated BODE index and the ADO index. Lancet. 2009;374(9691):704–711. | ||
Vassaux C, Torre-Bouscoulet L, Zeineldine S, et al. Effects of hyperinflation on the oxygen pulse as a marker of cardiac performance in COPD. Eur Respir J. 2008;32(5):1275–1282. | ||
Diaz O, Villafranca C, Ghezzo H, et al. Role of inspiratory capacity on exercise tolerance in COPD patients with and without tidal expiratory flow limitations at rest. Eur Respir J. 2000;16(2):1256–1276. | ||
O’Donnell DE, Reville SM, Webb KA. Dynamic hyperinflation and exercise intolerance in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2001;164(5):770–777. | ||
Crapo RO, Morris AH, Clayton PD, Nixon CR. Lung volumes in healthy nonsmoking adults. Bull Eur Physiopathol Respir. 1982; 18(3):419–425. | ||
Miller A, Thorton JC, Warshaw R, Anderson H, Teirstein AS, Selikoff IJ. Single breath diffusing capacity in a representative sample of the population of Michigan, a large industrial state. Predicted values, lower limits of normal, and frequencies of abnormality by smoking history. Am Rev Respir Dis. 1983;127(3):270–277. | ||
Pauwels RA, Buist AS, Calverley PM, Jenkins CR, Hurd SS; GOLD Scientific Committee. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) Workshop summary. Am J Respir Crit Care Med. 2001;163(5):1256–1276. | ||
Rationale and Design of The National Emphysema Treatment Trial: a prospective randomized trial of lung volume reduction surgery. The National Emphysema Treatment Trial Research Group. Chest. 1999; 116(6):1750–1761. | ||
Anthonisen NR, Skeans MA, Wise RA, Manfred J, Kanner RE, Connett JE; Lung Health Study Research Group. The effects of smoking cessation intervention on 14.5-year mortality. Ann Intern Med. 2005; 142(4):233–239. | ||
Ferguson GT. Why does the lung hyperinflate? Proc Am Thorac Soc. 2006;3(2):176–179. | ||
National Heart, Lung, and Blood Institute. Global Strategy for the Diagnosis, Management and Prevention of Chronic Obstructive Pulmonary Disease: NHLBI/NIH Workshop Report. Bethesda, MD: National Heart, Lung, and Blood Institute; 2001. Available from: http://www.goldcopd.org/uploads/users/files/GOLDWkshp2001.pdf. Accessed January 20, 2015. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.