Back to Journals » Diabetes, Metabolic Syndrome and Obesity » Volume 14
The β3 Adrenergic Receptor Agonist CL316243 Ameliorates the Metabolic Abnormalities of High-Fat Diet-Fed Rats by Activating AMPK/PGC-1α Signaling in Skeletal Muscle
Authors Ding LN, Cheng Y, Xu LY, Zhou LQ, Guan L , Liu HM, Zhang YX, Li RM, Xu JW
Received 17 December 2020
Accepted for publication 2 March 2021
Published 18 March 2021 Volume 2021:14 Pages 1233—1241
DOI https://doi.org/10.2147/DMSO.S297351
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Juei-Tang Cheng
Li-Na Ding,1,* Ya Cheng,1,* Lu-Yao Xu,1 Le-Quan Zhou,1,2 Li Guan,1,2 Hai-Mei Liu,1,2 Ya-Xing Zhang,1,2 Run-Mei Li,1 Jin-Wen Xu1,2
1The Research Center of Basic Integrative Medicine, Basic Medical College, Guangzhou University of Chinese Medicine, University Town, Guangzhou, 510006, People’s Republic of China; 2Department of Physiology, Basic Medical College, Guangzhou University of Chinese Medicine, University Town, Guangzhou, 510006, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Jin-Wen Xu
Guangzhou University of Chinese Medicine, University Town, Waihuan East Road 232, Guangzhou, 510006, People’s Republic of China
Tel +86-20-39358028
Fax +86-20-39358020
Email [email protected]
Purpose: Skeletal muscle has a major influence on whole-body metabolic homeostasis. In the present study, we aimed to determine the metabolic effects of the β 3 adrenergic receptor agonist CL316243 (CL) in the skeletal muscle of high-fat diet-fed rats.
Methods: Sprague-Dawley rats were randomly allocated to three groups, which were fed a control diet (C) or a high-fat diet (HF), and half of the latter were administered 1 mg/kg CL by gavage once weekly (HF+CL), for 12 weeks. At the end of this period, the serum lipid profile and glucose tolerance of the rats were evaluated. In addition, the phosphorylation and protein and mRNA expression of AMP-activated protein kinase (AMPK), peroxisome proliferator-activated receptor γ coactivator (PGC)-1α, and carnitine palmitoyl transferase (CPT)-1b in skeletal muscle were measured by Western blot analysis and qPCR. The direct effects of CL on the phosphorylation (p-) and expression of AMPK, PGC-1α, and CPT-1b were also evaluated by Western blotting and immunofluorescence in L6 myotubes.
Results: CL administration ameliorated the abnormal lipid profile and glucose tolerance of the high-fat diet-fed rats. In addition, the expression of p-AMPK, PGC-1α, and CPT-1b in the soleus muscle was significantly increased by CL. CL (1 μM) also increased the protein expression of p-AMPK, PGC-1α, and CPT-1b in L6 myotubes. However, the effect of CL on PGC-1α protein expression was blocked by the AMPK antagonist compound C, which suggests that CL increases PGC-1α protein expression via AMPK.
Conclusion: Activation of the β 3 adrenergic receptor in skeletal muscle ameliorates the metabolic abnormalities of high-fat diet-fed rats, at least in part via activation of the AMPK/PGC-1α pathway.
Keywords: CL316243, AMPK, PGC-1α, L6 myotubes, carnitine palmitoyl transferase
Introduction
Skeletal muscle accounts for 40% of body mass and plays an important role in homeostasis.1,2 It is quantitatively the most significant site of peripheral insulin resistance in obesity and diabetes, which are associated with a higher risk of cardiovascular disease.3,4 Furthermore, the loss of skeletal muscle mass and function can contribute to metabolic syndrome,5,6 while the exercise-induced restoration of muscle function is paralleled by an improvement in skeletal muscle insulin sensitivity.7,8 Skeletal muscle insulin resistance is a predictor of the development of type 2 diabetes, and the maintenance of appropriate muscle glucose and lipid disposal helps prevent obesity, diabetes, and metabolic syndrome.9
The β-adrenergic receptor (AR) is one of the key regulators of skeletal muscle mass and function,10 and β-AR agonists may represent a means of preventing or ameliorating the muscle wasting that is associated with aging.11,12 Knowledge of the role of β-AR signaling in skeletal muscle is mainly based on the results of studies of the effects of β2-AR agonists because the β2-AR is the predominant subtype in skeletal muscle.13 However, β3-ARs are also expressed in human and rodent skeletal muscles,14,15 and the selective activation of β3-ARs has been shown to induce muscle hypertrophy and inhibit protein breakdown in rat skeletal muscle.16,17 Although the role of β3-ARs in the regulation of skeletal muscle structure and function are recognized, less is known regarding the metabolic effects of β3-AR activation in skeletal muscle.
UCP is one of a protein groups participating thermogenesis and there are 5 main well-known isoforms including UCP-1 to UCP-5.18 UCP-3, which is the predominant isoform expressed in skeletal muscle, increases metabolic rate in skeletal muscle.19,20. Administration of the β3-adrenergic agonist CL316243 (CL) has been shown to increase the expression of uncoupling proteins 3 (UCP-3) in the skeletal muscle of mice.21,22 This suggests that the activation of the β3-AR in skeletal muscle might have beneficial metabolic effects and help maintain energy homeostasis. In the present study, we first aimed to determine whether the administration of the β3-AR agonist CL ameliorates the metabolic abnormalities in high-fat diet-fed rats. Then, to gain more insight into the direct effects of β3-AR in skeletal muscle, we aimed to determine whether CL activates the AMP-activated protein kinase (AMPK)/peroxisome proliferator-activated receptor gamma coactivator (PGC)-1α pathway in L6 myotubes.
Materials and Methods
Reagents
CL was obtained from Sigma-Aldrich (St. Louis, MO, USA). Dulbecco’s-Modified Eagle’s Medium (DMEM), Opti-MEM, and fetal bovine serum were obtained from Invitrogen (Carlsbad, CA, USA). All other chemicals were of analytical grade and obtained from Guangzhou Chemical Reagents (Guangzhou, China).
Animals
All the experimental procedures were performed in accordance with the Guidelines for Animal Experiments of the Committee of Medical Ethics, National Health Department of China. The animal experiments were approved by the Animal Research Center of Guangzhou University of Chinese Medicine. The ethical approval certificate number is 2,016,139. Male Sprague-Dawley rats (220–250 g) were obtained from the Laboratory Animal Center at Guangzhou University of Chinese Medicine. The rats were randomly allocated to three groups, which were fed a control diet and orally administered physiological saline (C group; n=10); fed a high-fat diet (60% hydrogenated coconut oil; D12492; Research Diets, Inc; New Brunswick, NJ, USA) and orally administered physiological saline (HF group; n=10); or fed the high-fat diet and orally administered CL (1 mg/kg once weekly; HF+CL, n=10) for 12 weeks. CL was dissolved in dimethyl sulfoxide (DMSO) and then diluted in physiological saline.
Intraperitoneal Glucose Tolerance Testing
Intraperitoneal glucose tolerance testing was performed as previously described.23 Briefly, rats were administered 25% w/v glucose solution intraperitoneally at a dose of 1 g/kg body mass. Tail vein blood samples were collected immediately and 30, 60, 90, and 120 min later. Blood glucose concentrations were measured using a glucose meter (Johnson & Johnson).
Serum and Tissue Collection
Trunk blood was then obtained, left at room temperature for 30 min, and then centrifuged at 4500 × g and 4°C for 15 min. The lipid concentrations of the serum obtained were measured using a Hitachi clinical analyzer.
Histological Analysis
Tissue sections (5 mm) were obtained from adipose tissue samples that had been fixed and embedded in paraffin. These were then deparaffinized in xylene, rehydrated, and washed in phosphate-buffered saline, prior to hematoxylin and eosin (HE) staining. The diameter of the adipocyte in the HF and HF+CL groups was normalized to that of the C group.
Cell Culture
The L6 cell line was purchased from the cell bank of the Chinese Academy of Sciences (Shanghai, China). For differentiation into myotubes, L6 myoblasts were cultured at 37 °C with 95% humidity in DMEM supplemented with 2% horse serum, which was changed every other day. Myotube formation was achieved after 5 days of incubation, at which time the cells were used in the experiments.
Immunoblotting
Immunoblotting was performed as previously described.23 Briefly, tissue lysates were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then electrotransferred to membranes. The primary antibodies used were as follows: anti-PGC-1a (PA5-22,958; Thermo), anti-AMPK (5831; Cell Signaling Technology), anti-p-AMPK (2535; Cell Signaling Technology), anti-CPT-1b (ab134988, Abcam), and anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (5174; Cell Signaling Technology). Membranes were incubated with primary and secondary antibodies using standard techniques and the detection of specific protein bands was accomplished using enhanced chemiluminescence. Images of the bands were acquired and analyzed using a quantitative digital imaging system (Quantity One; Bio-Rad), avoiding saturation.
Real-Time Reverse Transcription-PCR
Real-time PCR was performed as previously described.24 cDNA was synthesized using a Transcriptor First Strand cDNA Synthesis Kit (Roche, Basel, Switzerland), according to the manufacturer’s instructions. For PGC1-α, the primers were 5′-CAACAATGAGCCTGCGAACA-3′ (forward) and 5′-TGAGGACCGCTAGCAAGTTTG-3′ (reverse), resulting in a 71-bp RT-PCR product. For CPT-1b, the primers were 5′-GGCTGCCGTGGGACATT-3′ (forward) and 5′-TGCCTTGGCTACTTGGTACGA-3′ (reverse), resulting in a 100-bp RT-PCR product. For GAPDH, the primers were 5′-AGACAGCCGCATCTTCTTGT-3′ (forward) and 5′-CTTGCCGTGGGTAGAGTCAT-3′ (reverse), resulting in a 207-bp RT-PCR product. RT-PCR reactions were performed using a CFX96™ Real-Time System (Bio-Rad, Hercules, CA, USA) in final volumes of 20 μL, which contained 10 μL of FastStart Universal SYBR Green Master (Rox) (Roche, Basel, Switzerland), 1 μL of each primer (10 μM), 2 μL of cDNA, and 7 μL of PCR-grade water. The RT-PCR products underwent melting point analysis and were quantified using the ΔΔCT method, with GAPDH as the reference gene. The expression in the HF and HF+CL groups was normalized to that of the C group.
Immunofluorescence
L6 myotubes were grown on coverslips, treated as described below, then fixed with 4% paraformaldehyde and permeabilized with 0.1% Triton-X. The cells were then blocked using 3% normal serum for 20 min and incubated with an antibody against p-AMPK (BD Transduction Laboratories) and a FITC-conjugated secondary antibody (K00018968, Dako North America Inc., Dako, Denmark). After washing, the nuclei were counterstained with 4′,6-diamidino-2-phenylindole (Sigma). Immunofluorescence was visualized using an Olympus BX41 microscope (Tokyo, Japan) and images were obtained using a high-resolution DP70 Olympus digital camera.
Statistical Analysis
Data are presented as mean ± standard deviation (SD) and represent the results of at least three independent experiments. Statistical comparisons were made using Student’s t-test or one-way analysis of variance, followed by Tukey’s test where applicable, to identify significant differences between mean values. P < 0.05 was considered to represent statistical significance.
Results
CL Administration Reduces the Body Mass and Ameliorates the Serum Lipid Abnormalities and Glucose Intolerance of High-Fat Diet-Fed Rats
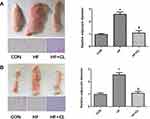
As expected, the body masses of the HF rats were significantly higher than those of the C rats, but CL administration reduced the body mass of the HF rats (Figure 1A). The HF rats had significantly higher total serum cholesterol, triglyceride, and low-density lipoprotein (LDL)-cholesterol concentrations than the C group, whereas the high-density lipoprotein (HDL)-cholesterol concentration in the HF rats was significantly lower. However, CL administration improved the lipid profile of the HF rats (Figure 1B). Intraperitoneal glucose tolerance testing showed the area under the glucose curve (AUC) was significantly larger in HF rats than C rats, and this difference was also eliminated by concurrent CL administration (Figure 1C and D).
CL Administration Reduces the Peri-Epididymal and Inguinal Fat Depot Masses of HFD-Fed Rats
HE staining of tissue sections showed that HF rats had larger lipid droplets in their peri-epididymal and inguinal adipocytes (Figure 2A and B). Concurrent CL administration eliminated these differences.
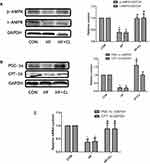
CL Administration Increases the Expression of p-AMPK, PGC-1α, and CPT-1b in the Soleus Muscles of HF Rats
Immunoblotting showed that the protein expression levels of AMPK, p-AMPK, PGC-1α, and CPT-1b in the soleus muscles of HF rats were significantly lower than those of C rats, and CL administration eliminated these differences (Figure 3A and B). Consistent with this, CL administration increased PGC-1α mRNA expression in the soleus muscles of HF rats (Figure 3C).
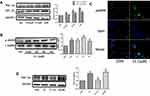
CL Treatment Increases AMPK, PGC-1α, and CPT-1b Expression in L6 Myotubes
Immunoblotting showed that CL increased PGC-1α and CPT-1b protein expression levels in a dose-dependent manner in L6 myotubes (Figure 4A). Treatment with CL (1 µM) for 4 h also increased AMPK phosphorylation, as demonstrated using Western blotting and immunofluorescence (Figure 4B and C). 5-Aminoimidazole-4-carboxamide ribonucleotide (AICAR/acadesine, a cell-permeable activator of AMPK, 0.5 mM/L) increased PGC-1α expression, whereas the co-treatment of L6 myotubes with compound C (an inhibitor of AMPK, 10 μM/L) alongside CL significantly reduced PGC-1α expression (Figure 4D).
Discussion
Skeletal muscle plays an important role in the regulation of whole-body energy homeostasis, and energy production depends on mitochondrial function. The dysfunction or loss of skeletal muscle mass has been shown to contribute to obesity and type 2 diabetes.5,6 Mitochondrial dysfunction and/or a reduction in mitochondrial content has been reported in skeletal muscle and adipose tissue from obese and diabetic subjects.25,26 PGC-1α is a major transcriptional regulator of mitochondrial function in skeletal muscle,27,28 and AMPK acts as a key sensor of energy status in skeletal muscle and regulates glucose and fatty acid metabolism.29 The activation of AMPK regulates the expression and activity of PGC-1α in skeletal muscle,30 and high-fat diet-feeding has been shown to reduce the expression and activity of PGC-1 and AMPK and downstream signaling in skeletal muscle.31,32 Consistent with this, in the present study, high-fat diet-feeding induced metabolic abnormalities that were associated with lower expression of PGC-1α and p-AMPK in skeletal muscle (Figure 3).
The β3-AR is an established therapeutic target for obesity and metabolic disease.33,34 CL, a highly selective systemic β3-AR agonist, induces expression of UCP-1 in adipose tissue and has anti-obesity effects.35–37 Although β3-AR is principally expressed in adipose tissue, it is also expressed in human and rodent skeletal muscles.14,15 Recently, the β3-AR agonist mirabegron has been shown to improve glucose homeostasis and increase the expression of PGC-1α in the skeletal muscle of obese humans.38 Consistent with this, in the present study, we have shown that selective activation of β3-AR by CL in high-fat diet-fed rats reduces their body mass and ameliorates their glucose intolerance, as well as increasing the protein expression of PGC-1α and p-AMPK in skeletal muscle.
Long-chain fatty acids (LCFAs) are a major energy source in muscle. CPT-1 is a rate-limiting enzyme in the mitochondrial β-oxidation of LCFAs.39 The muscle isoform, CPT-1b, is mainly expressed in brown and white adipose tissue, the heart, and skeletal muscle.40,41 The activity of CPT-1b is lower in obese skeletal muscle, which contributes to the lower fatty acid oxidation.42 Furthermore, mice with CPT-1b deficiency become severely insulin resistant after 7 months of high-fat diet-feeding.43 Consistently, we found that high-fat diet-feeding reduced the expression of CPT-1b in skeletal muscle and CL administration increased the protein and mRNA expression of this enzyme. These results suggest that the beneficial effect of CL on the lipid profile of HF rats is mediated through higher protein and mRNA expression of CPT-1b in skeletal muscle.
Adipose tissue communicates with skeletal muscle through the secretion of adipokines, such as adiponectin, leptin, omentin, osteopontin, and cardiotrophin-1; and batokines, such as bone morphogenetic protein 8b, fibroblast growth factor-21, endothelin-1, and interleukin-6.44,45 Adiponectin activates the AMPK/PGC-1α pathway and stimulates fatty acid oxidation.46,47 Furthermore, a 7-day infusion of CL316243 increases adiponectin expression in white adipose tissue and the serum concentration of adiponectin.48 Therefore, the effects of CL on the skeletal muscle of the mice in the present study might have been secondary to effects on adipose tissue. However, the in vitro data confirm that CL has a direct dose-dependent effect to increase PGC-1α protein expression in muscle cells. Because PGC-1α expression is regulated by AMPK,49,50 we next determined whether AMPK inhibition would prevent the CL-induced activation of PGC-1α in L6 myotubes. Indeed, CL (1 µM) treatment for 4 h increased AMPK phosphorylation, and compound C, an AMPK inhibitor, reduced PGC-1α protein expression in CL-treated L6 myotubes. These results suggest that CL increases PGC-1α protein expression by activating AMPK.
In addition to adipose tissue and skeletal muscle, β3-AR are also found in the brain, in areas such as the brain stem and the hypothalamus.51 Acute central injection of CL potentially reduced food intake and increased hypothalamic neuronal activity in rats.52 One limitation of our study is that we did not detect the food intake of the rats. Chronic effect of CL on the animal feeding habits needs to be further studied.
Conclusion
Our study suggests that CL improves glucose tolerance and lowers lipid profiles in the high-fat diet-feeding of rats by activating the AMPK/PGC-1α pathway. This finding supports the development of novel anti-obesity therapies that involve the activation of β3-ARs in skeletal muscle.
Acknowledgments
We thank Mark Cleasby, PhD, from Liwen Bianji, Edanz Group China, for editing the English text of a draft of this manuscript. We also thank the rats sacrificed in this study.
Funding
This study was supported by the National Natural Science Foundation of China, the Natural Science Foundation of Guangdong Province, and the Guangzhou University of Chinese Medicine (grant numbers81774107, 81673717, and QNYC20170101).
Disclosure
The authors declare that they have no conflicts of interest relevant to this work.
References
1. Frontera WR, Ochala J. Skeletal muscle: a brief review of structure and function. Calcif Tissue Int. 2015;96(3):183–195. doi:10.1007/s00223-014-9915-y
2. Gan Z, Fu T, Kelly DP, Vega RB. Skeletal muscle mitochondrial remodeling in exercise and diseases. Cell Res. 2018;28(10):969–980. doi:10.1038/s41422-018-0078-7
3. Garneau L, Aguer C. Role of myokines in the development of skeletal muscle insulin resistance and related metabolic defects in type 2 diabetes. Diabetes Metab. 2019;45(6):505–516. doi:10.1016/j.diabet.2019.02.006
4. Townsend LK, Brunetta BS, Mori MA. Mitochondria-associated ER membranes in glucose homeostasis and insulin resistance. Am J Physiol Endocrinol Metab. 2020;319(6):E1053–E1060. doi:10.1152/ajpendo.00271.2020
5. Cho J, Choi Y, Sajgalik P, et al. Exercise as a therapeutic strategy for sarcopenia in heart failure: insights into underlying mechanisms. Cells. 2020;9(10):E2284. doi:10.3390/cells9102284
6. Cleasby ME, Jamieson PM, Atherton PJ. Insulin resistance and sarcopenia: mechanistic links between common co-morbidities. J Endocrinol. 2016;229(2):R67–R81. doi:10.1530/JOE-15-0533
7. Sjøberg KA, Frøsig C, Kjøbsted R, et al. Exercise increases human skeletal muscle insulin sensitivity via coordinated increases in microvascular perfusion and molecular signaling. Diabetes. 2017;66(6):1501–1510. doi:10.2337/db16-1327
8. Cartee GD. Mechanisms for greater insulin-stimulated glucose uptake in normal and insulin-resistant skeletal muscle after acute exercise. Am J Physiol Endocrinol Metab. 2015;309(12):E949–E959. doi:10.1152/ajpendo.00416.2015
9. Richter-Stretton GL, Fenning AS, Vella RK. Skeletal muscle - a bystander or influencer of metabolic syndrome? Diabetes Metab Syndr. 2020;14(5):867–875. doi:10.1016/j.dsx.2020.06.006
10. Ryall JG, Church JE, Lynch GS. Novel role for ß-adrenergic signalling in skeletal muscle growth, development and regeneration. Clin Exp Pharmacol Physiol. 2010;37(3):397–401. doi:10.1111/j.1440-1681.2009.05312.x
11. Koziczak-Holbro M, Rigel DF, Dumotier B, et al. Pharmacological characterization of a novel 5-hydroxybenzothiazolone-derived β 2-adrenoceptor agonist with functional selectivity for anabolic effects on skeletal muscle resulting in a wider cardiovascular safety window in preclinical studies. J Pharmacol Exp Ther. 2019;369(2):188–199. doi:10.1124/jpet.118.255307
12. Ryall JG, Plant DR, Gregorevic P, Sillence MN, Lynch GS. Beta 2-agonist administration reverses muscle wasting and improves muscle function in aged rats. J Physiol. 2004;555(Pt 1):175–188. doi:10.1113/jphysiol.2003.056770
13. Ito A, Ohnuki Y, Suita K, et al. Role of β-adrenergic signaling in masseter muscle. PLoS One. 2019;14(4):e0215539. doi:10.1371/journal.pone.0215539
14. Kim YS, Sainz RD, Molenaar P, Summers RJ. Characterization of β1- and β2-adrenoceptors in rat skeletal muscles. Biochem Pharmacol. 1991;42(9):1783–1789. doi:10.1016/0006-2952(91)90516-8
15. Evans BA, Papaioannou M, Bonazzi VR, Summers RJ. Expression of beta 3-adrenoceptor mRNA in rat tissues. Br J Pharmacol. 1996;117(1):210–216. doi:10.1111/j.1476-5381.1996.tb15176.x
16. Puzzo D, Raiteri R, Castaldo C, et al. CL316,243, a β3-adrenergic receptor agonist, induces muscle hypertrophy and increased strength. Sci Rep. 2016;5:37504. doi:10.1038/srep37504
17. Navegantes LC, Resano NM, Baviera AM, Migliorini RH, Kettelhut IC. CL 316,243, a selective beta3-adrenergic agonist, inhibits protein breakdown in rat skeletal muscle. Pflugers Arch. 2006;451(5):617–624. doi:10.1007/s00424-005-1496-1
18. Adams SH. Uncoupling protein homologs: emerging views of physiological function. J Nutr. 2000;130(4):711–714. doi:10.1093/jn/130.4.711
19. Busiello RA, Savarese S, Lombardi A. Mitochondrial uncoupling proteins and energy metabolism. Front Physiol. 2015;6:36. doi:10.3389/fphys.2015.00036
20. Choi CS, Fillmore JJ, Kim JK, et al. Overexpression of uncoupling protein 3 in skeletal muscle protects against fat-induced insulin resistance. J Clin Invest. 2007;117(7):1995–2003. doi:10.1172/JCI13579
21. Nakamura Y, Nagase I, Asano A, et al. Beta 3-adrenergic agonist up-regulates uncoupling proteins 2 and 3 in skeletal muscle of the mouse. J Vet Med Sci. 2001;63(3):309–314. doi:10.1292/jvms.63.309
22. Nagase I, Yoshida T, Kumamoto K, et al. Expression of uncoupling protein in skeletal muscle and white fat of obese mice treated with thermogenic beta 3-adrenergic agonist. J Clin Invest. 1996;97(12):2898–2904. doi:10.1172/JCI118748
23. Li RM, Chen SQ, Zeng NX, et al. Browning of abdominal aorta perivascular adipose tissue inhibits adipose tissue inflammation. Metab Syndr Relat Disord. 2017;15(9):450–457. doi:10.1089/met.2017.0074
24. Chen SQ, Ding LN, Zeng NX, et al. Icariin induces irisin/FNDC5 expression in C2C12 cells via the AMPK pathway. Biomed Pharmacother. 2019;115:108930. doi:10.1016/j.biopha.2019.108930
25. Kelley DE, He J, Menshikova EV, Ritov VB. Dysfunction of mitochondria in human skeletal muscle in type 2 diabetes. Diabetes. 2002;51(10):2944–2950. doi:10.2337/diabetes.51.10.2944
26. Ritov VB, Menshikova EV, Jing H, Ferrell RE, Goodpaster BH, Kelley DE. Deficiency of subsarcolemmal mitochondria in obesity and type 2 diabetes. Diabetes. 2005;54(1):8–14. doi:10.2337/diabetes.54.1.8
27. Puigserver P, Spiegelman BM. Peroxisome proliferator-activated receptor-gamma coactivator 1 alpha (PGC-1 alpha): transcriptional coactivator and metabolic regulator. Endocr Rev. 2003;24(1):78–90. doi:10.1210/er.2002-0012
28. Dillon LM, Rebelo AP, Moraes CT. The role of PGC-1 coactivators in aging skeletal muscle and heart. IUBMB Life. 2012;64(3):231–241. doi:10.1002/iub.608
29. Hardie DG, Sakamoto K. AMPK: a key sensor of fuel and energy status in skeletal muscle. Physiology. 2006;21:48–60. doi:10.1152/physiol.00044.2005
30. Leick L, Fentz J, Biensø RS, et al. PGC-1{alpha} is required for AICAR-induced expression of GLUT4 and mitochondrial proteins in mouse skeletal muscle. Am J Physiol Endocrinol Metab. 2010;299(3):E456–E465. doi:10.1152/ajpendo.00648.2009
31. Shen S, Liao Q, Zhang T, Pan R, Lin L. Myricanol modulates skeletal muscle-adipose tissue crosstalk to alleviate high-fat diet-induced obesity and insulin resistance. Br J Pharmacol. 2019;176(20):3983–4001. doi:10.1111/bph.14802
32. Yao S, Yuan Y, Zhang H, et al. Berberine attenuates the abnormal ectopic lipid deposition in skeletal muscle. Free Radic Biol Med. 2020;159:66–75. doi:10.1016/j.freeradbiomed.2020.07.028
33. Pan R, Zhu X, Maretich P, Chen Y. Combating obesity with thermogenic fat: current challenges and advancements. Front Endocrinol (Lausanne). 2020;11:185. doi:10.3389/fendo.2020.00185
34. Mund RA, Frishman WH. Brown adipose tissue thermogenesis: β3-adrenoreceptors as a potential target for the treatment of obesity in humans. Cardiol Rev. 2013;21(6):265–269. doi:10.1097/CRD.0b013e31829cabff
35. Ghorbani M, Claus TH, Himms-Hagen J. Appearance of brown adipocytes in white adipose tissue during CL 316,243-induced reversal of obesity and diabetes in zucker fa/fa rats. Int J Obes Relat Metab Disord. 1997;21(6):465–475. doi:10.1038/sj.ijo.0800432
36. Ghorbani M, Claus TH, Himms-Hagen J. Effect of CL-316,243, a thermogenic beta 3-agonist, on energy balance and brown and white adipose tissues in rats. Am J Physiol. 1994;266(4 Pt 2):R1371–R1382. doi:10.1152/ajpregu.1994.266.4.R1371
37. Clookey SL, Welly RJ, Shay D, et al. Beta 3 adrenergic receptor activation rescues metabolic dysfunction in female estrogen receptor alpha-null mice. Front Physiol. 2019;10:9. doi:10.3389/fphys.2019.00009
38. Finlin BS, Memetimin H, Zhu B, et al. The beta3-adrenergic receptor agonist mirabegron improves glucose homeostasis in obese humans. J Clin Invest. 2020;130(5):2319–2331. doi:10.1172/JCI134892
39. Schlaepfer IR, Joshi M. CPT1A-mediated fat oxidation, mechanisms, and therapeutic potential. Endocrinology. 2020;161(2):bqz046. doi:10.1210/endocr/bqz046
40. Esser V, Brown NF, Cowan AT, Foster DW, McGarry JD. Expression of a cDNA isolated from rat brown adipose tissue and heart identifies the product as the muscle isoform of carnitine palmitoyltransferase I (M-CPT I). M-CPT I is the predominant CPT I isoform expressed in both white (epididymal) and brown adipocytes. J Biol Chem. 1996;271(12):6972–6977. doi:10.1074/jbc.271.12.6972
41. Price N, van der Leij F, Jackson V, et al. A novel brain-expressed protein related to carnitine palmitoyltransferase I. Genomics. 2002;80(4):433–442. doi:10.1006/geno.2002.6845
42. Kim JY, Hickner RC, Cortright RL, Dohm GL, Houmard JA. Lipid oxidation is reduced in obese human skeletal muscle. Am J Physiol Endocrinol Metab. 2000;279:E1039–E1044. doi:10.1152/ajpendo.2000.279.5.E1039
43. Kim T, Moore JF, Sharer JD, Yang K, Wood PA, Yang Q. Carnitine palmitoyltransferase 1b deficient mice develop severe insulin resistance after prolonged high fat diet feeding. J Diabetes Metab. 2014;5:1000401. doi:10.4172/2155-6156.1000401
44. Li F, Li Y, Duan Y, Hu CA, Tang Y, Yin Y. Myokines and adipokines: involvement in the crosstalk between skeletal muscle and adipose tissue. Cytokine Growth Factor Rev. 2017;33:73–82. doi:10.1016/j.cytogfr.2016.10.003
45. Coles CA. Adipokines in healthy skeletal muscle and metabolic disease. Adv Exp Med Biol. 2016;900:133–160.
46. Yamauchi T, Kamon J, Minokoshi Y, et al. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat Med. 2002;8(11):1288–1295. doi:10.1038/nm788
47. Yoon MJ, Lee GY, Chung JJ, Ahn YH, Hong SH, Kim JB. Adiponectin increases fatty acid oxidation in skeletal muscle cells by sequential activation of AMP-activated protein kinase, p38 mitogen-activated protein kinase, and peroxisome proliferator-activated receptor alpha. Diabetes. 2006;55(9):2562–2570. doi:10.2337/db05-1322
48. Zhang Y, Matheny M, Zolotukhin S, Tumer N, Scarpace PJ. Regulation of adiponectin and leptin gene expression in white and brown adipose tissues: influence of beta3-adrenergic agonists, retinoic acid, leptin and fasting. Biochim Biophys Acta. 2002;1584(2–3):115–122. doi:10.1016/S1388-1981(02)00298-6
49. Lira VA, Brown DL, Lira AK, et al. Nitric oxide and AMPK cooperatively regulate PGC-1 in skeletal muscle cells. J Physiol. 2010;588(Pt 18):3551–3566. doi:10.1113/jphysiol.2010.194035
50. Okamoto S, Asgar NF, Yokota S, Saito K, Minokoshi Y. Role of the alpha2 subunit of AMP-activated protein kinase and its nuclear localization in mitochondria and energy metabolism-related gene expressions in C2C12 cells. Metabolism. 2019;90:52–68. doi:10.1016/j.metabol.2018.10.003
51. Castillo-Meléndez M, McKinley MJ, Summers RJ. Intracerebroventricular administration of the beta(3)-adrenoceptor agonist CL 316243 causes fos immunoreactivity in discrete regions of rat hypothalamus. Neurosci Lett. 2000;290(3):161–164. doi:10.1016/S0304-3940(00)01359-8
52. Richard JE, López-Ferreras L, Chanclón B, et al. CNS β 3-adrenergic receptor activation regulates feeding behavior, white fat browning, and body weight. Am J Physiol Endocrinol Metab. 2017;313(3):E344–E358. doi:10.1152/ajpendo.00418.2016
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.