Back to Journals » International Journal of Nanomedicine » Volume 12
Targeted concurrent and sequential delivery of chemotherapeutic and antiangiogenic agents to the brain by using drug-loaded nanofibrous membranes
Authors Tseng YY , Yang TC, Wang YC, Lee WH, Chang TM, Kau YC, Liu SJ
Received 13 October 2016
Accepted for publication 2 December 2016
Published 14 February 2017 Volume 2017:12 Pages 1265—1276
DOI https://doi.org/10.2147/IJN.S124593
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Lei Yang
Yuan-Yun Tseng,1,2 Tao-Chieh Yang,3 Yi-Chuan Wang,4 Wei-Hwa Lee,5 Tzu-Min Chang,4 Yi-Chuan Kau,6 Shih-Jung Liu4,7
1Division of Neurosurgery, Department of Surgery, Shuang Ho Hospital, 2Department of Surgery, School of Medicine, College of Medicine, Taipei Medical University, Taipei, 3Department of Neurosurgery, Asia University Hospital, Taichung, 4Department of Mechanical Engineering, Chang Gung University, Taoyuan, 5Department of Pathology, Shuang Ho Hospital, Taipei Medical University, Taipei, 6Department of Anesthesiology, 7Department of Orthopedic Surgery, Chang Gung Memorial Hospital, Taoyuan, Taiwan
Abstract: Glioblastoma is the most frequent and devastating primary brain tumor. Surgery followed by radiotherapy with concomitant and adjuvant chemotherapy is the standard of care for patients with glioblastoma. Chemotherapy is ineffective, because of the low therapeutic levels of pharmaceuticals in tumor tissues and the well-known tumor-cell resistance to chemotherapy. Therefore, we developed bilayered poly(d,l)-lactide-co-glycolide nanofibrous membranes that enabled the sequential and sustained release of chemotherapeutic and antiangiogenic agents by employing an electrospinning technique. The release characteristics of embedded drugs were determined by employing an in vitro elution technique and high-performance liquid chromatography. The experimental results showed that the fabricated nanofibers showed a sequential drug-eluting behavior, with the release of high drug levels of chemotherapeutic carmustine, irinotecan, and cisplatin from day 3, followed by the release of high concentrations of the antiangiogenic combretastatin from day 21. Biodegradable multidrug-eluting nanofibrous membranes were then dispersed into the cerebral cavity of rats by craniectomy, and the in vivo release characteristics of the pharmaceuticals from the membranes were investigated. The results suggested that the nanofibrous membranes released high concentrations of pharmaceuticals for more than 8 weeks in the cerebral parenchyma of rats. The result of histological analysis demonstrated developmental atrophy of brains with no inflammation. Biodegradable nanofibrous membranes can be manufactured for long-term sequential transport of different chemotherapeutic and antiangiogenic agents in the brain, which can potentially improve the treatment of glioblastoma multiforme and prevent toxic effects due to systemic administration.
Keywords: nanofibrous membrane, poly(d,l)-lactide-co-glycolide (PLGA), glioblastoma multiforme (GBM), chemotherapy, targeted therapy, antiangiogenesis
Introduction
Gliomas are the most frequent intracranial tumors and account for more than 50% of all primary brain tumors, with glioblastoma multiforme (GBM) being by far the most common and aggressive.1–3 GBM presents severe management challenges: it is difficult to treat, devastating in its progressive and disabling manifestations, and highly lethal, with a median survival of 9–12 months.3,4 At present, surgical debulking followed by radiotherapy with concomitant and adjuvant chemotherapy with temozolomide is the standard of care for patients with GBM aged <70 years.4,5 Despite advances in surgical and medical technologies, the prognosis for patients with GBM remains poor, with recurrence of GBM being almost universal. In addition, GBM has been long considered chemoresistant, because the blood–brain barrier (BBB) and systemic toxicity present major limitations to the systemic administration of chemotherapeutic drugs.6,7 Although inadequate drug delivery may limit the efficacy of intracerebral chemotherapy, treatment failure occurs primarily because of tumor resistance to chemotherapeutic agents.8,9 Therefore, a degradable delivery device that provides targeted multiple adjuvant chemotherapeutic agents with differing antitumor mechanisms to the target site for preventing tumor resistance is highly desired.10,11
In this study, we developed bilayered poly(D,L)-lactide-co-glycolide (PLGA) nanofibers that enabled the sequential and sustained release of multiple anticancer pharmaceuticals to the brain parenchyma. Three chemotherapeutic agents (carmustine [bis-chloroethylnitrosourea {BCNU}], irinotecan, and cisplatin) with differing antitumor mechanisms were incorporated into 50:50 PLGA nanofibers, and an antiangiogenic agent (combretastatin) was loaded onto 75:25 PLGA nanofibers. BCNU was the first tested and approved drug for treating GBM, and has yielded modest improvements in patient survival since the 1960s. Belonging to the class of nitrosoureas, BCNU alkylates the O6-guanine position and cross-links the DNA, thereby inhibiting cancer growth.12 Cisplatin produces a strong cytotoxic effect, especially when administered directly into a tumor, for the treatment of malignant gliomas. The existence of the BBB, however, rather limits drug absorption by the perifocal tumor area.13,14 Conversely, irinotecan is a camptothecin derivative that restrains topoisomerase I. It is currently approved for treating metastatic colorectal cancer.3 Irinotecan, which crosses the BBB, is active against a range of central nervous system (CNS) tumor xenografts in animal models.3,15 Previous reports have demonstrated its anticancer effect toward human glioblastoma cells that exhibit resistance for multiple drugs.3,15,16 Therefore, we developed biodegradable multidrug-eluting nanofibers able to release various chemotherapeutic agents concurrently.
Furthermore, GBM characteristically exhibits angiogenesis (neovascularization). The development of antiangiogenic therapeutic strategies for treating patients with malignant glioma tumors has garnered considerable attention.17,18 Combretastatin is a vascular system-disrupting agent that has a high affinity for tubulin and destabilizes tubulin polymers of the cytoskeleton. It selectively damages such tumoral vasculature, particularly its capillary endothelium, causing vascular collapse and shutdown, depriving the tumor of blood and oxygen supply, and subsequently leading to rapid hemorrhagic necrosis inside the tumor.19,20
After electrospinning, the release behaviors of the pharmaceuticals were evaluated using an in vitro elution technique and a high-performance liquid chromatography (HPLC) assay. The multidrug-eluting nanofibrous membranes were positioned on the brain parenchyma by craniectomy. The in vivo pharmaceutical release patterns of the membranes were examined. Tissue reactions to the biodegradable drug-eluting nanofibers were histologically studied.
Materials and methods
Fabrication of pharmaceutical-embedded PLGA nanofibers
Polymeric materials, including 50:50 and 75:25 PLGA copolymers, and pharmaceuticals, including BCNU, irinotecan, cisplatin, and combretastatin (BICC), were purchased from Sigma-Aldrich (St Louis, MO, USA). To prepare the nanofibrous multidrug-eluting membranes, 50:50 PLGA–BCNU–irinotecan–cisplatin and 75:25 PLGA–combretastatin were separately dissolved in 1,1,1,3,3,3-hexafluoro-2-propanol (HFIP). Two polymer:drug weight ratios, 6:1 and 4:1, were employed in the experiments. The mixed PLGA–BIC solution was first electrospun into nanofibrous membranes, followed by the electrospinning of the PLGA–combretastatin solution. A previously developed lab-scale electrospinning device,21 which consists of a power supply, a needle with an internal diameter of 0.42 mm, a needle pump, a ground electrode, and a collection plate, was employed to fabricate the nanofibrous membranes. Before the experiments, a few test trials were completed to identify the optimal processing parameters. Table 1 lists the parameters applied to manufacture the nanofibrous membranes. To electrospin the nanofibers, PLGA materials and drugs were first dissolved in HFIP. The solution was then delivered and electrospun by a syringe pump with a volumetric flow rate of 1.8 mL/h to obtain nanofibrous membranes on the collection plate. The distance between the needle tip and the plate (ground electrode) was 12 cm, and the positive voltage applied to the polymer solutions was 17 kV. All electrospinning experiments were performed at room temperature. The total time for electrospinning a membrane was approximately 48 hours: 24 hours for each layer. The electrospun nanofibers were collected in unwoven membrane form on the collection plate. A dual-layered membrane with PLGA–BIC as the top layer and PLGA–combretastatin as the bottom layer was obtained; the membrane thus obtained was approximately 0.11 mm in thickness.
Scanning electron microscopy observations
The surface morphology of the prepared nanofibers was examined employing field-emission scanning electron microscopy (JAM-7500F; JEOL, Tokyo, Japan). The samples for observation were first sputter-coated with gold to render them electrically conductive before the experiments.
In vitro elution characteristics of chemotherapeutic agents
Membrane-release features of BICC were assessed employing an in vitro elution technique. Specimens (1×1 cm each) obtained from the multidrug-embedded membranes were deposited in glass test tubes (one specimen for each tube, n=3) containing 1 mL of a phosphate-buffered solution with a concentration of 0.15 mol/L and a pH value of 7.4. After being placed in an oven at 37°C for 24 hours, the solution in the glass tube was collected and replaced by 1 mL fresh phosphate-buffered solution for the following 24 hours. This procedure was repeated daily for 8 weeks. Drug concentrations in the collected solutions were evaluated employing an HPLC assay (L-2200 multisolvent delivery system; Hitachi, Tokyo, Japan).
To evaluate the concentrations of released BCNU, a Waters Symmetry C18 4.6×150 mm, 5 μm HPLC column was employed to separate the chemotherapeutic agents. Purified water (85/15, v/v) and methanol (Sigma-Aldrich) were adopted as the mobile phase. The monitoring absorbance was set at 230 nm, while the flow rate was 1 mL/min. To evaluate irinotecan, a Symmetry C18 4.6×250 mm column was employed. The mobile phase included acetonitrile (Mallinckrodt, Chesterfield, UK), 0.01 M potassium dihydrogen phosphate, and methanol in a volume ratio of 55:27:18. Monitoring absorbance was set at 225 nm, while the flow rate was 1.5 mL/min. To evaluate cisplatin, the Symmetry 4.6×250 mm column was used. The mobile phase contained acetonitrile, methanol (90/10, v/v), and distilled water. The absorbance was monitored and set at 300 nm, while the flow rate was 2 mL/min. Combretastatin was characterized using the Symmetry 4.6×150 mm, 5 μm HPLC column. The mobile phase consisted of methanol and distilled water (65/35, v/v). The monitoring absorbance was set at 300 nm, and the flow rate was 1 mL/min. A calibration curve for each drug was first determined by five standard concentrations (1, 10, 100, 500, and 1,000 μg/L). The released concentrations of the drugs were then determined by interpreting the curves. All experiments were repeated three times.
Surgical procedure
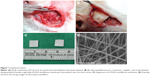
All animal experimental procedures received institutional approval (Taipei Medical University LAC-2013-0172), and all studied animals were cared for in line with the regulations of the Ministry of Health and Welfare of Taiwan under the supervision of a licensed veterinarian. In total, 70 adult Wistar rats weighing 200–300 g were treated for the in vivo drug-concentration experiments. All the rats were housed in standard facilities with no more than four rats per cage, and were given free access to water and pellets. The rats were anesthetized with an intraperitoneal injection that contained 6% chloral hydrate (0.6 mL/kg body weight), and were randomly divided into nine groups (3 days and 1–8 weeks each) containing six to seven rats. After the postorbital region was shaved and sterilized, a 1.5 cm scalp incision was made. After dissection of the scalp fascia and muscle by employing a scalpel, craniectomy (about 10×10 mm with a weight of approximately 20 mg) was performed using an electric burr (Figure 1A). After local hemostasis was attained, the BICC–PLGA nanofibrous membrane with a polymer:drug ratio of 4:1 was put on the surface of the brain parenchyma after craniectomy (Figure 1B). The incision was then closed with 3-0 nylon sutures. After recovering from anesthesia, the rats were returned to the housing facility. If any intraoperative brain injury or infection (including scalp, skull bone, and brain tissue) was observed in the rats, they were excluded from the study.
Chemotherapeutic agent pharmacokinetics
Overdoses of anesthesia were given to the rats intraperitoneally (over 1.2 mL/kg of body weight). Blood specimens were collected via syringes through heart puncture. The ipsilateral brain tissue beneath the BICC–PLGA nanofibrous membrane was also removed. The brain tissue (8×8 mm and 8–10 mm thick) was sliced into five layers (namely layers 1–5, from the surface beneath the membrane toward the center of the brain, with each layer approximately 1.5 mm thick) by employing a rodent-brain slicer (Zivic Instruments, Pittsburgh, PA, USA). Approximately 50 mg of the tissue from each sliced layer was collected. All samples (brain tissue and blood of rats) were gathered on day 3 and in each of weeks 1–8. Tissue specimens were sonically extracted for 20 seconds and then centrifuged, while plasma was collected and kept at −80°C before analysis. Drug concentrations in the samples were evaluated employing HPLC. Furthermore, the brain tissue at each time point was extirpated for histological analysis. Specimens were stained with hematoxylin and eosin and GFAP, and examined by light microscopy.
Statistical analysis
Experimental data were recorded as means ± standard deviation. Statistically significant difference analysis was completed by performing a paired-sample t-test with SPSS software (version 12.0; SPSS Inc, Chicago, IL, USA).
Results
Electrospinning drug-loaded PLGA solutions resulted in an ultrafine membrane with a smooth surface (Figure 1C). Field-emission scanning electron microscopy of the electrospun nanofibers (under a magnification of 20,000×) showed that the diameter of the 50:50 PLGA–BIC fibers was 270–850 nm, and the nanofibrous membrane was highly porous (Figure 1D). Meanwhile, the diameter of electrospun 75:25 PLGA–combretastatin nanofibers ranged from 580 nm to 1,120 nm. Both the 50:50 and 75:25 nanofibers exhibited a pore size of 1–3 μm and a porosity of approximately 70%.
In vitro release patterns of chemotherapeutic agents
Figure 2A presents the accumulated release curves of BCNU, irinotecan, cisplatin, and combretastatin from the biodegradable nanofibers with a polymer:drug ratio of 6:1. BCNU, irinotecan, and cisplatin were loaded onto 50:50 PLGA nanofibers, which enabled the sustained release of the drugs for 30 days. Furthermore, BCNU and cisplatin exhibited triphasic release profiles from the electrospun nanofibrous membranes, ie, an initial burst from days 0 to 3, a moderate release from days 4 to 14, and an accelerated release from days 15 to 30. Irinotecan exhibited a biphasic drug release behavior, ie, a rapid release before day 12 and a gradual release thereafter until day 30. Combretastatin was loaded onto 75:25 PLGA nanofibers and observed for 56 days. Only a small amount (less than 5%) of combretastatin was released from the nanofibers within the first 18 days. Thereafter, combretastatin was rapidly released from days 18 to 23. More than 50% of the combretastatin was released during this stage. In total, 82% of the drug was released by day 56. However, Figure 2B presents the release curves of electrospun nanofibers with a polymer:drug ratio of 4:1. The nanofibrous membranes exhibited similar release patterns (triphasic release profiles) to those of the 6:1 polymer:drug ratio.
During manufacturing, most drugs were embedded in the bulk of the PLGA matrix; nevertheless, some pharmaceuticals might not have been encapsulated completely by the polymeric matrix and were distributed on the surface of the membranes. This led to the burst release of the drugs. Following the burst elution, drug release was dominated mainly by diffusion. This might explain why all drugs exhibited an initial burst followed by a more gradual release. Finally, when polymer molecular weight decreases sufficiently, polymer loss begins. The drugs will then be released along with this polymer loss. The release rate was thus accelerated. In addition, since combretastatin was loaded onto 75:25 PLGA nanofibers made from polymers with higher molecular weight, it thus exhibited delayed and sustained drug release.
In vivo elution patterns of pharmaceuticals from nanofibers
In vivo drug concentrations were evaluated for 7 weeks employing HPLC. After excluding the rats that perished during surgery due to anesthesia overdose, vast blood loss, brain injury, or infections, a total of seven rats on postsurgery day (PSD) 3, seven rats on PSD 7 (week 1), six rats on PSD 14 (week 2), six rats on PSD 21 (week 3), seven rats on PSD 28 (week 4), seven rats on PSD 35 (week 5), seven rats on PSD 42 (week 6), seven rats on PSD 49 (week 7), and six rats on PSD 56 (week 8) were obtained for the subsequent drug-concentrations analysis. Figure 3A shows the drug-release curves of BCNU in different layers of the brain and plasma. The mean BCNU concentration in the brain was 48.63±48.15 μg/mL on PSD 3 and ranged from 27.51±4.5 to 134.37±72.85 μg/mL over 8 weeks. The average BCNU concentration value in the plasma on PSD 3 was 0.45±0.03 μg/mL. With the degradation of PLGA, the BCNU level had increased gradually to 2.62±0.26 μg/mL (also the highest concentration) in the plasma by the end of this study. The local drug levels at the target site (brain tissue) were all apparently greater than those in the plasma at any time point (P<0.001). Furthermore, in the in vitro investigation, the chemotherapeutic agents were completely released by day 28, whereas in the in vivo investigation the drug concentrations were still available on PSD 56. For all pharmaceuticals, the in vivo environment seemed to provide a slower metabolic rate than the in vitro environment does. This might explain why the total period of in vivo drug release was longer than that of in vitro drug release.
Figure 3B presents the release patterns of irinotecan in different layers of the brain and plasma. The irinotecan concentrations were high (59.97±44.78 μg/mL) on PSD 3, and ranged from 31.84±9.95 to 181.77±121.94 μg/mL over eight weeks. Drug level in the plasma was relatively low (0.22±0.03–0.77±0.04 μg/mL). Figure 3C shows the drug-release curves of cisplatin in different layers of the brain and plasma. The cisplatin level in the brain tissue quickly peaked (660.47±131.31 μg/mL) on PSD 3, and remained higher than 200 μg/mL during the first 7 weeks, followed by a minor decrease to 70.01±20.73 μg/mL at eight weeks. Again, cisplatin levels were markedly lower (0.67±0.13–3.35±0.54 μg/mL) in the plasma.
Figure 3D presents the release patterns of combretastatin in different layers of the brain and plasma. Combretastatin was loaded onto 75:25 PLGA nanofibers, which have a lower degradation rate than 50:50 PLGA nanofibers. The combretastatin concentration was relatively lower (10.69±3.2 μg/mL) than the concentration of other chemotherapeutic agents on PSD 3, and then was mildly decreased (6.88±1.09 μg/mL) in week 1. The drug concentration increased from week 3, and became maximal (274.68±51.99 μg/mL) in week 4. All drug levels (including BCNU, irinotecan, cisplatin, and combretastatin) were substantially higher in the brain than in the blood (systemic). The drug levels of chemotherapeutic agents differed significantly between the brain and plasma at different times (P<0.01).
Figure 3 also shows the concentrations of the chemotherapeutic (BCNU, irinotecan, and cisplatin) and antiangiogenic (combretastatin) drugs in different layers of the brain. As expected, due to drug diffusion, higher concentrations were achieved in the superficial layer (namely layers 1 and 2) of brain tissues for the first few weeks. Drug concentrations of BCNU in the superficial layer were greater than those in the deep region (ie, layers 4 and 5) of the brain during the first 3 weeks. This might have been due to the fact that as polymers degraded, the drugs were discharged and accumulated in the brain parenchyma. Drug levels in the deep layer were greater than those in the superficial layer from week 4 (Figure 3A). On the other hand, irinotecan levels in the superficial layer of the brain were greater than those in the deep region during the first 3 weeks and showed no statistical difference during weeks 4 and 5. Drug levels in the deep layer were greater than those in the superficial layer after 6 weeks (Figure 3B). Furthermore, cisplatin drug levels in the superficial layer of the brain were greater than those in the deep layer during the first 4 weeks, while drug levels in the deep layer were greater than those in the superficial layer after 5 weeks (Figure 3C). Finally, combretastatin levels in the superficial layer of the brain were greater than those in the deep layer during the first 3 weeks, and showed no statistical difference during week 4. Drug levels in the deep layer were greater than those in the superficial layer after 5 weeks (Figure 3D).
Histological examination of brain tissue
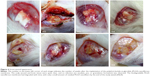
Figure 4 depicts the gross wound appearance at different times. The scalp wound and brain parenchyma were clear. No infection (exudate, pus, or granulation formation) was noticed grossly. The BICC–PLGA nanofibers gradually degraded after implantation, and only a small amount of residual nanofibers were observed in week 8.
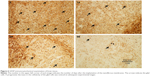
Figure 5 presents hematoxylin and eosin-stained sections of the brain tissues in weeks 1–8. No inflammation response (migration of leukocytes to brain tissue), tissue necrosis, or thickness of dura mater was noticed. A progressive decrease in cellularity (brain atrophy) was observed at subsequent experimental stages. Cell numbers were approximately 304.5±29.5 mm2 at week 1 and had progressively decreased to 125.5±19.8 mm2 by the end of the study (week 8). Figure 6 shows GFAP immunocytochemical examinations of brain tissues; progressively decreased cellularity of the glial cells was noticed.
Discussion
The basic treatment for brain tumors is surgery, in which the bulk of the tumor is removed and the peripheral infiltrating part is the target of supplemental treatment.22 Many promising biopharmaceutical agents have been developed; however, few (<5%) can be used to treat the CNS, because the BBB prevents their access to the desired site of action in therapeutically adequate quantities.23,24 In vitro studies have demonstrated that locally delivered chemotherapeutic agents lead to higher local drug concentrations for longer periods compared with systemic bolus therapy, and significantly reduce systemic toxicity.21,25 The Gliadel® wafer (polifeprosan 20 with carmustine implant; Arbor Pharmaceuticals, LLC, Atlanta, GA, USA) is implanted intracranially after surgical debulking of the malignant glioma. Although interstitial chemotherapy delivered with polymers directly to brain tumors at the time of surgery is a safe treatment for GBM, patients receiving Gliadel-wafer treatment survive about 2 months longer than patients without the treatment.10,21,25 Traditional therapeutic methods, such as disruption of the BBB, intraventricular pharmaceutical diffusion, and local treatment, are extremely invasive; therefore, they are not appropriate for long-term therapy regimens. Moreover, diffusional distance cannot exceed 5 mm in the brain by using such systems; therefore, tumor areas beyond this distance remain unattainable.26,27
Nanotechnology in treating glioblastoma multiforme
GBM is not easily targeted; however, advances in nanotechnology have improved the prospects of delivering therapeutics to GBM. In recent years, engineered tunable nanoscale devices have been proposed as valuable tools that can potentially solve the unresolved problem of enhancing drug transport across the BBB.28,29 Polymeric nanoparticles have been shown to be promising carriers for CNS drug delivery, because of their potential in encapsulating drugs, thereby protecting them from excretion and metabolism, and in delivering active agents across the BBB without inflicting any damage to the barrier.30 By contrast, nanoscale fibers have an extremely high surface area:volume ratio and a three-dimensional open porous structure, which facilitate reducing the constraint toward drug diffusion, resulting in a more efficient drug-release system.31,32 Local drug delivery by using electrospun nanofibers is extremely advantageous, because of its site specificity and lower overall medical dosage, leading to less systemic absorption and reducing unwanted toxicity and side effects. Moreover, electrospun membranes are thin, can be cut into any size and shape, and can conform completely to the tissue after the removal of tumors, thus achieving improved drug transport without interfering with the normal functions of the brain.32,33 Various drugs predominantly used to treat infections and cancers can easily be incorporated into these membranes and released at therapeutic dosages.32,34
Targeted therapy
The importance of the vasculature in tumor growth, development, and metastasis has generated substantial interest in strategies for interfering with the angiogenic process or destroying the existing tumor-vessel network.35 Vascular proliferation, or neoangiogenesis, is a distinct histopathological characteristic of GBM, and is correlated with prognosis.36,37 Bevacizumab, recently approved by the US Food and Drug Administration (FDA), is a humanized monoclonal antibody that inhibits VEGF, and is the first antiangiogenic therapy for use in patients with GBM.18,38 Combretastatin induces significant tumor necrosis as a single agent, and when combined with an anti-VEGF antibody can diminish the surviving tumor rim, thus significantly increasing the antitumor activity.20
Combretastatin is a microtubule-depolymerizing agent; the mechanism of action of the drug is thought to involve the binding of combretastatin to tubulin, leading to cytoskeletal and then morphological changes in endothelial cells.35 Combretastatin can induce potent and selective antivascular effects against tumor-associated endothelia. The apparent selectivity of the effects observed now warrant considerably more detailed study of the cellular and molecular mechanisms involved, in addition to investigation of the therapeutic potential of the drug in single- and multiple-dose schedules, both alone or in combination with other chemotherapeutic agents.19,35
Drug cocktails
GBM is one of the most difficult human malignancies to manage. The mean survival rate of patients with GBM has increased by only 7 months during the past 70 years.4,25 The current standard of care involves a combination of various modalities of treatment, such as surgery, chemotherapy, and radiation. A combinatorial drug approach involving drugs within a specific group, and those from various groups would be effective. With only rare exceptions, monochemotherapy has yielded little success in treating human malignancies compared with multiagent therapy.10,11 Tumor resistance to chemotherapy remains the primary cause of treatment failure. To prevent tumor resistance, a team from Duke University treated patients with primary GBM with a standard regimen consisting of radiation therapy and concurrent temozolomide, followed by adjuvant multiagent rotational chemotherapy; the results demonstrated that adjuvant multiagent rotational chemotherapy was more effective compared with single-agent adjuvant regimens after radiation and concurrent temozolomide therapy.10 Subsequent studies also demonstrated that the consolidation of irinotecan and alkylating agents, especially BCNU, increases antitumor efficacy to a level markedly higher than that of the additive efficacy of the individual drugs.39 The chemotherapy regimen (combinations of BCNU and cisplatin) appears to have salient activity, and may prolong survival in adults newly diagnosed with high-grade astrocytoma.40,41 The combination of bevacizumab and irinotecan was strikingly active against recurrent GBM, and toxicity was significant but acceptable.18,38 In other experimental studies,35 vascular-targeting agents (combretastatin) enhanced the activities of chemotherapeutic agents (cisplatin) but increased the toxicity associated with anticancer drugs, thus giving rise to therapeutic gain. These agents have exhibited only modest efficacy in monotherapy; however, new multitargeted kinase inhibitors and combinations of single-targeted kinase inhibitors in combination with radiation and cytotoxic chemotherapy will likely play an increasingly crucial role in GBM management. Several randomized prospective studies are ongoing.42
Biodegradable polymer
PLGA, an FDA-approved polymer, is highly biocompatible and biodegradable, and has been among the most attractive polymeric candidates used in fabricating devices for drug-delivery and tissue-engineering applications.43,44 PLGA polymers degrade through the autocatalytic hydrolysis of the ester bond into monomers of lactic acid (LA) and glycolic acid (GA), and eventually degrade via the Krebs cycle into CO2 and H2O.21,23 The degradability of PLGA varies among organisms, depending on the PLA:PGA ratio. The presence of methyl side groups in PLA makes it more hydrophobic than PGA, and hence lactide-rich PLGA copolymers are less hydrophilic, absorb less water, and subsequently degrade more slowly.43 One exception is copolymers with a 50:50 monomer ratio, which exhibit quicker degradation and can be entirely resorbed in about 2 months.21,43 By contrast, 75:25 PLGA maintained its three-dimensional morphology at a physiological pH for over 6 months.45 In addition, the overall physical properties of the polymer–drug matrix can be tuned by controlling the relevant parameters, such as polymer molecular weight, ratio of lactide to glycolide, and drug concentration, to achieve a desired dosage and release interval depending on the drug type.43,46,47
In this study, we incorporated three chemotherapeutic agents (BCNU, irinotecan, and cisplatin) onto rapidly degrading 50:50 PLGA nanofibers and loaded the antiangiogenic agent (combretastatin) onto 75:25 PLGA nanofibers. High drug levels of BCNU, irinotecan, and cisplatin were released 2 weeks earlier than those of combretastatin. By using PLGA with various LA/GA compositions that resulted in different degradation profiles of PLGA, we successfully controlled the sequential elution of chemotherapeutic and antiangiogenic pharmaceuticals from biodegradable nanofibers. The histologic examinations of postimplanted brain tissues demonstrated that the PLGA did not induce inflammation response and delivered a high concentration of chemotherapeutic and antiangiogenic agents to the brain tissues, thus resulting in brain atrophy (decreased cellularity in neurons and glial cells).
Furthermore, in contrast to other concurrent (combined) chemotherapy regimens such as the procarbazine–lomustine–vincristine regimen, which demands an intricate therapy process: administration of lomustine (110 mg/m2) on day 1, administration of procarbazine (60 mg/m2) daily for 14 days, beginning on day 8, and administration of vincristine (1.4 mg/m2) on days 8 and 29 of each 6-week therapy cycle48,49 the BICC–PLGA nanofibers developed in this study were able simultaneously to transport three chemotherapeutic agents and sequentially deliver antiangiogenic agents in a single step (the membrane can simply be deployed to the brain tissue after the surgical removal of tumors).
One major limitation of the current study lies in that a nondiseased animal model was enrolled. It remains unknown whether or not the nanofibrous membranes would perform differently in a diseased cerebral space. Further assessment of the chemotherapeutic and antiangiogenic agent-loaded PLGA-copolymer nanofibers in animal models with tumors will be needed to better illustrate this question. Although the simultaneous administration of four drugs is expected to be more efficient for tumor treatment locally, the associated side effects and toxicity also can be exaggerated. More experimental work will be needed to examine the influence of combinatorial therapies on animals’ hepatic and renal functions. The potential synergies of the side effects also need to be further investigated. Additionally, the relationship of the findings in this study to humans with brain tumors is still unclear, and needs further exploration.
Conclusion
In this study, we successfully fabricated biodegradable multiagent nanofibrous membranes by employing the electrospinning process, which provided the sequential and sustained release of BCNU, irinotecan, cisplatin, and combretastatin. All biodegradable nanofibers discharged high levels of BCNU, irinotecan, cisplatin, and combretastatin over 8 weeks in the brains of rats. Furthermore, electrospun membranes can more effectively conform to brain-tissue geometry and more completely cover the tissue after the removal of tumors, achieving higher drug transport without interfering with the normal functions of the brain. Histological examination revealed no obvious inflammatory reaction in the brain tissues.
Acknowledgments
This work was supported by Taipei Medical University (TMU102-AE1-B08), Chang Gung Memorial Hospital (CMRPD2E0042), and the Ministry of Science and Technology, Taiwan (104-2221-E-182-048-MY3). The authors thank Ms Ying-Chun Chen for her assistance with the experiments and preparation of the figures.
Disclosure
The authors report no conflicts of interest in this work.
References
Black PM. Brain tumors: part 1. N Engl J Med. 1991;324(21):1471–1476. | ||
DeAngelis LM. Brain tumors. N Engl J Med. 2001;344(2):114–123. | ||
Vredenburgh JJ, Desjardins A, Reardon DA, Friedman HS. Experience with irinotecan for the treatment of malignant glioma. Neuro Oncol. 2009;11(1):80–91. | ||
Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–996. | ||
Knisely JP, Baehring JM. A silver lining on the horizon for glioblastoma. Lancet Oncol. 2009;10(5):434–435. | ||
Lesniak MS, Brem H. Targeted therapy for brain tumours. Nat Rev Drug Discov. 2004;3(6):499–508. | ||
Sonabend AM, Stuart RM, Yun J, et al. Prolonged intracerebral convection-enhanced delivery of topotecan with a subcutaneously implantable infusion pump. Neuro Oncol. 2011;13(8):886–893. | ||
Reardon DA, Wen PY. Therapeutic advances in the treatment of glioblastoma: rationale and potential role of targeted agents. Oncologist. 2006;11(2):152–164. | ||
Choueiri TK, Raghavan D. Chemotherapy for muscle-invasive bladder cancer treated with definitive radiotherapy: persisting uncertainties. Nat Clin Pract Oncol. 2008;5(8):444–454. | ||
Affronti ML, Heery CR, Herndon JE 2nd, et al. Overall survival of newly diagnosed glioblastoma patients receiving carmustine wafers followed by radiation and concurrent temozolomide plus rotational multiagent chemotherapy. Cancer. 2009;115(15):3501–3511. | ||
Debinski W. Drug cocktails for effective treatment of glioblastoma multiforme. Expert Rev Neurother. 2008;8(4):515–517. | ||
Reithmeier T, Graf E, Piroth T, Trippel M, Pinsker MO, Nikkhah G. BCNU for recurrent glioblastoma multiforme: efficacy, toxicity and prognostic factors. BMC Cancer. 2010;10:30. | ||
Donelli MG, Zucchetti M, D’Incalci M. Do anticancer agents reach the tumor target in the human brain? Cancer Chemother Pharmacol. 1992;30(4):251–260. | ||
Sheleg SV, Korotkevich EA, Zhavrid EA, et al. Local chemotherapy with cisplatin-depot for glioblastoma multiforme. J Neurooncol. 2002;60(1):53–59. | ||
Vassal G, Terrier-Lacombe MJ, Bissery MC, et al. Therapeutic activity of CPT-11, a DNA-topoisomerase I inhibitor, against peripheral primitive neuroectodermal tumour and neuroblastoma xenografts. Br J Cancer. 1996;74(4):537–545. | ||
Nakatsu S, Kondo S, Kondo Y, et al. Induction of apoptosis in multi-drug resistant (MDR) human glioblastoma cells by SN-38, a metabolite of the camptothecin derivative CPT-11. Cancer Chemother Pharmacol. 1997;39(5):417–423. | ||
Ahluwalia MS, Gladson CL. Progress on antiangiogenic therapy for patients with malignant glioma. J Oncol. 2010;2010:689018. | ||
Vredenburgh JJ, Desjardins A, Herndon JE 2nd, et al. Bevacizumab plus irinotecan in recurrent glioblastoma multiforme. J Clin Oncol. 2007;25(30):4722–4729. | ||
Dark GG, Hill SA, Prise VE, Tozer GM, Pettit GR, Chaplin DJ. Combretastatin A-4, an agent that displays potent and selective toxicity toward tumor vasculature. Cancer Res. 1997;57(10):1829–1834. | ||
Nathan P, Zweifel M, Padhani AR, et al. Phase I trial of combretastatin A4 phosphate (CA4P) in combination with bevacizumab in patients with advanced cancer. Clin Cancer Res. 2012;18(12):3428–3439. | ||
Tseng YY, Liao JY, Chen WA, Kao YC, Liu SJ. Sustainable release of carmustine from biodegradable poly[(D,L)-lactide-co-glycolide] nanofibrous membranes in the cerebral cavity: in vitro and in vivo studies. Expert Opin Drug Deliv. 2012;10(7):879–888. | ||
Jain KK. Role of nanobiotechnology in the personalized management of glioblastoma multiforme. Nanomedicine (Lond). 2011;6(3):411–414. | ||
Barbu E, Molnar E, Tsibouklis J, Gorecki DC. The potential for nanoparticle-based drug delivery to the brain: overcoming the blood-brain barrier. Expert Opin Drug Deliv. 2009;6(6):553–565. | ||
Pardridge WM. Drug targeting to the brain. Pharm Res. 2007;24(9):1733–1744. | ||
Gallia GL, Brem S, Brem H. Local treatment of malignant brain tumors using implantable chemotherapeutic polymers. J Natl Compr Canc Netw. 2005;3(5):721–728. | ||
Roullin VG, Deverre JR, Lemaire L, et al. Anti-cancer drug diffusion within living rat brain tissue: an experimental study using [3H](6)-5-fluorouracil-loaded PLGA microspheres. Eur J Pharm Biopharm. 2002;53(3):293–299. | ||
Kreuter J, Gelperina S. Use of nanoparticles for cerebral cancer. Tumori. 2008;94(2):271–277. | ||
Holmes D. The next big things are tiny. Lancet Neurol. 2013;12(1):31–32. | ||
Jain KK. Nanobiotechnology-based strategies for crossing the blood-brain barrier. Nanomedicine (Lond). 2012;7(8):1225–1233. | ||
Tosi G, Costantino L, Ruozi B, Forni F, Vandelli MA. Polymeric nanoparticles for the drug delivery to the central nervous system. Expert Opin Drug Deliv. 2008;5(2):155–174. | ||
Luo X, Xie C, Wang H, Liu C, Yan S, Li X. Antitumor activities of emulsion electrospun fibers with core loading of hydroxycamptothecin via intratumoral implantation. Int J Pharm. 2012;425(1–2):19–28. | ||
Hadjiargyrou M, Chiu JB. Enhanced composite electrospun nanofiber scaffolds for use in drug delivery. Expert Opin Drug Deliv. 2008;5(10):1093–1106. | ||
Zamani M, Prabhakaran MP, Ramakrishna S. Advances in drug delivery via electrospun and electrosprayed nanomaterials. Int J Nanomedicine. 2013;8:2997–3017. | ||
Tseng YY, Wang YC, Su CH, Liu SJ. Biodegradable vancomycin-eluting poly[(d,l)-lactide-co-glycolide] nanofibres for the treatment of postoperative central nervous system infection. Sci Rep. 2015;5:7849. | ||
Siemann DW, Mercer E, Lepler S, Rojiani AM. Vascular targeting agents enhance chemotherapeutic agent activities in solid tumor therapy. Int J Cancer. 2002;99(1):1–6. | ||
Plate KH, Breier G, Weich HA, Risau W. Vascular endothelial growth factor is a potential tumour angiogenesis factor in human gliomas in vivo. Nature. 1992;359(6398):845–848. | ||
Reijneveld JC, Voest EE, Taphoorn MJ. Angiogenesis in malignant primary and metastatic brain tumors. J Neurol. 2000;247(8):597–608. | ||
Friedman HS, Prados MD, Wen PY, et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol. 2009;27(28):4733–4740. | ||
Friedman HS, Keir ST, Houghton PJ. The emerging role of irinotecan (CPT-11) in the treatment of malignant glioma in brain tumors. Cancer. 2003;97(9S):2359–2362. | ||
Grossman SA, Wharam M, Sheidler V, et al. Phase II study of continuous infusion carmustine and cisplatin followed by cranial irradiation in adults with newly diagnosed high-grade astrocytoma. J Clin Oncol. 1997;15(7):2596–2603. | ||
Dazzi C, Cariello A, Giannini M, et al. A sequential chemo-radiotherapeutic treatment for patients with malignant gliomas: a phase II pilot study. Anticancer Res. 2000;20(1B):515–518. | ||
Minniti G, Muni R, Lanzetta G, Marchetti P, Enrici RM. Chemotherapy for glioblastoma: current treatment and future perspectives for cytotoxic and targeted agents. Anticancer Res. 2009;29(12):5171–5184. | ||
Makadia HK, Siegel SJ. Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers (Basel). 2011;3(3):1377–1397. | ||
Xie H, Smith JW. Fabrication of PLGA nanoparticles with a fluidic nanoprecipitation system. J Nanobiotechnology. 2010;8:18. | ||
Holy CE, Dang SM, Davies JE, Shoichet MS. In vitro degradation of a novel poly(lactide-co-glycolide) 75/25 foam. Biomaterials. 1999;20(13):1177–1185. | ||
Mohamed F, van der Walle CF. Engineering biodegradable polyester particles with specific drug targeting and drug release properties. J Pharm Sci. 2008;97(1):71–87. | ||
Mundargi RC, Babu VR, Rangaswamy V, Patel P, Aminabhavi TM. Nano/micro technologies for delivering macromolecular therapeutics using poly(D,L-lactide-co-glycolide) and its derivatives. J Control Release. 2008;125(3):193–209. | ||
Levin VA, Edwards MS, Wright DC, et al. Modified procarbazine, CCNU, and vincristine (PCV 3) combination chemotherapy in the treatment of malignant brain tumors. Cancer Treat Rep. 1980;64(2–3):237–244. | ||
Kappelle AC, Postma TJ, Taphoorn MJ, et al. PCV chemotherapy for recurrent glioblastoma multiforme. Neurology. 2001;56(1):118–120. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.