Back to Journals » Infection and Drug Resistance » Volume 13
Targeting Polyamine Metabolism for Control of Human Viral Diseases
Authors Huang M, Zhang W, Chen H , Zeng J
Received 9 May 2020
Accepted for publication 27 October 2020
Published 1 December 2020 Volume 2020:13 Pages 4335—4346
DOI https://doi.org/10.2147/IDR.S262024
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Mingyuan Huang,1,2,* Weijian Zhang,1,2,* Haiyong Chen,3 Jincheng Zeng1,4,5
1Dongguan Key Laboratory of Medical Bioactive Molecular Developmental and Translational Research, Guangdong Provincial Key Laboratory of Medical Molecular Diagnostics, Guangdong Medical University, Dongguan 523808, People’s Republic of China; 2Dongguan Key Laboratory of Environmental Medicine, School of Public Health, Guangdong Medical University, Dongguan 523808, People’s Republic of China; 3School of Chinese Medicine, Li Ka Shing Faculty of Medicine, The University of Hong Kong, Hong Kong, People’s Republic of China; 4Key Laboratory of Medical Bioactive Molecular Research for Department of Education of Guangdong Province, Guangdong Medical University, Dongguan 523808, People’s Republic of China; 5Collaborative Innovation Center for Antitumor Active Substance Research and Development, Guangdong Medical University, Zhanjiang, Guangdong 524023, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Jincheng Zeng Email [email protected]
Abstract: A virus is an infectious particle which generally contains nucleic acid genome (DNA or RNA inside a protein shell), except for human immunodeficiency virus (HIV). Viruses have to reproduce by infecting their host cells. Polyamines are ubiquitous compounds in mammalian cells and play key roles in various cellular processes. The metabolic pathways of polyamines have been well studied. Targeting these metabolic pathways can reduce infections caused by viruses. In the study, we systematically reviewed the association of polyamine metabolic pathways and viruses including coxsackievirus B3 (CVB3), enterovirus 71 (EV71), poliovirus (PV), Zika virus (ZKV), hepatitis C virus (HCV), hepatitis B virus (HBV), dengue virus (DENV), Japanese encephalitis virus (JEV), yellow fever virus (YFV), Ebola virus (EBOV), marburgvirus (MARV), chikungunya virus (CHIKV), sindbis virus (SINV), Semliki Forest virus (SFV), Epstein–Barr virus (EBV), herpes simplex virus 1 (HSV), human cytomegalovirus (HCMV), vesicular stomatitis virus (VSV), Rabies virus (RABV), Rift Valley fever virus (RVFV), La Crosse virus (LACV), human immunodeficiency virus (HIV), Middle East respiratory syndrome virus (MERS-CoV), and coronavirus disease 2019 (SARS-CoV-2). This review revealed that targeting polyamine metabolic pathways may be a potential approach to control human viral infection.
Keywords: polyamine metabolism, virus, infection, diseases
Introduction
A virus is one of the greatest threats to human health. It generally is a small, simple, non-cellular organism containing one or two nucleic acid genomes made of DNA or RNA, and some viruses contain a protein shell called capsid. Viruses have to live and reproduce in infecting living cells, most of them resulting in damage of host cells. Viral infections have enormous impacts on the health, social, and economic burden globally. For example, HIV and Rabies virus can bring danger to human life.1,2 Ebola Hemorrhagic Fever is a disease with high infectivity, high pathogenicity, and high mortality, which causes the Ebola virus, and, in the outbreak of west Africa in early 2014, there were 2,240 infectious cases and 1,229 deaths.3 Recently, a new species of coronavirus called SARS-CoV-2 has infected millions of people worldwide and killed hundreds of thousands.4 However, the diverse mechanisms of viral infection are not fully understood. A virus, lacking its own metabolic machinery and enzyme system, has to infect host cells to complete the genome replication and transcription, and reproduction of a new infectious virus.5 In this process, polyamines may play an essential role in the viral life cycle.
Polyamines are small positively charged amine molecules in mammalian cells, including putrescine, spermidine, and spermine. They are important metabolic regulatory substances that participate in a variety of physiological and pathological processes, such as promoting cell differentiation, proliferation, and growth, and regulating nucleic acid and protein synthesis.6 In the metabolic pathway of biogenic polyamines, ornithine is converted to putrescine by ornithine decarboxylase 1(ODC1), and it follows that putrescine is converted to spermidine by spermine synthetase (SRM) and spermine by spermine synthetase (SMS),7,8 as shown in Figure 1. Spermine can be further degraded into spermine and putrescine through the action of spermine/spermine acetyltransferase (SAT1), spermine oxidase (SMOX), and polyamine oxidase (PAOX). The polyamines metabolic enzymes are specifically modulated by the enzyme regulators, including difluoromethornithine (DFMO) inhibiting ODC1, methylglyoxal bis(guanylhydrazone) (MGBG) inhibiting S-adenosylmethionine decarboxylase (SAMDC), N1,N11-diethylnorspermine (DENspm) activating SAT1, and N,N1-bis (2,3-butadienyl)-1,4-butanediamine (MDL) inhibiting PAOX. In eukaryotes, spermidines act as a substrate for the hypusination of a specific lysine residue of the eukaryotic initiation factor 5A (eIF5A) through two enzymes, deoxyhypusine synthase (DHPS) and deoxyhypusine hydroxylase (DOHH). The eukaryotic initiation factor 5A (eIF5A) and its hypusine modification were important in the elongation phase of protein synthesis and facilitated the production of proteins.9 Besides, the hypusine of eIF5A may be involved in transcription, translation, and nucleocytoplasmic transport in cellular processes.10 Emerging studies revealed that polyamine metabolic pathways were involved in the process of viral invasion and replication in the host.11 Understanding how viruses utilize host cell polyamines to complete self-replication may lead to the development of a novel strategy to reduce viral infection threats. In this review, we illustrate roles of polyamine metabolic pathways for diverse viruses as well as the potential drugs targeting this pathway.
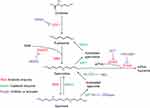 |
Figure 1 The metabolic pathways of biogenic polyamines. |
Picornaviridae
Coxsackievirus B3
Coxsackievirus B3 (CVB3) is the common cause of myocarditis, pancreatitis, and central nervous system infections. It is transmitted by fecal–oral route or across the placenta.12 The epidemiology of CVB3 has been well understood.13 The pathogenic mechanism of CVB3 remains largely unclear. Previous studies showed that CVB3 was sensitive to polyamine depletion by DFMO, as shown in Table 1, and the virus titers decreased in varying degrees.14–16 CVB3 utilized polyamines to adhere to susceptible cells and initiated the infection process, and the inhibition of polyamines biosynthesis protected against viral infection by preventing the virus from attaching to susceptible cells.14 It can be inferred that decreasing affinity for polyamines may coincidentally preclude virus binding to host cells. Polyamines enhanced viral protease (2A and 3C) activity and promoted viral replication.15 In the polyamine-depleted conditions, protease (2A and 3C) mutations promoted cleavage of cellular eIF4G during the virus infection. In a Vero-E6 cells model, there was a dose-response inhibition of CVB3 replication by reducing polyamine biosynthesis with DFMO and the virus titers were rescued by exogenous polyamines.16 And in a BALB/c mice infected with CVB3 and treated with DFMO, there were significant differences with 3–7-fold reduction in viral titer in the organs (like lung, liver, kidneys). These findings revealed that polyamines play an essential role in CVB3 infection and drugs suppressing polyamines may prevent CVB3 infection.
 |  |  |
Table 1 Role of Polyamines in Viral Infection |
Enterovirus 71 and Poliovirus
Enterovirus 71 (EV71) and poliovirus are both enteroviruses, which are also prevalent in infants and children. The common symptoms of EV71 infection are fever, oral ulcers, and skin herpes, and severe cases can cause central nervous complications.17 Similarly, poliovirus (PV) invades the central nervous system in severe cases, which eventually develops into paralysis.18 Mounce et al16 found that there was a significant reduction in viral titers in DFMO-treated Hela cells. The viral titers dropped 12-fold in the EV71 group and declined 20-fold in the poliovirus group. The viral titers were rescued with exogenous polyamine supplement, which suggested that EV71and poliovirus rely on polyamines for their replication in host cells.
Flaviviridae
Zika Virus
Zika virus (ZKV) is an insect-borne virus mainly transmitted by mosquitoes. ZKV can go through blood–brain barrier and damage the central nervous system. Symptoms of the virus infection are generally mild. The recent evidence revealed a possible association between maternal ZKV infection and adverse fetal outcomes, such as congenital microcephaly, as well as a possible association with Guillain-Barrésyndrome.19 There are no specific treatments for ZKV at present currently. Pilot studies indicated that drugs (eg, DENspm) targeting SAT1 may be effective against the virus, as shown in Table 1.16,20,21 Polyamine analogs, such as symmetrically substituted bus(ethyl) norspermine (BENSpm), can compete with natural polyamines for transport and biosynthesis, which decreased polyamine availability for viral replication and translation.21 Studies uncovered that polyamine metabolism enzymes were influenced by type I interferon (IFN I), particularly in upregulating SAT1.22,23 In this way, IFN I may play an important role in antiviral signaling through upregulating SAT1, which decreased polyamines biosynthesis and restricted infectious viral replication, like ZKV.20 It was reported that viral infection caused IFN I production, which gave rise to expression of SAT1 that reduced spermidine and spermine levels, resulting in disability for ZKV replication.
Hepatitis C Virus
Hepatitis C virus (HCV) is a blood borne virus. Globally, there are (estimated) 71 million people who have chronic HCV, the major risk factor for cirrhosis and hepatocellular carcinoma.24 It was reported that polyamines promoted HCV replication and increased HCV titers by activating HCV RNA polymerase and inhibiting viral helicase.25 In addition, HCV core proteins and NS5A suppressed ODC1and SAT1 levels but elevated SMOX, which lead to decreased concentrations of spermidine and spermine.26 Masalova et al,27 demonstrated that polyamines depletion by DFMO in HCV infected Huh7.5 cells suppressed the production of cytokines (such as TNF-α, TGF-β, and IL-1β) and declined the levels of expression of NS5A and NS5B viral proteins involved in HCV replication which implied that DFMO could be a potential anti-HCV therapeutic agent.
Hepatitis B Virus
Hepatitis B virus (HBV) is a pathogen containing HBV core protein (HBc), HBV X protein (HBx), HBV-DNA polymerase, and envelope proteins, which can cause a global public health problem. There are more than 257 million people throughout the world who are chronically infected with HBV and chronic HBV infection has a high risk of developing cirrhosis and hepatocellular carcinoma.28 Current therapies act mainly in the HBV life cycle, but drugs that target viral protein might be effective in inhibiting HBV infection. A recent study showed that polyamine inhibitor DFMO impeded HBV core-related DNA synthesis (reduced 34%) by reducing HBc protein levels at 84% and viral capsids.29 Followed by replenishing exogenous polyamines, the HBc protein and capsids levels significantly rescued, suggesting that polyamines may promote HBc protein levels and HBV replication.
Dengue Virus
Dengue virus (DENV) is transmitted mainly through vector insects such as Aedes aegypti and Aedes albopictus, causing dengue hemorrhagic fever and Dengue-shock syndrome with circulatory collapse, which have high morbidity and mortality rates.30 It was reported that DENV was sensitive to polyamine depletion by DFMO or DENspm that reduced viral titers by 60-fold.16 To understand the mechanism of polyamine metabolizing enzyme SAT1 on virus inhibition, Pozzi et al31 found that protein RBM10 accelerating SAT1 splicing factor exon 4 skipping had lower expression after DENV infection due to RBM10 interaction with Dengue polymerase NS5. In contrast, overexpressed RBM10 restrained virus replication by preventing SAT1 from splicing, which uncovered the antiviral effect of RBM10 targeting SAT1.
Other Viruses
Flaviviruses, such as Japanese encephalitis virus (JEV) and yellow fever virus (YFV), are transmitted by arthropods, in particular mosquito and ticks. Clinical manifestation caused by these viruses infection are usually mild, but the severe infections can be fatal; for example, JEV infection leads to gradually acute encephalitis with persistent fever and deep coma.32 Severe YFV infection results in internal bleeding, dehydration, and acidosis.33 The infection of the viruses might be closely related to polyamine metabolism. When JEV incubated after 24 hours infection of BHK-21 cells and YFV at 96 hpi, depletion polyamines by DFMO or DENspm reduced viral titers of JEV and YFV by 5-fold and 90-fold, respectively (seen in Table 1).16 The virus titers were rescued by the addition of exogenous polyamines.
Filoviridae
Ebola Virus
Ebola virus (EBOV) has high morbidity and mortality in humans and primates. There have been several major epidemics since its discovery, mainly in Africa, for example, there were 3,707 cases, including 1,848 deaths between 2013 and August 2014.3 EBOV infections causes severe bleeding symptoms, eg, hemorrhagic conjunctivitis, gingival bleeding, and blood stool, which occurs 4–5 days after onset. Olsen et al34 reported that the blockade of polyamine synthesis and the inhibition of hypusination of eukaryotic initiation factor 5A (eIF5A) prevented EBOV replication. EBOV was an inhibited infectious virus replication (3-log viral titers reduction) by DFMO inhibiting ODC1 and MDL blocking SMOX and PAOX which decreased the level of spermidine. Besides, CPX and DEF (inhibitors of DOHH) hindering the hypusination of eIF5A reduced EBOV gene expression and viral infection by 61% and 90%, respectively. Most importantly, antihypusination drug GC7 (inhibitor of DHPS) obstructed the expression of EBOV by decreasing the level of VP30 protein accumulation. Olsen et al35 further showed that polyamines supported the virus gene transcription/mRNA accumulation and the translation of EBOV mRNAs were supported by introducing spermidine into the translation factor eIF5A which promoted translation elongation by inhibiting eIF5A hypusination which resulted in a defect in the gene expression of EBOV at the level of transcription, translation, or protein stability.
Marburgvirus
Marburgvirus (MARV) is highly contagious and manifests symptoms of high fever, diarrhea, vomiting, and severe bleeding in the body.36 There is no cure at present. A study showed that CPX, a antihypusination compound, reduced MARV glycoprotein expression by at least 85% and declined MARV infectious titers by almost 3-log units in HepG2 cells,34 which suggested that inhibiting hypusination of eIF5A which played roles in the extension phase of protein synthesis, especially in stimulating the production of proteins containing continuous proline residues restrained the replication of infectious MARV.
Togaviridae
Chikungunya Virus
Chikungunya virus (CHIKV), an acute infectious pathogen, is transmitted to humans by mosquitoes, which cause sustained high fever and severe pain in the joints of the limbs and spine.37 But there is no specific drug therapy for the viral infection. Guan et al38 showed that aaNAT5b, a molecular and biochemical identification of SAT (a key catabolic enzyme), was vigorous to spermine and spermidine. The key residues of aaNAT5b were involved in spermine binding in the virus infection model, which indicated that polyamines were required in CHIKV replication. In addition, depletion of spermidine and spermine with DENSpm via IFN I signaling-mediated induction of SAT1 suppressed the virus replication.20 As mentioned previously, IFN I played a crucial role in upregulating SAT1. Viral titers were enhanced when the expression of SAT1 was knockout. These findings indicated that SAT1 played a key role in the disease progression of CHIKV infection, and the development of SAT1 drug could control CHIKV infection. However, CHIKV has appeared to develop resistance to polyamine deletions through two mutations in the non-structural protein 1 (nsP1).39 The mutations resulted in the increase of viral replication not only in the polyamine depleted cells but also in vivo. In a C57/Bl6 mice model provided with DFMO and infected with CHIKV, there were statistically significant changes in viral titer in tissues (like liver, lung, and kidney) and it tended to be lower.16 The finding suggested that it is likely for DFMO suppressed polyamines in vivo to have the potential to reduce CHIKV replication.
Sindbis Virus
Sindbis virus (SINV) is one of the arboviruses, and is one the alphavirusus in the family of togaviridae. SINV infection has major symptoms of fever, joint pain, rash, and encephalitis.40 SINV was suppressed by the DFMO induced polyamine depletion in a dose-dependent manner in BHK-21 infection cells.16 More importantly, DFMO was effective in a mammalian model. It was reported that the addition of several model organisms (such as Drosophila and zebrafish systems) with DFMO restricted SINV infection.20 The finding indicated that DFMO could be a candidate drug for SINV infection.
Semliki Forest Virus
Semliki Forest virus (SFV) is one of the alphaviruses, which belong to the alphavirus-like superfamily containing multiple +RNA virus families from plants and animals. SFV is also an arthropod-borne that induces host cell membrane depression.41 Tuomi et al42,43 insisted that polyamines were required to maintain the maximum initial rate of SFV replication and polyamine deficiency by DFMO-treatment and could significantly decrease the activity of viral RNA polymerase. More importantly, the activity of viral RNA polymerase but not the enzyme activity was rescued by external polyamines replenish, especially spermidine.
Herpesviridae
Epstein–Barr Virus
Epstein–Barr virus (EBV) is a DNA herpesvirus with oncogenic potential and global distribution and high infection rate in the population.44 The EBV infection is associated with several diseases, eg, infectious mononucleosis, hemophagocytic lymphohistiocytosis, and certain malignant lymphomas (Burkitt’s lymphoma (BL) and Hodgkin’s disease). Polyamines play an important role in the development of tumors, for example, polyamines promoted the expression of key components of the SCFSkp2 complex (ubiquitin ligase complex) and led to degradation of p27Kip1 and finally promoted cancer,45 which bring to assumption that EBV is closely related to polyamines. The SAT1 activity was decreased in EBV-infected cells, which increased the levels of polyamines and the occurrence of BL.46 On the contrary, it could enhance infection of EBV by increasing the activity of SAT1 with DEMspm treatment.
Herpes Simplex Virus
Herpes simplex virus (HSV) is highly prevalent in populations worldwide, and humans are the only natural reservoir of HSV. HSV-1 mainly causes the infection of skin, mucous membrane (oral mucous membrane) and organs (brain) and HSV-2 mainly infects the genital area skin mucous membrane.47 Based on the frequent recurrence of HSV and emergence of drug-resistant virus strains, it is urgent to develop antiviral drugs to inhibit the spread of virus. A study reported that the virions in HSV infected cells contained more spermidine and spermine. Besides, the suppression of polyamine with DFMO treatment resulted in a reduction in HSV DNA,48 which led to a decrease in the infectivity of HSV.49 Greco et al50 discovered that spermidine and spermine, component of HSV-1 virions, were involved in the process of viral infection. Subsequently, addition of MGBG targeting SAMDC effectively suppressed the virus infection.
Human Cytomegalovirus
Human cytomegalovirus (HCMV) infection is very common in people and can be contracted by close contact and sexual contact between people with the virus. Although the majority of patients with HCMV infection are asymptomatic, about 20% of them developed neurological deficits. The HCMV infection in immunocompromised patients have more severe outcomes. The infection suppressed cellular immunity in the body by decreasing the activity of killer T cells and NK cells.51 A study prepared human foreskin fibroblast (HFF) cells for the virus infected model,52 and another one used human diploid fibroblasts (MRC5) cells to infect with HCMV.53 The intracellular polyamine (spermidine and spermine) levels reduced by DFMO in both CMV-infected cells. And they reported that polyamine-depletion with DFMO supplement reduced infectious HCMV replication by 6-log and 4-log units, respectively52,53 (Table 1). The antiviral effect of polyamine depletion was rescued with exogenous polyamines addition.53 The finding implied that polyamines were essential for viral replication. In this way, drugs suppressing polyamines could be an approach to an-HCMV infection.
Rhabdoviridae
Vesicular stomatitis virus (VSV) is mainly distributed in South Africa and the Americas as well as in hot and humid areas. It infects humans and animals such as cows and pigs.54 Rabies virus (RABV) is also a member of the rhabdoviridae family, but different than VSV, without complications or deaths in humans, the case of RABV fatality rate is 100% after being bitten by a sick or poisonous animal with RABV.55 As shown in Table 1, running out of polyamines through DFMO or DENSpm could significantly decline the infectious viral production by 20-fold in VSV and 2-fold in RABV.16 When it treated Vero-E6 cells with DFMO to infection with VSV in combination with IFNβ, the viral titers were synergistically reduced. It means that DFMO combined with IFNβ may be a potential therapeutic intervention of these viruses.
Bunyaviridae
Bunyaviruses such as Rift Valley fever virus (RVFV) and La Crosse virus (LACV) are emerging arthropod-borne viruses that cause serious symptoms. RVFV, mainly endemic in Africa and the Middle East, is transmission to humans through mosquito bites or contact with infected animals. RVFV infection causes encephalitis, meningitis, and hemorrhagic fever in humans.56 Similar to RVFV, LACV also cause encephalitis following infection transmitted by mosquito bite, and is prevalent in the Midwest and mid-Atlantic US states.57 Bunyaviruses were sensitive to polyamine depletion through either DFMO or DENSpm.58,59 There were significant declines in viral titers over a series of infections and there was a meaningful increase in the number of viral genomes per infectious virus. However, polyamine depletion had no physical effect on the virions produced from infected cells, which revealed that RVFV and LACV in the infected cells with polyamine depletion produced non-infectious particles. Mastrodomenico et al58 thought that the noninfectious particles interfered with virus reproduction and activated type I interferon signaling to resist viral infection in the infected cells. They also showed that RVFV and LACV replication was promoted by polyamines, especially spermidine, that bound to purified virions and transmitted from one cell to another during infection.59 Therefore, polyamines may have a unique role in maintaining Bunyaviruses infectivity.
Retroviridae
Human immunodeficiency virus (HIV) is transmitted through sexual contact, blood, and mother-to-child transmission, infecting tens of millions of patients globally.60 HIV not only makes it difficult for the body’s immune system to resist attack, but also brings difficulties in the development of specific therapeutic drugs and preventive vaccines. Eukaryotic initiation factor 5A (elF5A) is essential for cell viability and an important component of cellular protein.9 Besides, the process of elF5A-hypusine requires the participation of the spermidine (seen in Figure 1). Studies have shown that polyamines may indirectly affect HIV replication, and elF5A was essential for viral gene expression.61–63 HIV-1 Rev was required for elF5A to function in HIV-1 mRNA translation and viral production. And there was a decrease in virus production by inhibiting RNA translation and Rev-induced gene expression through HIV-1 siRNA interfering with the formation of elF5A-hypusine, which implied that activation of the hypusine of elF5A to the viral RNA transcripts was likely mandated by their specific interaction with the HIV-1 Rev protein.63 The hypusine of elF5A was involved in the process of polyamines (spermidine) metabolism. Another study claimed that CPX and DEF, the inhibitors of DOHH (the key anabolic enzyme of elF5A-hypusine), prevented the maturation of eIF5A and reduced the levels of viral RNAs in the nucleus and cytoplasm by 2–3-fold, suggested that CPX and DEF may have potential as an antiviral drug.62 Further, supplements of CPX and DEF suppressed the expression of HIV-1 promoter at transcription initiation. The virus replication was inhibited (>93%) in viral infection cells expressing the eIF5A mutants which blocked HIV-1 Rev trans-activation and Rev-mediated nuclear output.61 The findings supported the importance of eIF5A and hypusine formation in HIV-1 gene expression, indicating that inhibition of the elF5A-hypusine related pathway may have potential effect for anti-HIV infection.
Coronaviridae
Coronavirus Middle East respiratory syndrome virus (MERS-CoV), a novel coronavirus, is mainly found in Saudi Arabia. The spread of MERS-CoV is from person-to-person and community-based.64 The common clinical manifestations of MERS-CoV infection are fever accompanied by chills, cough, shortness of breath, and muscle soreness. However, there is no vaccine to prevent MERS yet. An experiment showed that MERS-CoV was infected in Vero81 cells and treated with DFMO, and the viral titers were reduced 30-fold, which suggested that MERS-CoV was sensitive in depletion-polyamine with DFMO supplement.16 Following the supplement of exogenous polyamine, the viral titers were rescued. It appears that the virus relies on polyamines for its replication and the inhibition of polyamines has an anti-MERS-CoV effect.
Since December 2019, a series of unexplained pneumonia cases have been reported in Wuhan, China.65 On January 12, 2020, the World Health Organization (WHO) temporarily named the new virus 2019 New Coronavirus (2019-nCoV). On February 11, 2020, the World Health Organization officially named the disease caused by 2019-nCoV as SARS-CoV-2. Now, the SARS-CoV-2 epidemic is spreading worldwide. It was reported that endogenous polyamines inhibited innate immune responses by blocking monocyte function.66 Besides, the coronavirus destroyed macrophages and thus attenuated the innate immune response.67 Therefore, we speculate that targeting polyamine metabolism may be a useful tool for controlling SARS-CoV-2, although there are no reports on polyamine metabolism and SARS-CoV-2.
Conclusion
The polyamines play critical roles in viral infection, replication, and reproduction. In recent years, more and more studies have been conducted on the effect of targeting polyamines as well as the pathway in viral infection, suggesting that the important role of polyamines in the occurrence and development of viral replication has been recognized. Viruses may reveal mechanisms for using polyamines in a similar manner. Because such a diverse group of viruses rely on polyamines for replication, it may be due to the more general function of polyamines. Additionally, polyamines are important for cellular translation via the hypusine of eIF5A. In this review, several human viruses such as CVB3, EV71, PV, ZKV, HCV, DENV, JEV, YFV, EBOV, MARV, CHIKV, SINV, SFV, EBV, HSV, HCMV, VSV, RABV, RVFV, LACV, HIV, and MERS-CoV that were or are still circulating were closely related to polyamines. Therefore, targeting the polyamine pathway in viral infection is of great value for diseases to develop antiviral drugs. However, most of the compounds targeting polyamine metabolism are still under preliminary research, and more research is needed to determine their potential clinical antiviral effects. Existing studies needs to pay attention to the precise mechanism of polyamines in virus replication and how they participate in the immune response of different organisms, so as to further study the role of polyamines in viral infections and provide a basis for revealing the important role of developing antiviral drugs that target the polyamine pathway.
Acknowledgments
This study was supported by grants from the Natural Science Foundation of Guangdong Province (2019A1515011713), the Medical Science Foundation of Guangdong Province (A2020425), “Group-type” Special Supporting Project for Educational Talents in Universities (4SG19221,4SG19218), Research Foundation of Guangdong Medical University for Ph.D. Staff (B2019038) and the Key Cultivation Project of Guangdong Medical University (Z2016005).
Disclosure
The authors have no conflicts of interest to declare.
References
1. Davis BM, Rall GF, Schnell MJ. Everything you always wanted to know about rabies virus (but were afraid to ask). Annu Rev Virol. 2015;2(1):451–471. doi:10.1146/annurev-virology-100114-055157
2. Cilliers K, Muller CJF, Page BJ. Human immunodeficiency virus in cadavers: a review. Clin Anat. 2019;32(4):603–610. doi:10.1002/ca.23358
3. Jadav SS, Kumar A, Ahsan MJ, Jayaprakash V. Ebola virus: current and future perspectives. Infect Disord Drug Targets. 2015;15(1):20–31. doi:10.2174/1871526515666150320162259
4. Sohrabi C, Alsafi Z, O’Neill N, et al. World Health Organization declares global emergency: a review of the 2019 novel coronavirus (COVID-19). Int J Surg. 2020;76:71–76. doi:10.1016/j.ijsu.2020.02.034
5. Florin L, Lang T. Tetraspanin assemblies in virus infection. Front Immunol. 2018;9:1–9. doi:10.3389/fimmu.2018.01140
6. Miller-Fleming L, Olin-Sandoval V, Campbell K, Ralser M. Remaining mysteries of molecular biology: the role of polyamines in the cell. J Mol Biol. 2015;427(21):3389–3406. doi:10.1016/j.jmb.2015.06.020
7. Igarashi K, Kashiwagi K. Modulation of protein synthesis by polyamines. IUBMB Life. 2015;67(3):160–169. doi:10.1002/iub.1363
8. Hesterberg RS, Cleveland JL, Epling-Burnette PK. Role of polyamines in immune cell functions. Med Sci (Basel). 2018;6(22):1–19.
9. Dever TE, Gutierrez E, Shin BS. The hypusine-containing translation factor eIF5A. Crit Rev Biochem Mol Biol. 2014;49(5):413–425. doi:10.3109/10409238.2014.939608
10. Puleston DJ, Buck MD, Klein Geltink RI, et al. Polyamines and eIF5A hypusination modulate mitochondrial respiration and macrophage activation. Cell Metab. 2019;30(2):352–363.e358. doi:10.1016/j.cmet.2019.05.003
11. Mounce BC, Olsen ME, Vignuzzi M, Connor JH. Polyamines and their role in virus infection. Microbiol Mol Biol Rev. 2017;81(4):e00029–00017. doi:10.1128/MMBR.00029-17
12. Rivadeneyra L, Charó N, Kviatcovsky D, et al. Role of neutrophils in CVB3 infection and viral myocarditis. J Mol Cell Cardiol. 2018;125:149–161. doi:10.1016/j.yjmcc.2018.08.029
13. Han Z, Zhang Y, Huang K, et al. Two coxsackievirus B3 outbreaks associated with hand, foot, and mouth disease in China and the evolutionary history worldwide. BMC Infect Dis. 2019;19(1):1–10. doi:10.1186/s12879-019-4107-z
14. Kicmal TM, Tate PM, Dial CN, et al. Polyamine depletion abrogates enterovirus cellular attachment. J Virol. 2019;1–37.
15. Dial CN, Tate PM, Kicmal TM, Mounce BC. Coxsackievirus B3 responds to polyamine depletion via enhancement of 2A and 3C protease activity. Viruses. 2019;11(403):1–15. doi:10.3390/v11050403
16. Mounce BC, Cesaro T, Moratorio G, et al. Inhibition of polyamine biosynthesis is a broad-spectrum strategy against RNA viruses. J Virol. 2016;90(21):9683–9692. doi:10.1128/JVI.01347-16
17. Chen Z, Li R, Xie Z, et al. IL-6, IL-10 and IL-13 are associated with pathogenesis in children with enterovirus 71 infection. Int J Clin Exp Med. 2014;7(9):2718–2723.
18. Ma Y, Wang X, Zhou X, et al. Poliovirus detection and genetic characteristic from sewage in Heilongjiang province from 2013 to 2016. Jpn J Infect Dis. 2018;71(6):442–447. doi:10.7883/yoken.JJID.2017.338
19. Musso D, DJ G. Zika virus. Clin Microbiol Rev. 2016;29(3):487–524. doi:10.1128/CMR.00072-15
20. Mounce BC, Poirier EZ, Passoni G, et al. Interferon-induced spermidine-spermine acetyltransferase and polyamine depletion restrict zika and chikungunya viruses. Cell Host Microbe. 2016;20(2):167–177. doi:10.1016/j.chom.2016.06.011
21. Routhu NK, Xie Y, Dunworth M, et al. Polymeric prodrugs targeting polyamine metabolism inhibit zika virus replication. Mol Pharm. 2018;1–45.
22. de Weerd NA, Vivian JP, Nguyen TK, et al. Structural basis of a unique interferon-β signaling axis mediated via the receptor IFNAR1. Nat Immunol. 2013;14(9):901–907. doi:10.1038/ni.2667
23. Henig N, Avidan N, Mandel I, et al. Interferon-beta induces distinct gene expression response patterns in human monocytes versus T cells. PLoS One. 2013;8(4):e62366. doi:10.1371/journal.pone.0062366
24. Zeisel MB, Felmlee DJ, Baumert TF. Hepatitis C virus entry. Curr Top Microbiol Immunol. 2013;369:87–112.
25. Korovina AN, Tunitskaya VL, Khomutov MA, et al. Biogenic polyamines spermine and spermidine activate RNA polymerase and inhibit RNA helicase of hepatitis C virus. Biochemistry. 2012;77(10):1413–1422. doi:10.1134/S0006297912100094
26. Smirnova OA, Keinanen TA, Ivanova ON, et al. Hepatitis C virus alters metabolism of biogenic polyamines by affecting expression of key enzymes of their metabolism. Biochem Biophys Res Commun. 2017;483(2):904–909. doi:10.1016/j.bbrc.2017.01.032
27. Masalova OV, Lesnova EI, Samokhvalov EI, et al. Low-molecular-weight regulators of biogenic polyamine metabolism affect cytokine production and expression of hepatitis С virus proteins in Huh7.5 human hepatocarcinoma cells. Mol Biol. 2017;51(3):453–464. doi:10.1134/S0026893317030128
28. Chen Y, Tian Z. HBV-induced immune imbalance in the development of HCC. Front Immunol. 2019;10:2048. doi:10.3389/fimmu.2019.02048
29. Mao B, Wang Z, Pi S, et al. Difluoromethylornithine, a decarboxylase 1 inhibitor, suppresses hepatitis B virus replication by reducing hbc protein levels. Front Cell Infect Microbiol. 2020;10:158. doi:10.3389/fcimb.2020.00158
30. Muller DA, Depelsenaire ACI, Young PR. Clinical and laboratory diagnosis of dengue virus infection. J Infect Dis. 2017;215(suppl_2):S89–S95. doi:10.1093/infdis/jiw649
31. Pozzi B, Bragado L, Mammi P, et al. Dengue virus targets RBM10 deregulating host cell splicing and innate immune response. Nucleic Acids Res. 2020;48(12):6824–6838. doi:10.1093/nar/gkaa340
32. Wang X, Li S-H, Zhu L, et al. Near-atomic structure of Japanese encephalitis virus reveals critical determinants of virulence and stability. Nat Commun. 2017;8(1):14. doi:10.1038/s41467-017-00024-6
33. Klitting R, Roth L, Rey FA, de Lamballerie X. Molecular determinants of yellow fever virus pathogenicity in Syrian golden hamsters: one mutation away from virulence. Emerg Microbes Infect. 2018;7(1):51. doi:10.1038/s41426-018-0053-x
34. Olsen ME, Filone CM, Rozelle D, et al. Polyamines and hypusination are required for ebolavirus gene expression and replication. mBio. 2016;7(4):e00882–00816. doi:10.1128/mBio.00882-16
35. Olsen ME, Cressey TN, Muhlberger E, Connor JH. Differential mechanisms for the involvement of polyamines and hypusinated eif5a in ebola virus gene expression. J Virol. 2018;1–34.
36. Amman BR, Nyakarahuka L, McElroy AK, et al. Marburgvirus resurgence in kitaka mine bat population after extermination attempts, Uganda. Emerg Infect Dis. 2014;20(10):1761–1764. doi:10.3201/eid2010.140696
37. Weaver SC, Lecuit M. Chikungunya virus and the global spread of a mosquito-borne disease. N Engl J Med. 2015;372(13):1231–1239. doi:10.1056/NEJMra1406035
38. Guan H, Wang M, Liao C, et al. Identification of aaNAT5b as a spermine N-acetyltransferase in the mosquito, Aedes aegypti. PLoS One. 2018;13(3):e0194499. doi:10.1371/journal.pone.0194499
39. Mounce BC, Cesaro T, Vlajnić L, et al. Chikungunya virus overcomes polyamine depletion by mutation of nsP1 and the opal stop codon to confer enhanced replication and fitness. J Virol. 2017;91(15):1–45. doi:10.1128/JVI.00344-17
40. Sigei F, Nindo F, Mukunzi S, Ng’ang’a Z, Sang R. Evolutionary analyses of Sindbis virus strains isolated from mosquitoes in Kenya. Arch Virol. 2018;163(9):2465–2469. doi:10.1007/s00705-018-3869-8
41. Pietilä MK, Albulescu IC, MJv H, Ahola T. Polyprotein processing as a determinant for in vitro activity of semliki forest virus replicase. Viruses. 2017;9(10):292. doi:10.3390/v9100292
42. Tuomi K, Mantyjarvi R, Raina A. Inhibition of Semliki Forest and herpes simplex virus production in alpha-difluoromethylornithinetreated cells: reversal by polyamines. FEBS Lett. 1980;121(2):292–294. doi:10.1016/0014-5793(80)80365-6
43. Tuomi K, Rainat A, Mantyjarvi R. Synthesis of Semliki-Forest virus in polyamine-depleted baby-hamster kidney cells. Biochem Soc. 1982;206:113–119.
44. Nowalk A, Green M. Epstein-barr virus. Microbiol Spectr. 2016;4(3):
45. Casero RA
46. Shi M, Gan Y-J, Davis TO, Scott RS. Downregulation of the polyamine regulator spermidine/spermine N1-acetyltransferase by epstein–barr virus in a Burkitt’s lymphoma cell line. Virus Res. 2013;177(1):11–21. doi:10.1016/j.virusres.2013.07.004
47. Kumru OS, Joshi SB, Thapa P, et al. Characterization of an oncolytic herpes simplex virus drug candidate. J Pharm Sci. 2015;104(2):485–494.
48. Gibson W, Roizman B. Compartmentalization of spermine and spermidine in the herpes simplex virion. Proc Nat Acad Sci USA. 1971;68(11):2818–2821. doi:10.1073/pnas.68.11.2818
49. Pohjanpelto P, Sekki A, Hukkanen V, Bonsdorff C-HV. Polyamine depletion of cells reduces the infectivity of herpes simplex virus but not the infectivity of virus. Life Sci. 1988;42(20):2011–2018. doi:10.1016/0024-3205(88)90501-2
50. Greco A, Callé A, Morfin F, et al. S-adenosyl methionine decarboxylase activity is required for the outcome of herpes simplex virus type 1 infection and represents a new potential therapeutic target. FASEB J. 2005;19(9):1128–1130. doi:10.1096/fj.04-2108fje
51. Lv Y-L, Han -F-F, Gong -L-L, et al. Human cytomegalovirus infection and vascular disease risk: a meta-analysis. Virus Res. 2017;227:124–134. doi:10.1016/j.virusres.2016.09.010
52. Tyms AS, Williamson JD. Inhibitors of polyamine biosynthesis block human cytomegalovirus replication. Nature. 1982;297(5868):690–691. doi:10.1038/297690a0
53. Gibson W, Breemen RV, Fields A, Lafemina R, Irmiere A. D,L-alpha-difluoromethylornithine inhibits human cytomegalovirus replication. J Virol. 1984;50(1):145–154. doi:10.1128/JVI.50.1.145-154.1984
54. Rodríguez LL. Emergence and re-emergence of vesicular stomatitis in the United States. Virus Res. 2002;85(2):211–219. doi:10.1016/S0168-1702(02)00026-6
55. Correa-Giron EP, Allen R, Sulkin SE. The infectivity and pathogenesis of rabiesvirus administered orally. Am J Epidemiol. 1970;91(2):203–215.
56. Linthicum KJ, Britch SC, Anyamba A. Rift valley fever: an emerging mosquito-borne disease. Annu Rev Entomol. 2016;61(1):395–415. doi:10.1146/annurev-ento-010715-023819
57. McJunkin JE, Khan RR, Tsai TF. California-la crosse encephalitis. Infect Dis Clin North Am. 1998;12(1):83–93. doi:10.1016/S0891-5520(05)70410-4
58. Mastrodomenico V, Esin JJ, Graham ML, et al. Polyamine depletion inhibits bunyavirus infection via generation of noninfectious interfering virions. J Virol. 2019;93(14):e00530–00519. doi:10.1128/JVI.00530-19
59. Mastrodomenico V, Esin J, Qazi S, et al. Virion-associated polyamines transmit with bunyaviruses to maintain infectivity and promote entry. ACS Infect Dis. 2020;6(9):2490–2501. doi:10.1021/acsinfecdis.0c00402
60. Margolis DM, Archin NM, Latency P. Persistent human immunodeficiency virus infection, and the development of latency reversing agents. J Infect Dis. 2017;215(suppl_3):S111–S118. doi:10.1093/infdis/jiw618
61. Bevec D, Jaksche H, Oft M, et al. Inhibition of HIV-1 replication in lymphocytes by mutants of the rev cofactor eIF-5A. Science. 1996;271(5257):1858–1860. doi:10.1126/science.271.5257.1858
62. Hoque M, Hanauske-Abel HM, Palumbo P, et al. Inhibition of HIV-1 gene expression by ciclopirox and deferiprone, drugs that prevent hypusination of eukaryotic initiation factor 5A. Retrovirology. 2009;6(1):90. doi:10.1186/1742-4690-6-90
63. Liu J, Henao-Mejia J, Liu H, Zhao Y, He JJ. Translational regulation of HIV-1 replication by HIV-1 rev cellular cofactors Sam68, eIF5A, hRIP, and DDX3. J Neuroimmune Pharmacol. 2011;6(2):308–321. doi:10.1007/s11481-011-9265-8
64. Chafekar A, Fielding BC. MERS-CoV: understanding the latest human coronavirus threat. Viruses. 2018;10(2):93.
65. Palacios Cruz M, Santos E, Velázquez Cervantes MA, León Juárez M. COVID-19, a worldwide public health emergency. Rev Clin Esp. 2020.
66. Zhang M, Lyudmila V, Borovikova HW, Metz C, Tracey KJ. Spermine inhibition of monocyte activation and inflammation. Mol Med. 1999;5(9):595–605. doi:10.1007/BF03402072
67. Qin C, Zhou L, Hu Z, et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin Infect Dis. 2020.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
