Back to Journals » Neuropsychiatric Disease and Treatment » Volume 11
The impact of internalizing symptoms on autistic traits in adolescents with restrictive anorexia nervosa
Authors Calderoni S, Fantozzi P, Balboni G, Pagni V, Franzoni E, Apicella F, Narzisi A, Maestro S, Muratori F
Received 26 August 2014
Accepted for publication 17 October 2014
Published 5 January 2015 Volume 2015:11 Pages 75—85
DOI https://doi.org/10.2147/NDT.S73235
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 5
Editor who approved publication: Dr Roger Pinder
Sara Calderoni,1,* Pamela Fantozzi,1,* Giulia Balboni,2 Veronica Pagni,1 Emilio Franzoni,3 Fabio Apicella,1 Antonio Narzisi,1 Sandra Maestro,1 Filippo Muratori1,4
1IRCCS Stella Maris Foundation, 2Department of Surgery, Medical, Molecular and Critical Area Pathology, University of Pisa, Pisa, Italy; 3Child Neuropsychiatric Unit, Women, Children and Adolescents Health Department, University Hospital S Orsola-Malpighi, Bologna, Italy; 4Department of Clinical and Experimental Medicine, University of Pisa, Pisa, Italy
*These authors contributed equally to this work
Background: Although previous studies indicated a positive association between restrictive anorexia-nervosa (AN-R) and autistic traits, the potential interference of psychiatric internalizing comorbidity on this association is not yet fully investigated.
Materials and methods: The aim of this study was to explore autistic traits and internalizing psychopathology in adolescents (age range: 11.7–17.2 years) with AN-R. Twenty-five patients referred to two tertiary-care hospitals were compared to a large control group (N=170) with no differences in age and sex. AN-R patients and controls filled out instruments assessing autistic traits (autism spectrum quotient [AQ]), psychopathology (youth self-report [YSR] 11–18), and eating patterns (eating attitude test [EAT]). In order to disentangle the possible mediating role of internalizing symptoms on autistic traits, two separate control groups (called True and False healthy control, both composed of 25 eating-problem-free participants) were derived from the whole control group on the basis of the presence or absence of internalizing problems in the YSR.
Results: AN-R patients scored significantly higher on AQ compared to the whole control group and to controls without internalizing problems (True HC), but these differences disappeared when only controls with internalizing problems (False HC) were considered.
Conclusion: Autistic traits in AN-R individuals may have been overestimated and may partly be due to comorbid internalizing symptoms in investigated patients.
Keywords: anorexia nervosa-restricting type, youth self-report, evolutive age, autism spectrum quotient
Introduction
Anorexia nervosa restricting type (AN-R) is a severe psychiatric disorder with a typical adolescent onset and a marked preponderance of females among the patients affected.1 The essential features for a diagnosis of AN-R include 1) restriction of energy intake relative to requirements, leading to a significantly low body weight in the context of age, sex, developmental trajectory, and physical health; 2) an intense fear of gaining weight or persistent behavior that interferes with weight gain; and 3) disturbance in experiencing body weight and shape. In addition, during the last 3 months, the person has not engaged in recurrent episodes of binge eating or purging behavior (ie, self-induced vomiting or the misuse of laxatives, diuretics, or enemas).2 Conversely, autism spectrum disorders (ASDs) are a heterogeneous group of neurodevelopmental conditions that appear in the early developmental period and show significant male bias in their incidence. The clinical presentation involves impairment in the socio-communicative abilities, as well as the presence of restrictive and repetitive patterns of behavior, interests, or activities that must be present in early developmental period.2 Despite these macroscopic epidemiological and phenotypic differences, an overlap between AN-R and ASD is strongly suggested. The link between these two disorders was first proposed by Gillberg.3 He indicated a similarity between AN-R and ASD patients both from the genotypic (co-presence of AN-R and ASD among members of the same family) and phenotypic (obsessive symptoms and difficulties in social relationship) points of view. Subsequently, a number of studies focused on AN-R individuals frequently expressing cognitive profiles similar to those observed in ASD subjects and characterized by an impairment in theory of mind (ToM) and empathy,4–6 in set shifting or cognitive flexibility,7,8 as well as in central coherence.9 Moreover, high levels of autistic-like behavior have been demonstrated in AN, such as the presence of rigid attitudes, narrow interests focused on food and weight, and social anhedonia.10–12 The co-occurrence of autistic and anorexic symptomatology has been summarized in a recent systematic review that has pointed to an average ASD prevalence of 22.9% in individuals with eating disorders.13 Finally, recent functional magnetic resonance imaging reports have provided empirical support for a different neuroanatomical involvement in task-induced activity in AN patients compared to healthy controls. In particular, similar to ASD patients, individuals with AN show altered activation in brain networks underlying ToM,14 cognitive and behavioral flexibility,15 and central coherence.16 In a similar vein, a recent study performed through the eye-tracking measure found that weight-restored females with AN-R spent significantly less time looking in the eye region than did controls,17 in accordance with analogous data on ASD patients.18 Conversely, other investigations failed to detect the presence of autism spectrum pathology in patients with AN-R,19–21 or have found autistic features only in a subgroup of the investigated anorexic patients.22,23 In order to examine to what extent patients with AN-R show an autism spectrum phenotype, several recent studies used the autism spectrum quotient (AQ),24 a standardized self-report measure for the evaluation of autistic traits. Hambrook et al20 first reported that adult patients with AN, compared to healthy control individuals, scored significantly higher (ie, were more impaired) on total AQ scores and on three of the five subscales (social skills, attention switching, and imagination). Furthermore, in a prospective study on teenage-onset AN individuals evaluated over a period of 18 years,25 patients described themselves as more autistic-like than controls on total score AQ and on social skills, attention switching, and attention-to-details subscales; moreover, a substantial subgroup of AN-R patients (32%) meet or have met criteria for an ASD. A very recent investigation on adolescents with AN26 suggested that autistic traits, measured through the AQ questionnaire, were overrepresented in AN individuals and that 2.9% of these patients (vs 0.4% of controls) belong to the “narrow autism phenotype”, while 47.8% is assigned to the “broader autism phenotype” and the “medium autism phenotype” categories, following the Wheelwright et al criteria.27 A direct comparison between AQ questionnaires fulfilled by AN and ASD patients was performed by Courty et al.28 The authors found that a third of the AN participants is classified as belonging to the broader/medium autism phenotype; in addition, AN patients had higher AQ total, attention switching, and communication skills scores than matched typical controls; on the other hand, participants with AN showed similar attention-to-details scores of ASD individuals. Finally, Tchanturia et al29 have recently replicated the finding of higher autistic traits (evaluated through the short version of the AQ) in AN patients compared with controls, highlighting the independence of eating disorder symptoms from autistic traits. Notwithstanding the repeated finding of autistic traits in AN patients, the nature of this association requires further investigation. In particular, to our knowledge, no study has deeply explored the relationship between internalizing symptoms and autistic traits in a group exclusively composed of adolescent with restrictive-type AN. Investigating this correlation is fundamental because anxious/depressive psychopathology is a frequent comorbid feature of anorexia30 and could have a major impact on social functioning of subjects with AN.31 Thus, this study aims to explore whether autistic traits in adolescent with purely AN-R are really part of an autism spectrum condition or, vice versa, represent a component of the internalizing psychopathology.
Specifically, 1) in order to investigate whether patients with AN-R present autistic traits (and therefore to replicate other investigations), a group of adolescent females with AN-R was compared on AQ with a large control group of healthy adolescent females of comparable age and education, and 2) in order to reveal whether autistic traits were specific to AN-R or were associated to the internalizing symptoms, the AN group was compared to two matched control subgroups of healthy adolescents with or without internalizing problems. The hypothesis was that internalizing symptoms directly interfere with the level of autistic traits, and, consequently, that differences in autistic traits usually found when comparing AN-R to healthy controls would decrease when AN-Rs are compared to adolescents with internalizing symptoms.
Methods
Participants
Twenty-five adolescent females (11–18 years old) with AN-R, characterized by restrictive behaviors with the absence of binge eating or purging behaviors, were recruited at the inpatient eating disorders unit of two tertiary Italian developmental neuropsychiatry hospitals (IRCCS Stella Maris, Pisa, and Polyclinic Sant’Orsola-Malpighi, Bologna, Italy). Patients were selected from all consecutive participants admitted to the two units from March 2012 to July 2013 who satisfied all the following criteria: diagnosis of AN-R type according to DSM-IV-TR criteria,32 female sex, and age range between 11 years and 18 years. Exclusion criteria were presence of psychotic symptoms, Wechsler Full Scale IQ <80, internistic pathology not correlated with the eating disorder, current or history of substance abuse, significant internistic instability (eg, severe hypotension or bradycardia, dehydration, or electrolyte imbalance) requiring constant medical care supervision. Twenty-one patients (80.8%) fulfilled the criteria for an Axis I anxiety and/or mood disorder (assessed through the Italian version of the Schedule for Affective Disorders and Schizophrenia for School-Age Children/Present and Lifetime Version: K-SADS-PL)33 and two out of these 21 patients met also the criteria for an Axis II Personality Disorder (evaluated by the Structured Clinical Interview for DSM-IV Axis II Personality Disorders: SCID-II).34 Seventeen patients (65.4%) received a psychopharmacological treatment with selective serotonin reuptake inhibitors and/or atypical antipsychotics and/or a mood stabilizer, while the remaining eight participants were medication-naïve. The control group (HC) consisted of 170 healthy adolescent females of comparable age and level of education. They were recruited from a group of students of three different middle schools and one high school of the metropolitan area of Pisa (Tuscany, Italy). In this group, the height and weight were self-reported.
In order to verify the association between autistic traits and internalizing symptoms, participants from the HC group were selected to create two subgroups of 25 participants (True and False HC), both 1-to-1 age matched with the AN-R patients, but with or without internalizing behaviors, measured by the youth self-report 11–18 (YSR 11–18).35 The participants of the True HC subgroup had a T-score <60 on the YSR summary scales, while those of the False HC had a T-score ≥60 on both internalizing and total problems summary scales.
Assessment instruments
To assess the presence and severity of autistic traits, eating disorder, and internalizing symptoms, the AQ,24 the eating attitude test-26 (EAT-26)35 and the YSR were used, respectively.
Autism spectrum quotient
The AQ is a 50-item self-report questionnaire, made up of ten questions organized in five subscales assessing five different areas: social skill, attention switching, attention to detail, communication, and imagination. Participants are asked to indicate the grade of agreement with each statement in a 4-point Likert scale ranging from “strongly agree” to “strongly disagree”. To measure the total AQ score, each item scores “1” if the respondent records the item/behavior either mildly or strongly. In all the other cases, the score is “0”. Possible total scores range from 0 to 50. The scale has high test/retest reliability and sound internal reliability with Cronbach’s-alpha for all subscales above 63.24 The total scores were classified into one out of five categories:27 low (<20), average (between 20 and 23), broader autism phenotype (BAP: AQ scores between 1 and 2 SDs above the mean; 23–28), medium autism phenotype (MAP: AQ scores between 2 and 3 SDs above the mean; 29–34), or narrow autism phenotype (NAP: AQ scores 3 SDs or higher above the mean; 35+). Globally, the score under the cutoff of 23 is considered as typical (non-ASD), while a score above the cutoff of 23 is considered in the autism phenotype.
Eating attitude test-26
The EAT-26 consists of 26 questions designed to measure attitudes and behaviors associated with eating disorder. It comprises three subscales: dieting, bulimia, and oral control. It is rated using a 5-point Likert scale ranging from “always” to “never” to measure total levels of ED psychopathology. Possible total scores range from 0 to 78; a higher score reflects a greater degree of eating pathology, with a score over 20 indicating a possible eating problem. The EAT-26 has very good internal reliability with a Cronbach’s-alpha of 90.36
Youth self-report 11–18
The YSR 11–18 is a 119-item self-report measure designed to record problems in youth of ages 11 to 18. The measure provides scores for three summary scales (internalizing, externalizing, total problems), eight syndrome scales, and six DSM-oriented scales. The individual is asked to rate the frequency of each behavior on a 3-point Likert scale (0, “not true”; 1, “somewhat or sometimes true”; 2, “very true or often true”). A T-score of 63 and above for summary scales is 70 and above for syndrome and DSM-oriented scales are generally considered clinically significant. Values between 60 and 63 for summary scales or between 65 and 70 for syndrome and DSM-oriented scales identify a borderline clinical range. Values <60 for the summary scales or under 65 for other scales are not considered clinically significant.35
Procedure
First, we assessed the prevalence of autistic traits among AN-R participants. For this, the AN-R group was compared on the AQ scale with the HC group made up of all the 170 healthy adolescent females. Then, the association between autistic traits and internalizing symptoms was evaluated. For this, the AN-R participants were compared on AQ with the two matched False and True HC subgroups with and without internalizing symptoms, respectively.
Moreover, to investigate the relationship between autistic traits, severity of the eating disorder, and psychopathology, correlation was computed between AQ, body mass index (BMI), EAT-26, and YSR 11–18 internalizing in AN-R group and in False and True HC subgroups.
All the instruments (AQ, EAT-26, and YSR 11–18) were filled out by the AN-R patients during the first days of the clinical assessment and by the HC individuals during school hours. The protocol received ethical clearance from the Health Research Ethics Committee at IRCCS Stella Maris Foundation (Italy), and was approved by the educational board of each school for the HC group. The study was carried out in accordance with the guidelines of the Declaration of Helsinki. Written informed consent from participants and a parent or guardian of the patients and from healthy individuals and their parents was obtained.
Statistical analyses
Student’s t-test for independent samples and ANOVA for dependent samples with Bonferroni’s post hoc, respectively, were used to compare the AN-R group with the overall HC group and with the two True and False HC subgroups. In case of statistically significant differences, Cohen’s-d for independent and dependent samples was computed37,38 and was evaluated, in agreement with the author’s criteria: as a negligible (<0.20), small (0.20–0.49), medium (0.50–0.79), and large (≥0.80) effect size. Pearson correlations were used to study the relationships between autistic traits, severity of eating disorder, and psychopathology.
Results
Comparisons between AN-R and HC groups
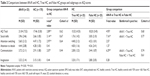
Table 1 shows no significant differences between AN-R and HC in age (P=0.156). As expected, AN-R patients, compared to HC, had a statistically significant lower BMI (P<0.001), a significantly higher score on the EAT-26,36 total, and subscales (P<0.001), and on the psychopathology behavior scale YSR,35 total problems, and internalizing and withdrawn scales (P<0.001; the effect sizes were always large).
There were no differences in age between AN-R and both True and False HC subgroups. AN-R participants had a statistically significant lower BMI and higher EAT-26 total and subscale scores than both True and False HC subgroups (with large or [just in two cases] medium effect sizes). On the contrary, regarding the YSR scale scores, AN-R had a different score compared with the True HC (with large or [just in one case] medium effect sizes) but not with the False HC subgroup (Table 1). As expected, the only differences found between the True and False HC subgroups were on the YSR scales, with the False HC having a higher score (with large effect sizes).
The mean (SD) scores obtained on the AQ by the AN-R and HC groups are reported in Table 2. AN-R had a statistically significant higher score on AQ total scale as well as on social skills, attention switching, and communication AQ subscales; the effect sizes were medium or (just for the communication) small. On the contrary, no differences were found between AN-R and HC groups on the attention-to-details and imagination AQ subscales.
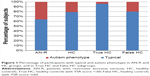
Using AQ categories, 16 (64%) AN-R vs 135 (79.41%) HC were classified as typical; 7 (28%) AN-R vs 30 (17.65%) HC were classified as BAP; 5 (2.94%) HC were classified as MAP and 2 (8%) AN-R were classified as NAP. When classified as typical (< the cutoff of 23) vs autistic phenotype (> the cutoff of 23; Figure 1), no difference was found in the classification between the two groups (χ2(1)=2.96, P=0.08).
Comparisons between AN-R group and True and False HC subgroups
The mean (SD) scores obtained on the AQ by the AN-R group and True and False HC subgroups participants are reported in Table 2. Compared with the True HC individuals, AN-R participants had higher scores on AQ total scale and on social skills and communication subscales with a medium effects size. On the contrary, no differences were found on AQ scales and subscales between AN-R and False HC participants. Between the True and False HC subgroups, the only difference found was on AQ communication subscale (with a medium effect size). Classification of the AN-R, True, and False HC participants is reported in Figure 1. AN-R participants more than True HC participants had an autistic phenotype over the cutoff of 23 (χ2(1)=8.00, P=0.005), while no difference was found if compared with False HC participants (χ2(1)=1.59, P=0.21).
Correlation between autistic traits, severity of eating disorder, and psychopathology
Correlation coefficients between the AQ scores, EAT-26, and YSR internalizing scales in AN-R group, True HC, and False HC subgroups are reported in Table 3. Within the AN-R group, the EAT-26 total score was significantly correlated with the autistic traits communication (P<0.05), whereas within the False HC group the EAT-26 total score was significantly correlated with the autistic traits’ social skills (P<0.05). Moreover, Pearson correlation analysis did not reveal any association also between AQ scores (total and subscales) and BMI in all the three groups.
As regards the relationship between autistic traits and psychopathology, a positive, statistically significant correlation was found between YSR internalizing and AQ total, AQ attention switching, and AQ communication in AN-R. In the False HC group, a positive, significant correlation was found between YSR and AQ total, social skills, and attention switching. No statistically significant correlations were found in the True HC subgroup.
Discussion
The primary goal of this study was to investigate whether adolescents with AN-R have higher rates of autistic traits and BAP in comparison to a matched control group in an Italian sample. Our results confirmed that AN-R patients have significantly higher scores on the AQ than their typical peers. This finding supports the hypothesis that females with anorexia share social and communication difficulties usually described in participants with ASD and replicates in a narrow group of adolescents the evidence of previous studies on more aged patients.20,26,29 If we consider AQ categories,27 more than a third of our AN-R enter in the broader, narrow autism phenotype and two out of these cases have a more clear autistic phenotype. As in previous investigations,26,29 patients with AN are more likely to show the broader, medium or narrow phenotype: in Tchanturia et al’s study,29 more than a quarter of their AN group scored over the clinical cutoff; an even higher percentage is reported by Baron-Cohen et al.26 While BAP refers to subthreshold autism-related traits,39 NAP is composed of a group of characteristics (language, personality, cognitive, and behavioral) mirroring the autism symptoms domains.40 Thus, we can support the hypothesis that autistic traits regard a significant minority of AN-R and that in some more rare cases they can be a core part of the clinical presentation or a real comorbidity, where autistic traits may exacerbate and maintain the eating disorder.29 In addition, the presence of NAP individuals within the AN-R group invites speculation that a minority of high-functioning subjects with a global good functioning arrive at the clinical observation only for eating disorders rather than for typical ASD symptomatology. In these cases, only an accurate and in-depth assessment, performed by an autism expert multidisciplinary team and supported by gold standard diagnostic evaluations (ie, the Autism Diagnostic Observation Schedule [ADOS]41 and the Autism Diagnostic Interview–Revised [ADI-R])42 could highlight the co-occurring autistic disorder.
Recent studies support the hypothesis that ASD is difficult to identify in females: girls are frequently diagnosed later in life than boys and especially if coexistent intellectual or behavioral problems are present.43–45 Therefore, ASD females with a higher IQ or with a milder phenotype may be underdiagnosed or misdiagnosed, and some of these “missed” patients could lie in AN-R population.
The second objective of this study was to explore the relationship between autistic traits in adolescents with AN-R and the internalizing psychopathology. To reach this aim, we compared the AN-R patients not only to a large HC group but also to the adolescents within the control group showing levels of internalizing problems similar to those observed in AN-R patients, hereinafter referred to as the “False HC”. When performing this new comparison, the AQ differences tend to disappear, ie, higher levels of internalizing problems in the “False HC” group were associated with higher AQ scores, and these latter scores were not significantly different from those of AN-R patients. This finding supports the hypothesis that autistic traits are more linked to anxiety and depression than to AN-R per se. Furthermore, similar to a previous study,29 we found a positive correlation between AQ (total and some subscales) scores and YSR internalizing in AN-R and False HC groups. On the other hand, our research documented only a marginal association between autistic traits and eating disorder symptoms, in keeping with previous investigations.28,29
These two findings regarding autistic traits, ie, their close relationship with internalizing psychopathology and their extremely limited association with eating disorder symptoms, would suggest that aspects of depression such as low mood, social withdrawal, and social anhedonia could have a major role in increasing autistic traits in AN-R. We could speculate that these traits are exacerbated by starvation, which has been shown to reduce the quality of interpersonal relationships, interest, and pleasure from social interaction, and libido.46 According with this view, the association of anxiety disorders/depression with eating disorders accounted for a large portion of difficulties with social functioning in patients with eating disorder.47 On the other hand, the overlap between some autistic symptoms and depressive symptoms has been known for a long time. Hoekstra et al48 reported that the YSR withdrawn scale, a component of the internalizing scale, was a predictor of endorsement of autistic traits. The withdrawn syndrome scale captures shy, introvert, and withdrawn behavior and includes items such as “prefer to be alone”, “secretive”, or “withdrawn”, but also contains items such as “lacks energy” and “sad” more indicative of depression. These authors48 have also suggested that social problems related to anxiety and depression may be common in people highly endorsed of autistic traits; a similar interpretation has been proposed by a study on the Child Behavior Check List 1.5–5,49 whose structure is analogous to that of YSR.
It is worth noting that the associations between autistic and anorexic symptomatology in the current study were primarily accounted for by three autistic traits. In fact, the AN-R adolescents significantly differ from the whole HC group as far as social skills impairment, attention switching problems, and communication difficulties are concerned; on the contrary, no significant differences between patients and controls were found in the attention-to-details and imagination AQ subscales. Not all studies exploring autistic traits in AN-R have considered AQ scores in each subscale, but when this analysis was performed, results similar to ours were obtained.28 Several authors have highlighted impairment in social skills and communication abilities in AN-R individuals,50,51 but these characteristics could be attributed to the internalizing symptoms frequently associated with the anorexic psychopathology,31,52,53 and do not necessarily reflect a phenotypic comorbidity with ASD.
The third significantly impaired subscale in AN-R patients, ie, attention switching, refers to the ability to shift between attentional or response sets. An impairment in this domain reflects poor cognitive flexibility (ie, the ability to intentionally shift thoughts and actions in response to contextual changes)54 and perseveration, namely behavioral characteristics typical for ASD patients. But the comorbid psychopathology may play a role also in this case. In fact, the same poor mental flexibility could be found in obsessive-compulsive personality disorder (OCPD), a maladaptive personality pattern characterized by excessive rigidity, need for order/control, preoccupation with details, and excessive perfectionism,2 and consistently overrepresented in AN-R.55 In particular, traits of OCPD in childhood may represent an important risk endophenotype for developing AN-R,54 frequently persist after recovery,57 and tend to be related with a poorer prognosis.58 Thus, high levels of attention switching problems could be an expression to the personality traits associated with AN-R59 rather than being the manifestation of an autistic feature. According to the view that the associated internalizing psychopathology may account for the AQ traits in AN-R patients, a recent investigation60 found impaired set shifting abilities (largely reconductable to attention switching domain) in individuals with unipolar depression, but not in AN patients without a comorbid mood disorder.
Differently, AQ subscales scores showed that AN-R patients do not differ from their non-anorexic peers in attention to details and imagination domains. This would mean that the cognitive style composed of higher attention to detail (ascribable to the weak central coherence theory [WCC])61,62 and lower imagination,63 commonly described in ASD individuals, do not characterize a norectic patients. Existing studies provide mixed support for these findings. In fact, a superior performance on attention to details has been documented as a specific AN-R trait in several neuropsychological studies on AN-R patients9,64,65 and on recovered AN individuals.66,67 On the other hand, recent investigations assessing the neuropsychological profile of AN-R patients relative to HC individuals using a large set of tests failed to detect significant differences in specific measures referable to attention-to-detail ability.19 Conversely, literature exploring the imagination skills in AN-R individuals is scarce, and a studies on this topic (eg, through the investigation of early pretend play ability of individuals who develop AN-R) are required in the future.68
Taken together, the observation of AQ subscales seems to prove main difficulties in social interaction and communication abilities, some difficulties in attention switching, but typical performance in attention to detail and imagination, that is, the absence of superior local processing of information and the weak imagination that are core characteristics of ASD. Crucially, these latter aspects of cognitive profile might account for the ASD non-social symptoms, including repetitive behavior, a narrow repertoire of interests, or resistance to change. All these symptoms are considered core deficits of ASD and now necessary for an ASD diagnosis.2
In conclusion, the strong positive correlation between AQ and YSR, as well as the profile emerging from the analysis of AQ subscales, suggests the importance of not jumping to easy conclusions of interpreting AQ traits in AN-R patients as an ASD phenotype expression.
Clinical implications
We replicated previous findings of elevated AQ scores also in patients with AN-R; however, our investigation appeared to indicate that internalizing problems, in particular depression, may account for the high autistic traits detected in AN-R patients. Since autistic traits are frequently associated with illness chronicity and poor outcome, and persist long after recovery,25,69,70 an effective treatment of internalizing comorbidity could be the first step to healing. In the absence of a clear understanding of the pathophysiology and cognitive alterations underlying mental disorders, it is, however, not possible to conclude whether the clinical, personality, and neurocognitive features reported here are “truly” autistic or rather intrinsic characteristics of the AN-R (if such a distinction can be made). Overall, our study links disturbed eating, emotional regulation, and problems in social interactions. Nevertheless, our results suggest that difficulties in AN are only superficially similar to those found in ASD. Our mixed findings encourage the exploration of transdiagnostic similarities between anorexic and autistic disorders. In particular, longitudinal studies could help elucidate whether or not the autistic features observed in AN patients are a mere effect of the acute phase of starvation.
Limitations and future directions
The results of this study need to be considered in light of a number of limitations. First, the information obtained from the AQ questionnaires was subject to limitations inherent in self-report measures; consequently, it should be important to integrate these findings with experimental paradigms. Second, the relatively small sample size of the clinical group limited the power of our study and precluded the examination of the possible influence of age on autistic traits. Third, the inclusion of an inpatient or day-patient AN-R population referred to two tertiary care centers limits the representativeness of the group and reduces the generalizability of results to the full population of people with restrictive eating disorder. In fact, whether our findings could be generalized to individuals with Eating Disorder Not Otherwise Specified (EDNOS) with restrictive eating habits, a disease presentation markedly more prevalent than purely AN-R,71 remains an open question for further empirical investigation. Fourth, the possible impact of some variables (eg, socioeconomic status, disease duration) on the results obtained has not been evaluated, and it deserves further investigation. Fifth, the large variability on AQ and EAT-26 scores in the patient group reflects high heterogeneity in autistic traits and severity of eating disorder within the AN-R subjects. This issue implies that results cannot be generalized with confidence to the entire AN-R population and suggests future analyses in larger subgroups, more homogeneous in terms of AQ and EAT-26 scores.
Future studies need to identify the underlying cognitive strategies and neuroanatomical circuits that account for co-occurring features in AN-R and ASD patients, in order to inform whether the two disorders share common cerebral mechanisms. Further investigations should also determine whether the relationship between autistic traits and anxiety/depression is specific to AN-R only or, vice versa, could be extended to other types of eating disorders in which the internalizing psychopathology is highly represented (eg, anorexia nervosa binge-eating/purging type, bulimia nervosa). Moreover, the next step to better understand the relationship between AN-R and autistic traits is to collect longitudinal data that assess AN-R patients in the acute phase of malnutrition and after their recovery. In this way, it will be possible to investigate 1) whether autistic traits persist in recovered AN-R individuals, 2) the role of internalizing comorbidity for autistic traits in AN-R, since the anxious and depressive symptoms are known to be related with a state of starvation,46,72 3) the influence of autistic traits on the outcome of AN-R patients (ie, their the prognostic value). Finally, our findings highlight the importance of measuring ASD traits in the clinical assessment of AN-R patients, in order to provide, if necessary, an integrated intervention tailored to the autistic features.
Disclosure
The authors received no specific grant from any funding agency, commercial or not-for-profit sectors, for this publication. The authors report no conflicts of interest in this work.
References
Herpertz-Dahlmann B. Adolescent eating disorders: definitions, symptomatology, epidemiology and comorbidity. Child Adolesc Psychiatr Clin N Am. 2008;18(1):31–47. | ||
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Publishing; 2013. | ||
Gillberg C. Are autism and anorexia nervosa related? Br J Psychiatry. 1983;142:428. | ||
Calderoni S, Fantozzi P, Maestro S, et al. Selective cognitive empathy deficit in adolescents with restrictive anorexia nervosa. Neuropsychiatr Dis Treat. 2013;9:1583–1589. | ||
Russell TA, Schmidt U, Doherty L, Young V, Tchanturia K. Aspects of social cognition in anorexia nervosa: affective and cognitive theory of mind. Psychiatry Res. 2009;168(3):181–185. | ||
Oldershaw A, Hambrook D, Tchanturia K, Treasure J, Schmidt U. Emotional theory of mind and emotional awareness in recovered anorexia nervosa patients. Psychosom Med. 2010;72(1):73–79. | ||
Friederich HC, Herzog W. Cognitive-behavioral flexibility in anorexia nervosa. Curr Top Behav Neurosci. 2011;6:111–123. | ||
Tchanturia K, Davies H, Roberts M, et al. Poor cognitive flexibility in eating disorders: examining the evidence using the Wisconsin Card Sorting Task. PLoS One. 2012;7(1):e28331. | ||
Lopez C, Tchanturia K, Stahl D, Treasure J. Weak central coherence in eating disorders: a step towards looking for an endophenotype of eating disorders. J Clin Exp Neuropsychol. 2009;31(1):117–125. | ||
Holliday J, Uher R, Landau S, Collier D, Treasure J. Personality pathology among individuals with a lifetime history of anorexia nervosa. J Pers Disord. 2006;20(4):417–430. | ||
Oldershaw A, Treasure J, Hambrook D, Tchanturia K, Schmidt U. Is anorexia nervosa a version of autism spectrum disorders? Eur Eat Disord Rev. 2011;19(6):462–474. | ||
Tchanturia K, Davies H, Harrison A, Fox JR, Treasure J, Schmidt U. Altered social hedonic processing in eating disorders. Int J Eat Disord. 2012;45(8):962–969. | ||
Huke V, Turk J, Saeidi S, Kent A, Morgan JF. Autism spectrum disorders in eating disorder populations: a systematic review. Eur Eat Disord Rev. 2013;21(5):345–351. | ||
Schulte-Rüther M, Mainz V, Fink GR, Herpertz-Dahlmann B, Konrad K. Theory of mind and the brain in anorexia nervosa: relation to treatment outcome. J Am Acad Child Adolesc Psychiatry. 2012;51(8):832–841. | ||
Zastrow A, Kaiser S, Stippich C, et al. Neural correlates of impaired cognitive-behavioral flexibility in anorexia nervosa. Am J Psychiatry. 2009;166(5):608–616. | ||
Fonville L, Lao-Kaim NP, Giampietro V, et al. Evaluation of enhanced attention to local detail in anorexia nervosa using the embedded figure test; an fMRI study. PLoS One. 2013;8(5):e63964. | ||
Watson KK, Werling DM, Zucker NL, Platt ML. Altered social reward and attention in anorexia nervosa. Front Psychol. 2010;1:36. | ||
Dalton KM, Nacewicz BM, Johnstone T, et al. Gaze fixation and the neural circuitry of face processing in autism. Nat Neurosci. 2005;8(4):519–526. | ||
Calderoni S, Muratori F, Leggero C, et al. Neuropsychological functioning in children and adolescents with restrictive-type anorexia nervosa: an in-depth investigation with NEPSY-II. J Clin Exp Neuropsychol. 2013;35(2):167–179. | ||
Hambrook D, Tchanturia K, Schmidt U, Russell T, Traesure J. Empathy, systemizing, and autistic traits in anorexia nervosa: a pilot study. Br J Clin Psychol. 2008;47(pt 3):335–339. | ||
Medina-Pradas C, Blas Navarro J, Álvarez-Moya EM, Grau A, Obiols JE. Emotional theory of mind in eating disorders. Int J Clin Health Psychol. 2012;12(2):189–202. | ||
Gillberg IC, Gillberg C, Råstam M, Johansson M. The cognitive profile of anorexia nervosa: a comparative study including a community-based sample. Compr Psychiatry. 1996;37(1):23–30. | ||
Tchanturia K, Happé F, Godley J, Treasure J, Bara-Carril N, Schmidt U. “Theory of Mind” in anorexia nervosa. Eur Eat Disord Rev. 2004;12(6):361–366. | ||
Baron-Cohen S, Wheelwright S, Skinner R, Martin J, Clubley E. The Autism-spectrum quotient (AQ): evidence from Asperger syndrome/high-functioning autism, males and females, scientists and mathematicians. J Autism Dev Disord. 2001;31(1):5–17. | ||
Anckarsäter H, Hofvander B, Billstedt E, et al. The sociocommunicative deficit subgroup in anorexia nervosa: autism spectrum disorders and neurocognition in a community-based, longitudinal study. Psychol Med. 2012;42(9):1957–1967. | ||
Baron-Cohen S, Jaffa T, Davies S, Auyeung B, Allison C, Wheelwright S. Do girls with anorexia nervosa have elevated autistic traits? Mol Autism. 2013;4(1):24. | ||
Wheelwright S, Auyeung B, Allison C, Baron-Cohen S. Defining the broader, medium and narrow autism phenotype among parents using the Autism Spectrum Quotient (AQ). Mol Autism. 2010;1(1):10. | ||
Courty A, Maria AS, Lalanne C, et al. Levels of autistic traits in anorexia nervosa: a comparative psychometric study. BMC Psychiatry. 2013;13: 222. | ||
Tchanturia K, Smith E, Weineck F, et al. Exploring autistic traits in anorexia: a clinical study. Mol Autism. 2013;4(1):44. | ||
Godart NT, Perdereau F, Rein Z, et al. Comorbidity studies of eating disorders and mood disorders. J Affect Disord. 2007;97(1–3):37–49. | ||
Tchanturia K, Hambrook D, Curtis H, et al. Work and social adjustment in patients with anorexia nervosa. Compr Psychiatry. 2013;54(1): 41–45. | ||
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed, Text Revision. Washington: American Psychiatric Association; 2000. | ||
Kaufman J, Birmaher B, Brent D, et al. Schedule for affective disorders and schizophrenia for school-age children-present and lifetime version (K-SADS-PL): initial reliability and validity data. J Am Acad Child Adolesc Psychiatry. 1997;36(7):980–988. | ||
First MB, Gibbon M, Spitzer RL, Williams JBW, Benjamin LS. Structured Clinical Interview for DSM-IV Axis II Personality Disorders (SCID-II). Washington: American Psychiatric Press; 1997. | ||
Achenbach T, Rescorla L. The Manual for the ASEBA School-Age Forms and Profiles. Burlington: University of Vermont, Research Center for Children, Youth, and Families of Vermont Department of Psychiatry; 2001. | ||
Garner DM, Olmsted MP, Bohr Y, Garfinkel P. The eating attitude test: psychometric features and clinical correlates. Psychol Med. 1982;12(4):871–878. | ||
Cohen J. Statistical power analysis for the behavioral sciences. 2nd ed. Hillsdale: Erlbaun; 1988. | ||
Morris SB, DeShon RP. Combing effect size estimates in meta-analysis with repeated measures and independent-groups designs. Psychol Methods. 2002;7(1):105–125. | ||
Virkud YV, Todd RD, Abbacchi AM, Zhang Y, Constantino JN. Familial aggregation of quantitative autistic traits in multiplex versus simplex autism. Am J Genet B Neuropsychiatr Genet. 2009;150B(3):328–334. | ||
Ruta L, Mazzone D, Mazzone L, Wheelwright S, Baron-Cohen S. The autism-spectrum quotient-Italian version: a cross cultural confirmation of the broader autism phenotype. J Autism Dev Disord. 2011;42(4):625–633. | ||
Lord C, Rutter M, DiLavore PS, Risi S. Autism Diagnostic Observation Schedule (ADOS). Los Angeles: Western Psychological Services; 1999. | ||
Le Couteur A, Lord C, Rutter M. Autism Diagnostic Interview-Revised (ADI-R). Los Angeles: Autism Genetic Resource Exchange; 2009. | ||
Begeer S, Mandell D, Wijnker-Holmes B, et al. Sex differences in the timing of identification among children and adults with autism spectrum disorders. J Autism Dev Disord. 2013;43(5):1151–1156. | ||
Dworzynski K, Ronald A, Bolton P, Happé F. How different are girls and boys above and below the diagnostic threshold for autism spectrum disorders? J Am Acad Child Adolesc Psychiatry. 2012;51(8):788–797. | ||
Giarelli E, Wiggins LD, Rice CE, et al. Sex differences in the evaluation and diagnosis of autism spectrum disorders among children. Disabil Health J. 2010;3(2):107–116. | ||
Keys A, Brožek J, Henschel A, Mickelsen O, Taylor HL. The Biology of Human Starvation. Oxford: University of Minnesota Press; 1950. | ||
Godart NT, Perdereau F, Curt F, et al. Predictive factors of social disability in anorexic and bulimic patients. Eat Weight Disord. 2004;9(4):249–257. | ||
Hoekstra R, Bartels M, Hudziak JJ, Van Beijsterveldt TCEM, Boomsma D. Genetic and environmental covariation between autistic traits and behavioral problems. Twin Res Hum Genet. 2007;10(6):853–860. | ||
Narzisi A, Calderoni S, Maestro S, Calugi S, Mottes E, Muratori F. Child behavior check list 11/2–5 as a tool to identify toddlers with autism spectrum disorders: a case control study. Res Dev Disabil. 2012;34(4): 1179–1189. | ||
Schmidt U, Evans K, Tiller J, Treasure J. Puberty, sexual milestones and abuse: how are they related in eating disorder patients? Psychol Med. 1995;25(2):413–417. | ||
Hartmann A, Zeeck A, Barrett MS. Interpersonal problems in eating disorders. Int J Eat Disord. 2010;43(7):619–627. | ||
Bühren K, Schwarte R, Fluck F, et al. Comorbid psychiatric disorders in female adolescents with first-onset anorexia nervosa. Eur Eat Disord Rev. 2014;22(1):39–44. | ||
Godart NT, Flament MF, Perdereau F, Jeammet P. Comorbidity between eating disorders and anxiety disorders: a review. Int J Eat Disord. 2002;32(3):253–270. | ||
Monsell S. Task switching. Trends Cogn Sci. 2003;7(3):134–140. | ||
Serpell L, Livingstone A, Neiderman M, Lask B. Anorexia nervosa: obsessive-compulsive disorder, obsessive-compulsive personality disorder, or neither? Clin Psychol Rev. 2002;22(5):647–669. | ||
Anderluh MB, Tchanturia K, Rabe-Hesketh S, Treasure J. Childhood obsessive-compulsive personality traits in adult women with eating disorders: defining a broader eating disorder phenotype. Am J Psychiatry. 2003;160(2):242–247. | ||
Tchanturia K, Morris RG, Anderluh MB, Collier DA, Nikolaou V, Treasure J. Set shifting in anorexia nervosa: an examination before and after weight gain, in full recovery and relationship to childhood and adult OCPD traits. J Psychiatr Res. 2004;38(5):545–552. | ||
Crane AM, Roberts ME, Treasure J. Are obsessive-compulsive personality traits associated with a poor outcome in anorexia nervosa? A systematic review of randomized controlled trials and naturalistic outcome studies. Int J Eat Disord. 2007;40(7):581–588. | ||
Wagner A, Barbarich-Marsteller NC, Frank GK, et al. Personality traits after recovery from eating disorders: do subtypes differ? Int J Eat Disord. 2006;39(4):276–284. | ||
Giel KE, Wittorf A, Wolkenstein L, et al. Is impaired set-shifting a feature of “pure” anorexia nervosa? Investigating the role of depression in set-shifting ability in anorexia nervosa and unipolar depression. Psychiatry Res. 2012;200(2–3):538–543. | ||
Happé F, Frith U. The neuropsychology of autism. Brain. 1996;119(pt 4):1377–1400. | ||
Happé F, Frith U. The weak central coherence account: detail-focused cognitive style in autism spectrum disorders. J Autism Dev Disord. 2006;36(1):5–25. | ||
Wing L, Gould J. Severe impairments of social interaction and associated abnormalities in children: epidemiology and classification. J Autism Child Schizophr. 1979;9(1):11–29. | ||
Lopez C, Tchanturia K, Stahl D, Treasure J. Central coherence in eating disorders: a systematic review. Psychol Med. 2008;38(10):1393–1404. | ||
Southgate L, Tchanturia K, Treasure J. Information processing bias in anorexia nervosa. Psychiatry Res. 2008;160(2):221–227. | ||
Lindner SE, Fichter MM, Quadflieg N. Central coherence in full recovery of anorexia nervosa. Eur Eat Disord Rev. 2013;21(2):115–120. | ||
Tenconi E, Santonastaso P, Degortes D, et al. Set-shifting abilities, central coherence, and handedness in anorexia nervosa patients, their unaffected siblings and healthy controls: exploring putative endophenotypes. World J Biol Psychiatry. 2010;11(6):813–823. | ||
Pepin G, Stagnitti K. Come play with me: an argument to link autism spectrum disorders and anorexia nervosa through early childhood pretend play. Eat Disord. 2012;20(3):254–259. | ||
Zucker NL, Losh M, Bulik CM, LaBar KS, Piven J, Pelphrey KA. Anorexia nervosa and autism spectrum disorders: guided investigation of social cognitive endophenotypes. Psychol Bull. 2007;133(6):976–1006. | ||
Gillberg IC, Billstedt E, Wentz E, Anckarsäter H, Rastam M, Gillberg C. Attention, executive functions, and mentalizing in anorexia nervosa eighteen years after onset of eating disorder. J Clin Exp Neuropsychol. 2010;32(4):358–365. | ||
Smink FR, van Hoeken D, Hoek HW. Epidemiology, course, and outcome of eating disorders. Curr Opin Psychiatry. 2013;26(6):543–548. | ||
Pollice C, Kaye WH, Greeno CG, Weltzin TE. Relationship of depression, anxiety, and obsessionality to state of illness in anorexia nervosa. Int J Eat Disord. 1997;21(4):367–376. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.