Back to Journals » Drug Design, Development and Therapy » Volume 12
The influence of prebiotic or probiotic supplementation on antibody titers after influenza vaccination: a systematic review and meta-analysis of randomized controlled trials
Authors Yeh TL , Shih PC , Liu SJ , Lin CH, Liu JM , Lei WT , Lin CY
Received 25 October 2017
Accepted for publication 14 December 2017
Published 25 January 2018 Volume 2018:12 Pages 217—230
DOI https://doi.org/10.2147/DDDT.S155110
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Qiongyu Guo
Tzu-Lin Yeh,1 Pei-Ching Shih,1 Shu-Jung Liu,2 Chao-Hsu Lin,3 Jui-Ming Liu,4,5 Wei-Te Lei,3,* Chien-Yu Lin3,*
1Department of Family Medicine, Hsinchu MacKay Memorial Hospital, Hsinchu, 2Department of Medical Library, MacKay Memorial Hospital, Tamsui Branch, New Taipei City, 3Department of Pediatrics, Hsinchu MacKay Memorial Hospital, Hsinchu, 4Department of Surgery, Taoyuan General Hospital, Ministry of Health and Welfare, Taoyuan, 5Department of Medicine, National Yang-Ming University, Taipei, Taiwan
*These authors contributed equally to this work
Background: Influenza infection is a common disease with a huge disease burden. Influenza vaccination has been widely used, but concerns regarding vaccine efficacy exist, especially in the elderly. Probiotics are live microorganisms with immunomodulatory effects and may enhance the immune responses to influenza vaccination.
Methods: We conducted a systematic review and meta-analysis to determine the influence of prebiotics/probiotics/synbiotics supplementation on vaccine responses to influenza vaccination. Studies were systematically identified from electronic databases up to July 2017. Information regarding study population, influenza vaccination, components of supplements, and immune responses were extracted and analyzed. Twelve studies, investigating a total of 688 participants, were included in this review.
Results: Patients with prebiotics/probiotics supplements were found to have higher influenza hemagglutination inhibition antibody titers after vaccination (for A/H1N1, 42.89 vs 35.76, mean difference =7.14, 95% CI =2.73, 11.55, P=0.002; for A/H3N2, 105.4 vs 88.25, mean difference =17.19, 95% CI =3.39, 30.99, P=0.01; for B strain, 34.87 vs 30.73, mean difference =4.17, 95% CI =0.37, 7.96, P=0.03).
Conclusion: Supplementation with prebiotics or probiotics may enhance the influenza hemagglutination inhibition antibody titers in all A/H1N1, A/H3N2, and B strains (20%, 19.5%, and 13.6% increases, respectively). Concomitant prebiotics or probiotics supplementation with influenza vaccination may hold great promise for improving vaccine efficacy. However, high heterogeneity was observed and further studies are warranted.
Keywords: influenza, influenza vaccine, probiotics, prebiotics, synbiotics, antibody titer, immune response
Introduction
Influenza is a common infectious disease with a huge disease burden worldwide. It is estimated to be responsible for 250,000–500,000 deaths annually, especially among the elderly.1 Influenza vaccination prevents influenza infection. Usually, the influenza vaccine is composed of split virions with 2 A strains (A/H1N1 and A/H3N2) and 1 B strain (Victoria or Yamagata lineages). Influenza vaccines are widely used, but concerns regarding vaccine efficacy exist, especially in the elderly. In a meta-analysis published in 2012, the pooled efficacy was 59% in adults aged 18–65 years, and evidence of protection in the elderly was lacking.2 Low vaccine efficacy leads to inadequate protection, breakthrough infection, and influenza-related morbidity and mortality. Efforts have been made to improve the immune responses to influenza vaccines, such as adding adjuvant supplements, nutritional interventions, or increasing the vaccine dose.3,4 In summary, the efficacy of the current influenza vaccine is not satisfactory.
The human intestine is host to a vast variety of microbes. Probiotics are microorganisms that have beneficial properties for the host and are known to alter the intestinal microflora.5,6 Prebiotics are defined as dietary components that stimulate the growth and metabolic activity of probiotics. Synbiotics are the combination of prebiotics and probiotics. Application of prebiotics/probiotics/synbiotics suppresses the growth of pathogenic bacteria and improves the intestinal barrier function, and is widely used in patients with gastrointestinal infections and inflammation.7,8 In addition to the beneficial effects on the intestinal tract, probiotics also have immunomodulatory effects by inducing production of protective cytokines and suppressing pro-inflammatory cytokines.9–12 Extraintestinal benefits of probiotics include immune regulations in allergic diseases, cardiovascular diseases, and suppression of tumor growth.13–15 Adjuvant probiotic use in these diseases is a potential target for future development.
The beneficial properties of immune modulation that follow probiotics consumption may enhance the immune responses to influenza vaccines.16–20 Several randomized controlled trials (RCTs) have been conducted to investigate the influence of probiotics on influenza vaccines, but the results were inconsistent and inconclusive. Therefore, we conducted this systematic review and meta-analysis to evaluate the impacts of prebiotics/probiotics/synbiotics on immune responses after influenza vaccination.
Materials and methods
Study design and study selection
This study was approved by the Ethics Committees of MacKay Memorial Hospital, Taiwan (IRB No: 16MMHIS174e) and conducted in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols guidelines.21 We systematically searched for all relevant articles in the following online databases: Embase, PubMed, the Cochrane Library, the Cumulative Index to Nursing and Allied Health, the Airiti Library, and the PerioPath Index to the Taiwan Periodical Literature in Taiwan, from the earliest record to July 2017. The Cochrane Collaboration Central Register of Controlled Clinical Trials, Cochrane Systematic Reviews, and ClinicalTrials.gov were manually searched for additional references. The key terms used for the search were “influenza vaccine”, “probiotics”, “prebiotics”, and “synbiotics”. Keywords were combined using Boolean searches and the search was made using keywords, Boolean operators, and MeSH descriptor. The detailed search strategy is enclosed as Box S1. Two authors (P-CS and S-JL) conducted the search independently, and disagreements were resolved through discussion with the third author (W-TL).
After the initial search, 2 independent reviewers (P-CS and T-LY) assessed the eligibility of each publication. The inclusion criteria of selected RCTs were as follows: 1) studies in adults; 2) inclusion of a control group in the study design; 3) use of influenza vaccination and supplementation of probiotics, prebiotics, or synbiotics in the intervention group; 4) reporting of at least 1 immunological response to influenza vaccination. We excluded the following: 1) articles irrelevant to the topic; 2) duplicate publications; 3) trials of a cross-over study design; and 4) studies in which the control arm received an effective intervention rather than a placebo.
Data extraction and quality assessment
Two authors (W-TL and T-LY) independently evaluated the quality assessment of all eligible articles using the Cochrane Review risk of bias assessment tool. We assessed the adequacy of randomization, allocation concealment, blinding methods, implementation of the intent-to-treat analysis, dropout rate, complete outcome data, selective data reporting, and other biases of each enrolled publication.
The articles were scrutinized, and data regarding study population, influenza vaccine components, protocols of probiotics consumption, details of vaccine immune responses, and adverse effects from the selected studies were extracted. Discrepancies between the 2 independent evaluations for potential articles were resolved through discussion and consensus. The primary outcome was the immunogenicity of influenza vaccination, presented as hemagglutination inhibition (HI) antibody titers. The HI antibody titer equals the maximum dilution capable of inhibiting the agglutination of guinea pig red blood cells, with the influenza viruses under standardized conditions.22 Other comparative variables included the components of the vaccine and probiotics, the protocols of probiotics consumption, and the serious adverse effects.
Data synthesis and analysis
Immunogenicity data from all the studies were extracted, analyzed, and compared to determine differences in the efficacy of influenza vaccination in the groups receiving prebiotics/probiotics/synbiotics supplementation and the placebo groups. Due to significant (and expected) heterogeneity among the studies, a random effects model was employed.23 The results were represented by a point estimate with a 95% CI. The heterogeneity across studies was tested using I2 and Cochran’s Q tests. A P-value <0.10 for chi-square testing of the Q statistic or an I2>50% was considered as statistically significant heterogeneity.24 A sensitivity analysis was performed by removing some studies to observe whether the action caused serious changes in the overall results. The potential publication bias was assessed by observing the symmetry of funnel plots and using Egger’s test.25 Review Manager (version 5.3.5) was used for our analyses.
Results
Description of studies and quality assessment
Of the 22 non-duplicate citations identified from the literature, 2 studies were not RCTs and 20 were ultimately assessed for eligibility (Figure 1). Finally, 11 publications with 12 RCTs were included in our qualitative synthesis after critical review (Table 1).26–36 Two trials (a pilot and a confirmatory study) with different patient numbers, treatment protocols, and years of study were published in the same article.34 Seven studies investigated the effects of probiotics, and five studies investigated the effects of prebiotics. One study investigating synbiotics was excluded, after critical review, for using a different outcome parameter.37 The included studies were conducted in the USA, France, Japan, and the UK. In total, 780 patients were enrolled in these studies with female predominance (M:F =1:2.1). Five different probiotics and 5 different prebiotics were used in the intervention arm. The trivalent inactivated influenza vaccines (TIV) were used in most studies (10/12). Most of the included studies had a low bias, as shown by our quality assessment using the Cochrane assessment tool. The detailed quality assessment of each included study is shown in Table S1.
Data synthesis and meta-analysis
Ultimately, 688 patients were enrolled in our meta-analysis. By comparing the HI titers of strain A/H1N1 after influenza vaccination, we found a significantly higher HI titers in the probiotics/prebiotics group (42.89 vs 35.76, mean difference =7.14, 95% CI =2.73, 11.55, P<0.001, I2=96%) (Figure 2). For strain A/H3N2, similar increase in HI titers was observed (105.4 vs 88.25, mean difference =17.19, 95% CI =3.39, 30.99, P<0.001, I2=100%) (Figure 3). In patients with prebiotics/probiotics supplement, higher immune responses after influenza vaccination was noticed for strain B (34.87 vs 30.73, mean difference =4.17, 95% CI =0.37, 7.96, P<0.001, I2=94%) (Figure 4). The percentages of increases were 20% (A/H1N1), 19.5% (A/H3N2), and 13.6% (B strain); the mean HI antibody titers are summarized in Table 2. Subgroup analysis of prebiotics and probiotics showed similar results. The heterogeneity was high in all analyses. We found no significant differences in serious adverse effects in either arm (Figure 5). The funnel plots were also assessed (Figures S1–S3).
  | Figure 4 Forest plot of the HI titers of B strain between prebiotic or probiotic group, and placebo group. |
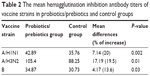  | Table 2 The mean hemagglutination inhibition antibody titers of vaccine strains in probiotics/prebiotics and control groups |
  | Figure 5 Forest plot of the incidence of adverse effect between prebiotic or probiotic group, and placebo group. |
Discussion
Our systematic review and meta-analysis support the beneficial effects of prebiotic/probiotic supplementation on humoral responses to influenza vaccination. We found that supplementation with pre- or probiotics enhanced the HI titers in all A/H1N1, A/H3N2, and B strains (20%, 19.5%, and 13.6% increases in HI antibody titers, respectively). Concomitant prebiotics/probiotics supplementation potentially improved the protection of influenza vaccination and decreased the subsequent risk of influenza-related morbidity and mortality. However, high heterogeneity was noted and further studies are warranted to consolidate this suggestion.
Influenza is highly contagious and virulent. Despite widespread use of influenza vaccination, it remains an important health threat. Currently, the effectiveness of influenza vaccination is not satisfactory and multiple factors contribute to the low effectiveness, including antigen drift, season mismatch, and manufacture technique limitations.2,38,39 Elderly individuals have both the highest burden of disease and the lowest immune responses to vaccination.40–42 The protection rate may be as low as 30% in elderly people after vaccination and little evidence is found supporting the benefits of influenza vaccination in the elderly.40,41 Immunosenescence, gradual deterioration of the immune system brought on by natural aging, also plays an important role in the hyporesponsiveness of influenza vaccination.43 Poorer nutritional status and higher rates of comorbid diseases are also important reasons for the nearly inevitable weak immune responses after vaccination in the elderly.44,45 The TIV with high doses (4× the standard dose) induced significantly higher antibody responses in elderly people, but are not widely used.3 Supplementation with prebiotics/probiotics may provide a simple, convenient, and practical solution.16–18,20,46,47 Besides, probiotics consumption may have beneficial effects in preventing respiratory tract infections and influenza-related illnesses.48,49 Our study provided comprehensive evidence that prebiotic/probiotic use will enhance the HI antibody titer after influenza vaccination. In addition, the immunogenicity of influenza vaccination may be affected by the components of vaccine strains. Compared with A/H1N1 and A/H3N2 strains, poorer antigen immunogenic responses in B strain were reported in previous studies.50–52 Our studies also showed relatively lower HI antibody titers in B strain (Table 2). However, the beneficial effects of prebiotic/probiotic supplementation were observed in all A/H1N1, A/H3N2, and B strains. A 20% (A/H1N1), 19.5% (A/H3N2), and 13.6% (B strain) increase in HI antibody titers was observed in individuals with prebiotics/probiotics use.
Consumption of “good bacteria” could suppress the growth of pathogenic bacteria in the gastrointestinal tract and improve the intestinal barrier function.6,7 The use of prebiotics/probiotics in patients with bacterial diarrhea is well known.8,53–55 Probiotics are also used to prevent necrotizing enterocolitis and sepsis in preterm neonates and may also contribute to adjuvant therapy in eradication of Helicobacter pylori.54,56–60 In addition to the beneficial effects in the gastrointestinal tract, systemic immunomodulatory effects, toll-like receptor-mediated pathways, regulatory T cell induction, natural killer cells, soluble proteins, and various cytokines were involved in the probiotic immune regulatory mechanism.5,9–12,61 Therefore, manipulation of the gut microbiota may benefit patients with systemic diseases, such as allergic diseases.14 Reduced risks of subsequent cardiovascular diseases and metabolic outcomes were also observed.15,62 In a report published in 2016, it was stated that probiotic-modulated gut microbiota may suppress hepatocellular carcinoma growth in mice via regulation of T cell and pro-inflammatory cytokines.13 The use of prebiotics/probiotics/synbiotics may hold great promise for preventing and treating many extraintestinal diseases.
Probiotics are “live” bacteria, which help human to fight against pathogenic bacteria. Although the benefits of probiotics in preterm neonates are well documented, safety of probiotics in immunocompromised individuals remains a major concern.56–58 Bacteremia caused by probiotics strains was reported in some immunocompromised patients.63–66 Elderly people are at increased risk of being immunocompromised and the issue of safety remains important. In our meta-analysis studies, more than half of the participants were bedridden, fed with nasogastric tubes, or nursing home residents; no documented probiotics-related sepsis was reported.26–28,30,31,34,35 Furthermore, in the subgroup analysis of our study, prebiotics were also beneficial for enhancing immune responses after influenza vaccination. Prebiotic use may be a reasonable choice for immunocompromised patients at increased risk for infection.
Our study had some limitations. First, the study design, study participants, and study period were highly heterogeneous. Further large-scale studies are warranted to confirm our findings. Second, the strain, doses, and the duration of prebiotics/probiotics supplementation differed among studies. The immune responses may vary in different supplement protocol. Further studies are required to investigate the optimal strain, dosage, and duration of probiotic consumption. Finally, the components of the influenza vaccine and prevalent influenza strains were different each year. It may be more valuable to explore the effects of probiotics with the same influenza vaccine.
Conclusion
Our study suggests that concomitant prebiotics/probiotics use might be an effective intervention to enhance the HI antibody titer following influenza vaccination (13.6%–20% increases in HI antibody titers). Adjuvant prebiotics/probiotics use may hold great promise for the improvement of immune responses following influenza vaccination. However, high heterogeneity was observed and further studies are warranted to elucidate the effectiveness and decide the optimal strains, dose, timing, and duration of supplementation.
Acknowledgment
Our manuscript has been edited for English language, grammar, punctuation, and spelling by Enago, the editing brand of Crimson Interactive Pvt, Ltd.
Author contributions
All authors contributed toward data analysis, drafting and critically revising the paper, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Jordan RE, Hawker JI. Influenza in elderly people in care homes. BMJ. 2006;333(7581):1229–1230. | ||
Osterholm MT, Kelley NS, Sommer A, Belongia EA. Efficacy and effectiveness of influenza vaccines: a systematic review and meta-analysis. Lancet Infect Dis. 2012;12(1):36–44. | ||
DiazGranados CA, Dunning AJ, Kimmel M, et al. Efficacy of high-dose versus standard-dose influenza vaccine in older adults. N Engl J Med. 2014;371(7):635–645. | ||
Pae M, Meydani SN, Wu D. The role of nutrition in enhancing immunity in aging. Aging Dis. 2012;3(1):91–129. | ||
Yan F, Cao H, Cover TL, Whitehead R, Washington MK, Polk DB. Soluble proteins produced by probiotic bacteria regulate intestinal epithelial cell survival and growth. Gastroenterology. 2007;132(2):562–575. | ||
Markowiak P, Śliżewska K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients. 2017;9(9):E1021. | ||
Jones SE, Versalovic J. Probiotic Lactobacillus reuteri biofilms produce antimicrobial and anti-inflammatory factors. BMC Microbiol. 2009;9:35. | ||
Hojsak I, Szajewska H, Canani RB, et al. Probiotics for the prevention of nosocomial diarrhea in children. J Pediatr Gastroenterol Nutr. 2018;66(1):3–9. | ||
Atarashi K, Tanoue T, Oshima K, et al. Treg induction by a rationally selected mixture of Clostridia strains from the human microbiota. Nature. 2013;500(7461):232–236. | ||
Round JL, Lee SM, Li J, et al. The Toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science. 2011;332(6032):974–977. | ||
Pathmakanthan S, Li CK, Cowie J, Hawkey CJ. Lactobacillus plantarum 299: beneficial in vitro immunomodulation in cells extracted from inflamed human colon. J Gastroenterol Hepatol. 2004;19(2):166–173. | ||
Lee A, Lee YJ, Yoo HJ, et al. Consumption of dairy yogurt containing Lactobacillus paracasei ssp. paracasei, Bifidobacterium animalis ssp. lactis and heat-treated Lactobacillus plantarum improves immune function including natural killer cell activity. Nutrients. 2017;9(6):E558. | ||
Li J, Sung CY, Lee N, et al. Probiotics modulated gut microbiota suppresses hepatocellular carcinoma growth in mice. Proc Natl Acad Sci U S A. 2016;113(9):E1306–E1315. | ||
Chang YS, Trivedi MK, Jha A, Lin YF, Dimaano L, Garcia-Romero MT. Synbiotics for prevention and treatment of atopic dermatitis: a meta-analysis of randomized clinical trials. JAMA Pediatr. 2016;170(3):236–242. | ||
Wu L, Sun D. Consumption of yogurt and the incident risk of cardiovascular disease: a meta-analysis of nine cohort studies. Nutrients. 2017;9(3):E315. | ||
Praharaj I, John SM, Bandyopadhyay R, Kang G. Probiotics, antibiotics and the immune responses to vaccines. Philos Trans R Soc Lond B Biol Sci. 2015;370(1671):20140144. | ||
Maidens C, Childs C, Przemska A, Dayel IB, Yaqoob P. Modulation of vaccine response by concomitant probiotic administration. Br J Clin Pharmacol. 2013;75(3):663–670. | ||
Licciardi PV, Tang ML. Vaccine adjuvant properties of probiotic bacteria. Discov Med. 2011;12(67):525–533. | ||
Zimmermann P, Curtis N. The influence of probiotics on vaccine responses – a systematic review. Vaccine. 2018;36(2):207–213. | ||
Lei WT, Shih PC, Liu SJ, Lin CY, Yeh TL. Effect of probiotics and prebiotics on immune response to influenza vaccination in adults: a systematic review and meta-analysis of randomized controlled trials. Nutrients. 2017;9(11):E1175. | ||
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339:b2700. | ||
de Jong JC, Palache AM, Beyer WE, Rimmelzwaan GF, Boon AC, Osterhaus AD. Haemagglutination-inhibiting antibody to influenza virus. Dev Biol (Basel). 2003;115:63–73. | ||
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539–1558. | ||
Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–560. | ||
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–634. | ||
Maruyama M, Abe R, Shimono T, Iwabuchi N, Abe F, Xiao JZ. The effects of non-viable Lactobacillus on immune function in the elderly: a randomised, double-blind, placebo-controlled study. Int J Food Sci Nutr. 2016;67(1):67–73. | ||
Akatsu H, Nagafuchi S, Kurihara R, et al. Enhanced vaccination effect against influenza by prebiotics in elderly patients receiving enteral nutrition. Geriatr Gerontol Int. 2016;16(2):205–213. | ||
Nagafuchi S, Yamaji T, Kawashima A, et al. Effects of a formula containing two types of prebiotics, bifidogenic growth stimulator and galacto-oligosaccharide, and fermented milk products on intestinal microbiota and antibody response to influenza vaccine in elderly patients: a randomized controlled trial. Pharmaceuticals (Basel). 2015;8(2):351–365. | ||
Lomax AR, Cheung LV, Noakes PS, Miles EA, Calder PC. Inulin-type beta2–1 fructans have some effect on the antibody response to seasonal influenza vaccination in healthy middle-aged humans. Front Immunol. 2015;6:490. | ||
Akatsu H, Iwabuchi N, Xiao JZ, et al. Clinical effects of probiotic Bifidobacterium longum BB536 on immune function and intestinal microbiota in elderly patients receiving enteral tube feeding. JPEN J Parenter Enteral Nutr. 2013;37(5):631–640. | ||
Akatsu H, Arakawa K, Yamamoto T, et al. Lactobacillus in jelly enhances the effect of influenza vaccination in elderly individuals. J Am Geriatr Soc. 2013;61(10):1828–1830. | ||
Davidson LE, Fiorino AM, Snydman DR, Hibberd PL. Lactobacillus GG as an immune adjuvant for live-attenuated influenza vaccine in healthy adults: a randomized double-blind placebo-controlled trial. Eur J Clin Nutr. 2011;65(4):501–507. | ||
Namba K, Hatano M, Yaeshima T, Takase M, Suzuki K. Effects of Bifidobacterium longum BB536 administration on influenza infection, influenza vaccine antibody titer, and cell-mediated immunity in the elderly. Biosci Biotechnol Biochem. 2010;74(5):939–945. | ||
Boge T, Remigy M, Vaudaine S, Tanguy J, Bourdet-Sicard R, van der Werf S. A probiotic fermented dairy drink improves antibody response to influenza vaccination in the elderly in two randomised controlled trials. Vaccine. 2009;27(41):5677–5684. | ||
Langkamp-Henken B, Wood SM, Herlinger-Garcia KA, et al. Nutritional formula improved immune profiles of seniors living in nursing homes. J Am Geriatr Soc. 2006;54(12):1861–1870. | ||
Langkamp-Henken B, Bender BS, Gardner EM, et al. Nutritional formula enhanced immune function and reduced days of symptoms of upper respiratory tract infection in seniors. J Am Geriatr Soc. 2004;52(1):3–12. | ||
Enani S, Przemska-Kosicka A, Childs CE, et al. Impact of ageing and a synbiotic on the immune response to seasonal influenza vaccination; a randomised controlled trial. Clin Nutr. Epub 2017 Jan 28. | ||
Manzoli L, Ioannidis JP, Flacco ME, De Vito C, Villari P. Effectiveness and harms of seasonal and pandemic influenza vaccines in children, adults and elderly: a critical review and re-analysis of 15 meta-analyses. Hum Vaccin Immunother. 2012;8(7):851–862. | ||
Nicoll A, Sprenger M. Low effectiveness undermines promotion of seasonal influenza vaccine. Lancet Infect Dis. 2013;13(1):7–9. | ||
Puig-Barbera J, Mira-Iglesias A, Tortajada-Girbes M, et al. Waning protection of influenza vaccination during four influenza seasons, 2011/2012 to 2014/2015. Vaccine. 2017;35(43):5799–5807. | ||
Bellei NC, Carraro E, Castelo A, Granato CF. Risk factors for poor immune response to influenza vaccination in elderly people. Braz J Infect Dis. 2006;10(4):269–273. | ||
Jefferson T, Rivetti D, Rivetti A, Rudin M, Di Pietrantonj C, Demicheli V. Efficacy and effectiveness of influenza vaccines in elderly people: a systematic review. Lancet. 2005;366(9492):1165–1174. | ||
Haq K, McElhaney JE. Immunosenescence: influenza vaccination and the elderly. Curr Opin Immunol. 2014;29:38–42. | ||
Chen WH, Kozlovsky BF, Effros RB, Grubeck-Loebenstein B, Edelman R, Sztein MB. Vaccination in the elderly: an immunological perspective. Trends Immunol. 2009;30(7):351–359. | ||
Brydak LB, Machala M, Mysliwska J, Mysliwski A, Trzonkowski P. Immune response to influenza vaccination in an elderly population. J Clin Immunol. 2003;23(3):214–222. | ||
Yaqoob P. Ageing, immunity and influenza: a role for probiotics? Proc Nutr Soc. 2014;73(2):309–317. | ||
Youngster I, Kozer E, Lazarovitch Z, Broide E, Goldman M. Probiotics and the immunological response to infant vaccinations: a prospective, placebo controlled pilot study. Arch Dis Child. 2011;96(4):345–349. | ||
Hao Q, Dong BR, Wu T. Probiotics for preventing acute upper respiratory tract infections. Cochrane Database Syst Rev. 2015;2:CD006895. | ||
Leyer GJ, Li S, Mubasher ME, Reifer C, Ouwehand AC. Probiotic effects on cold and influenza-like symptom incidence and duration in children. Pediatrics. 2009;124(2):e172–e179. | ||
Talbot HK, Keitel W, Cate TR, et al. Immunogenicity, safety and consistency of new trivalent inactivated influenza vaccine. Vaccine. 2008;26(32):4057–4061. | ||
Del Porto F, Lagana B, Biselli R, et al. Influenza vaccine administration in patients with systemic lupus erythematosus and rheumatoid arthritis. Safety and immunogenicity. Vaccine. 2006;24(16):3217–3223. | ||
Salemi S, Picchianti-Diamanti A, Germano V, et al. Influenza vaccine administration in rheumatoid arthritis patients under treatment with TNFα blockers: safety and immunogenicity. Clin Immunol. 2010;134(2):113–120. | ||
Floch MH, Walker WA, Sanders ME, et al. Recommendations for probiotic use – 2015 update: proceedings and consensus opinion. J Clin Gastroenterol. 2015;49(Suppl 1):S69–S73. | ||
Vandenplas Y, Veereman-Wauters G, De Greef E, et al. Probiotics and prebiotics in prevention and treatment of diseases in infants and children. J Pediatr. 2011;87(4):292–300. | ||
Szajewska H, Canani RB, Guarino A, et al. Probiotics for the prevention of antibiotic-associated diarrhea in children. J Pediatr Gastroenterol Nutr. 2016;62(3):495–506. | ||
Pammi M, Suresh G. Enteral lactoferrin supplementation for prevention of sepsis and necrotizing enterocolitis in preterm infants. Cochrane Database Syst Rev. 2017;6:CD007137. | ||
Hu HJ, Zhang GQ, Zhang Q, Shakya S, Li ZY. Probiotics prevent candida colonization and invasive fungal sepsis in preterm neonates: a systematic review and meta-analysis of randomized controlled trials. Pediatr Neonatol. 2017;58(2):103–110. | ||
Rao SC, Athalye-Jape GK, Deshpande GC, Simmer KN, Patole SK. Probiotic supplementation and late-onset sepsis in preterm infants: a meta-analysis. Pediatrics. 2016;137(3):e20153684. | ||
Aceti A, Maggio L, Beghetti I, et al. Probiotics prevent late-onset sepsis in human milk-fed, very low birth weight preterm infants: systematic review and meta-analysis. Nutrients. 2017;9(8):E904. | ||
Aceti A, Gori D, Barone G, et al. Probiotics for prevention of necrotizing enterocolitis in preterm infants: systematic review and meta-analysis. Ital J Pediatr. 2015;41:89. | ||
McCarthy J, O’Mahony L, O’Callaghan L, et al. Double blind, placebo controlled trial of two probiotic strains in interleukin 10 knockout mice and mechanistic link with cytokine balance. Gut. 2003;52(7):975–980. | ||
Taylor B, Woodfall G, Sheedy K, et al. Effect of probiotics on metabolic outcomes in pregnant women with gestational diabetes: a systematic review and meta-analysis of randomized controlled trials. Nutrients. 2017;9(5):E461. | ||
Esaiassen E, Hjerde E, Cavanagh JP, Simonsen GS, Klingenberg C. Bifidobacterium bacteremia: clinical characteristics and a genomic approach to assess pathogenicity. J Clin Microbiol. 2017;55(7):2234–2248. | ||
Sherid M, Samo S, Sulaiman S, Husein H, Sifuentes H, Sridhar S. Liver abscess and bacteremia caused by lactobacillus: role of probiotics? Case report and review of the literature. BMC Gastroenterol. 2016;16(1):138. | ||
Cohen SA, Woodfield MC, Boyle N, Stednick Z, Boeckh M, Pergam SA. Incidence and outcomes of bloodstream infections among hematopoietic cell transplant recipients from species commonly reported to be in over-the-counter probiotic formulations. Transpl Infect Dis. 2016;18(5):699–705. | ||
Meini S, Laureano R, Fani L, et al. Breakthrough Lactobacillus rhamnosus GG bacteremia associated with probiotic use in an adult patient with severe active ulcerative colitis: case report and review of the literature. Infection. 2015;43(6):777–781. | ||
Olivares M, Diaz-Ropero MP, Sierra S, et al. Oral intake of Lactobacillus fermentum CECT5716 enhances the effects of influenza vaccination. Nutrition. 2007;23(3):254-260.68. French PW, Penny R. Use of probiotic bacteria as an adjuvant for an influenza vaccine. Int J Probiotics Prebiotics. 2009;4(3):175–182. | ||
Van Puyenbroeck K, Hens N, Coenen S, et al. Efficacy of daily intake of Lactobacillus casei Shirota on respiratory symptoms and influenza vaccination immune response: a randomized, double-blind, placebo-controlled trial in healthy elderly nursing home residents. Am J Clin Nutr. 2012;95(5):1165–1171. | ||
Rizzardini G, Eskesen D, Calder PC, Capetti A, Jespersen L, Clerici M. Evaluation of the immune benefits of two probiotic strains Bifidobacterium animalis ssp. lactis, BB-12® and Lactobacillus paracasei ssp. paracasei, L. casei 431® in an influenza vaccination model: a randomised, double-blind, placebo-controlled study. Br J Nutr. 2012; 107(6):876–884. | ||
Bosch M, Méndez M, Pérez M, Farran A, Fuentes MC, Cuñé J. Lactobacillus plantarum CECT7315 and CECT7316 stimulate immunoglobulin production after influenza vaccination in elderly. Nutr Hosp. 2012;27(2):504–509. | ||
Jespersen L, Tarnow I, Eskesen D, et al. Effect of Lactobacillus paracasei subsp. paracasei, L. casei 431 on immune response to influenza vaccination and upper respiratory tract infections in healthy adult volunteers: a randomized, double-blind, placebo-controlled, parallel-group study. Am J Clin Nutr. 2015;101(6):1188–1196. | ||
Bunout D, Hirsch S, Pía de la Maza M, et al. Effects of prebiotics on the immune response to vaccination in the elderly. J Parenter Enteral Nutr. 2002;26(6):372–376. |
Supplementary materials
  | Figure S1 Funnel plot of strain A/H1N1. |
  | Figure S2 Funnel plot of strain A/H3N2. |
  | Figure S3 Funnel plot of strain B. |
References
Olivares M, Diaz-Ropero MP, Sierra S, et al. Oral intake of Lactobacillus fermentum CECT5716 enhances the effects of influenza vaccination. Nutrition. 2007;23(3):254–260. | ||
French PW, Penny R. Use of probiotic bacteria as an adjuvant for an influenza vaccine. Int J Probiotics Prebiotics. 2009;4(3):175–182. | ||
Boge T, Remigy M, Vaudaine S, Tanguy J, Bourdet-Sicard R, van der Werf S. A probiotic fermented dairy drink improves antibody response to influenza vaccination in the elderly in two randomised controlled trials. Vaccine. 2009;27(41):5677–5684. | ||
Namba K, Hatano M, Yaeshima T, Takase M, Suzuki K. Effects of Bifidobacterium longum BB536 administration on influenza infection, influenza vaccine antibody titer, and cell-mediated immunity in the elderly. Biosci Biotechnol Biochem. 2010;74(5):939–945. | ||
Davidson LE, Fiorino AM, Snydman DR, Hibberd PL. Lactobacillus GG as an immune adjuvant for live-attenuated influenza vaccine in healthy adults: a randomized double-blind placebo-controlled trial. Eur J Clin Nutr. 2011;65(4):501–507. | ||
Van Puyenbroeck K, Hens N, Coenen S, et al. Efficacy of daily intake of Lactobacillus casei Shirota on respiratory symptoms and influenza vaccination immune response: a randomized, double-blind, placebo-controlled trial in healthy elderly nursing home residents. Am J Clin Nutr. 2012;95(5):1165–1171. | ||
Rizzardini G, Eskesen D, Calder PC, Capetti A, Jespersen L, Clerici M. Evaluation of the immune benefits of two probiotic strains Bifidobacterium animalis ssp. lactis, BB-12® and Lactobacillus paracasei ssp. paracasei, L. casei 431® in an influenza vaccination model: a randomised, double-blind, placebo-controlled study. Br J Nutr. 2012;107(6):876–884. | ||
Bosch M, Méndez M, Pérez M, Farran A, Fuentes MC, Cuñé J. Lactobacillus plantarum CECT7315 and CECT7316 stimulate immunoglobulin production after influenza vaccination in elderly. Nutr Hosp. 2012; 27(2):504–509. | ||
Akatsu H, Arakawa K, Yamamoto T, et al. Lactobacillus in jelly enhances the effect of influenza vaccination in elderly individuals. J Am Geriatr Soc. 2013;61(10):1828–1830. | ||
Akatsu H, Iwabuchi N, Xiao JZ, et al. Clinical effects of probiotic Bifidobacterium longum BB536 on immune function and intestinal microbiota in elderly patients receiving enteral tube feeding. JPEN J Parenter Enteral Nutr. 2013;37(5):631–640. | ||
Jespersen L, Tarnow I, Eskesen D, et al. Effect of Lactobacillus paracasei subsp. paracasei, L. casei 431 on immune response to influenza vaccination and upper respiratory tract infections in healthy adult volunteers: a randomized, double-blind, placebo-controlled, parallel-group study. Am J Clin Nutr. 2015;101(6):1188–1196. | ||
Maruyama M, Abe R, Shimono T, Iwabuchi N, Abe F, Xiao JZ. The effects of non-viable Lactobacillus on immune function in the elderly: a randomised, double-blind, placebo-controlled study. Int J Food Sci Nutr. 2016;67(1):67–73. | ||
Bunout D, Hirsch S, Pía de la Maza M, et al. Effects of prebiotics on the immune response to vaccination in the elderly. J Parenter Enteral Nutr. 2002;26(6):372–376. | ||
Langkamp-Henken B, Bender BS, Gardner EM, et al. Nutritional formula enhanced immune function and reduced days of symptoms of upper respiratory tract infection in seniors. J Am Geriatr Soc. 2004;52(1):3–12. | ||
Langkamp-Henken B, Wood SM, Herlinger-Garcia KA, et al. Nutritional formula improved immune profiles of seniors living in nursing homes. J Am Geriatr Soc. 2006;54(12):1861–1870. | ||
Nagafuchi S, Yamaji T, Kawashima A, et al. Effects of a formula containing two types of prebiotics, bifidogenic growth stimulator and galacto-oligosaccharide, and fermented milk products on intestinal microbiota and antibody response to influenza vaccine in elderly patients: a randomized controlled trial. Pharmaceuticals (Basel). 2015;8(2):351–365. | ||
Lomax AR, Cheung LV, Noakes PS, Miles EA, Calder PC. Inulin-type beta2–1 fructans have some effect on the antibody response to seasonal influenza vaccination in healthy middle-aged humans. Front Immunol. 2015;6:490. | ||
Akatsu H, Nagafuchi S, Kurihara R, et al. Enhanced vaccination effect against influenza by prebiotics in elderly patients receiving enteral nutrition. Geriatr Gerontol Int. 2016;16(2):205–213. | ||
Enani S, Przemska-Kosicka A, Childs CE, et al. Impact of ageing and a synbiotic on the immune response to seasonal influenza vaccination; a randomised controlled trial. Clin Nutr. Epub 2017 Jan 28. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.






