Back to Journals » Infection and Drug Resistance » Volume 12
The first report of a novel IncHI1B blaSIM-1-carrying megaplasmid pSIM-1-BJ01 from a clinical Klebsiella pneumoniae isolate
Authors Lü Y , Zhao S, Liang H, Zhang W, Liu J, Hu H
Received 15 April 2019
Accepted for publication 3 July 2019
Published 19 July 2019 Volume 2019:12 Pages 2103—2112
DOI https://doi.org/10.2147/IDR.S212333
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Yang Lü,1 Shulong Zhao,2 Hui Liang,3 Wei Zhang,3 Jia Liu,3 Hongyan Hu3
1State Key Laboratory of Mycology, Institute of Microbiology, Chinese Academy of Sciences, Beijing, People’s Republic of China; 2Department of Laboratory Medicine, The Affiliated Hospital of Xuzhou Medical University, Xuzhou, Jiangsu, People’s Republic of China; 3Department of Laboratory Medicine, The Third Medical Center, Chinese PLA General Hospital, Beijing, People’s Republic of China
Background: A rare member of metallo-β-lactamases genes, blaSIM-1, carried by a 316-kb plasmid designated pSIM-1-BJ01 was isolated from a clinical cephalosporins- and carbapenem-resistant Klebsiella pneumoniae 13624. This is the first sequence report of a transferable blaSIM-1-carrying conjugative plasmid isolated from K. pneumoniae.
Purpose: The sequence analysis of pSIM-1-BJ01 will help us to identify genes responsible for conjugation, plasmid maintenance and drug resistance, to understand the evolution and control the dissemination of resistance plasmids.
Patients and methods: K. pneumoniae 13624 was isolated from the urine specimen of a patient. Bacterial genomic DNA was sequenced with PacBio RSII platform.
Results: Most of the pSIM-1-BJ01 backbone matches that of pRJA166a, which was isolated from a clinical hypervirulent K. pneumoniae ST23 strain at Shanghai, China, recently. The highly homologous backbones between the two plasmids imply the close relationship of evolution. Two different multidrug-resistant regions both carrying the class 1 integrons with different resistance genes have been assembled into the pSIM-1-BJ01. Besides, the other two resistance plasmids, pKP13624-1 carrying blaTEM-1 and blaCTX-M-15 and pKP13624-2 carrying blaCTX-M-14 and blaLAP-2 were also identified.
Conclusion: The emergence of the blaSIM-1-carrying IncHI1B pSIM-1-BJ01 suggests the spread of blaSIM among Enterobacteriaceae is possible. We should pay more attention to supervise and control the dissemination of hypervirulent carbapenem-resistant K. pneumonia in public hospitals.
Keywords: SIM-1, carbapenemase, Klebsiella pneumoniae, multidrug-resistant, China
Introduction
SIM, a rare member of metallo-β-lactamases (MBLs), belongs to Class B1 MBLS. It can hydrolyze a broad array of β-lactams, including penicillin, narrow to extended spectrum cephalosporins, and carbapenems, but not monobactams. The SIM-1 protein exhibits 64–69% identity with the IMP-type MBLs, which are its closest relatives. The first reported bacteria carrying blaSIM-1 were Acinetobacter baumannii isolated from Korea in 2005.1 In blaSIM-1-harboring Acinetobacter isolates collected from 2003 to 2008 in Korea, the ca. 280-kb plasmid carrying the blaSIM-1 gene was the major transfer mechanism between different Acinetobacter spp., but these plasmids were untypable.2 In 2011, an A. baylyi strain with a 360-kb plasmid carrying both blaSIM-1 and blaOXA-23 was isolated from a Chinese patient in Ningbo, China, who had no history of visiting Korea, but the sequence of the 360-kb plasmid was not determined.3 In 2012, a Pseudomonas aeruginosa strain with a 282-kb blaSIM-2-harbouring plasmid pHN39-SIM (Genbank: KU254577) was isolated from a Chinese patient in Zhengzhou, China; the 282-kb blaSIM-2-harbouring plasmid was sequenced, showing the SIM-2 protein differs from SIM-1 due to only a single amino acid substitution Gly198Asp.4 Overall, the detection of blaSIM is rare and mainly reported by Korea and China, mostly confined to Acinetobacter spp. and P. aeruginosa.
In Acinetobacter spp., the reported blaSIM-1 genes, derived from chromosome or plasmids, were always carried on a gene cassette inserted into a class 1 integron with three additional resistance genes (arr-3, catB3 and aadA1);1 the blaSIM-2 gene in the pHN39-SIM from P. aeruginosa was also inserted into a class 1 integron but in a different cassette array(ereA1, catB3q, gcu161, arr-3, aadA1a).4 Except for the pHN39-SIM, none of these plasmids is fully sequenced.
Here, we present a 316-kb plasmid designated pSIM-1-BJ01 carrying blaSIM-1 isolated from a clinical carbapenem-resistant Klebsiella pneumoniae 13624 strain harboring three resistance plasmids in Beijing, China. To the best of our knowledge, this is the first sequence report of a transferable plasmid with blaSIM-1 isolated from K. pneumoniae, the sequence analysis of pSIM-1-BJ01 reveals a mosaic-like structure and identifies genes responsible for conjugation, plasmid maintenance and drug resistance.
Materials and methods
Bacterial isolates and identification
K.pneumoniae 13624 was isolated in 2013 from the urine specimen of a 79-year-old female patient with acute onset of cerebral infarction and type 2 diabetes mellitus in a tertiary hospital in Beijing, China. The urinary tract infection was cured by levofloxacin after 2 weeks. The bacterial species identification was performed using BioMérieux Vitek 2, Bruker MALDI Biotyper and 16S rRNA gene sequencing.
Antimicrobial resistance
Susceptibility tests with the Vitek 2(bioMérieux, France) system were performed using AST-GN cards according to the manufacturer’s instructions, and the antimicrobial susceptibility results were evaluated according to 2014 CLSI guidelines. Metallo-β-lactamase activity was detected by Modified Hodge test. The carbapenemase genes (blaGES, blaKPC, blaSME, blaIMI, blaBIC, blaIMP, blaVIM, blaNDM, blaTMB, blaFIM, blaSPM, blaDIM, blaGIM, blaSIM, blaAIM, blaSMB, blaOXA) were screened for by PCR with specific primers described by Chen.5
Plasmid transfer
Conjugal transfer experiment was performed with Escherichia coli J53 Azir as the recipient strain. Overnight cultures of the bacteria was diluted to 1.5×108 cells/mL. Donor and recipient cells were mixed at 1:10 donor-to-recipient ratio, after 18 h of incubation of donor-recipient mixtures on blood plates at 35 °C, cells were washed by normal saline solution and diluted to be cultured selectively on MacConkey agar plates supplemented with sodium azide (100 μg/mL) and imipenem (1 μg/mL) for 24 h. Transconjugants were confirmed by PCR amplifying blaSIM with primers (SIM-F: 5ʹ TACAAGGGATTCGGCATCG 3ʹ; SIM-R: 5ʹ TAATGGCCTGTTCCCATGTG 3ʹ).5
Whole genome sequencing and data analysis
Bacterial genomic DNA was sequenced with PacBio RSII platform. Raw Pacbio reads were de novo assembled by SMRT Portal program with default settings. The coverage of the sequences was 177. The genes were predicted with GeneMarkS and further annotated by BLASTP and BlASTN against UniPort and NR databases. The complete sequence of pSIM-1-BJ01, pKP13624-1, and pKP13624-2 was submitted to GenBank under accession number MH681289, MK158080 and MK158081, respectively.
Ethical approval
The urine specimen was part of the routine hospital laboratory procedure. The use of human specimens and all related experimental protocols was approved by the Committee on Human Research of the indicated institutions, and was carried out in accordance with the approved guidelines.
Results
Antibiotic resistance of the blaSIM-1-carrying K. pneumoniae
The isolated strain was resistant to ampicillin, cephalosporin, and carbapenem, but sensitive to ciprofloxacin, levofloxacin, and colistin; see Table S1. We screened for the carbapenemase genes by PCR, only the blaSIM-1 gene was amplified.
In the conjugation experiment, we also detected the blaSIM-1 by PCR in the recipient E.coli J53 Azir, suggesting that a transferable plasmid (designated pSIM-1-BJ01) might exist in the clinically isolated strain. The transferable pSIM-1-BJ01 might give the recipient E.coli J53 Azir the carbapenem resistance, as shown in Table S1.
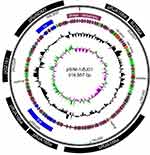
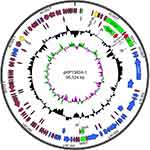
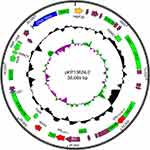
We sequenced the whole genome of the isolated strain and found that there were three drug-resistant plasmids in it. They are respectively the blaSIM-1-harboring pSIM-1-BJ01 (Figure S1), pKP13624-1 (Figure S2) carrying resistance genes blaCTX-M-15 and blaTEM-1, and pKP13624-2 (Figure S3) carrying the extended-spectum β-lactamase (ESBL) gene blaCTX-M-14 and blaLAP-2.
Overview and comparative analysis of pSIM-1-BJ01
The pSIM-1-BJ01 is a 316,557 bp circular plasmid containing 342 putative ORFs (153 hypothetical proteins), the average GC content is 46%. The pSIM-1-BJ01 possesses two replicons (RepFIB and RepHI1B), it is classified to IncHI1B type according to PlasmidFinder (https://cge.cbs.dtu.dk/services/PlasmidFinder-1.0/). The RepHI1B (encoded from 230,320 bp to 231,204 bp) identified in pSIM-1-BJ01 shared 87% amino acid identity with the RepHI1B (Protein ID AFB82850.1) encoded by the IncHI1B type pNDM-MAR (Genbank: JN420336) isolated from a clinical K.pneumoniae ST15.6
The IncH family plasmids can produce the H-pilus, a long and flexible conjugation pilus similar to the F-pilus. Here, the pSIM-1-BJ01 possess two transfer regions, carrying the H-pilus assembly and transfer related protein genes (trhL, trhE, traK, traB, dsbC, traV, traC, trhZ, trhO, htdA, htdK, traW, traU, traN, traD, traI, traF, traH and traG). Most of these proteins have high similarity with those encoded by the IncHI1B plasmid pNDM-MAR, see Table 1; strongly suggest that the pSIM-1-BJ01 can be classified into the IncHI1B type.
 |
Table 1 The homology analysis for pilus assembly and transfer related proteins |
The backbone of pSIM-1-BJ01(18,028–35,486 bp; 36,998–51,411 bp; 107,310–168,501 bp; 173,574–196,369 bp; 197,420–222,610 bp) highly matched that of the plasmid RJA166a (Genbank: CP019048.1) isolated from a clinical hypervirulent K. pneumoniae ST23 strain at Shanghai, China, recently.7 In addition, a small part of the backbone (233,359–254,569 bp) highly matched that of the pOZ181 (Genbank: CP016764.1) isolated from Citrobacter freundii strain B38 at Guangzhou, China,8 and the other small part of the backbone (265,886–305,806 bp) highly matched that of the pRpNDM1(Genbank: JX515588.1) isolated from Raoultella planticola strain RpNDM1 at Gansu, China,9 as shown in the outermost circle of Figure S1. Besides, the pSIM-1-BJ01 also contains two class 1 integron carrying multidrug-resistant genes and one unique region related to phage invasion and assembly, see Figure S1.
A mosaic clinical class 1 integron harboring blaSIM-1
The region from 34,290 bp to 84,861 bp was a mosaic structure of a novel clinical class 1 integrons,10 see Figure 1A. As reported in Acinetobacter baylyi (Genbank: JF731030) or Acinetobacter baumannii (Genbank: AY887066), the blaSIM-1 was embedded in the conserved intI1-blaSIM-1-arr3-catB3-aadA1 gene cassette array.1 In the pSIM-1-BJ01, the blaSIM-1 is also embedded in the conserved cassette array, but the aadA1 was replaced by aadA17. AadA17, displaying two amino acids substitution (Met126Thr and Thr136Ile), has 93% similarity to the AadA1, so the aadA17 cassette seems to be hybrid of aadA1 and aadA2 cassettes.
Another three resistance genes dfrA3b, aph(3ʹ)-Ia and qnrS1 are closely adjacent to the conserved intI1-blaSIM-1-arr3-catB3-aadA17-qacEΔ1-sul1 gene arrays. The dfrA3b is flanked by ISCR1 at its 5ʹ-end and IS26 at its 3ʹ-end. The aph(3ʹ)-Ia is flanked by directly oriented IS26 at both ends, which is most likely Tn6023(Genbank: GU562437). Tn6023 is a 2.6 kb composite transposon, found in plasmid pSRC125 from a multiply antibiotic-resistant Salmonella enterica serovar Typhimurium isolate of bovine origin, it comprised of the aph(3ʹ)-Ia gene flanked by inversely oriented IS26 at both ends.11 The qnrS1 is flanked by IS26 at its 5ʹ-end and ISKpn19 at its 3ʹ-end; as shown in Figure 1A. These suggest the region is variable and might capture the different resistance genes following exposure to a sophisticated antibiotic selective environment.
The reported blaSIM-2-harboring pHN39-SIM carries intI1-blaSIM-2-ereA1-catB3q-arr3-aadA1a-qacEΔ1-sul1 gene array and two different mercury resistance gene arrays a and b, respectively harbored in Tn5046 and Tn512; the CatB3q is a derivative of CatB3.4 The mercury resistance gene array of pSIM-1-BJ01 (from 50,597 to 54,673 bp) only has 78% homology to the mer gene array b of pHN39-SIM, but has 99.3% identity to that of a truncated mercury resistance transposon in pMER610(GenBank: Y08993.1), as shown in Figure 1A. The mer resistance transposon in pMER610 was firstly found in England in a large Inc II-2 plasmid in a wide range of strains belonging of Pseudomonas, Acinetobacter, Alcaligenes and Enterobacter.12 In pSIM-1-BJ01, the left-flank insertion site (5ʹ-AATAAAGCAC-3ʹ) of IS4321R is located at the 5ʹ-end of the mer gene array, the 3ʹ-end of the mer array is flanked by IS4321R, indicating the mer gene array in pSIM-1-BJ01 might origin from the reported mer resistance transposon in pMER610 and be recombined into the pSIM-1-BJ01 by IS4321.
The tellurium resistance gene array(from 36,998 to 51,411 bp) in the pSIM-1-BJ01 is 99% identical to that of pRJA166a (from 78,751 bp to 93,171 bp), as shown in Figure 1A. The pRJA166a was also isolated from a K. pneumoniae strain, which caused a fatal outbreak of hypervirulent carbapenem-resistant K. pneumoniae (hv CRKP) in a Chinese hospital.7 In pRJA166a, the terF was interrupted by the insertion of two transposase genes.
The relB between the tel and mer arrays, belongs to type II antitoxin systems which are expected to increase the longevity of plasmids in bacterial populations even in the absence of selection pressure due to antibiotics and metals;13 besides, a DNA starvation and stationary phase protection protein (ferritin) is also located between the mer array and the drug-resistant gene array in the clinical class 1 integron, see Figure 1A.
Here, the blaSIM-1-carrying clinical class 1 integron, carrying the mer array, the tel array and different drug resistance genes, suggests the synergistic gene combination under clinical selection.
A Tn1696-associated transposon harboring armA and other resistance genes
The other class 1 integron in the pSIM-1-BJ01 has the dfrA1-gcu37-aadA5-qacEΔ1-sul1 gene arrays, as shown in Figure 1B. The 16s rRNA methylase gene armA has been observed in a number of plasmids,14 among these plasmids, two direct repeats of IS26 and the resistance gene backbone consisting of qacEΔ1-sul1-ISCR1-tnpU-armA-tnpD-msr(E)-mph(E) are well conserved, here, the armA surrounding region is consistent with the reported conserved backbone. Compared with the Tn1548-associated armA backbone (GenBank: JN225877),14 as shown in Figure 1B, the sequence analysis showed that the IS26 at the upstream of the conserved backbone was replaced by IS5075 in the pSIM-1-BJ01; IS5075 is normally found interrupting the terminal inverted repeats of Tn21-family transposeons, here it is to be Tn1696 (Genbank: U12338).15
The region (from 258,498 to 265,885 bp) carrying resistance genes catA2 and tet(D) with three direct repeats of IS26 is also conserved and consistent with that of the blaIMP-4-carrying pMS7884A (Genbank: CP022533.1, from 143,213 to 135,889 bp) isolated from an Enterobacter cloacae, see Figure 1B. The antitoxin gene higA, toxin gene higB as well as the DNA protection protein (ferritin) gene, flanked by IS26 and ISKpn26, are also captured here to help bacteria to tolerate various antibiotic treatments.16
Overview of pKP13624-1
The pKP13624-1 is a 95,324 bp circular plasmid containing 124 putative ORFs (54 hypothetical proteins), and the average GC content is 52%, as shown in Figure S2. The pKP13624-1 carrying blaTEM-1 and blaCTX-M-15 belongs to the IncFⅡK type according to the PubMLST website and it is almost identical to the pL22-5 (Genbank: CP031262.1) isolated from a hypermucoviscous strain Klebsiella quasi ST367 and the pKF3-94 (Genbank: FJ876826) isolated from a clinical drug-resistant strain K. pneumoniae KF3.17 The blaCTX-M-15 was flanked by ΔTn2 at the downstream and ISEcp1(interrupted by a truncated IS1) at the upstream, along with the blaTEM-1 carried by ΔTn2 transposon, which increasing the mobility of the two resistance genes.
Overview of pKP13624-2
The pKP13624-2 is a 38,669 bp circular plasmid containing 29 putative ORFs (8 hypothetical proteins), and the average GC content is 49%, as shown in Figure S3. The pKP13624-2 belongs to IncFІA type and has never been reported before. The region (from 12,339 to 27,055 bp) containing three resistance genes blaCTX-M-14, blaLAP-2, and qnrS1 flanked by ISEcp1 and ΔISKpn19, is 99% identical to that of pE66An (Genbank: HF545433.1, from 79,443 to 64,716 bp) isolated from E.coli in Vietnam. The blaCTX-M-14 is flanked by ISEcp1 at its 5ʹ-end and IS903B at its 3ʹ-end, while the blaLAP-2 is flanked by IS26 at its 5ʹ-end and ΔIS2 at its 3ʹ-end and the qnrS1 is flanked by ΔIS2 at the upstream and ΔISKpn19 at the downstream, as shown in Figure S3.
Discussion
The infection rate of carbapenem-resistant K. pneumoniae (CRKP) has increased substantially in the past 10 years, even a fatal outbreak of hypervirulent carbapenem-resistant K. pneumoniae (hv CRKP) in a Chinese hospital.18–20 Here, we presented a clinically isolated K. pneumoniae with three resistance plasmids. Among them, the pSIM-1-BJ01 carried the rare carbapenemase gene blaSIM-1. This is the first report of the blaSIM-1-carrrying megaplasmid isolated from K. pneumoniae. Most of the pSIM-1-BJ01 backbone matches that of pRJA166a, which was isolated from a clinical hypervirulent K. pneumoniae ST23 strain at Shanghai, China, recently.7 The highly homologous backbones between the two plasmids imply the close relationship of evolution. Two different multidrug-resistant regions both carrying the class 1 integrons with different resistance genes have been assembled into the pSIM-1-BJ01 under the continuous selection exerted by human antibiotic use, made the novel pSIM-1-BJ01 become a significant environmental contaminant.21 Now there was one case report about the blaSIM-1 carrying E.coli from India.22 Except for the pSIM-1-BJ01, the pKP13624-2 carrying blaCTX-M-14 and blaLAP-2 has never been reported before. We should pay more attention to supervise and control the dissemination of carbapenem-resistant K. pneumonia in public hospitals.
Acknowledgment
This work was supported by the Research Fund of the General Hospital of Chinese People’s Armed Police Forces (WZ2014013).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Lee K, Yum JH, Yong D, et al. Novel acquired metallo-beta-lactamase gene, blaSIM-1, in a class 1 integron from Acinetobacter baumannii clinical isolates from Korea. Antimicrob Agents Chemother. 2005;49:4485–4491. doi:10.1128/AAC.49.11.4485-4491.2005
2. Kim Y, Roh KH, Lee Y, et al. Clonal change of blaSIM-1-carrying Acinetobacter spp. from 2003 to 2008 in the hospital where it was initially discovered. Microb Drug Resist. 2013;19:37–41. doi:10.1089/mdr.2012.0038
3. Zhou Z, Du X, Wang L, et al. Clinical carbapenem-resistant Acinetobacter baylyi strain coharboring blaSIM-1 and blaOXA-23 from China. Antimicrob Agents Chemother. 2011;55:5347–5349. doi:10.1128/AAC.00425-11
4. Sun F, Zhou D, Wang Q, et al. The first report of detecting the blaSIM-2 gene and determining the complete sequence of the SIM-encoding plasmid. Clin Microbiol Infect. 2016;22:347–351. doi:10.1016/j.cmi.2015.12.001
5. Chen Z, Li H, Feng J, et al. NDM-1 encoded by a pNDM-BJ01-like plasmid p3SP-NDM in clinical Enterobacter aerogenes. Front Microbiol. 2015;6:294. doi:10.3389/fmicb.2015.00294
6. Villa L, Poirel L, Nordmann P, Carta C, Carattoli A. Complete sequencing of an IncH plasmid carrying the blaNDM-1, blaCTX-M-15 and qnrB1 genes. J Antimicrob Chemother. 2012;67:1645–1650. doi:10.1093/jac/dks114
7. Xie Y, Tian L, Li G, et al. Emergence of the third-generation cephalosporin-resistant hypervirulent Klebsiella pneumoniae due to the acquisition of a self-transferable blaDHA-1-carrying plasmid by an ST23 strain. Virulence. 2018;9:838–844. doi:10.1080/21505594.2018.1456229
8. Xiong J, Deraspe M, Iqbal N, et al. Genome and plasmid analysis of blaIMP-4-carrying Citrobacter freundii B38. Antimicrob Agents Chemother. 2016;60:6719–6725. doi:10.1128/AAC.00588-16
9. Li J, Lan R, Xiong Y, et al. Sequential isolation in a patient of Raoultella planticola and Escherichia coli bearing a novel ISCR1 element carrying blaNDM-1. PLoS One. 2014;9:e89893. doi:10.1371/journal.pone.0089893
10. Gillings MR. Class 1 integrons as invasive species. Curr Opin Microbiol. 2017;38:10–15. doi:10.1016/j.mib.2017.03.002
11. Cain AK, Hall RM. Transposon Tn5393e carrying the aphA1-containing transposon Tn6023 upstream of strAB does not confer resistance to streptomycin. Microb Drug Resist. 2011;17:389–394. doi:10.1089/mdr.2011.0037
12. Yurieva O, Kholodii G, Minakhin L, et al. Intercontinental spread of promiscuous mercury-resistance transposons in environmental bacteria. Mol Microbiol. 1997;24:321–329.
13. Pal C, Bengtsson-Palme J, Kristiansson E, Larsson DGJ. Co-occurrence of resistance genes to antibiotics, biocides and metals reveals novel insights into their co-selection potential. BMC Genomics. 2015;16:964. doi:10.1186/s12864-015-2153-5
14. Du XD, Li DX, Hu GZ, et al. Tn1548-associated armA is co-located with qnrB2, aac(6ʹ)-Ib-cr and blaCTX-M-3 on an IncFII plasmid in a Salmonella enterica subsp. enterica serovar Paratyphi B strain isolated from chickens in China. J Antimicrob Chemother. 2012;67:246–248. doi:10.1093/jac/dkr407
15. Wohlleben W, Arnold W, Bissonnette L, et al. On the evolution of Tn21-like multiresistance transposons: sequence analysis of the gene(aacC1) for gentamicin acetyltransferase-3-I(AAC(3)-I), another member of the Tn21-based expression cassette. Mol Gen Genet. 1989;217:202–208.
16. Zhai Y, He Z, Kang Y, et al. Complete nucleotide sequence of pH11, an IncHI2 plasmid conferring multi-antibiotic resistance and multi-heavy metal resistance genes in a clinical Klebsiella pneumoniae isolate. Plasmid. 2016;86:26–31. doi:10.1016/j.plasmid.2016.04.001
17. Zhao F, Bai J, Wu J, et al. Sequencing and genetic variation of multidrug resistance plasmids in Klebsiella pneumoniae. PLoS One. 2010;5:e10141. doi:10.1371/journal.pone.0010141
18. Gu D, Dong N, Zheng Z, et al. A fatal outbreak of ST11 carbapenem-resistant hypervirulent Klebsiella pneumoniae in a Chinese hospital: a molecular epidemiological study. Lancet Infect Dis. 2018;18:37–46. doi:10.1016/S1473-3099(17)30489-9
19. Wong MHY, Shum H-P, Chen JHK, et al. Emergence of carbapenem-resistant hypervirulent Klebsiella pneumoniae. Lancet Infect Dis. 2018;18:24. doi:10.1016/S1473-3099(17)30629-1
20. Yao H, Qin S, Chen S, Shen J, Du X-D. Emergence of carbapenem-resistant hypervirulent Klebsiella pneumoniae. Lancet Infect Dis. 2018;18:25. doi:10.1016/S1473-3099(17)30628-X
21. Gillings MR. Integrons: past, present, and future. Microbiol Mol Biol Rev. 2014;78:257–277. doi:10.1128/MMBR.00056-13
22. Gopalakrishnan S, Kamalanathan A, Rajan S, et al. Emergence of armA and rmtB genes among VIM, NDM, and IMP metallo-beta-lactamase-producing multidrug-resistant Gram-negative pathogens. Acta Microbiol Immunol Hung. 2018;65:107–118. doi:10.1556/030.64.2017.027
Supplementary materials
 |
Table S1 Antimicrobial drug susceptibility profiles |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.