Back to Journals » Neuropsychiatric Disease and Treatment » Volume 12
The impact of synapsin III gene on the neurometabolite level alterations after single-dose methylphenidate in attention-deficit hyperactivity disorder patients
Authors Başay, Kabukçu Başay B, Alacam H, Öztürk, Büber A, Yilmaz SG , Kıroğlu Y, Erdal ME, Herken H
Received 9 February 2016
Accepted for publication 22 March 2016
Published 10 May 2016 Volume 2016:12 Pages 1141—1149
DOI https://doi.org/10.2147/NDT.S106076
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Roger Pinder
Ömer Başay,1 Burge Kabukcu Basay,1 Huseyin Alacam,2 Onder Ozturk,1 Ahmet Buber,1 Senay Gorucu Yilmaz,3 Yılmaz Kıroğlu,4 Mehmet Emin Erdal,5 Hasan Herken2
1Department of Child and Adolescent Psychiatry, Faculty of Medicine, Pamukkale University, Denizli, 2Department of Psychiatry, Faculty of Medicine, Pamukkale University, Denizli, 3Department of Nutrition and Dietetics, Faculty of Health Sciences, Gaziantep University, Gaziantep, 4Department of Radiology, School of Medicine, Pamukkale University, Denizli, 5Department of Medical Biology and Genetics, Faculty of Medicine, Mersin University, Mersin, Turkey
Objective: To investigate the neurometabolite level changes according to synapsin III gene rs133945G>A and rs133946C>G polymorphisms by using magnetic resonance spectroscopy (MRS) in patients with attention-deficit hyperactivity disorder (ADHD).
Methods: Fifty-seven adults diagnosed with ADHD were recruited for the study. The participants were examined by single-voxel 1H MRS when medication naïve and 30 minutes after oral administration of 10 mg methylphenidate (Mph). Those who had been on a stimulant discontinued the medication 48 hours before MRS imaging. Spectra were taken from the anterior cingulate cortex, dorsolateral prefrontal cortex, striatum, and cerebellum, and N-acetylaspartate (NAA), choline, and creatine levels were examined. For genotyping of the synapsin III gene polymorphisms, DNA was isolated from peripheral blood leukocytes. The effects of age, sex, and ADHD subtypes were controlled in the analyses.
Results: After a single dose of Mph, choline levels increased significantly in the striatum of rs133945G>A polymorphism-GG genotypes (P=0.020) and NAA levels rose in the anterior cingulate cortex of rs133946C>G polymorphism-CG genotypes (P=0.014). Both rs133945G>A and rs133946C>G polymorphisms were found to statistically significantly affect the alteration of NAA levels in response to Mph in dorsolateral prefrontal cortex with two-way repeated measure of analysis of variance. Post hoc comparisons revealed a significant difference between CG and GG genotypes of rs133946C>G polymorphisms after Bonferroni adjustment (P=0.016).
Conclusion: Synapsin III gene polymorphisms may be affecting the changes in neurometabolite levels in response to Mph in adult ADHD patients. Future studies are needed to confirm our findings.
Keywords: adult ADHD, magnetic resonance spectroscopy, N-acetylaspartate, genotype
Introduction
Attention-deficit hyperactivity disorder (ADHD) is a neurodevelopmental disorder that is characterized by inattention, impulsiveness, and hyperactivity. The prevalence of ADHD is reported to be 7.2% in childhood and estimated to be 5% in adulthood.1,2 Neuropsychological evaluations and structural and functional brain imaging studies have supported that ADHD is often characterized by structural and functional brain alterations in comparison to healthy controls.3 Similarly, ADHD treatment has been shown to cause alterations in brain regions by magnetic resonance imaging (MRI) studies. Methylphenidate (Mph) increases frontal and striatal metabolism as seen from functional MRI scans.4 Long-term ADHD treatment with Mph was shown to normalize structural brain changes in the white matter, cerebellum, anterior cingulate cortex (ACC), and thalamus, and a single-dose of Mph has been found to increase the frontostriatal network for inhibitory control in ADHD-diagnosed children.4,5
Studies on ADHD etiology have used magnetic resonance spectroscopy (MRS) to investigate the possible neurometabolic changes in the brain, which could be related to the pathogenesis of the disorder. MRS is a noninvasive method for measuring different metabolites including N-acetylaspartate (NAA), choline (Cho), creatine (Cr), and others in the human brain.6 NAA and Cr concentrations have been shown to be reduced in the left dorsolateral prefrontal cortex (DLPFC) in adult patients with ADHD.7,8 Colla et al9 found increased Cho values in the ACC of adults diagnosed with ADHD.
The MRS technique also provides an opportunity for further investigations of metabolite changes in response to ADHD drug treatments. Studies conducted in children and adolescents have investigated the effect of acute or long-term ADHD treatment and have reported altered or unchanged neurometabolite levels.10–12 In adult patients with ADHD, increased NAA and reduced Cho levels in the anterior cingulum were reported following 5–6 weeks of Mph treatment.13 In another study, Maltezos et al found that adult ADHD patients had significantly lower concentrations of glutamate + glutamine (Glx), Cr, and NAA in the basal ganglia and Cr in the DLPFC compared to controls. They showed no differences between treatment-naïve and stimulant-treated ADHD patients.8
In recent years, pharmacogenetic investigations have indicated the presence of a relationship between catecholaminergic-system-related genes and ADHD in children.14 However, in adults diagnosed with ADHD, most findings obtained in the pharmacogenetic area have been negative so far, and all of these studies focused on the response to Mph. Only one positive result was reported, which is for a polymorphism at the DAT1 gene.15 It has been suggested that the integration of neuroimaging methods and neuropsychological tests with these studies may be helpful in the understanding of the mechanisms of drug action and ADHD pathophysiology.15 But there are very few studies conducted in this field. One study by Öner et al16 is an example, and the authors suggested that SNAP-25 genotypes (DdeI T/T and MnlI T/T) or (DdeI T/T and MnlI G/G or T/G) might be associated with Mph-induced brain hemodynamic changes in children with ADHD.
Dopamine is a clinically important neurotransmitter that has implications in many neuropsychiatric disorders including ADHD.17 Synapsins (synapsin I, II, and III) are a family of three neuron-specific genes encoding for phosphoproteins, which have important roles in the regulation of neurodevelopment and neurotransmission.18 Among them, synapsin III has recently been demonstrated to be specifically involved in the release of dopamine in the striatum. To support this, Porton et al19 showed a similar magnitude of dopamine release in the slices derived from synapsin III knockout mice, suggesting that the regulation of dopamine release in the striatum is primarily related to synapsin III. Synapsin III polymorphism is mostly implicated in schizophrenia.20,21 However, its association with ADHD has been studied only very rarely. Makkar et al22 failed to find a significant relation between synapsin III gene polymorphisms and ADHD. In contrast, Kenar et al23 found an association between the 631 C>G polymorphism of the synapsin III gene and ADHD in adult patients. As far as we know, the relationship between synapsin III polymorphisms and brain metabolites in response to Mph has not been investigated.
In this study, we aimed to investigate the alterations in neurometabolic variables according to synapsin III gene 196 G>A (rs133945G>A) and 631 C>G (rs133946C>G) polymorphisms by using MRS, following the administration of a single dose of Mph, in DLPFC, ACC, striatum, and cerebellum in adult ADHD patients.
Methods
Subjects
Fifty-seven voluntary adult patients 18–60 years of age and meeting criteria for ADHD according to Diagnostic and Statistical Manual of Mental Disorders, Fourth-Edition (DSM-IV), Text Revision24 participated in this study. The patients were recruited from the outpatient clinic of the Department of Psychiatry, Faculty of Medicine, Pamukkale University, Turkey. Illiterate patients or patients with any neurologic or chronic disorder, psychotic disorder, organic mental disorder, or mental retardation were excluded from the study. Ethical approvals were obtained from the Pamukkale University Ethics Committe. All the patients were informed about the aim and the procedures of the study, and signed written informed consents were obtained.
Instruments
Sociodemographic data form
This is a data sheet developed by the researchers for studying the sociodemographic characteristics of the study group.
Adult Attention-Deficit Disorder/ADHD DSM-IV-based diagnostic screening and rating scale
In the diagnostic assessment of patients, the Turkish version of Adult Attention-Deficit Disorder/ADHD DSM-IV based diagnostic screening and rating scales25,26 was used. When developing the adult Attention-Deficit Disorder/ADHD scale, 18 symptoms of the diagnostic criteria in DSM-IV were reframed so that patients could understand them. The first part of this scale had nine questions relating to inattention, and the second part also had nine questions on hyperactivity/impulsivity. The third part of the scale consisted of the most frequently associated symptoms in ADHD that were not in the DSM-IV ADHD diagnostic criteria. Their Turkish validity and reliability were established by Günay et al.25
MRS
Patients were examined by single-voxel 1H MRS, and spectra were taken from ACC, DLPFC, the striatum, and the cerebellum. A clinical 1.5 T MR scanner (GE Medical System, Milwaukee, WI, USA) was used with the following parameters: point-resolved spectroscopy sequence with water suppression (chemical-shift-selective imaging sequence), echo time/repetition time of 35/3,000 ms, and 128 averages. Conventional spin echo sequence with T2-weighted fast spin echo parameters were horizontal 10 mm slice thickness with repetition time/echo time 3,000/88, field-of-view (FOV): 10 cm, matrix: 512×512, and next: 1. The whole Magnetic Resonance (MR) session took ~45 minutes.
Patients who had been on Mph treatment discontinued the medication 48 hours before the neuroimaging procedures. NAA, Cho, and Cr levels were examined by single-voxel 1H MRS when medication naïve and 30 minutes after the administration of 10 mg oral Mph.
DNA isolation and molecular analysis
For the amplification of the synapsin III gene rs133945G>A polymorphism, DNA was isolated from peripheral blood leukocytes by the standard phenol/chloroform method and genotyped by the polymerase chain reaction (PCR)-restriction fragment length polymorphism method. PCR was performed with a personal thermal cycler (Techgene, NJ, USA) using SYN2 F-5′-T CCTTTCCAGAAGGATGTCC-3′/SYN2 R-5′-AAGCCAACA AATACATAAGTGGAGA-3′ primers. For the amplification of the synapsin III gene rs133946C>G polymorphism, DNA was isolated from peripheral blood leukocytes by the standard phenol/chloroform method and genotyped by the PCR-restriction fragment length polymorphism method. PCR was performed with a personal thermal cycler (Techgene) using SYN1 F-5′-AGGCATGTACTTGCGTTACC-3′/SYN1 R-5′-CCAAATGACTACAAAGATGTACCA-3′ primers.
Statistical analysis
The findings of the study were analyzed with the SPSS 17.0 package. The comparison of the categorical variables was made by Pearson’s chi-square test. The normality of dispersion of the numeric variables was evaluated with the Kolmogorov–Smirnov test. One-way analysis of variance (ANOVA) was used for three group comparisons of numeric variables. The significance of the neurometabolite level alterations before and after the medication was assessed by two related sample tests (paired-sample t-test). Statistical significance was accepted at the P<0.05 level. Two-way repeated measures of ANOVA were performed to compare the effects of single-dose Mph on brain metabolite levels between the genotypes of rs133945G>A and rs133946C>G polymorphisms of synapsin III gene while taking sex, age, and subtypes of ADHD as covariates. Outliers were controlled. Post hoc comparisons were performed, Bonferroni correction was made, and P-value <0.017 (0.05/3) was accepted as statistically significant.
Results
A total of 57 adult ADHD patients aged between 18 years and 60 years (12 women [21.1%] and 45 men [78.9%]; mean age: 29.05±7.85 years) were recruited for the study. Thirty-three (57.9%) of them had been on stimulant treatment for ADHD (Table 1).
  | Table 1 Sociodemographic characteristics |
Hardy–Weinberg equilibrium P-values for rs133945G>A and rs133946C>G were 0.674 (X2=0.175) and 0.501 (X2=0.452), respectively. For the rs133945G>A polymorphism, 30.4% (n=14) of the male participants had GG genotype, 47.8% (n=22) had GA genotype, and 21.7% (n=10) had AA genotype and 9.1% (n=1) of the female participants had GG genotype, 72.7% (n=8) had GA genotype, and 18.2% (n=2) had AA genotype. For the rs133946C>G polymorphism, 28.3% (n=13) of the male participants had CC genotype, 50% (n=23) had CG genotype, and 21.7% (n=10) had GG genotype and 9.1% (n=1) of the female participants had CC genotype, 72.7% (n=8) had CG genotype, and 18.2% (n=2) had GG genotype. No significant difference was detected regarding the patients’ sex distribution according to synapsin III gene 196 G>A polymorphism (X2=2.22, P=0.26) and synapsin III gene 631 C>G polymorphism (X2=2.63, P=0.39). Age distribution was not statistically different among the GG (mean age 32.1±10.2 years), GA (mean age 28.5±6.6 years), and AA (mean age 26.4±6.4 years) genotype groups of rs133945G>A polymorphism (F=2.35; P=0.10) and CC (mean age 32.6±10.4 years), CG (mean age 28.4±6.5 years), and GG (mean age 26.4±6.3 years) genotype groups of rs133946C>G polymorphism (F=1,96; P=0.15). Age distribution was not statistically different among the GG, GA, and AA genotype groups of rs133945G>A polymorphism (F=2,35; P=0.10) and CC, CG, and GG genotype groups of rs133946C>G polymorphism (F=1,96; P=0.15). No significant difference was found regarding ADHD subtypes according to rs133945G>A polymorphism and rs133946C>G polymorphism (Table 2).
  | Table 2 Distribution of synapsin III genotypes among ADHD subtypes |
When we investigated the Cho levels, we found that its levels had increased significantly after a single dose of Mph in the striatum of rs133945G>A polymorphism-GG genotype (P=0.020). Another statistically significant difference was recorded for NAA values in the ACC of rs133946C>G polymorphism-CG genotype: after a single dose of 10 mg Mph, the NAA levels were found to have increased (P=0.014). In the other brain regions, we did not find any medication-related, statistically significant metabolite level change in any of the polymorphism groups with paired sample t-test (Table 3).
The difference between rs133945G>A polymorphism GG, GA, and AA genotypes and rs133946C>G polymorphism CC, CG, and GG genotypes in terms of changes of NAA level was found to be statistically significantly different in DLPFC with two-way repeated measure of ANOVA, while the sex, age, and subtypes of ADHD were taken as covariates. Two outliers with CG and CC genotypes were detected and removed from the analysis.
The NAA values before the administration of Mph in the DLPFC were 63.6±9.9 in the GG genotype, 73.0±15.7 in the GA genotype, and 61.8±9.7 in the AA genotype. The values in the DLPFC post-Mph administration were 64.5±13.5 in the GG genotype, 74.9±16.0 in the GA genotype, and 62.7±7.2 in the AA genotype. After controlling for the effects of age, sex, and ADHD subtypes, rs133945G>A polymorphism was found to be statistically significantly affecting the alteration of NAA levels in response to Mph in the DLPFC (F[2,49]=4,72; P=0.013; ɲ2=0.162). Post hoc comparisons were conducted to reveal the statistically significant group differences in the rs133945G>A genotypes, and GA and AA genotypes were found to be different regarding the NAA levels (P=0.029). However, this difference was not found to be statistically significant according to Bonferroni adjustment.
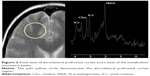
The values of NAA in the DLPFC before Mph administration were 64.5±9.5 in the CC genotype, 69.5±12.3 in the CG genotype, and 61.8±9.7 in the GG genotype. The corresponding values of post-Mph administration were 65.5±13.4 in the CC genotype, 71.4±12.8 in the CG genotype, and 62.7±7.4 in the GG genotype. Similarly, when the effects of age, sex, and ADHD subtypes were controlled, rs133946C>G polymorphism was found to be statistically significantly affecting the alteration of NAA levels in response to Mph in the DLPFC (F[2,51]=3,56; P=0.036; ɲ2=0.125). Post hoc comparisons revealed a statistically significant difference between CG and GG genotypes after Bonferroni adjustment (P=0.016; Figure 1). In the other brain regions, the polymorphism groups were not found to have any statistically significant effect on metabolite level alterations in response to Mph. The axial view of DLPFC and metabolite peaks are shown in Figure 2.
Discussion
In this study, we found that the variations of synapsin III gene rs133945G>A and rs133946C>G polymorphisms affected the NAA levels in the DLPFC after a single dose of Mph, while the age, sex, and ADHD subtypes were controlled. In the post hoc tests, we could not find a statistically significant difference among the rs133945G>A genotypes. But regarding rs133946C>G genotypes, we found a significant level of increase in the NAA concentrations for the CG genotype in comparison to the GG genotype.
To the best of our knowledge, this is the first study to investigate postmedication metabolite alterations among ADHD patients with regard to synapsin III polymorphism. Thus, we did not encounter any data that we could compare our results with regarding the impact of synapsin polymorphism genotypes on the medication-related neurometabolite alterations. Nevertheless, the NAA level alterations have been studied in relation to ADHD. Increased NAA levels in the PFC cortex of ADHD children have been reported.27–29 It has been suggested that the increased levels might represent the compensatory increased metabolism based on “feedback mechanisms” in children in the PFC,30 or an indicator of hyperactivation in the resting state.29 On the other hand, Hesslinger et al have found decreased NAA levels in the left DLPFC of combined ADHD subtypes compared to inattentive subtypes and healthy controls in an MRS study in adults. The authors suggested that decreased NAA levels were associated with neuronal loss and neuronal function impairment.7 Husarova et al30 proposed that this finding might be related to the lower metabolism or decreased density of neurons due to the long-lasting hypermetabolism in the DLPFC. When we interpret these results together, it may be concluded that in children diagnosed with ADHD the NAA levels in the PFC may be increased in comparison with controls; however in adults, the differences in NAA levels between ADHD patients and healthy controls may disappear, or combined ADHD subtypes may have lower levels of NAA in comparison to controls.
A literature search revealed inconsistent data about the alterations in NAA levels in response to medications in ADHD patients: For example, Carrey et al31 treated children diagnosed with ADHD with atomoxetine, Mph, and dexedrine for 13 weeks and reported no significant change in NAA levels in response to medication in the PFCs of the participants. In another study conducted with children diagnosed with ADHD, again, Husarova et al reported decreased NAA levels and NAA/Cr ratio in the left DLPFC after 8 weeks of atomoxetine treatment but no change in response to Mph treatment.32 In contrast, Wiguna et al33 found increased NAA/Cr ratio in the PFCs of children with ADHD after 20 mg daily Mph in their study and suggested that this change may be a result of functional amelioration and increase in neuroplasticity.
Our results regarding increased NAA concentrations after Mph administration are inconsistent with those of Wiguna et al.33 In the literature, it has been proposed that NAA is a marker of overall neuronal and axonal integrity, and while a low level of NAA is accepted as an indicator of neuronal loss and/or dysfunction, increased NAA may indicate neuroplasticity.7,10,30,33,34 Thus, increased NAA concentration in DLPFC that we reported may be an indicator of the alterations that contribute to the treatment effects of Mph. Microdialysis studies have demonstrated that clinically relevant doses of Mph increase dopamine efflux preferentially within the PFC.35 Synapsin III is involved in slow synaptic transmission, particularly dopamine release,36 and it has a significant role in neurogenesis and axon formation.19 The impact of synapsin III polymorphism on the alterations of post-Mph DLPFC NAA levels that we have seen seems to be consistent with this knowledge. Based on this, we speculate that synapsin III gene may be one of the factors that contribute to the treatment effects of Mph in the DLPFC. It is unclear whether this effect is by the direct impact of synapsin III gene in the DLPFC or by indirect effects via striatofrontal networks. In a postmortem study conducted with schizophrenia patients, several synaptic proteins were found to be decreased in DLPFC, characterizing a synaptic pathology.37 More specifically, Porton and Wetsel38 reported reduced levels of synapsin III in schizophrenia patients. This data from the literature may lend support to the disturbances in synapsin III and other synaptic proteins especially in the DLPFC, which could be related to the pathogenesis of brain disorders. Hyman39 hypothesized that neural misconnectivity contributes to schizophrenia and other neuropsychiatric disorders, which is also in line with this view. There is need for future studies with animals and humans to find the answer to this question or to confirm this speculation.
According to post hoc comparisons, while there was no significant difference among the synapsin III gene rs133945G>A polymorphism genotypes, a statistical significance was detected between the rs133946C>G polymorphism genotypes: CG genotype was found to act to alter NAA concentrations more than the GG genotype. Kenar et al23 found in their study, which reported an association between ADHD and synapsin III gene rs133946C>G polymorphism, that the CG genotype was more frequent among ADHD patients and the GG genotype was more common among healthy controls. Thus, it may be suggested that, as indicated by increased NAA levels, an increase in post-Mph neuronal activation is more prominent among ADHD patients with the CG genotype in comparison to ADHD patients with the GG genotype. However, to confirm this view, more future studies are needed.
Another finding of our study was that NAA levels had significantly increased after a single dose of Mph in the ACC of rs133946C>G polymorphism CG genotype. Hammerness et al12 treated children diagnosed with ADHD for 6 weeks with Mph but could not find any significant change in the level of any metabolite, including NAA in the ACC of children. However, Kronenberg et al found a significant increase in NAA levels in the ACC of adult ADHD patients after administration of Mph for 5–6 weeks. They claimed that NAA is synthesized in the mitochondria in an energy-dependent manner and is therefore regarded as an indicator of neuronal energy status.13 A study investigated the effect of Mph on hypoperfusion and reported that blood flow increased after the treatment in the bilateral prefrontal, caudate, and thalamic areas.40 Similarly, chronic Mph application was found to increase the neuronal mitochondrial activity in rats.41 Thus, our finding of increased NAA post-Mph may be an indicator of improved perfusion as well as increased neuronal activation in the CG genotype of rs133946C>G polymorphism.
Finally, we found a significant increase in the Cho levels after a single dose of Mph in the striatum of patients with GG genotype of synapsin III gene rs133945G>A polymorphism. The Cho peak is considered to be related to membrane metabolism and believed to be reflecting membrane turnover, ie, either increased membrane degradation or synthesis.42,43 Also the role of Cho-containing compounds has been indicated in the energy metabolism,44,45 and a possible relationship to cholinergic neurotransmission has been suggested.46,47
Elevated Cho or Cho/Cr levels in the striatum, ACC, and PFC have been shown in patients with ADHD in comparison to controls.9,10,28,48 However, in a more recent meta-analysis, Aoki et al29 could not find a significant difference in Cho levels in the striatum of ADHD patients in comparison to controls. In studies searching for treatment effects, significant Cho and Cho/Cr decreases after 5–6 weeks of Mph treatments have been shown in the ACC in adults13 and in the PFC of children after 12 weeks of long-acting Mph.33
Findings of those studies that showed either elevated or unchanged Cho levels in ADHD patients or reduced levels after Mph treatment are in contrast to the increased Cho postmedication that we found. This difference could be a consequence of the medication (long acting vs short acting, treatment duration, dose, etc) as well as the age, prior medication status of patients (treatment naïve or a prior history of medication use) or variable genetic structure of the participants. As support for probable medication-related alterations, it has been suggested that Mph may have different acute effects on particular metabolites, and therefore, the reactivity rate of particular metabolites to drugs may be potentially different.32 Nonetheless, we should add that Husarova et al found increased Cho concentrations in the right DLPFC after atomoxetine medication but not Mph. The authors argued that, although this result was not in accordance with their hypothesis, increased Cho-containing compounds could indicate an increased membrane synthesis due to the restoration of membranes after the decrease in ADHD-related hyperexcitation.32 In our study, the GG genotype of rs133945G>A polymorphism showed a trend toward having markedly lower (although not at statistically significant level) premedication levels of Cho in comparison to other genotypes (Cho values for GG: 31.8±5, GA: 35.9±6.9, and AA: 37.8±7.2; P=0.056). Thus, we hypothesize, similar to Husarova et al, that in participants with the GG genotype, Mph might have acted to decrease the hyperexcitation, and as a consequence of the restoration of membranes, Cho synthesis might have increased. The precise meaning of altered Cho signals needs to be resolved with future studies.48
Soliva et al found no significant correlations between dose or time duration of medication and neurometabolite levels in the frontal gyrus of pediatric ADHD subjects with an 18.2-month mean medication time.49 Similarly, in a study conducted with ADHD diagnosed children, one single dose of Mph had no significant effect on brain metabolite levels.10 However, imaging studies have demonstrated the effect of a single dose and shown that Mph enhances attentional processing and modulates the activity in brain areas associated with inhibition and attention. In line with this, short-term Mph and atomoxetine treatment were found to affect the neurometabolic changes in some studies.32,33,50,51 Also, Mph exposure has been reported to cause long-term changes in MRI studies.52,53
In the one study conducted with an adolescent age group, treated ADHD patients were found to have lower metabolite ratios in ACC than the untreated patients, although the difference was not statistically significant.12 In adult ADHD patients, increased NAA and reduced Cho levels in the anterior cingulum were reported following 5–6 weeks of Mph treatment.13 It has been suggested that the increase in NAA can be related to the induction of mitochondrial respiratory chain enzyme activities by chronic Mph exposure, especially in the PFC and striatum.41 However, Maltezos et al8 could not find any differences between treatment-naïve and stimulant-treated ADHD patients. Collectively, although the results are controversial, Mph may have short-term and long-term effects in pediatric, adolescent, and adult ADHD patients. It should be kept in mind that 58% of our study participants had been on stimulant treatment and had not received medication in the last 48 hours.
To the best of our knowledge, this is the first study that investigated metabolite changes in the brain structure after a single dose of 10 mg Mph according to synapsin III polymorphisms in adult ADHD patients. The total number of patients who participated in the study was considerable when compared to previous studies. However, there are several limitations of the present study that should be noted. First, a good percentage of the patients had a prior history of long-term use of stimulant medication, although a washout period of 48 hours was provided. This prior treatment might have affected the MRS metabolite levels. Second, cigarette smoking was not ruled out, which is a possible factor that might affect Mph response. Third, we did not correlate the metabolite changes with clinical outcome measures, such as neuropsychological tests. The fourth limitation is related to technical procedures: we used relatively low-Tesla MR and a unilateral area was evaluated. Finally, a control group was not present in the study.
Conclusion
The results of this study suggest that synapsin III gene polymorphisms may be affecting the changes in neurometabolite levels in response to Mph in adult ADHD patients. Although the role of synaptic proteins in the etiopathogenesis of brain disorders has been shown in the literature, still there is need for more extensive and robust data. With future studies, the alterations in neurometabolite levels that are thought to be related to Mph response should be correlated with clinical improvement measures. While doing this, knowing the plasma concentrations of Mph may help in the interpretation of the findings and also contribute to a better understanding, at least, of some of the pharmacogenetic factors that have probable effects. Lastly, future studies should investigate age-related alterations in child, adolescent, and adult age groups of participants to better understand the treatment responses and pathophysiology of ADHD.
Acknowledgment
We thank to Gonca Unal for her contribution related to participants, and we thank to all the study participants.
Disclosure
The authors report no conflicts of interests in this work.
References
Willcutt EG. The prevalence of DSM-IV attention-deficit/hyperactivity disorder: a meta-analytic review. Neurotherapeutics. 2012;9(3):490–499. | ||
Thomas R, Sanders S, Doust J, Beller E, Glasziou P. Prevalence of attention-deficit/hyperactivity disorder: a systematic review and meta-analysis. Pediatrics. 2015;135(4):e994–e1001. | ||
Weyandt L, Swentosky A, Gudmundsdottir BG. Neuroimaging and ADHD: fMRI, PET, DTI findings, and methodological limitations. Dev Neuropsychol. 2013;38(4):211–225. | ||
Rubia K, Halari R, Cubillo A, et al. Methylphenidate normalizes fronto-striatal underactivation during interference inhibition in medication-naïve boys with attention-deficit hyperactivity disorder. Neuropsychopharmacology. 2011;36(8):1575–1586. | ||
Cubillo A, Smith AB, Barrett N, et al. Drug-specific laterality effects on frontal lobe activation of atomoxetine and methylphenidate in attention deficit hyperactivity disorder boys during working memory. Psychol Med. 2014;44(3):633–646. | ||
Soares DP, Law M. Magnetic resonance spectroscopy of the brain: review of metabolites and clinical applications. Clin Radiol. 2009;64(1):12–21. | ||
Hesslinger B, Thiel T, Tebartz van Elst L, Hennig J, Ebert D. Attention-deficit disorder in adults with or without hyperactivity: where is the difference? A study in humans using short echo 1H-magnetic resonance spectroscopy. Neurosci Lett. 2001;304(1–2):117–119. | ||
Maltezos S, Horder J, Coghlan S, et al. Glutamate/glutamine and neuronal integrity in adults with ADHD: a proton MRS study. Transl Psychiatry. 2014;4:e373. | ||
Colla M, Ende G, Alm B, Deuschle M, Heuser I, Kronenberg G. Cognitive MR spectroscopy of anterior cingulate cortex in ADHD: elevated choline signal correlates with slowed hit reaction times. J Psychiatr Res. 2008;42(7):587–595. | ||
Jin Z, Zang YF, Zeng YW, Zhang L, Wang YF. Striatal neuronal loss or dysfunction and choline rise in children with attention-deficit hyperactivity disorder: a 1H-magnetic resonance spectroscopy study. Neurosci Lett. 2001;315(1–2):45–48. | ||
Carrey NJ, MacMaster FP, Gaudet L, Schmidt MH. Striatal creatine and glutamate/glutamine in attention-deficit/hyperactivity disorder. J Child Adolesc Psychopharmacol. 2007;17(1):11–17. | ||
Hammerness P, Biederman J, Petty C, Henin A, Moore CM. Brain biochemical effects of methylphenidate treatment using proton magnetic spectroscopy in youth with attention-deficit hyperactivity disorder: a controlled pilot study. CNS Neurosci Ther. 2012;18(1):34–40. | ||
Kronenberg G, Ende G, Alm B, Deuschle M, Heuser I, Colla M. Increased NAA and reduced choline levels in the anterior cingulum following chronic methylphenidate. A spectroscopic test-retest study in adult ADHD. Eur Arch Psychiatry Clin Neurosci. 2008;258(7):446–450. | ||
Wilens TE. Effects of methylphenidate on the catecholaminergic system in attention-deficit/hyperactivity disorder. J Clin Psychopharmacol. 2008;28(3 suppl 2):S46–S53. | ||
Contini V, Rovaris DL, Victor MM, Grevet EH, Rohde LA, Bau CHD. Pharmacogenetics of response to methylphenidate in adult patients with attention-deficit/hyperactivity disorder (ADHD): a systematic review. Eur Neuropsychopharmacol. 2013;23(6):555–560. | ||
Öner Ö, Akin A, Herken H, et al. Association among SNAP-25 gene DdeI and MnlI polymorphisms and hemodynamic changes during methylphenidate use: a functional near-infrared spectroscopy study. J Atten Disord. 2011;15(8):628–637. | ||
Viggiano D, Vallone D, Sadile A. Dysfunctions in dopamine systems and ADHD: evidence from animals and modeling. Neural Plast. 2004;11(1–2):97–114. | ||
Kao H-T, Porton B, Czernik AJ, et al. A third member of the synapsin gene family. Proc Natl Acad Sci U S A. 1998;95(8):4667–4672. | ||
Porton B, Wetsel WC, Kao H-T. Synapsin III: role in neuronal plasticity and disease. Semin Cell Dev Biol. 2011;22(4):416–424. | ||
Stöber G, Meyer J, Nanda I, et al. Linkage and family-based association study of schizophrenia and the synapsin III locus that maps to chromosome 22q13. Am J Med Genet. 2000;96(3):392–397. | ||
Chen Q, Che R, Wang X, et al. Association and expression study of synapsin III and schizophrenia. Neurosci Lett. 2009;465(3):248–251. | ||
Makkar R, Gomez L, Wigg KG, et al. The gene for synapsin III and attention-deficit hyperactivity disorder. Psychiatr Genet. 2007;17(2):109–112. | ||
Kenar AN, Edgünlü T, Herken H, Erdal ME. Association of synapsin III gene with adult attention deficit hyperactivity disorder. DNA Cell Biol. 2013;32(8):430–434. | ||
American Psychiatric Association. DSM-IV-TR. Arlington, VA: APA; 2000. | ||
Günay Ş, Savran C, Aksoy U, Maner F, Turgay A, Yargiç I. The norm study, transliteral equivalence, validity, reliability of adult hyperactivity scale in Turkish adult population. Psychiatry Turkey. 2006;8(2):98–107. | ||
Turgay A. Adult Hyperactivity Assessment Scale based on DSM IV (Unpublished Scale). Toronto, ON: Integrative Therapy Institute; 1995. | ||
Fayed N, Modrego PJ, Castillo J, Dávila J. Evidence of brain dysfunction in attention deficit-hyperactivity disorder: a controlled study with proton magnetic resonance spectroscopy. Acad Radiol. 2007;14(9):1029–1035. | ||
Courvoisie H, Hooper SR, Fine C, Kwock L, Castillo M. Neurometabolic functioning and neuropsychological correlates in children with ADHD-H: preliminary findings. J Neuropsychiatry Clin Neurosci. 2004;16(1):63–69. | ||
Aoki Y, Inokuchi R, Suwa H, Aoki A. Age-related change of neurochemical abnormality in attention-deficit hyperactivity disorder: a meta-analysis. Neurosci Biobehav Rev. 2013;37(8):1692–1701. | ||
Husarova V, Ondrejka I, Tonhajzerova I. Potential pathomechanisms of ADHD based on neurometabolite changes. Neuro Endocrinol Lett. 2010;31(4):438–445. | ||
Carrey N, MacMaster FP, Fogel J, et al. Metabolite changes resulting from treatment in children with ADHD: a 1H-MRS study. Clin Neuropharmacol. 2003;26(4):218–221. | ||
Husarova V, Bittsansky M, Ondrejka I, Dobrota D. Prefrontal grey and white matter neurometabolite changes after atomoxetine and methylphenidate in children with attention deficit/hyperactivity disorder: a (1)H magnetic resonance spectroscopy study. Psychiatry Res. 2014;222(1–2):75–83. | ||
Wiguna T, Guerrero APS, Wibisono S, Sastroasmoro S. Effect of 12-week administration of 20-mg long-acting methylphenidate on Glu/Cr, NAA/Cr, Cho/Cr, and mI/Cr ratios in the prefrontal cortices of school-age children in Indonesia: a study using 1H magnetic resonance spectroscopy (MRS). Clin Neuropharmacol. 2012;35(2):81–85. | ||
Dautry C, Vaufrey F, Brouillet E, et al. Early N-acetylaspartate depletion is a marker of neuronal dysfunction in rats and primates chronically treated with the mitochondrial toxin 3-nitropropionic acid. J Cereb Blood Flow Metab. 2000;20(5):789–799. | ||
Berridge CW, Devilbiss DM. Psychostimulants as cognitive enhancers: the prefrontal cortex, catecholamines, and attention-deficit/hyperactivity disorder. Biol Psychiatry. 2011;69(12):e101–e111. | ||
Kile BM, Guillot TS, Venton BJ, Wetsel WC, Augustine GJ, Wightman RM. Synapsins differentially control dopamine and serotonin release. J Neurosci. 2010;30(29):9762–9770. | ||
Pennington K, Beasley CL, Dicker P, et al. Prominent synaptic and metabolic abnormalities revealed by proteomic analysis of the dorsolateral prefrontal cortex in schizophrenia and bipolar disorder. Mol Psychiatry. 2008;13(12):1102–1117. | ||
Porton B, Wetsel WC. Reduction of synapsin III in the prefrontal cortex of individuals with schizophrenia. Schizophr Res. 2007;94(1–3):366–370. | ||
Hyman S. Mental illness: genetically complex disorders of neural circuitry and neural communication. Neuron. 2000;28(2):321–323. | ||
Kim BN, Lee JS, Cho SC, Lee DS. Methylphenidate increased regional cerebral blood flow in subjects with attention deficit/hyperactivity disorder. Yonsei Med J. 2001;42(1):19–29. | ||
Fagundes AO, Rezin GT, Zanette F, et al. Chronic administration of methylphenidate activates mitochondrial respiratory chain in brain of young rats. Int J Dev Neurosci. 2007;25(1):47–51. | ||
Mason GF, Krystal JH. MR spectroscopy: its potential role for drug development for the treatment of psychiatric diseases. NMR Biomed. 2006;19(6):690–701. | ||
Gujar SK, Maheshwari S, Björkman-Burtscher I, Sundgren PC. Magnetic resonance spectroscopy. J Neuroophthalmol. 2005;25(3):217–226. | ||
Bittšanský M, Výbohová D, Dobrota D. Proton magnetic resonance spectroscopy and its diagnostically important metabolites in the brain. Gen Physiol Biophys. 2012;31(1):101–112. | ||
Purdon AD, Rosenberger TA, Shetty HU, Rapoport SI. Energy consumption by phospholipid metabolism in mammalian brain. Neurochem Res. 2002;27(12):1641–1647. | ||
Freeman JJ, Jenden DJ. The source of choline for acetylcholine synthesis in brain. Life Sci. 1976;19(7):949–961. | ||
Barker PB, Breiter SN, Soher BJ, et al. Quantitative proton spectroscopy of canine brain: in vivo and in vitro correlations. Magn Reson Med. 1994;32(2):157–163. | ||
Perlov E, Philipsen A, Matthies S, et al. Spectroscopic findings in attention-deficit/hyperactivity disorder: review and meta-analysis. World J Biol Psychiatry. 2009;10(4 pt 2):355–365. | ||
Soliva JC, Moreno A, Fauquet J, et al. Cerebellar neurometabolite abnormalities in pediatric attention/deficit hyperactivity disorder: a proton MR spectroscopic study. Neurosci Lett. 2010;470(1):60–64. | ||
Benamor L. (1)H-Magnetic resonance spectroscopy study of stimulant medication effect on brain metabolites in French Canadian children with attention deficit hyperactivity disorder. Neuropsychiatr Dis Treat. 2014;10:47–54. | ||
Carrey N, MacMaster FP, Sparkes SJ, Khan SC, Kusumakar V. Glutamatergic changes with treatment in attention deficit hyperactivity disorder: a preliminary case series. J Child Adolesc Psychopharmacol. 2002;12(4):331–336. | ||
Nakao T, Radua J, Rubia K, Mataix-Cols D. Gray matter volume abnormalities in ADHD: voxel-based meta-analysis exploring the effects of age and stimulant medication. Am J Psychiatry. 2011;168(11):1154–1163. | ||
Frodl T, Skokauskas N. Meta-analysis of structural MRI studies in children and adults with attention deficit hyperactivity disorder indicates treatment effects. Acta Psychiatr Scand. 2012;125(2):114–126. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.