Back to Journals » Diabetes, Metabolic Syndrome and Obesity » Volume 13
The Effects of Puerarin on Autophagy Through Regulating of the PERK/eIF2α/ATF4 Signaling Pathway Influences Renal Function in Diabetic Nephropathy
Authors Xu X, Chen B, Huang Q, Wu Y, Liang T
Received 1 April 2020
Accepted for publication 2 July 2020
Published 20 July 2020 Volume 2020:13 Pages 2583—2592
DOI https://doi.org/10.2147/DMSO.S256457
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Prof. Dr. Juei-Tang Cheng
Xiaohui Xu,1,2,* Biao Chen,3,* Qichun Huang,1 Yani Wu,4 Tao Liang5
1Department of Pharmacy, Affiliated Tumor Hospital of Guangxi Medical University, Nanning, Guangxi 530021, People’s Republic of China; 2Institute of Cancer Prevention and Treatment of Guangxi Zhuang Autonomous Region, Nanning, Guangxi 530021, People’s Republic of China; 3The First Nanning People’s Hospital, Nanning, Guangxi 530022, People’s Republic of China; 4Pharmaceutical College, Guangxi Medical University, Nanning, Guangxi 530021, People’s Republic of China; 5College of Stomatology of Guangxi Medical University, Nanning, Guangxi 530021, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xiaohui Xu
Department of Pharmacy, Affiliated Tumor Hospital of Guangxi Medical University, No. 71, Hedi Road, Nanning 530021, People’s Republic of China
Tel/ Fax +86 771-5778582
Email [email protected]
Tao Liang
College of Stomatology of Guangxi Medical University, No. 10, Shuangyong Road, Nanning 530021, People’s Republic of China
Tel/ Fax +86 771-5358635
Email [email protected]
Background and Purpose: Autophagy is the main protective mechanism against aging in podocytes, which are terminally differentiated cells that have a very limited capacity for mitosis and self-renewal. Here, a streptozotocin-induced DN C57BL/6 mouse model was used to investigate the effects of puerarin on the modulation of autophagy under conditions associated with endoplasmic reticulum stress (ERS). In addition, this study aimed to identify the potential underlying molecular mechanisms.
Methods and Results: DN C57BL/6 mouse model was induced by streptozotocin (150 mg/kg) injection. The mice were administered rapamycin and puerarin, respectively, daily for up to 8 weeks. After the serum and kidney samples were collected, the fasting blood glucose (FBG), parameters of renal function, histomorphology, and the podocyte functional proteins were analyzed. Moreover, the autophagy markers and the expressions of PERK/ATF4 pathway were studied in kidney. Results found that the FBG level in DN mice was significantly higher than in normal mice. Compared with DN model mice, puerarin-treated mice showed an increased expression of podocyte functional proteins, including nephrin, podocin, and podocalyxin. Furthermore, the pathology and structure alterations were improved by treatment with rapamycin and puerarin compared with the DN control. The results indicated an elevated level of autophagy in rapamycin and puerarin groups compared with the DN model, as demonstrated by the upregulated expression of autophagy markers Beclin-1, LC3II, and Atg5, and downregulated p62 expression. In addition, the levels of PERK, eIF2α, and ATF4 were reduced in the DN model, which was partially, but significantly, prevented by rapamycin and puerarin.
Conclusion: This study emphasizes the renal-protective effects of puerarin in DN mice, particularly in the modulation of autophagy under ERS conditions, which may be associated with activation of the PERK/eIF2α/ATF4 signaling pathway. Therefore, PERK may be a potential target for DN treatment.
Keywords: diabetic nephropathy, autophagy, PERK, puerarin
Introduction
Diabetes mellitus (DM) is a globalized chronic endocrine system disease that significantly threatens the safety and quality of human life. Diabetic nephropathy (DN) is one of the major causes of end-stage renal disease and is a characteristic of glomerular hypertrophy, which is the thickening of the basement membrane and accumulation of extracellular matrix (ECM).1 Although several approaches, such as glucose control, lipid modulation, anti-oxidation, and angiotensin-converting enzyme inhibitors, are used clinically to treat DN patients, it is still difficult to prevent the continuous decline of renal function. Therefore, it is important to explore the pathogenesis of DN and develop new therapeutic drug targets.
Autophagy is the self-phagocytosis of cells, which is an important degradation mechanism that eliminates damaged organelles and harmful substances in the lysosome.2 A study found that the progression of DN was accompanied by decreased levels of autophagy in podocytes. In high glucose treated podocytes and DN animal models, the activation of autophagy contributed to the prevention of DN, whereas disruption of autophagy promoted glomerulosclerosis and tubulointerstitial fibrosis.3 Therefore, autophagy may play a critical role in DN. Protein kinase R-like endoplasmic reticulum kinase (PERK) is a key protease involved in the endoplasmic reticulum stress (ERS) response. Specifically, the PERK signaling pathway promotes temporary termination of protein synthesis and improves the physiological state of cells during ERS. Furthermore, it modulates the ERS mediated-CHOP pathway to prevent apoptosis.4–6
Puerarin (PR), 8-(β-D-Glucopyranosyl)-4ʹ,7-dihydroxyisoflavone, isolated from Puerarin lobata (Wild.) Ohwi, has been reported to exhibit positive effects on coronary heart disease and diabetic peripheral neuropathy.7,8 Recently, studies reported that puerarin has benefit effects on cancer cell apoptosis, cerebral ischemia reperfusion injury, neurological deficits and DN treatment.9–11 In our previous studies, puerarin not only attenuates the oxidative stress in DN, but also meliorates the glucolipid metabolism.12 Additionally, renal function and podocyte fusion were improved by puerarin intervention.13 Most importantly, autophagy activity was enhanced by the presence of puerarin through the activation of PERK activity. Therefore, these favorable changes induced by puerarin may be associated with modulation of the PERK/eIF2α/ATF4 signaling pathway.
To further shed light on these processes, we explored the effects of puerarin on the modulation of autophagy under conditions associated with ERS and investigated the potential underlying molecular mechanisms.
Materials and Methods
Experimental Animals
Eight-week-old male C57BL/6 mice weighing 18–22 g provided by Nanjing Biomedical Research Institute at the Nanjing University (Nanjing, China) were housed under temperature-controlled laboratory conditions of 22–25 °C with a 12 h light-dark cycle and had unlimited access to standard rodent chow and water provided by the Laboratory Animal Centre. All the animal protocols followed the guidelines of the Guiding Opinions for the care and use of laboratory animals of Guangxi Medical University and were approved by the Ethics Committee of Affiliated Tumor Hospital of Guangxi Medical University (approval number: LW2020034).
Materials
Puerarin (purity>99%) was provided by the Department of Pharmaceutical Chemistry at Guangxi Medical University (Nanning, China), and the extraction procedures have been previously published [10]. Streptozotocin (STZ) was purchased from Sigma Co., Ltd. Missouri, USA. Fasting blood glucose (FBG) were detected using the Roche ACCU-CHEK® Performa Kit (Strip lot: 470664, Switzerland), and blood levels were measured with an automatic biochemical analyzer (Hitachi Model 7100 Automatic Analyzer). Other materials are outlined in the following sections.
Experimental Design13
The 8-week-old male C57BL/6 mice underwent adaptive feeding for one week. Subsequently, the mice were tail vein injected with 150 mg/kg body weight of streptozotocin after 12 hours of fasting. Seventy-two hours later, DN mice were established with an FBG level ≥11.1 mmol/L. The experimental groups were as follows: the normal control group consisted of healthy C57BL/6 mice (n=10) with unlimited access to water, the DN control group consisted of DN C57BL/6 mice (n=10) with unlimited access to water, the rapamycin control group consisted of DN C57BL/6 mice (n=10) who were given an intraperitoneal injection of 4 mg/kg/d rapamycin, and the puerarin groups consisted of DN C57BL/6 mice who were given an oral administration of 40 (n=10) and 80 (n=10) mg/kg/d puerarin. The treatments were administered daily for up to 8 weeks.
Biochemical Measurements
Blood samples for FBG detection were collected from the tail veins. Serum samples were obtained from whole blood by centrifugation at 1300 × g for 10 min. The FBG levels in tail vein blood were measured on weeks 4 and 8 using the Roche ACCU-CHEK® Performa Kit (Strip lot: 470664, Switzerland). Blood urea nitrogen (BUN), serum creatinine (Scr), urine creatinine (Ucr), and 24-hour urine protein levels were measured by an automatic biochemical analyzer (Hitachi Model 7100 Automatic Analyzer).
Histopathological Examination
Kidney samples were harvested and fixed in 10% formaldehyde. After dehydration, samples were embedded in paraffin. The samples were sectioned into 4 μm slices for hematoxylin-eosin (HE) staining. Pathological images were obtained using a light microscope (LEICA DM 2000 LED).
Transmission Electron Microscopy Evaluation
Electron microscopy studies were performed on kidney tissues, as previously described.12 Kidney samples were removed, cut into small pieces (about 2 mm × 2 mm × 2 mm), and immediately fixed in 2.5% pre-cooled glutaraldehyde for 2 hours at 0°C. After rinsing in buffer, kidney tissues were post-fixed in 1% osmium tetroxide for 2 hours at room temperature and dehydrated in a graded series of ethanol. The samples were infiltrated, embedded, and cut into sections for uranyl acetate and lead citric acid staining. Ultra-structure pictures were obtained with a transmission electron microscope (HITACHI H-7650).
Immunohistochemistry
Kidney tissue sections were prepared as described in section 2.5. Immunohistochemical staining was performed following the procedures noted in the instructions provided by the commercially available kits.
Mouse monoclonal antibodies were purchased from Proteintech Group, Inc., Wuhan, China: PERK/EIF2AK3 rabbit polyclonal antibody, eIF2α rabbit polyclonal antibody, ATF4 rabbit polyclonal antibody, P62/SQSTM1 rabbit polyclonal antibody, Beclin-1 rabbit polyclonal antibody, LC3B-specific rabbit polyclonal antibody, and Atg5 Rabbit Polyclonal Antibody.
Real-Time Quantitative Polymerase Chain Reaction (RT-qPCR)
Total RNA was extracted with the AxyPrep Multisource Total RNA Miniprep Kit (Corning Life Sciences (Wujiang) Co., Ltd) following the manufacturer’s protocol. The PCR primers were synthesized by Sangon Biotech Co., Ltd., and the primer sequences are listed in Table 1. For target gene expression analysis, cDNA was amplified using a PowerUp SYBR Green Master Mix (Takara Bio, Foster City, CA, USA). RT-qPCR reactions were performed in a final volume of 10 µL and run on an ABI7300 Real-Time PCR System. The reactions were initially denatured at 50°C for 2 min and 95°C for 2 min, followed by 40 cycles of 95°C for 15 sec, 55–60°C for 15 sec, and 72°C for 1 min. Gene expression was measured using the 2−ΔΔCt method.
 |
Table 1 Primer Sequences for RT-qPCR |
Statistical Analysis
All experiment data are presented as the mean ± SD. Differences between groups were analyzed by one-way ANOVA, followed by Tukey’s multiple comparisons test. A P < 0.05 was considered statistically significant. All statistical tests were performed using GraphPad Prism Version 7.0 (GraphPad Prism Software, Inc, CA, USA.).
Results
Biochemical Measurements
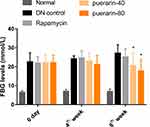
Seventy-two hours after the streptozotocin injection, FBG levels of streptozotocin-induced DN mice were significantly increased compared with the control. During the experiment, the FBG levels of the puerarin groups were lower than before administration, while the levels in the DN control group remained stable. After administration for 8 weeks, FBG levels in the puerarin groups were significantly decreased compared with the DN control group. However, a slight increase in FBG was observed in rapamycin-treated DN mice (Figure 1).
The renal function indexes BUN, Scr, Ucr, and 24-hour urine protein levels in normal control mice were considered as baseline levels. Compared with the control group, BUN, Scr, and 24-hour urine protein levels of DN mice were significantly enhanced, whereas they were significantly decreased in puerarin- and rapamycin-treated mice compared with the DN control. Conversely, Ucr levels in DN control mice (103.34 ±10.02) were significantly lower than in normal mice (265.98 ± 46.34), whereas the creatinine clearance in DN mice was enhanced after puerarin treatment (Figure 2). These results suggested that the mice injected with a single large dose of streptozotocin (150 mg/kg) were eventually progressed to DN.
Morphological Examination
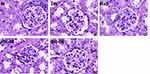
Representative graphs of renal morphology are shown in Figure 3. The analysis revealed substantial pathological alterations in DN mice, which included glomerular collapse and tubular vacuolar degeneration. Reduced matrix expansion, dilatation of the renal tubules, and glomerular collapse were observed in mice treated with rapamycin and puerarin.
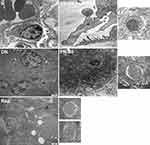
We also analyzed the ultrastructure of kidney tissues. Electron photomicrographs showed severe mitochondrial damage, nuclear deformation, and basement membrane fusion in the kidney samples of DN mice. However, mitochondrial damage and basement membrane fusion were alleviated by puerarin treatment. The mitochondrial cavitation in puerarin mice were less than that in DN control, and the mitochondrial cristae is clearly visible. In all cases, puerarin treatment prevented the changes caused by streptozotocin (the red white arrow indicates the basement membrane and mitochondria). Additionally, autophagosome (characterized by a bilayer membrane vesicle) accumulation was identified in both rapamycin and puerarin groups (Figure 4).
Expression of PERK, eIF2α, and ATF4
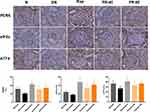
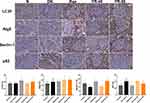
Immunohistochemistry analysis showed that the expression of PERK, eIF2α, and ATF4 in the kidney samples of DN control mice was significantly decreased compared to the control. However, the levels of these proteins in mice treated with different concentrations of puerarin were significantly upregulated (Figure 5).
Expression of Autophagy-Related Proteins
The autophagy markers Beclin-1, LC3II, Atg5, and p62 were assayed. The number of Beclin-1, LC3II, and Atg5 positive cells were obviously decreased in DN mice compared with control mice. However, there was an increase in the number of Beclin-1, LC3II, and Atg5 positive cells in mice treated with puerarin. Conversely, the expression of p62 in the DN control group was higher than in normal mice, whereas p62 expression was downregulated by puerarin treatment compared with the DN control (Figure 6).
Podocyte Functional Proteins
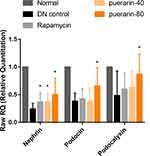
The gene expression of nephrin, podocin, and podocalyxin was analyzed. Their levels were reduced in DN control mice compared with normal healthy C57BL/6 mice, and this effect was prevented by rapamycin. The increase in the expression of nephrin following rapamycin treatment was significant, whereas the expression of podocin and podocalyxin was higher than in DN control mice, but these results were not significant. Additionally, the effect in 80 mg/kg/d puerarin group was significantly evaluated compared with control DN mice (Figure 7).
Discussion
Although the pathogenesis of DN is not fully understood, it may be associated with glucose lipid metabolic disturbance, inflammation, ERS, or autophagy. Abnormalities in the above processes can cause podocyte damage, leading to impaired glomerular filtration function induced by proteinuria.14,15 Podocytes play an important role as a glomerular filtration barrier. Foot process fusion caused by podocyte injury is the main mechanism of glomerular proteinuria.16,17 Clinical studies have shown that podocyte injury appeared in the early stage of the disease, and was accompanied by podocyte retraction, widening, fusion, and disappearance. Additionally, kidney biopsies of DN patients and animal models showed foot process disappearance and a decrease in podocytes by concomitant degrees of proteinuria.18 The above studies suggest that podocyte injury is involved in DN physiology and pathology.
Evidence from our previous studies revealed a hypoglycemic effect of puerarin on DM.12,13 In this study, the FBG levels at weeks 4 and 8 in the DN mice treated with puerarin were decreased compared with the FBG levels in DN control mice. Moreover, puerarin intervention also reduced the abnormally high levels of BUN, Scr, Ucr, and 24-hour urine protein in DN mice. Most importantly, our previous study found that puerarin activated autophagy. These findings suggest that the beneficial effects of puerarin on DN may be associated with the modulation of autophagy.
Recently, a study reported that podocyte dysfunction and apoptosis were related to ERS.19 There is a crosstalk mechanism between ERS and autophagy. Specifically, the unfolded protein response (UPR) is activated in ERS conditions, resulting in the activation of autophagy. Coordination between the UPR and autophagy is important to protect against protein misfolding and kidney damage.20 During the screening for novel DN therapeutic drugs, we discovered that puerarin exposure significantly increased the proliferation of podocytes induced by high sugar. The expression of ERS marker GRP78 and autophagy factor Atg5 were also affected. These findings suggest that ERS/autophagy crosstalk is likely to be a new target of puerarin for the treatment of DN. PERK is an endoplasmic reticulum resident protein with a cytoplasmic kinase domain and an N-terminal intraluminal stress signaling domain.21 Multiple pathways in mammalian cells, including the PERK/eIF2α/ATF4 signaling pathway, are involved in the regulation of ERS/autophagy crosstalk. Persistent ERS activates the PERK-eIF2α signaling pathway, ATF4 is then upregulated in response to eIF2α phosphorylation, and CHOP and Beclin-1 are activated, resulting in the autophagy response.22,23 Our present study found that the levels of autophagy markers Beclin-1, LC3II, and Atg5 were increased by puerarin treatment compared with the DN control, and conversely, p62 was downregulated. These results further confirm that puerarin can induce autophagy in DN. Furthermore, PERK, eIF2α, and ATF4 were upregulated in the presence of puerarin. Given that Beclin-1, LC3II, Atg5, and p62 were modulated by puerarin in DN mice, it is possible that puerarin may induce mitophagy through regulation of the PERK/eIF2α/ATF4 pathway in DN under ERS conditions. In addition, autophagy was induced by rapamycin, but FBG levels remained high. These results indicated that the protective effect of rapamycin on DN in the kidney does not depend on FBG control.
The pathological analysis and transmission electron microscopy evaluation results further confirmed that the kidney damage alterations were evaluated by puerarin. HE staining enabled the visualization of glomerular structures. Glomerular collapse and atrophy were the major streptozotocin-induced changes observed in DN mice. The alterations were mainly due to the effects of streptozotocin on tissues. As we expected, glomerular collapse and atrophy were partially reversed by puerarin. More importantly, autophagosomes were visualized in the tissue sections of the puerarin treatment groups, whereas severe mitochondria damage was observed in DN mice. The gene expression analysis of podocyte functional proteins nephrin, podocin, and podocalyxin provide more evidence for podocyte cytoskeletal repair in the puerarin treatment group. Present study further validates improve effects of puerarin on DN kidney damage. The morphological alterations of kidney are similar with previous Xu et al study.13 Importantly, this study is a further investigation base on our previous work. These results suggest that autophagy is restored following puerarin treatment, and this is accompanied by pathological improvements, which are consistent with previous results.24
Conclusion
In conclusion, this study provides valuable information regarding the mechanisms contributing to the modulation of ERS/autophagy crosstalk by regulating PERK in experimental animal models. Our data suggest that puerarin regulates autophagy at the molecular level, and in some context, is helpful to protect against DN in the kidney. Therefore, puerarin may be a good candidate to treat DN. Based on the present study, we aim to further confirm the activation of ERS-mediated PERK pathway mediated by puerarin promotes autophagy to protect from kidney damage in DN.
Abbreviations
DM, diabetes mellitus; DN, diabetic nephropathy; ECM, extracellular matrix; PERK, protein kinase R-like endoplasmic reticulum kinase; ERS, endoplasmic reticulum stress; PR: puerarin; STZ, streptozotocin; FBG, fasting blood glucose; BUN, blood urea nitrogen; Scr, serum creatinine; Ucr, urine creatinine; HE, hematoxylin-eosin; RT-qPCR, real-time quantitative polymerase chain reaction; UPR, unfolded protein response.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kumar A, Mittal R. Mapping Txnip: key connexions in progression of diabetic nephropathy. Pharmacol Rep. 2018;70(3):614–622. doi:10.1016/j.pharep.2017.12.008
2. Song S, Tan J, Miao Y, Li M, Zhang Q. Crosstalk of autophagy and apoptosis: involvement of the dual role of autophagy under ER stress. J Cell Physiol. 2017;232(11):2977–2984. doi:10.1002/jcp.25785
3. Lenoir O, Jasiek M, Hénique C, et al. Endothelial cell and podocyte autophagy synergistically protect from diabetes-induced glomerulosclerosis. Autophagy. 2015;11(7):1130–1145. doi:10.1080/15548627.2015.1049799
4. Wang X, Xia HY, Qin HY, et al. 20(S)-protopanaxadiol induces apoptosis in human umbilical vein endothelial cells by activating the PERK-eIF2alpha-ATF4 signaling pathway. J Cell Biochem. 2019;120(4):5085–5096. doi:10.1002/jcb.27785
5. Hughes D, Mallucci GR. The unfolded protein response in neurodegenerative disorders - therapeutic modulation of the PERK pathway. FEBS J. 2019;286(2):342–355. doi:10.1111/febs.14422
6. Mcquiston A, Diehl JA. Recent insights into PERK-dependent signaling from the stressed endoplasmic reticulum. F1000Res. 2017;6:1897. doi:10.12688/f1000research.12138.1
7. Xie B, Wang Q, Zhou C, Wu J, Xu D. Efficacy and safety of the injection of the Traditional Chinese Medicine Puerarin for the treatment of diabetic peripheral neuropathy: a systematic review and meta-analysis of 53 randomized controlled trials. Evid Based Complement Alternat Med. 2018;2018:2834650. doi:10.1155/2018/2834650
8. Wenjun H, Jing W, Tao L, et al. The protective effect of Puerarin on myocardial infarction reperfusion injury (MIRI): a meta-analysis of randomized studies in rat models. Med Sci Monit. 2015;21:1700–1706. doi:10.12659/MSM.894312
9. Liu XX, Zhao W, Wang W, Lin S, Yang L. Puerarin suppresses LPS-induced breast cancer cell migration, invasion and adhesion by blockage NF-κB and Erk pathway. Biomed Pharmacother. 2017;92:429–436. doi:10.1016/j.biopha.2017.05.102
10. Ling CL, Liang JM, Zhang C, et al. Synergistic effects of salvianolic acid B and Puerarin on cerebral ischemia reperfusion injury. Molecules. 2018;23(3):564. doi:10.3390/molecules23030564
11. Zhang Y, Yang X, Ge X, Zhang F. Puerarin attenuates neurological deficits via Bcl-2/Bax/cleaved caspase-3 and Sirt3/SOD2 apoptotic pathways in subarachnoid hemorrhage mice. Biomed Pharmacother. 2019;109:
12. Liang T, Xu X, Ye D, Chen W, Gao B, Huang Y. Caspase/AIF/apoptosis pathway: a new target of puerarin for diabetes mellitus therapy. Mol Biol Rep. 2019;46(5):4787–4797. doi:10.1007/s11033-019-04925-1
13. Xu X, Zheng N, Chen Z, Huang W, Liang T, Kuang H. Puerarin, isolated from Pueraria lobata (Willd.), protects against diabetic nephropathy by attenuating oxidative stress. Gene. 2016;591(2):411–416. doi:10.1016/j.gene.2016.06.032
14. Wang H, Zheng C, Lu Y, et al. Urinary fibrinogen as a predictor of progression of CKD. Clin J Am Soc Nephrol. 2017;12(12):1922–1929. doi:10.2215/CJN.01360217
15. Selim F, Wael A, Jackson KE. Diabetes-induced reactive oxygen species: mechanism of their generation and role in renal injury. J Diabetes Res. 2017;2017:8379327. doi:10.1155/2017/8379327
16. Denhez B, Geraldes P. Regulation of nephrin phosphorylation in diabetes and chronic kidney injury. Adv Exp Med Biol. 2017;966:149–161. doi:10.1007/5584_2017_62
17. Nagata M. Podocyte injury and its consequences. Kidney Int. 2016;89(6):1221–1230. doi:10.1016/j.kint.2016.01.012
18. Wolf G, Chen S, Ziyadeh FN. From the periphery of the glomerular capillary wall toward the center of disease podocyte injury comes of age in diabetic nephropathy. Diabetes. 2005;54(6):1626–1634. doi:10.2337/diabetes.54.6.1626
19. Kang S, Dahl R, Hsieh W, et al. Small molecular allosteric activator of the sarco/endoplasmic reticulum Ca2+-ATPase (SERCA) attenuates diabetes and metabolic disorders. J Biol Chem. 2016;291(10):5185–5198. doi:10.1074/jbc.M115.705012
20. Cybulsky AV. Endoplasmic reticulum stress, the unfolded protein response and autophagy in kidney diseases. Nat Rev Nephrol. 2017;13(11):681–696. doi:10.1038/nrneph.2017.129
21. Kong DQ, Li L, Liu Y, Zheng GY. Association between endoplasmic reticulum stress and risk factors of diabetic retinopathy. Int J Ophthalmol. 2018;11(10):1704–1710. doi:10.18240/ijo.2018.10.20
22. Hu X, Deng J, Yu T, et al. ATF4 deficiency promotes intestinal inflammation in mice by reducing uptake of glutamine and expression of antimicrobial peptides. Gastroenterology. 2019;156(4):1098–1111. doi:10.1053/j.gastro.2018.11.033
23. Rozpedek W, Markiewicz L, Diehl JA, Pytel D, Majsterek I. Unfolded protein response and perk kinase as a new therapeutic target in the pathogenesis of Alzheimer’s disease. Curr Med Chem. 2015;22(27):3169–3184. doi:10.2174/0929867322666150818104254
24. Zheng W, Xie W, Yin D, Luo R, Liu M, Guo F. ATG5 and ATG7 induced autophagy interplays with UPR via PERK signaling. Cell Commun Signal. 2019;17(1):42. doi:10.1186/s12964-019-0353-3
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.