Back to Journals » Diabetes, Metabolic Syndrome and Obesity » Volume 9
Symptom profiles of subsyndromal depression in disease clusters of diabetes, excess weight, and progressive cerebrovascular conditions: a promising new type of finding from a reliable innovation to estimate exhaustively specified multiple indicators–multiple causes (MIMIC) models
Authors Francoeur RB
Received 29 July 2016
Accepted for publication 12 September 2016
Published 8 December 2016 Volume 2016:9 Pages 391—416
DOI https://doi.org/10.2147/DMSO.S118432
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ming-Hui Zou
Richard B Francoeur
School of Social Work, Adelphi University, Garden City, NY, USA
Abstract: Addressing subsyndromal depression in cerebrovascular conditions, diabetes, and obesity reduces morbidity and risk of major depression. However, depression may be masked because self-reported symptoms may not reveal dysphoric (sad) mood. In this study, the first wave (2,812 elders) from the New Haven Epidemiological Study of the Elderly (EPESE) was used. These population-weighted data combined a stratified, systematic, clustered random sample from independent residences and a census of senior housing. Physical conditions included progressive cerebrovascular disease (CVD; hypertension, silent CVD, stroke, and vascular cognitive impairment [VCI]) and co-occurring excess weight and/or diabetes. These conditions and interactions (clusters) simultaneously predicted 20 depression items and a latent trait of depression in participants with subsyndromal (including subthreshold) depression (11≤ Center for Epidemiologic Studies Depression Scale [CES-D] score ≤27). The option for maximum likelihood estimation with standard errors that are robust to non-normality and non-independence in complex random samples (MLR) in Mplus and an innovation created by the author were used for estimating unbiased effects from latent trait models with exhaustive specification. Symptom profiles reveal masked depression in 1) older males, related to the metabolic syndrome (hypertension–overweight–diabetes; silent CVD–overweight; and silent CVD–diabetes) and 2) older females or the full sample, related to several diabetes and/or overweight clusters that involve stroke or VCI. Several other disease clusters are equivocal regarding masked depression; a couple do emphasize dysphoric mood. Replicating findings could identify subgroups for cost-effective screening of subsyndromal depression.
Keywords: depression, diabetes, overweight, cerebrovascular disease, hypertension, metabolic syndrome, stroke, vascular cognitive impairment
Introduction
Physicians and other health providers in primary care are more likely to suspect and detect depression in older adults when symptoms of depressed mood are prominent (especially feeling sad or blue or reporting crying spells) as part of a diagnosis of major depression.1 However, 20% of adults with clinically diagnosed depression do not endorse symptoms of depressed mood; this finding has shaped the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV and now DSM-V) diagnostic criteria for major depression to require the presence of either depressed mood or anhedonia (loss of interest or inability to derive pleasure from normal activities).2,3 Furthermore, dysthymic disorder without concurrent or prior major depression and subsyndromal depressive symptoms rival the prevalence of major depression in older adults,4–7 have similar risk factors as major depression, indicate older patients at risk of developing major depression, and worsen medical outcomes.8,9 As with major depression, these depressive conditions often are not recognized by health providers, or may be dismissed as a normal part of aging, unless dysphoric mood is prominent.
In their comprehensive review of depression in the context of medical illness, Gastelum et al10 conclude that although co-morbid major depression creates additional debilitation in many disease conditions, its treatment does not have a major impact on medical outcomes. These researchers acknowledge that treating depression only once it has developed into an episode of major depression may be “…too late in the game to meaningfully reverse course”. Instead, they suggest that there should be greater preventive efforts to screen and address psychosocial stressors in patients who are most at risk of developing major depression. However, other researchers challenge the pre-occupation with co-morbid major depression in diabetes, maintaining that in many situations, subsyndromal depressive symptoms, and even subthreshold symptoms that are approaching clinical significance, are non-pathological manifestations of distress in coping with illness-specific stressors and other life stressors that impinge on an individual’s capacity to manage their illness.11–14 Difficulties with glycemic control, for instance, occur at all levels of depressive symptoms, even at low levels, suggesting that they tap distress rather than depression in this context.15 On the other hand, clinically significant depression is more common when diabetes occurs with co-morbid disease.16
Co-morbid conditions of diabetes, such as cerebrovascular disease (CVD), may suggest illness contexts where a pathological process of depression, as well as distress, may coexist. Late-life depression (dysthymic, subthreshold, subsyndromal, and major depression) may present in atypical ways, involving low levels of dysphoric mood, such as sadness,17 masking the depression from detection. Although distress might be experienced, vascular depression may be the primary mental health condition. Vascular depression, a major category of “masked depression,” is characterized by anhedonia and social withdrawal and, in more progressed phases, by cognitive impairment and psychomotor retardation. The literature reveals that etiology is linked to CVD and magnetic resonance imaging evidence of lesions in frontal caudate circuits, which are associated with advancing age,18–20 and may be more prevalent in contexts of life stress and inadequate social support.21 In concert with this vascular dysregulation, chronic inflammation due to co-occurring obesity, diabetes, the metabolic syndrome, and vascular disease creates pernicious feedback effects involving sustained generation of proinflammatory cytokines that contribute to the vascular depression.22 Although proinflammatory cytokines such as interleukin (IL)-1 are known to generate sadness, alexithymia (low emotional self-awareness, discussed subsequently) in the context of vascular dysregulation may mean that dysphoric effects remain attenuated.
There is evidence that diabetes may be a significant risk factor for white matter lesions.23,24 Depressive symptoms were predicted by small basal ganglia lesions, but not by larger basal ganglia infarcts, suggesting an association with correlates of small-vessel vascular atherosclerosis, such as diabetes and hypertension.19,25 Indeed, advancing age and diabetes were among the independent risk factors for new silent atherothrombotic brain infarction among older Hispanic men and women.26 Diabetes can trigger episodes of hyperglycemia, which precipitates endothelial, vascular smooth muscle cell, and platelet dysfunction, and increase free fatty acids, insulin resistance,27 and cognitive impairment.28 After adjustment for effects from vascular disease, diabetes was found to increase risks especially for developing vascular dementia, as well as Alzheimer’s disease (which may be influenced by vascular factors).28
Similarly, obesity also magnifies the risk of developing late-life dementia, after adjusting for effects from diabetes and vascular disease.29 Obesity and diabetes may share similar pathways in contributing to late-life dementia since both conditions are associated with atrial fibrillation, in part through their role in precipitating obstructive sleep apnea.28,30 Thus, disease-specific effects of obesity and diabetes may aggravate existing vascular conditions that lead to small-vessel vascular atherosclerosis, such as hypertension, while precipitating other vascular effects. On the other hand, vascular conditions may aggravate pre-diabetes and diabetes progression through the metabolic syndrome (a condition of co-occurring excess weight, hypertension, and dyslipidemia) since it is a risk factor for pre-diabetes, insulin resistance, and glycemic dysregulation (hyperglycemia and hypoglycemia).31
These diverse pathways that aggravate pre-diabetes, diabetes, and vascular conditions lead us to ask whether certain depressive symptoms, or the overall level of vascular depression, may be more prominent when excess weight and/or diabetes are co-occurring conditions. This issue is critical because the incidence of age-related atherosclerosis is accelerated for individuals with diabetes,32 and co-occurring combinations of excess weight, diabetes, and hypertension are common in older adults.33 Indeed, different sources of evidence converge in implicating inflammatory processes in the interrelationships among excess weight, the metabolic syndrome, and type 2 diabetes.34
Although there has been little research to identify correlates or co-factors for symptom profiles of masked presentation of vascular depression, the neuropsychiatric literature reveals an interesting vein of empirical work on alexithymia (ie, inability to describe and identify one’s emotions related to reduced inner awareness of the physiological condition of the body). There is evidence, including that from several brain neuroimaging studies,35 that alexithymia reduces awareness of bodily sensation in autonomic nervous activity, particularly in the gut, which may lead to hypertension.36–38 The impaired ability to monitor bodily homeostasis in terms of perceptions of satiety may undermine glycemic control and precipitate hypoglycemia39–43 and increase risks for obesity and diabetes,44–46 including diabetes in the metabolic syndrome, a cluster condition of metabolic abnormalities, hypertension, and obesity.47 In more progressed CVD, alexithymia was detected in men and women with a right-hemisphere stroke, in addition to women with a left-hemisphere stroke.48 Furthermore, alexithymia was related to serum IL-18 levels in patients with right-hemisphere lesions but not to their stroke severity.49 The literature does not confirm that when obesity and/or diabetes is present, the characteristically low levels of sadness and dysphoria in vascular depression remain consistent in more progressed vascular diseases (stroke, post-stroke cognitive impairment, and vascular cognitive impairment [VCI]). Finally, the very definition of alexithymia suggests that it is likely to overlap masked depression characterized by low endorsed dysphoric mood since the concepts have in common a lack of endorsed emotions. Depression scale scores (not deliberately limited to masked depression) in patients with diabetes were positively correlated with scores on the Toronto Alexithymia Scale;45 however, much remains unknown about the relationship between alexithymia and depression in vascular-related conditions.
The propensity for masked depression is suggested by findings from a meta-analytic review of 19 laboratory studies of reduced emotional reactivity in persons with major depression, compared to non-depressed persons, to both positive stimuli (d=-0.53) and negative stimuli (d=-0.25).50 Persons with major depression report fewer positive life events,51,52 and moreover, low sadness reactivity is related to lower global assessment of functioning scores.53 The findings regarding reduced emotional reactivity to positive stimuli in individuals with major depression might be considered to imply that individuals with major depression who do not endorse items of dysphoric mood might still be detected based on their responses to items of low positive affect because they are likely to be less emotionally reactive to the positive emotional stimuli in their lives (ie, they report low positive affect regardless of whether the level of positive emotional stimuli is normal or deficient). Since older adults with cerebrovascular-related conditions often present with co-occurring subsyndromal, subthreshold, or dysthymic depression, the pattern of low-endorsed dysphoric mood items and endorsed positive affect items would need to hold in order to identify those at risk for worsening morbidity.
From this review of the literature, it is apparent that period(s) of subthreshold, subsyndromal, or dysthymic depression symptoms are common but may elude detection in CVD, diabetes, and obesity. They may reflect considerable distress from difficulties in disease management, represent a pathological condition that may indicate or precipitate clinical depression, or both. Research efforts should identify contexts of distinct medical conditions, co-occurring conditions, and patient subgroups characterized by 1) distinct symptom profiles and 2) indistinct symptom profiles where there is much symptom heterogeneity from patient-to-patient but aggravated overall levels of depressive symptoms. The present study contributes to the feasibility of this type of research because it demonstrates an advance discovered by the author for estimating a multiple indicators–multiple causes (MIMIC) structural equations model that specifies all possible causal pathways. This exhaustive specification makes it possible to reveal these diverse contexts without bias.
Distinct or indistinct symptom profiles may characterize subgroups experiencing alexithymia, vascular depression, or both. These individuals are not aware of experiencing negative affect and are, therefore, less likely to endorse dysphoric mood items in the Center for Epidemiologic Studies Depression Scale (CES-D). However, it may be hypothesized that risk for alexithymia, vascular depression, or both may be revealed when these individuals also endorse CES-D items of positive affect weakly, or not at all, since indications of low positive affect reflect anhedonia (lack of interest or pleasure). It is reasonable to expect that it would be easier to recognize that one cannot remember feeling happy, hopeful, or enjoying life compared to remembering that one actually did feel sad, blue, depressed, or had crying spells, as well as how frequently. The findings in the literature of vascular depression characterized by low dysphoric mood, and of depression and alexithymia in diabetes, CVD, and obesity, suggest that this masked depression, as revealed by low positive affect, may be prevalent, although it is not clear whether masked depression is more likely when these conditions occur separately or together.
Therefore, the present study aims to predict the symptoms and overall level of subthreshold and clinically significant depression, which may be masked in presentation, in older adult subgroups with progressive CVD (hypertension alone, silent CVD, VCI, stroke, and post-stroke cognitive impairment). More refined subgroups will target participants who are also overweight and/or diagnosed with diabetes to determine whether specific vascular–metabolic interactions contribute to masked depression. The inclusion of items that tap positive affect is a special feature of the CES-D Depression Scale that may help us detect masked depression characterized by anhedonia or alexithymia, as well as non-depressive distress.
Materials and methods
Although depression in which dysphoric mood is acknowledged poses no new methodological concerns, the detection of possible masked vascular depression presents a methodological challenge. Separate evidence suggests that the potential for masked depression may be revealed through screening approaches with items to detect low levels of positive mood, which are typically excluded from symptom scales and diagnostic measures of depression.54,55 An exception is the CES-D scale, which is used in the present study. Furthermore, CES-D total score thresholds are used to select participants with subthreshold (16> CES-D >10) or clinically significant (CES-D ≥16) depression. Based on the self-reported symptoms, participants with excess weight, diabetes without complications, and cerebrovascular conditions were not already likely to be experiencing major depression (CES-D >27).
Masked vascular depression occurs when participants are not likely to endorse depressed mood items that reveal self-awareness of dysphoria (felt sad, could not shake blues) and yet are likely to endorse low positive mood (including anhedonia; reverse-coded items for happy, enjoyed life, felt hopeful, as good as others), psychomotor retardation (including anhedonia; talked less than usual, bothered by things), interpersonal difficulties (people disliked me, people unfriendly), or remaining depressed mood items that may reflect apathy, fear, or alexithymia (felt depressed, crying spells, felt fearful, life a failure). When other depressed mood items are low, clinically significant endorsement of “crying spells” could be a signal that underlying dysphoria is denied or unconsciously experienced (alexithymia).
Sample
The present study analyzed data from the first wave (1982–1983) of the New Haven, Connecticut site of the Established Populations for the Epidemiologic Studies of the Elderly (EPESE), a representative community survey of non-institutionalized adults aged ≥65 years sponsored by the National Institute on Aging.56 The two-stage, stratified, systematic random sample design was conducted in separate clusters of elders living independently in the community. Furthermore, all residents of public senior housing (income restricted) and private senior housing were included (ie, a census).
The overall sample resulted in 1,169 men (85% response rate) and 1,643 women (86.4% response rate). Men were oversampled; other sampling weights for non-response and a census-based age-by-sex distribution were applied. The survey of demographic, psychosocial, and health-related characteristics was administered during face-to-face interviews. Participants (proxies for 2%) provided written consent. The institutional review boards of Columbia and Adelphi Universities exempted from review the deidentified, public version of these data used in the present study.
Measures
CES-D
The CES-D queries respondents about the frequency of 20 symptoms over the prior week using a four-category scale (“rarely or none of the time” to “most or all of the time”). Total scores range from 0 to 60. Research on sensitivity and specificity suggests that a total score of 16 or higher reveals “clinically significant” depression involving subsyndromal depression or a depression syndrome57 and a total score between 11 and 15 targets participants with “subthreshold” depression who may be at heightened risk of developing clinically significant depression.58 Four factors (depressed affect, positive affect, somatic activity and psychomotor retardation, and interpersonal problems) were detected across the adult age range in several studies, in addition to an overarching second-order factor for the overall level of depression.
A rigorous study of elderly medical inpatients revealed confounding of CES-D scores with symptoms of physical illness in the absence of depression.59 Consequently, in addition to using a total CES-D score cutoff of 16 to identify participants at risk for a depressive syndrome, the study applied a minimum score of 4 on the depressed affect factor (based on five items). Use of both criteria linked CES-D scores more closely with diagnostic criteria for depression (DSM-III-R), which improved the predictive power of the CES-D. A more stringent score of 6 was recommended for research studies without a confirmatory diagnostic interview and is adopted in the present study.
Cumulative Illness Rating Scale-Geriatrics (CIRS-G)
The CIRS-G is a measure of vascular disease that has been used to classify patients with depression as being at risk of experiencing a vascular-mediated depression.60 The CIRS-G classifies patients into one of five ordinal categories based on their most progressed vascular condition. The measure has demonstrated reliability and validity in heterogeneous samples.61,62
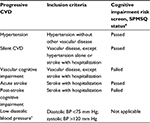
In the present study, similar categories approximating the CIRS-G were derived from other New Haven EPESE survey items (no hypertension; suspected coronary disease or suspected high blood pressure without antihypertensive medications; taking antihypertensive medications; coronary disease or confirmed high blood pressure without antihypertensive medications; and history of non-hospitalized stroke, angina, or claudication). The CIRS-G criteria were broadened within the last category to include criteria for vascular depression subtypes proposed by Steffens and Krishnan;20 intermittent claudication and angina are included in the final category due to the strong relationship of ischemic heart disease and atherosclerosis in cerebral and peripheral arteries. The scale appears valid in the New Haven EPESE site since those taking antihypertensive medications displayed higher readings for systolic and diastolic blood pressures than the first two non-treated subgroups in the scale.63
Short Portable Mental Status Questionnaire (SPMSQ)
Cognitive functioning was assessed using the SPMSQ. The SPMSQ is considered to be reliable and valid in different populations of older adults, including EPESE studies involving the New Haven and other sites.64 Evidence of impaired cognitive functioning is assessed based on the total error responses, “don’t know” responses, and response refusals. Four or more errors reflect moderate to severe cognitive impairment. The error rate increases with advancing age within each race–gender subgroup in the New Haven EPESE.56
In the present study, SPMSQ scores provide the basis for establishing the ordinal categories of progressive CVD, as revealed in Table 1. Based on whether a stroke with hospitalization was reported on the CIRS-G, participants with moderate to severe cognitive impairment were classified to be at risk for VCI or post-stroke cognitive impairment, the two major categories of cognitive impairment due to CVD. Other participants reporting a history of vascular disease on the CIRS-G who reveal intact through mild cognitive impairment (ie, less than four errors on the SPMSQ) were classified into categories for acute stroke (with hospitalization), silent CVD (other vascular diseases, except hypertension), or hypertension.
Blood pressure
The self-reported item of hypertension impacts the first three categories of the CIRS-G scale, which are based on elders’ self-reports to the items: “Has a doctor ever told you that you had high blood pressure? Are you currently taking any medication for this?” It is important to note the weakness in using this measure since its objectivity has not been established.
To correct for possible bias from this item, standardized measures of systolic and diastolic blood pressure were used to create a separate indicator for detecting potentially unreported hypertension (systolic, diastolic, or both). The average of the first and second readings for systolic and diastolic blood pressures was used. However, few participants were reclassified as having hypertension.
In addition, these standardized measures were used to derive a dummy variable for the vascular condition of low diastolic blood pressure in the context of normotensive systolic blood pressure, which some evidence suggests is implicated in co-occurring depression with masked presentation.65 Diastolic hypotension, which is common in the overtreatment of systolic hypertension, may be implicated in vascular depression since it may precipitate or aggravate cerebral ischemia and dementia.
Overweight and diabetes
There is some evidence that self-reported height and self-reported weight are each reliable,66 although proxies were used in some cases. In any event, both measures should be valid to distinguish respondents who are overweight from all others. The body mass index (BMI) was calculated as weight in kilograms divided by the square of height in meters. Participants scoring 25 or higher on the BMI were overweight (24.9 < BMI < 30) or obese (BMI = 30+); they were classified as a single group due to insufficient numbers of participants in either subgroup.
Participants were also asked whether they had “diabetes, sugar in the urine, or high blood sugar.” Despite the weaknesses of self-reported diabetes, the estimated gender-based prevalences of diabetes in the overall New Haven community of older adults (13.5% males; 13.2% females) appear valid since they are similar to those obtained from the other three EPESE sites.
Of those reporting diabetes, only participants with type II diabetes who had not progressed to require insulin and without evidence of diabetes complications were coded affirmatively within the dichotomous diabetes variable. Participants indicating use of insulin were excluded because they were assumed either diagnosed with type I diabetes (who are likely to present with a very different depression symptom profile) or contending with type II diabetes that had progressed to require insulin. All participants with indication of diabetes complication were excluded based on either vision loss (which may reflect retinopathy) or intermittent claudication (calf pain during ambulation due to peripheral vascular disease and often accompanied by peripheral neuropathy) because factors associated with diabetes complications may confound the masked profile of vascular depression in diabetes without complications. For instance, pronounced sadness is common in depression with co-morbid vision loss, which may be due to retinopathy.
Demographic correlates and age-associated chronic life stressors
Gender, race (black, white), and age were specified as co-factors. Several chronic life stressors that may be associated with advancing age are also accounted in order to distinguish them from developmental effects due to aging. Furthermore, vascular depression has been clinically observed to occur in contexts of low economic status and chronic life stressors, which could aggravate the condition.21
The present study uses an income equivalence scale to adjust annual family income to account for family size. A dummy variable was created to identify participants with low adjusted family income (≤$5,000).
A dummy variable was created to identify participants with low education, based on not having completed high school.
A dummy variable was created to identify participants at risk for social isolation. At-risk participants score two or less on an index calculated from survey items regarding the number of family members and adult children who are alive, as well as the number of close relatives and close friends seen at least once a month.
Recent widows (up to 2 years) were distinguished from non-recent widows to separate depressive symptoms related to recent grief and loss from chronic psychosocial stressors associated with widowhood.
There is direct empirical support for some chronic life stressors in the context of vascular depression. Family history of alcohol or drug use is a highly significant risk factor (odds ratio 4.42) in distinguishing patients with vascular depression from those with non-vascular depression.19 Therefore, a dummy variable, alcohol, was created to reflect high alcohol consumption based on the summative frequencies of beer, wine, and liquor consumed in the past month. In addition, dummy variables were created to indicate when participants were smokers or reported impairments in instrumental activities of daily living (impaired IADLs).
Finally, an ordinal variable (0–8) was created to control for confounding from the number (and not type) of cerebrovascular risk factors (CVRFs) in the explanatory analyses. The risk factors that contribute to this measure are hypertension, myocardial infarction, congestive heart failure, silent CVD, VCI, stroke, post-stroke cognitive impairment, any diabetes (type 1 with insulin, type 2 without complications, and diabetes with complications), and diastolic low blood pressure.
Analyses
The independent (x) variables were the progressive vascular conditions (hypertension, silent CVD, VCI, stroke, post-stroke cognitive impairment, and low diastolic blood pressure), the co-occurring metabolic conditions (overweight, type 2 diabetes without complications), and in some analyses, demographic correlates and chronic life stressors. All independent (x) variables were dummy variables, and multicollinearity was low across the set of x variables. The outcome (y) variables consisted of the individual items from the CES-D Depression Scale, which were four-category, ordinal variables, and in some analyses, the derivative latent factor for total depression. SPSS (Version 19) was used to derive most of the final variables and obtain frequencies and Mplus (Version 5.21) to derive the remaining final variables.
Depressive symptoms endorsed by subgroups of older adults with co-morbid metabolic and vascular conditions were modeled in Mplus (Version 5.21) using multivariate regression and MIMIC regression based on the MLR estimator (maximum likelihood parameter estimates with standard errors [SEs] that are robust to non-normality and non-independence in complex random samples67).
The analyses test all possible interactions of each of the progressive cerebrovascular conditions with overweight, diabetes, or both moderator variables. To distinguish whether influential contexts may depend on the absence of excess weight and/or diabetes, re-estimated analyses replace overweight and diabetes, one at a time, with their counterpart variable reflecting the absence of the condition; extended analyses also test smoker and non-smoker as interacting terms. Note that the analyses include both genders and ethnic groups (black and white), although when unique estimates were possible, models were run separately in older women.
Each of the multivariate regressions predicts the CES-D items simultaneously as 20 observed outcomes (ie, multiple y variables) without also incorporating them as loadings of a latent trait. This means that the multivariate regression may reveal whether specific CES-D items of dysphoric mood may be visible or masked during screening with the self-reported CES-D Depression Scale, but it does not also model the profile of depressive symptoms that occur within the specific, co-occurring overweight–diabetes–vascular condition itself nor adjust for the level of latent depression.
In contrast, the counterpart MIMIC regression does also incorporate this profile of depressive symptoms and adjusts for the level of latent depression. It comprises a regression (the multiple causes) that predicts a measurement model based on a latent trait and their individual observed (y) items (the multiple indicators). In the present study, a single latent factor for depression is specified, with each of the 20 CES-D items loading onto this factor.
Each descriptive MIMIC specifies the overweight and diabetes predictors, the progressive vascular condition of interest, and their interaction(s) to model the moderating effects by overweight condition and/or diabetes on relationships between a cerebrovascular variable and each of the 20 CES-D items (ie, direct effects), while also accounting for their relationship to latent depression (ie, indirect effects). These descriptive MIMIC analyses model visible effects while controlling for the level of latent depression; however, they do not account for potential confounding factors that may account for these relationships, which may vary in different samples.
It is important to account for these demographic correlates and chronic life stressors because they may be related not only to vascular depression but also to obesity, diabetes, and CVD.21 In the parallel explanatory MIMIC, the initial set of first-order correlates or co-factors (x variables) consists of demographic correlates and age-associated chronic life stressors as well as predictors for cancer, congestive heart failure, lost 10 pounds, and the number of CVRFs. These potential confounding predictors are specified prior to the overweight and diabetes predictors, the progressive vascular condition of interest, and their interaction(s). Finally, the specific interaction terms are specified to model the moderating effects by overweight condition and/or diabetes on relationships between a cerebrovascular variable and each of the 20 CES-D items (ie, direct effects) after accounting for their relationship to the latent factor of depression (ie, indirect effects). Since a number of descriptive MIMIC models are tested, each explanatory MIMIC also prevents accepting spurious findings that may arise from type 1 error. Figure 1 illustrates the explanatory MIMIC for the interactions among hypertension, overweight, and diabetes in older males.
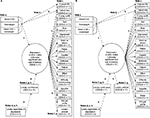  | Figure 1 Parallel multiple indicators–multiple causes (MIMIC) models with equivalent parameter estimates. Notes: Note a: The dichotomous predictors in (A) and (B) represent the presence/absence of the condition. The first-order terms in the upper left portion of (A) and (B) are global predictors of effects involving silent CVD or overweight, while the interaction term reveals the additional effect when both conditions co-occur. Note b: The descriptive MIMIC model in (A) and (B) reflects endorsement of the items enjoyed life (CES-D ≥ 11) and talked less (CES-D ≥ 11) that can be attributed to co-occurring atherosclerosis and being overweight (Path 2), after adjusting for endorsement that can be attributed instead to the overall level of the latent trait of depression in these conditions (Paths 1 and 3). Note c: While all 20 CES-D items are specified in the actual model, (A) and (B) shows only 17 items. Sixteen items are traditional symptoms of depression, and the final item, lonely (CES-D ≥ 11), is one of the four non-traditional symptoms in the CES-D scale. Although direct effects from silent CVD, overweight, and their interaction are specified to all 20 CES-D items, to keep (A) and (B) clear and simple, they are only drawn (Path 2) to the two CES-D items for which the direct effects are statistically significant: enjoyed life (CES-D ≥ 11) and talked less (CES-D ≥ 11). Also, arrows are not drawn to reflect the residual effects from each of the 17 CES-D items to minimize distracting complexity within the figure. Note d: Although each of the four non-traditional items (lonely, fearful, people were unfriendly, and people disliked me) contributes to the sensitivity and specificity of the CES-D scale in detecting real cases of subsequently confirmed clinical depression, they are not standard symptoms of depression. Corresponding instrumental variables (when CES-D ≥ 11) are specified to predict virtually all the variation within each of these four non-traditional items (when CES-D ≥ 0) because unpredicted residual variation within any of them may confound direct estimates (Path 2) to any of the remaining 16 standard items of depression. In addition, at least one of these instrumental variables is needed to resolve the problem of estimation indeterminacy that would otherwise result due to there being one too few degrees of freedom for estimation of a MIMIC model in which every direct effect is specified. Note e: The last predictor shown in (A) and (B), lonely regardless of depression (CES-D ≥ 0), is added 1) to account for unique variation from the last CES-D item, lonely–continuous (CES-D ≥ 11; Path 4); and 2) to permit estimation of the bi-directional effects of lonely (CES-D ≥ 11) by using two versions (ie, continuous and ordinal) of the variable (bi-directional Path 5). Because the instrumental variable lonely (CES-D ≥ 11) is a subset of the broader original variable, lonely regardless of depression (CES-D ≥ 0), the former accounts for virtually all the remaining variation in lonely (CES-D ≥ 0), which prevents confounding of direct paths to other items (Path 2). Although not shown, similar instrumental variables and bi-directional effects are specified for each of the three remaining non-traditional items (fearful, people were unfriendly, and people disliked me). Note f: In (A) and (B), part of the overall variation in lonely (CES-D ≥ 11), attributed to pre-existing lonely feelings, predicts the latent trait of depression. In (A), part of the overall variation in lonely (CES-D ≥ 11) manifests as an item from the measurement model portion of the MIMIC (ie, in bi-directional Path 5, the right path is an effect indicator with factor loading parameter λ), reflecting lonely feelings that manifest as a component of the latent factor for depression. However, in (B), this part of the overall variation in lonely (CES-D ≥ 11) occurs as a separate factor in the structural (regression) portion of the model; it is an outcome of the latent factor for depression (ie, with causal parameter B). Estimated values of the parameters for lonely, λ and B, are equivalent. Thus, estimates from both versions of the MIMIC model are equivalent. Note g: To prevent confounding of direct effects to the traditional symptoms of depression, the same type of specification used with the lonely item is used with each of the remaining three non-traditional CES-D depression items not shown in (A) and (B) (ie, fearful, people were unfriendly, and people disliked me). In the same way that lonely–continuous (CES-D ≥ 0) predicts its instrumental variable (ie, lonely–continuous [CES-D ≥ 11]), each of these three items predicts its corresponding instrumental variable when CES-D ≥ 11 (not shown). Although bi-directional arrows are only drawn (Path 5) to and from lonely (CES-D ≥ 11) in (A) and (B), all four items are estimated with bi-directional effects, and again, estimated values of the parameters λ and Β remain equivalent. Note h: David Kenny’s web page on identification discusses the conditions in which instrumental variables may be used.68 One important condition is that the instrumental variable must be highly correlated with the original variable, but cannot be correlated with the residual term when the original variable is regressed on the instrumental variable: Y = b(I) + U, where Y is the original variable, I is the instrumental variable, and U is the residual term. In the innovative instrumental variable approach for estimating an exhaustively specified MIMIC introduced in the present study, the original variable for a particular CES-D item, Y, includes the responses from participants with residual symptoms (CES-D < 11) as well as the responses from participants with subthreshold and clinically significant symptoms (CES-D ≥ 11). In contrast, the instrumental variable, I, only reflects subthreshold and clinically significant symptoms (CES-D ≥ 11); the original responses for participants with subthreshold or clinically significant symptoms are retained, while the responses for participants with residual symptoms are set to zero. This means that for participants with residual symptoms, I is always zero, while U is identical to Y, resulting in a low correlation between the instrumental variable (I) and the residual term (U). Similarly, for participants with subthreshold or clinically significant symptoms, I is identical to Y and U is always zero, again resulting in a low correlation between the instrumental variable (I) and the residual term (U). Abbreviations: CES-D, Center for Epidemiologic Studies Depression Scale; CVD, cerebrovascular disease. |
A limitation of MIMIC models in the literature concerns the lack of an approach to specify these models so that unique estimates can be obtained for all slope parameters and their SDs. In order to obtain an identified model, one of the pathways in the multiple causes portion of the model is not estimated; however, findings can shift considerably depending upon which pathway is not estimated. Usually, the pathway that is not estimated corresponds to a causal factor that is believed to have a minimal and restricted influence on the measurement items or on the overall latent trait. However, this choice is tricky and often implausible given the shared variation across the psychometric items and the multivariate nature of the estimates. There is no way to know for sure whether any of the possible identified models will yield valid estimates similar to those that would have been obtained if the exhaustive model could have been estimated.69,70 The present study overcomes this identification problem by demonstrating a unique way that the author has devised to specify an unbiased MIMIC model that completely links the confirmatory factor analysis portion of the model to the multiple regression portion of the model. It yields exhaustive profiles of a psychometric latent factor and all of its observed measures (the multiple indicators) within participant subgroups (the multiple causes) by estimating bidirectional relationships involving four of the 20 measurement model items using an instrumental variable approach.
This improved approach provides a novel and more valid way to specify non-traditional items within the measurement model. Four non-traditional items (lonely, unfriendly, disliked me, and fearful) were included when the CES-D Depression Scale was created in order to improve the specificity and sensitivity of the instrument in identifying actual cases of subsequently confirmed clinical depression.71 Unlike the remaining items, none of these items falls unambiguously into any of the specific symptom categories of diagnostic instruments, such as the DSM-V. Arguably, these items are closely related causes and effects of depression. Therefore, in addition to their traditional pathways as “effect indicators” within the measurement model, the opposite pathways as “causal indicators” are also specified. Strictly speaking, they are not bidirectional measurement items of the latent trait of depression and should be considered closely related variables in the structural model that cause, and are effects of, the latent trait (eg, Figure 1B). However, estimates from both specifications are equivalent, as Figure 1A and B show using the non-traditional item, lonely. For conceptual simplicity, the four bidirectional items, along with the remaining CES-D items, are all retained within the measurement portion of the model during estimation (eg, Figure 1A).
Furthermore, the improved approach employs a completely overlapping, and subsumed, instrumental variable for each of these four non-traditional items that completely accounts for variation in each of them. Recall that these four non-traditional items were included in the creation of the CES-D Depression Scale only to improve its sensitivity; they do not contribute legitimately to depression phenomenology. The instrumental variable approach excludes the variation from these non-traditional items from influencing the psychometric profile of depression within metabolic-vascular disease subgroups. Otherwise, this non-traditional variation would confound the unique contributions of the traditional items to the profile of depression items that distinguish each vascular/metabolic subgroup. This innovation is an advance over the common instrumental variable approach in which a different variable is sought, or an index of variables is created, which is highly correlated with the original variable. Some bias in slope estimates and SEs results in using the traditional approach since the instrumental variable is not completely overlapping, and subsumed, within the original variable (ie, there is no perfect overlap in the responses to the original and instrumental variables by the subgroup of interest in which participants exceed a certain threshold). More critically, however, the traditional procedure is not sufficiently reliable because the available data do not often yield a suitable variable or index of variables to serve as the instrumental variable. In contrast, using the original variable to create its own instrument, the improved approach is unbiased and reliable. Figure 1, note h, provides additional information.
In both the descriptive and explanatory MIMIC model specifications, the non-traditional ordinal items that contribute to the latent factor of depression (lonely, people unfriendly, people dislike me, and fearful) are also specified as continuous endogenous (y) variables that predict the latent factor of depression, allowing estimates of bidirectional relationships between each non-traditional item and total depression.
For instance, in panels A and B of Figure 1, lonely regardless of depression (CES-D ≥ 0) predicts lonely–continuous (CES-D ≥ 11), which permits us to estimate the relationship represented by the left dashed arrow in Path 5 that predicts the latent factor of depression. Essentially, lonely regardless of depression (CES-D ≥ 0) solely predicts an overlapping, and subsumed, instrumental variable, which is identical except for excluding values for residual depressive symptoms when CES-D <11 (ie, positive responses for residual symptoms are set to zero, such that positive responses are retained only in cases of subthreshold or clinically significant depression). In contrast to the stochastic or probabilistic relationships of the ordinal traditional items (where the “multiple causes” portion of the MIMIC is based on ordinal probit regression), this study is justified in creating a continuous variable for each of the non-traditional CES-D items because the relationship between the continuous variable and its identical, overlapping instrumental variable is deterministic in participants with CES-D scores ≥11 (the “multiple causes” portion of the MIMIC is based on continuous regression here). These instrumental variables constitute a necessary and non-biasing source of exogenous information (ie, from outside the model) in order to obtain an identified model with unique estimates for all parameters.
Results
Descriptive statistics
Table 2 summarizes diverse sample characteristics, including the prevalence of individual and co-occurring physical conditions and depression. More than one-third of the sample reported CVD limited to hypertension while an additional quarter of the sample reported a progressive CVD more advanced than hypertension. More than half of the sample was classified as being overweight based on a BMI of at least 25 (54.1%) and more than one in ten (11.2%) reported type II diabetes (without complications). Overweight or diabetes conditions co-occurred with progressive CVD in about one-third of all participants (32.3%), and close to half with these co-occurring conditions were at risk for subthreshold or clinically significant depression (CES-D ≥11; 46.4%).
Inspection of Table 2 reveals that across the overall categories of vascular conditions (hypertension, silent CVD, vascular cognitive impairment, stroke, post-stroke cognitive impairment, and low diastolic blood pressure), the overall categories of metabolic conditions (overweight and diabetes), and their co-occurring clustered conditions, participants reporting subthreshold or clinically significant depressive symptoms ranged from about one out of every three participants with the condition to about one out of every two participants with the condition.
The two exceptions are in the overall categories of metabolic conditions for older males. Between one out of every four and one out of every five older men who were overweight reported subthreshold or clinically significant depressive symptoms, and about one out of every six older men who disclosed diabetes reported subthreshold or clinically significant depressive symptoms. Since diabetes was self-disclosed and not medically assessed, the lower levels of depressive symptoms might indicate that undiagnosed diabetes may be more of an issue in older males; however, this lower rate did not also characterize any of the co-occurring clustered conditions with diabetes in older males.
Multivariate findings
Levels of evidence for masked depression
The potential for masked depression in each cerebrovascular–metabolic subgroup is based on a comparison of each multivariate regression reported in Table S1 with the counterpart descriptive MIMIC and explanatory MIMIC reported in Table 3A–D. (Table S1 reports multivariate regressions, which are not interpreted separately but are used in the assessment of evidence for masked depression [+++, ++, +, 0] in Table 3A–D). Table 3A–D reports the descriptive MIMICs and parallel explanatory MIMICs that were conducted; these specifications contribute to indices of excellent overall model fit, which are listed in a footnote to each table panel (A–D).
  | Table 3A CES-D depression items in co-occurring and synergistic hypertension subgroups based on overweight and diabetes status: multiple indicators–multiple causes (MIMIC) modelsa,b Notes: aThe measurement loading (λ) of the CES-D item depressed was fixed at 1 to set the metric of the measurement model. Estimated measurement loadings for the remaining CES-D items ranged from 0.293 (hopeful) to 0.811 (blues). Depending on the specific MIMIC model, 11–18 of the 20 measurement loadings were ≥0.450. bEach of the specific MIMIC models appear to fit the data adequately based on multiple fit indices: chi-square: 140.988–331.225; chi-square/degrees of freedom: 0.820–1.995; R2: 0.545–0.646; CFI: 0.988–1.000; TLI: 0.980–1.000; RMSEA: 0.000–0.014; and SRMR: 0.019–0.023. Except for R2, these fit indices are not available for MIMIC models with ordinal measurement items, such as the four-category CES-D items in the present study. They were derived from counterpart MIMIC models in which all CES-D measurement items of the latent trait of depression were specified to be continuous measures. In contrast to the ordinal models in which separate ordinal variables, along with their identical, overlapping instrumental variables, were specified for each of the non-traditional CES-D items, the specification in the continuous models differed in using a single continuous variable based on the sum of all four non-traditional CES-D measurement items, along with its identical, overlapping instrumental variable, as a “causal indicator” for estimating the pathway of the bi-directional relationship that predicts total depression. (Note that the four non-traditional CES-D items are still retained as individual items that serve as “effect indicators” for estimating the pathway of the bi-directional relationship that manifests as a result of the latent trait of depression.) In addition, the discrete nature of the continuous data (in contrast to the probabilistic nature of the data for analysis in the ordinal models) also allows residuals of CES-D items to be correlated, which contributes to model fit. It is unknown whether the need to collapse the four non-traditional items into a single variable in the continuous model tends to erode its fit; however, the fact that there is adequate fit in the continuous models across four fit indices despite this possibility may be taken to reveal that there is proper specification and adequate fit to the data within the ordinal models. cFor each subgroup, potential masked depression is based on a comparison of the multivariate regression reported in Table 2 with the counterpart MIMIC reported in Table S1. Refer to the main text for more information. dTwo-tailed test significance is as follows: 1) z=1.960 (p=0.05); 2) z=2.326 (p=0.025); 3) z=2.576 (p=0.01); 4) z=3.291 (p=0.005). Abbreviations: CES-D, Center for Epidemiologic Studies Depression Scale; CFI, Comparative Fit Index; RMSEA, Root Mean Square Error of Approximation; SE, standard error; SRMR, Standardized Root Mean Square Residual; TLI, Tucker–Lewis Index. |
  | Table 3B CES-D depression items in co-occurring and synergistic silent cerebrovascular disease subgroups based on overweight, diabetes, and smoking status: multiple indicators–multiple causes (MIMIC) modelsa,b Notes: aThe measurement loading (λ) of the CES-D item depressed was fixed at 1 to set the metric of the measurement model. Estimated measurement loadings for the remaining CES-D items ranged from 0.276 (good as others) to 0.985 (sad). Depending on the specific MIMIC model, 11–19 of the 20 measurement loadings were ≥0.450. bEach of the specific MIMIC models appear to fit the data adequately based on multiple fit indices: chi-square: 126.172–388.025; chi-square/degrees of freedom: 0.747–2.324; R2: 0.543–0.933; CFI: 0.969–1.000; TLI: 0.967–1.000; RMSEA: 0.000–0.023; and SRMR: 0.018–0.029. Except for R2, these fit indices are not available for MIMIC models with ordinal measurement items, such as the four-category CES-D items in the present study. They were derived from counterpart MIMIC models in which all CES-D measurement items of the latent trait of depression were specified to be continuous measures. In contrast to the ordinal models in which separate ordinal variables, along with their identical, overlapping instrumental variables, were specified for each of the non-traditional CES-D items, the specification in the continuous models differed in using a single continuous variable based on the sum of all four non-traditional CES-D measurement items, along with its identical, overlapping instrumental variable, as a “causal indicator” for estimating the pathway of the bi-directional relationship that predicts total depression. (Note that the four non-traditional CES-D items are still retained as individual items that serve as “effect indicators” for estimating the pathway of the bi-directional relationship that manifests as a result of the latent trait of depression.) In addition, the discrete nature of the continuous data (in contrast to the probabilistic nature of the data for analysis in the ordinal models) also allows residuals of CES-D items to be correlated, which contributes to model fit. It is unknown whether the need to collapse the four non-traditional items into a single variable in the continuous model tends to erode its fit; however, the fact that there is adequate fit in the continuous models across four fit indices despite this possibility may be taken to reveal that there is proper specification and adequate fit to the data within the ordinal models. cFor each subgroup, potential masked depression is based on a comparison of the multivariate regression reported in Table 2 with the counterpart MIMIC reported in Table S1. dTwo-tailed test significance is as follows: 1) z=1.960 (p=0.05); 2) z=2.326 (p=0.025); 3) z=2.576 (p=0.01); 4) z=3.291 (p=0.005). Abbreviations: CES-D, Center for Epidemiologic Studies Depression Scale; CFI, Comparative Fit Index; RMSEA, Root Mean Square Error of Approximation; SE, standard error; SRMR, Standardized Root Mean Square Residual; TLI, Tucker-Lewis Index. |
  | Table 3C CES-D depression items in co-occurring and synergistic stroke subgroups based on overweight and diabetes status: multiple indicators–multiple causes (MIMIC) modelsa,b Notes: aThe measurement loading (λ) of the CES-D item depressed was fixed at 1 to set the metric of the measurement model. Estimated measurement loadings for the remaining CES-D items ranged from 0.300 (hopeful) to 0.883 (sad). Depending on the specific MIMIC model, 11–16 of the 20 measurement loadings were ≥0.450. bEach of the specific MIMIC models appear to fit the data adequately based on multiple fit indices: R2: 0.554–0.985; CFI: 0.945–0.987; TLI: 0.875–0.975; RMSEA: 0.015–0.031; and SRMR: 0.021–0.031. Except for R2, these fit indices are not available in MLR estimation for MIMIC models with ordinal measurement items, such as the four-category CES-D items in the present study. They were derived from counterpart MLR-estimated MIMIC models in which all CES-D measurement items of the latent trait of depression were specified to be continuous measures. In contrast to the ordinal models in which separate ordinal variables, along with their identical, overlapping instrumental variables, were specified for each of the non-traditional CES-D items, the specification in the continuous models differed in using a single continuous variable based on the sum of all four non-traditional CES-D measurement items, along with its identical, overlapping instrumental variable, as a “causal indicator” for estimating the pathway of the bi-directional relationship that predicts total depression. (Note that the four non-traditional CES-D items are still retained as individual items that serve as “effect indicators” for estimating the pathway of the bi-directional relationship that manifests as a result of the latent trait of depression.) In addition, the discrete nature of the continuous data (in contrast to the probabilistic nature of the data for analysis in the ordinal models) also allows residuals of CES-D items to be correlated, which contributes to model fit. It is unknown whether the need to collapse the four non-traditional items into a single variable in the continuous model tends to erode its fit; however, the fact that there is adequate fit in the continuous models across four fit indices despite this possibility may be taken to reveal that there is proper specification and adequate fit to the data within the ordinal models. cFor each subgroup, potential masked depression is based on a comparison of the multivariate regression reported in Table 2 with the counterpart MIMIC reported in Table S1. Refer to the main text for more information. dTwo-tailed test significance is as follows: 1) z=1.960 (p=0.05); 2) z=2.326 (p=0.025); 3) z=2.576 (p=0.01); 4) z=3.291 (p=0.005). Abbreviations: CES-D, Center for Epidemiologic Studies Depression Scale; CFI, Comparative Fit Index; RMSEA, Root Mean Square Error of Approximation; SE, standard error; SRMR, Standardized Root Mean Square Residual; TLI, Tucker-Lewis Index. |
  | Table 3D CES-D depression items in co-occurring and synergistic vascular cognitive impairment subgroups based on overweight and diabetes status: multiple indicators–multiple causes (MIMIC) modelsa,b Notes: aThe measurement loading (λ) of the CES-D item depressed was fixed at 1 to set the metric of the measurement model. Estimated measurement loadings for the remaining CES-D items ranged from 0.294 (hopeful) to 0.867 (sad). Eleven to 16 of the 20 measurement loadings were ≥0.450. bEach of the specific MIMIC models appear to fit the data adequately based on multiple fit indices: chi-square: 175.477–341.392; chi-square/degrees of freedom: 1.427–2.253; R2: 0.563–0.624; CFI: 0.974–0.977; TLI: 0.945–0.961; RMSEA: 0.019–0.022; and SRMR: 0.021–0.027. Except for R2, these fit indices are not available for MLR estimation of MIMIC models with ordinal measurement items, such as the four-category CES-D items in the present study. They were derived from counterpart MLR-estimated MIMIC models in which all CES-D measurement items of the latent trait of depression were specified to be continuous measures. In contrast to the ordinal models in which separate ordinal variables, along with their identical, overlapping instrumental variables, were specified for each of the non-traditional CES-D items, the specification in the continuous models differed in using a single continuous variable based on the sum of all four non-traditional CES-D measurement items, along with its identical, overlapping instrumental variable, as a “causal indicator” for estimating the pathway of the bi-directional relationship that predicts total depression. (Note that the four non-traditional CES-D items are still retained as individual items that serve as “effect indicators” for estimating the pathway of the bi-directional relationship that manifests as a result of the latent trait of depression.) In addition, the discrete nature of the continuous data (in contrast to the probabilistic nature of the data for analysis in the ordinal models) also allows residuals of CES-D items to be correlated, which contributes to model fit. It is unknown whether the need to collapse the four non-traditional items into a single variable in the continuous model tends to erode its fit; however, the fact that there is adequate fit in the continuous models across four fit indices despite this possibility may be taken to reveal that there is proper specification and adequate fit to the data within the ordinal models. cFor each subgroup, potential masked depression is based on a comparison of the multivariate regression reported in Table 2 with the counterpart MIMIC reported in Table S1. Refer to the main text for more information. dTwo-tailed test significance is as follows: 1) z=1.960 (p=0.05); 2) z=2.326 (p=0.025); 3) z=2.576 (p=0.01); 4) z=3.291 (p=0.005). Abbreviations: CES-D, Center for Epidemiologic Studies Depression Scale; CFI, Comparative Fit Index; RMSEA, Root Mean Square Error of Approximation; SE, standard error; SRMR, Standardized Root Mean Square Residual; TLI, Tucker-Lewis Index. |
Although dysphoric mood is clearly indicated by items such as blue, crying, and sad, other closely related items of negative affect are ambiguous and may reflect masked depression. For instance, in the absence of items that clearly indicate dysphoric mood, the item depressed may reflect anhedonia in some participants. Items reflecting low positive affect (eg, reverse-coded items for happy, enjoyed life, or hopeful) or interpersonal symptoms (eg, talked less than others) also suggest the potential for masked depression when they occur in the absence of items that clearly indicate dysphoric mood.
In Table 3A–D, the symbol (+++) indicates the strongest level of evidence that reveals the potential for masked depression in participants. This evidence for a vascular condition or subgroup occurs when none of the negative affect items of dysphoric mood from the CES-D Depression Scale are statistically significant, or if statistically significant, reveal negative factor loadings. Furthermore, this evidence is more pronounced when negative affect or interpersonal items of anhedonia (eg, talked less than others) and/or low positive affect items are statistically significant. The same pattern of effects by identical items in the descriptive MIMIC must also occur in the corresponding explanatory MIMIC. Although the same or different items may occur in the corresponding multivariate regression, the same type of pattern must continue to hold; however, masked depression is also indicated if the multivariate regression yields no statistically significant items.
The symbol (++) indicates that there is some evidence that reveals the potential for masked depression in participants. It is similar to (+++); however, the overall depression score, depression, is positive and statistically significant in the descriptive MIMIC and corresponding explanatory MIMIC. (In one case, it is positive and statistically significant in the descriptive MIMIC only; however, dysphoric items are not statistically significant in the corresponding explanatory MIMIC.) When the latent factor of depression is statistically significant, none of the individual items from the CES-D Depression Scale are also found to be statistically significant. Thus, there is no pattern detected across the depression items within the vascular condition or subgroup, only that the overall level of depression is higher than in other participants. This ambiguity suggests that there may be potential for masked depression in some participants on account of the inconsistencies in the presentation or profile of depression symptoms across participants with the vascular condition or in the vascular subgroup.
The symbol (+) indicates that there is more limited, tentative evidence that there may be potential for masked depression in some participants. The pattern of evidence in the abovementioned paragraphs describing (+++) or (++) occurs in one or two, but not all three, types of statistical analyses (ie, the descriptive MIMIC, explanatory MIMIC, and multivariate regression) for a vascular condition or subgroup, such that one or more negative affect items of dysphoric mood are statistically significant in the remaining statistical analysis or analyses. The MIMIC models constitute the proper specifications for revealing the profile of depression items for a vascular condition or subgroup because they account for the overall level of depression (depression), along with the individual items of the CES-D Depression Scale. Therefore, the most sound of the scenarios reflected by (+) indicate potential for masked depression in the context of evidence for its consistent presentation or phenomenology based on the depression item profiles, or higher overall level of depression, within vascular conditions or subgroups. These situations occur when the pattern of evidence, as described in the abovementioned paragraphs describing (+++) or (++), occurs in the descriptive and explanatory MIMICs, and the negative affect item(s) of dysphoric mood are limited to the multivariate regression.
One might ask why the multivariate regressions were estimated at all, given that they do not model the precise profile of depression, or overall level of depression, within a vascular condition or subgroup. The multivariate regressions were conducted to reveal whether depression items may nonetheless stand out for some individuals and be detected during an assessment of the endorsed items from the CES-D Depression Scale, without also considering, and adjusting for, the overall level of depression (depression). In these scenarios, some – but not all – individuals with a vascular condition or from a vascular subgroup may still be detected during assessment to be depressed based on their sufficient endorsement of dysphoric items on the CES-D Depression Scale, even as the relationships across all individuals with the vascular condition, or from the vascular subgroup, that predict dysphoric mood items do not remain statistically significant in the descriptive and explanatory MIMIC analyses. These differences occur because the multivariate regressions estimate relationships that are based on the overall variation within each CES-D item, with all relationships across the CES-D items estimated simultaneously, while the MIMIC analyses estimate relationships that are based on the unique variation within each CES-D item after accounting for shared variation with other CES-D items and the overall level of depression (depression). On the other hand, (+) also includes scenarios in which the descriptive and/or explanatory MIMIC reveal dysphoric mood item(s), while the multivariate regression does not. These situations suggest that there may be individuals with masked depression who may be missed when clinicians depend only on inspecting the endorsed items from the CES-D Depression Scale, especially since the relationships across all individuals with the vascular condition, or from the vascular subgroup, that predict dysphoric mood items remain statistically significant in the descriptive and explanatory MIMIC analyses.
Finally, the symbol (0) reveals that there is no evidence to suggest the potential for masked depression. Negative affect item(s) of dysphoric mood from the CES-D Depression Scale are statistically significant in the descriptive and perhaps explanatory MIMIC, as well as the multivariate regression, conducted for the vascular condition or subgroup.
Overall patterns and specific findings
Table 3A–D reveals several CES-D items that are positive and statistically significant in the descriptive MIMIC regressions for each of the overall categories of vascular conditions (hypertension, silent CVD, stroke, and vascular cognitive impairment) but not in their respective explanatory MIMIC regressions. However, when overweight and/or diabetes co-occur with these progressive vascular conditions, all of these clustered conditions reveal the same CES-D items, or the latent trait of depression, to be positive and statistically significant in the descriptive and respective explanatory MIMIC regressions.
There is consistent evidence in Table 3A–D of masked vascular depression (+++ or ++) in older males with clustered conditions related to the metabolic syndrome. In older males, hypertension × overweight × diabetes predicts the latent factor of depression; silent CVD × overweight predicts enjoyed life and talked less than others; and silent CVD × diabetes predicts hopeful, everything an effort, difficult concentrating, lonely, unfriendly, and disliked me. When similar clustered conditions were also statistically significant in older females, the depression was not masked.
There is also consistent evidence of masked depression (+++ or ++) in the overall sample and/or in older females with clustered overweight conditions without diabetes involving more advanced vascular disease. In the overall sample and in older females, stroke × overweight × no diabetes predicts bothered by things, everything an effort, and not get going. In older females, vascular cognitive impairment × overweight × no diabetes predicts enjoyed life, poor appetite, unfriendly, and disliked me. In the overall sample, vascular cognitive impairment × not overweight × no diabetes predicts the latent factor of depression.
More tentative evidence of masked depression (+) occurs across the progression of vascular diseases in the following clustered conditions: hypertension × not overweight × diabetes in older females; silent CVD × no smoking × not overweight in older males; silent CVD × no smoking × no diabetes in older males; silent CVD × no smoking × no diabetes in older females; stroke; stroke × diabetes; stroke × overweight × diabetes; VCI; and VCI × overweight in older females.
Finally, the following vascular diseases and clustered conditions show evidence of non-masked depression (0): hypertension; silent CVD; silent CVD × overweight in older females; and vascular cognitive impairment × overweight. Recall that the explanatory MIMIC regressions for hypertension and silent CVD do not show positive and statistically significant CES-D items. Only the two clustered conditions reveal evidence of dysphoric mood in the descriptive and explanatory MIMIC regressions as well as the multivariate regression
Discussion
The collective findings of the present cross-sectional study of an epidemiological community sample of older White and Black Americans provide evidence that co-occurring conditions of excess weight, diabetes, and vascular disease interact to predict clinically significant vascular depression. When other findings from the literature were also considered, it becomes apparent that interactions are not limited to those among diabetes and co-occurring physical conditions that predict co-morbid depression experienced within the same timeframe, but that interactions among diabetes and co-morbid depression within the same timeframe may occur as well that in turn predict the eventual development or progression of vascular disease. For instance, the literature reveals that older Mexican Americans with type 2 diabetes experienced a gradient of risk in developing macro- and microvascular complications during a 7-year period when they experienced “minimal” depression (0 < CES-D < 16) or clinically significant depression (CES-D ≥ 16), and these findings were replicated based on clinical diagnostic criteria.72 Thus, interactions across physical and mental health conditions are likely to be ubiquitous; however, for the purposes of screening for participants with vascular depression, or at risk of developing vascular depression, interactions that manifest within the same time period among co-occurring physical conditions are most useful in identifying subgroups at risk for vascular depression.
The present study reveals that presentations of vascular depression may be hidden or “masked” because self-reported sadness or feeling blue is not prominently endorsed. Instead, clinically significant levels of low positive affect, anhedonia, and, potentially, social withdrawal are indicated. Although response biases to CES-D items of dysphoric mood, which older men and other subgroups may experience as stigmatizing, cannot be ruled out, the findings do not reveal masked depression in all subgroups of older men with metabolic/progressive vascular disease. If the only influence on masked depression in older men were response bias due to perceived stigma in endorsing dysphoric mood, masked depression in all of the metabolic/vascular subgroups of older men with depression would be expected. The fact that this outcome did not occur suggests that there may be significant scope for alexithymia within specific metabolic/vascular subgroups. The New Haven EPESE data did not include a measure for alexithymia, which future investigations should address.
The present study affords evidence that in older males, the less progressed stages of vascular disease (hypertension, silent CVD) in the context of co-occurring metabolic illness (overweight and diabetes) are more likely to present with masked depression and can present with a diverse or troubling pattern of symptoms.
The metabolic syndrome of hypertension, excess weight in the abdominal versus hip area, and diabetes is common in older males; however, the symptom profile of depression is likely to be very heterogeneous since the descriptive and explanatory MIMIC models only predicted the overall level of depression (depression). This heterogeneity in the symptom profile is also suggested by the evidence for a bi-directional relationship between the metabolic syndrome and depression in both cross-sectional and longitudinal meta-analyses.73 Different patterns and levels of symptoms may tend to be endorsed in any given situation depending on whether the metabolic syndrome is operating as a risk factor for depression or vice versa. Another possibility is that different hypertension medications, or classes of medications, may precipitate heterogeneous side effects that result in inconsistent symptom profiles. As alexithymia is linked to hypertension,36–38 it will be important for future research to determine whether the inconsistency in depression symptom endorsement by older males with hypertension is related to alexithymia.
The symptoms of masked depression in overweight, older males with silent CVD (enjoyed life, talked less than others) suggest anhedonia and perhaps alexithymia. In addition to serving as a risk factor for precipitating or accelerating hypertension and diabetes, overweight condition has a bi-directional relationship with depression according to the findings of a longitudinal meta-analysis.74 Overweight condition is associated with major and minor depression,75 and depression almost doubles the risk of developing diabetes.76 The symptom pattern of masked depression in older males when diabetes co-occurs with silent CVD (hopeful, everything an effort, difficulty concentrating, lonely, unfriendly, and disliked me) suggests distress; it may indicate poor glycemic control and poor adherence with diabetes medication, exercise, diet, and self-care regimens for other illness conditions such as hypertension and hyperlipidemia.11,77–79 Of course, these impaired outcomes may also be linked to depressive symptom profiles in other clusters of metabolic and vascular diseases.
Different symptom patterns of masked depression emerge in the more progressed stages of vascular disease. Masked depression is detected in older women, or in both genders, within specific metabolic/vascular subgroups with stroke or VCI. The findings of heightened alexithymia in women with either right- or left-hemisphere stroke48 support the present study finding of masked depression in overweight older women without diabetes who reported stroke (bothered by things, not get going, and everything an effort). (These symptoms of masked depression also occur in the non-gendered subgroup in the full sample.) Two items (unfriendly and disliked me) remained statistically significant in the descriptive and explanatory MIMIC models that revealed masked depression in overweight older women without diabetes who had VCI. It is possible that these two items reveal relationship difficulties that stem from aggressive behavior due to a lower tolerance threshold in patients with vascular dysfunction and depression, as reported recently by Turk et al.80 However, this interpretation would not apply universally, and it might be speculated, may not apply as much to the weight loss, wasting, and perhaps more extensive alexithymia in more advanced phases of vascular dysfunction. The basis for restricting how widely the interpretation may apply has to do with the heterogeneous symptom profile of depression when both genders with VCI were neither overweight nor diagnosed with diabetes – only the overall level of depression (depression) is statistically significant in the descriptive and explanatory MIMIC models. Finally, there is tentative evidence for masked depression in several metabolic/vascular subgroups across the stages of vascular disease, although no specific patterns emerge in these subgroups.
With the exception of the masked depression symptom pattern suggestive of anhedonia in overweight, older males with silent CVD (enjoyed life and talked less than others), the symptom patterns of masked depression among the participant subgroups in this study end up satisfying a stricter definition of masked depression proposed by Judd et al.81,82 These investigators revealed evidence of “subsyndromal symptomatic depression” in which two-thirds to three-fourths of depressed individuals do not meet DSM-IV criteria for major depression, dysthymia, minor depression, or recurrent brief depression because they do not endorse either dysphoric mood or anhedonia despite other depressive symptoms. It is disconcerting that these individuals with masked depression are likely to be missed in a diagnostic interview, which further supports the utility of screening targeted subgroups at risk using a self report depression scale such as the CES-D. These individuals would appear to be experiencing distress, alexithymia, and/or somatization (the tendency to experience and communicate psychological distress as physical symptoms), depending on their symptom profile of depression. Of course, the less strict definition of masked depression based only on low endorsed dysphoric mood remains useful since health providers are likely to miss depression characterized by anhedonia in the absence of screening.
Although much of the depression in participant subgroups with co-occurring cerebrovascular and metabolic conditions is clearly masked in its presentation, several additional disease clusters are equivocal regarding masked depression, which suggests a more variable symptom profile where depression may present as masked in some participants and non-masked in others. Finally, there is evidence of non-masked depression that clearly emphasizes dysphoric mood in only two participant subgroups (ie, overweight older women with silent CVD and overweight participants of both genders with VCI).
The present study uses a novel model specification strategy to estimate exhaustively specified latent trait or MIMIC models, affording a new kind of empirical evidence regarding the types and range of profiles of masked depression that manifest in co-occurring metabolic and progressive vascular conditions. Well-validated measures and a strict approach to partial out confounding factors were employed. Recall that in each of the overall cerebrovascular conditions (ie, hypertension, silent CVD, stroke, and vascular cognitive impairment) in Table 3A–D, several CES-D items are positive and statistically significant in the descriptive MIMIC regressions but not also in the respective explanatory MIMIC regressions. However, it is striking that these vascular conditions interact with overweight condition and/or diabetes within either of the gender subgroups, or within the sample overall, to predict multiple, positive CES-D items, or the latent factor of depression, which are statistically significant in both the descriptive and parallel explanatory MIMIC regressions. This highly consistent pattern suggests that there may be multiple profiles, or contributing phenomenology, of depression in progressive vascular disease that may be influenced by various co-occurring physical conditions and psychosocial factors. Therefore, in addition to using validated measures and accounting for confounding factors, it is important to model specific co-occurring conditions – in this study the metabolic and inflammatory conditions of obesity and diabetes – in order to identify specific profiles of depression related to these co-occurring conditions.
The highly consistent findings across the descriptive and corresponding explanatory MIMIC models support the contention that careful attention to modeling co-morbidity may help address the tradeoff dilemma in which it is feasible to use more extensive, gold standard measures only in smaller observational studies while comprehensive control of the range of confounding factors is possible only in larger studies.83,84 However, it might still be premature to assume that modeling co-morbidity will necessarily go far enough in addressing the tradeoff dilemma since the study data afford related unique strengths that may also have played an important role. For most adults, retirement and changes in social roles occur at the age of 65 years; thus, independent correlates for depression might shift at this age as well. Since the New Haven EPESE data are limited to older adults of age ≥65 years, minimal confounding of age-related correlates is an important strength of these cross-sectional data. Furthermore, the age distribution is evenly divided between the young–old (65–74 years) and old–old (75+ years). Finally, co-occurring illnesses may be more prevalent in senior housing facilities than among other older adults living independently, and the inclusion of all older adults in these facilities in the study sampling frame assures that these co-occurring illnesses are well represented.
The study has several limitations. In contrast to the overweight condition and progressive categories of CVD, which are based on objective health criteria, diabetes is a self-report measure; it is likely that the EPESE community survey missed undisclosed and undiagnosed diabetes. A larger overall sample is necessary to identify co-occurring cerebrovascular conditions that were more progressed and much less prevalent, as well as their co-occurrence both with being overweight and diabetes; this is especially true in the older male subsample. The secondary data did not include a validated measure of alexithymia. Therefore, CES-D profiles by individuals without alexithymia in coping with the distress of their illness conditions cannot be distinguished from CES-D profiles of individuals experiencing alexithymia, part of which may involve distress aggravated by the lack of emotional and physiological regulation from alexithymia. The prevalence of total CES-D scores between 16 and 27 (mild or minor depression or subsyndromal depression), and especially >27 (severe or major depression), was insufficient to estimate these separate, more restricted models. Finally, the lack of follow-up confirmatory diagnostic assessments is an important limitation, although a two-stage scoring method for research situations without follow-up confirmatory assessments was used to link CES-D scores more closely with DSM-III-R diagnostic criteria.59
However, the CES-D also affords a unique advantage for screening compared to other self-report inventories and diagnostic criteria. It may be more appropriate to conceive the four non-traditional items within the CES-D as tapping the closely related construct of distress rather than depression, although it may not be the case that these four items tap all forms of distress. Even so, the CES-D can reveal participants experiencing distress, depression, or depression with distress. With regard to at-risk subgroups with metabolic and vascular conditions, the use of the self-report CES-D to detect distress and depression, including masked vascular depression, could have critical utility in preventive and transdisciplinary care programs (involving social workers and other mental health clinicians, physicians, and nurses) in convenient settings such as primary care, assisted living facilities, home healthcare, and senior centers. These programs may be more cost-effective if there is a greater focus on larger patient subgroups with unmet medical care needs, as recommended by Gastelum et al.10 Older men at risk for undiagnosed diabetes and the metabolic syndrome constitute one large patient subgroup where targeted screening for masked depression may be cost-effective. Depressive symptoms may serve as a clue to undiagnosed diabetes, which was found to be 1) more common in individuals who were male, overweight, or hypertensive (ie, with the metabolic syndrome),85 2) an indicator of non-adherence to diabetes diet or care regimen,86 and 3) a predictor of dropout from weight loss programs.87 There is some evidence that alexithymia may lead to unhealthy dietary habits and sedentary lifestyles.88 In addition, alexithymia is a stronger predictor of emotional eating, a behavioral manifestation of distress, in obese men than obese women.44 Larsen et al44 attribute this finding to the greater means for emotional regulation available to women, and they call for interventions that are targeted to men to curb emotional eating.
Conclusion
The scope for targeting cost-effective screening may be especially promising in older men with the metabolic syndrome (either hypertension or silent CVD in the context of excess weight and/or diabetes), given their more pronounced frequency in the representative New Haven community. There is also scope for targeting older women or both genders in several diabetes and/or overweight clusters that involve stroke or VCI. Replicating the study findings in different data can help us strategize how to use screening instruments more cost-effectively by targeting their use within older adult subgroups who appear to be at greater risk for major depression, undiagnosed diabetes, or non-adherence to medical care regimens or dietary and lifestyle interventions, any of which may accelerate progression of metabolic and cerebrovascular conditions. Although the study has placed primary emphasis on identifying participant subgroups with clear evidence of masked depression, depression screening and follow-up should also include the targeted subgroups in this study with non-masked depression (ie, with clearly endorsed dysphoric mood) and with equivocal findings regarding masked depression.
Acknowledgments
The author received funding as the principal investigator from the Hartford Geriatric Social Work Faculty Scholar Initiative (no grant number) and the National Institute of Mental Health (grants 1R03MH064627-01 and 5R03MH064627-02). The author wishes to thank Drs Susan Hughes, Jeanne Teresi, and Karolynn Siegel for their consultation during grantsmanship and the early phase of this project. Randy Dexter assisted with table formatting.
Disclosure
The author reports no conflicts of interest in this work.
References
Roose SP, Devanand DP. Treatment of mood disorders in late life. In: Mann JJ, McGrath PJ, Roose SP, editors. Clinical Handbook for the Management of Mood Disorders. New York, NY: Cambridge; 2013:119–132. | ||
Montgomery SA. Late-life depression: rationalizing pharmacological treatment options. Gerontology. 2001;48(6):392–400. | ||
First MB, Endicott J. The diagnosis of mood disorders. In: Mann JJ, McGrath PJ, Roose SP, editors. Clinical Handbook for the Management of Mood Disorders. New York, NY: Cambridge; 2013:7–19. | ||
Devanand DP, Nobler MS, Singer T, et al. Is dysthymia a different disorder in the elderly? Am J Psychiatry. 1994;151(11):1592–1599. | ||
Devanand DP, Adorno E, Cheng J, et al. Late onset dysthymic disorder and major depression differ from early onset dysthymic disorder and major depression in elderly outpatients. J Affect Disord. 2004;78(3):259–267. | ||
Fisher L, Skaff MM, Mullan JT, Arean P, Glasgow R, Masharani U. A longitudinal study of affective and anxiety disorders, depressive affect, and diabetes distress in adults with Type 2 diabetes. Diabet Med. 2008;25(9):1096–1101. | ||
Meeks TW, Vahia IV, Lavretsky H, Kulkarni G, Jeste DV. A tune in “a minor” can “b major”: a review of epidemiology, illness course, and public health implications of subthreshold depression in older adults. J Affect Disord. 2011;129(1–3):126–142. | ||
Lin EH, Katon W, Von Korff M, et al. Effect of improving depression care on pain and functional outcomes among older adults with arthritis: a randomized controlled trial. JAMA. 2003;290(18):2428–2429. | ||
Ayuso-Mateos JL, Nuevo R, Verdes E, Naidoo N, Chatterji S. From depressive symptoms to depressive disorders: the relevance of thresholds. Br J Psychiatry. 2010;195(5):365–371. | ||
Gastelum E, Muskin PR, Shapiro PA. Depression in the context of physical illness. In: Mann JJ, McGrath PJ, Roose SP, editors. Clinical Handbook for the Management of Mood Disorders. New York, NY: Cambridge; 2013:181–194. | ||
Gonzalez JS, Safren SA, Cagliero E, et al. Depression, self-care, and medication adherence in type 2 diabetes: relationships across the full range of symptom severity. Diabetes Care. 2007;30(9):2222–2227. | ||
Gonzalez JS, Peyrot M, McCarl LA, et al. Depression and diabetes treatment nonadherence: a meta-analysis. Diabetes Care. 2008;31(12):2398–2403. | ||
Esbitt SA, Tanenbaum MA, Gonzalez JS. Disentangling clinical depression from diabetes-specific distress: making sense of the mess we’ve made. In: Lloyd CE, Pouwer F, Hermans N, editors. Screening for Depression and Other Psychological Problems in Diabetes. London: Springer; 2013:27–46. | ||
Fisher L, Gonzalez JS, Polonsky WH. The confusing tale of depression and distress in patients with diabetes: a call for greater clarity and precision. Diabet Med. 2014;31(7):764–772. | ||
Fisher L, Mullan JT, Arean P, Glasgow RE, Hessler D, Masharani U. Diabetes distress and not clinical depression or depressive affect is associated with glycemic control in both cross-sectional and longitudinal analyses. Diabetes Care. 2010;33:23–28. | ||
Pouwer F, Beekman AT, Nijpels G, et al. Rates and risks for co-morbid depression in patients with Type 2 diabetes mellitus: results from a community-based study. Diabetologia. 2003;46(7):892–898. | ||
Gallo JJ, Rabins PV, Lyketsos CG, Tien AY, Anthony JC. Depression without sadness: functional outcomes of nondysphoric depression in later life. J Am Geriatr Soc. 1997;45(5):570–578. | ||
Kales HC, Maixner DF, Mellow AF. Cerebrovascular disease and late-life depression. Am J Geriatr Psychiatry. 2005;13(2):88–98. | ||
Krishnan KRR, Hays JC, Blazer DG. MRI-defined vascular depression. Am J Psychiatry. 1997;154(4):497–501. | ||
Steffens DC, Krishnan KR. Structural neuroimaging and mood disorders: recent findings, implications for classification, and future directions. Biol Psychiatry. 1998;43(10):705–712. | ||
Krishnan KRR, Steffens DC. The pathophysiological basis of late-life depression. Psychiatr Times. 1998;15(2). | ||
Slavich GM, Irwin MR. From stress to inflammation and major depressive disorder: a social signal transduction theory of depression. Psychol Bull. 2014;140(3):774–815. | ||
Longstreth WT, Diehr P, Manolio TA, Beauchamp NJ, Jungreis CA, Lefkowitz D. Cluster analysis and patterns of findings on cranial Magnetic Resonance Imaging of the elderly. Arch Neurol. 2001;58:635–640. | ||
Taylor WD, MacFall JR, Provenzale JM, et al. Serial MR imaging of volumes of hyperintense white matter lesions in elderly patients. Am J Roentgenol. 2003;181:571–576. | ||
Steffens DC, Krishnan KRR, Crump C, Burke GL. Cerebrovascular disease and the evolution of depressive symptoms in the Cardiovascular Health Study. Stroke. 2002;33:1636–1644. | ||
Aronow WS, Ahn C. Risk factors for new atherothrombotic brain infarction in older Hispanic men and women. J Gerontol. 2002;57(1):M61–M63. | ||
Kirpichnikov D, Sowers JR. Diabetes mellitus and diabetes-associated vascular disease. Trends Endocrinol Metab. 2001;12(5):225–230. | ||
Stewart R, Liolitsa D. Type 2 diabetes mellitus, cognitive impairment, and dementia. Diabet Med. 1999;16(2):93–112. | ||
Whitmer RA, Gunderson EP, Barrett-Connor E, Quesenberry CP Jr, Yaffe K. Obesity in middle age and future risk of dementia: a 27 year longitudinal population based study. BMJ. 2005;330(7504):1360. | ||
Kannel WB, Wolf PA, Benjamin EJ, Levy D. Prevalence, incidence, prognosis, and predisposing conditions for atrial fibrillation: population-based estimates. Am J Cardiol. 1998;82(8A):2N–9N. | ||
Reaven GM. Role of insulin resistance in human disease. Diabetes. 1988;37:1595–1607. | ||
Kuusisto J, Mykkänen L, Pyörälä K, Laakso M. NIDDM and its metabolic control predict coronary heart disease in elderly subjects. Diabetes. 1994;43:960–967. | ||
Okosun IS, Chandra KMD, Choi S, Christman J, Dever GE, Prewitt TE. Hypertension and type 2 diabetes comorbidity in adults in the United States: risk of overall and regional adiposity. Obes Res. 2001;9(1):1–9. | ||
Esser N, Legrand-Poels S. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res Clin Pract. 2014;105(2):141–150. | ||
Lemche E, Brammer MJ, David AS, et al. Interoceptive-reflective regions differentiate alexithymia traits in depersonalization disorder. Psychiat Res. 2013;214(1):66–72. | ||
Todarello O, Taylor GJ, Parker JDA, Fanelli M. Alexithymia in essential hypertensive and psychiatric outpatients: a comparative study. J Psychosom Res. 1995;39(8):987–994. | ||
Jula A, Salminen JK, Saarijärvi S. Alexithymia: a facet of essential hypertension. Hypertension. 1999;33(4):1057–1061. | ||
Grabe HJ, Schwahn C, Barnow S, et al. Alexithymia, hypertension, and subclinical atherosclerosis in the general population. J Psychosom Res. 2010;68(2):139–147. | ||
Chatzi L, Bitsios P, Solidaki E, et al. Type 1 diabetes is associated with alexithymia in nondepressed, non-mentally ill diabetic patients: a case-control study. J Psychosom Res. 2009;67(4):307–313. | ||
Housiaux M, Luminet O, VanBroeck N, Dorchy H. Alexithymia is associated with glycaemic control of children with type1 diabetes. Diabetes Metab. 2010;36(6 pt 1):455–462. | ||
Luminet O, de Timary P, Buysschaert M, Luts A. The role of alexithymia factors in glucose control of persons with type 1 diabetes: a pilot study. Diabetes Metab. 2006;32:417–424. | ||
Meunier J, Dorchy H, Luminet O. Does family cohesiveness and parental alexithymia predict glycaemic control in children and adolescents with diabetes? Diabetes Metab. 2008;34(5):473–481. | ||
Topsever P, Filiz TM, Salman S, et al. Alexithymia in diabetes mellitus. Scott Med J. 2006;51:15–20. | ||
Larsen JK, van Striena T, Eisingac R, Engels RCME. Gender differences in the association between alexithymia and emotional eating in obese individuals. J Psychosom Res. 2006;60:237–243. | ||
Faccioli J, Guinzbourg M, Alvarez A, Finkelsztein C, Job A, Litwak L. Diabetes, alexithymia and depression. Vertex. 2012;23(106):433–438. | ||
Hintistan S, Cilingir D, Birinci N. Alexithymia among elderly patients with diabetes. Pak J Med Sci. 2013;29(6):1344–1348. | ||
Lemche AV, Chaban OS, Lemche E. Alexithymia as a risk factor for type 2 diabetes mellitus in the metabolic syndrome: a cross-sectional study. Psychiatry Res. 2014;215(2):438–443. | ||
Spalletta G, Pasini A, Costa A, et al. Alexithymic features in stroke: effects of laterality and gender. Psychosom Med. 2001;63:944–950. | ||
Bossu P, Salani F, Cacciari C, et al. Disease outcome, alexithymia, and depression are differently associated with serum IL-18 levels in acute stroke. Curr Neurovasc Res. 2009;6(3):163–170. | ||
Bylsma LM, Morris BH, Rottenberg J. A meta-analysis of emotional reactivity in major depressive disorder. Clin Psychol Rev. 2008;28(4):676–691. | ||
Bylsma LM, Rottenberg J. Uncovering the dynamics of emotion regulation and dysfunction in daily life with ecological momentary assessment. In: Nyklicek I, Vingerhoets AJJM, Zeelenberg M, editors. Emotion Regulation and Well-being: Part 3. New York: Springer; 2011:225–244. | ||
Peeters F, Nicolson NA, Berkhof J, Delespaul P, deVries M. Effects of daily events on mood states in major depressive disorder. J Abnorm Psychol. 2003;112(2):203–211. | ||
Rottenberg J, Kasch KL, Gross JJ, Gotlib IH. Sadness and amusement reactivity differentially predict concurrent and prospective functioning in major depressive disorder. Emotion. 2002;2:135–146. | ||
Papassotiropoulos A, Heun R, Maier W. The impact of dementia on the detection of depression in elderly subjects from the general population. Psychol Med. 1999;29(1):113–120. | ||
Penninx BWJH, Guralnik JM, Bandeen-Roche K, et al. The protective effect of emotional vitality on adverse health outcomes in disabled older women. J Am Geriatr Soc. 2000;48:1359–1366. | ||
Taylor JO, Wallace RB, Ostfeld AM, Blazer DG. Established Populations for Epidemiologic Studies of the Elderly, 1981-1993: [East Boston, Massachusetts, Iowa and Washington Counties, Iowa, New Haven, Connecticut, and North Central North Carolina]. 3rd ICPSR version ed. Bethesda, MD: National Institute on Aging; 1997. [Ann Arbor, MI, Inter-university Consortium for Political and Social Research; 1998]. | ||
Boyd JH, Weissman MM. Screening for depression in a community sample. Arch Gen Psychiatry. 1982;39:1195–1200. | ||
Hybels CF, Blazer DG, Pieper CF. Toward a threshold for subthreshold depression: an analysis of correlates of depression by severity of symptoms using data from an elderly community sample. Gerontologist. 2001;41(3):357–365. | ||
Schein RL, Koenig HG. The Center for Epidemiological Studies-Depression (CES-D) scale: assessment of depression in the medically ill elderly. Int J Geriatr Psychiatry. 1997;12(4):436–446. | ||
Alexopoulos GS, Meyers BS, Young RC, Kakuma T, Silbersweig D, Charlson M. Clinically defined vascular depression. Am J Psychiatry. 1997;154(4):562–565. | ||
Extermann M, Overcash J, Lyman GH, Parr J, Balducci L. Comorbidity and functional status are independent in older cancer patients. J Clin Oncol. 1998;16(4):1582–1587. | ||
Miller MD, Paradis CF, Houck PR, et al. Rating chronic medical illness burden in geropsychiatric practice and research: application of the Cumulative Illness Rating Scale. Psychiatry Res. 1992;41:237–248. | ||
Guralnik JM, Farmer ME, Blazer DG, et al. Chronic conditions. In: Cornoni-Huntley J, Blazer DG, Lafferty ME, Everett DF, Brock DB, Farmer ME, editors. Established Populations for Epidemiologic Studies of the Elderly (Volume II): Resource Data Book. Bethesda, MD: National Institute on Aging; 1990:95–128. | ||
Bassuk SS, Berkman LF, Wypij D. Depressive symptomatology and incident cognitive decline in an elderly community sample. Arch Gen Psychiatry. 1998;55(12):1073–1081. | ||
Stroup-Benham CA, Markides KS, Black SA, Goodwin JS. Relationship between low blood pressure and depressive symptomatology in older people. J Am Geriatr Soc. 2000;48(3):250–255. | ||
Palta M, Prineas RJ, Berman R, Hannan P. Comparison of self-reported and measured height and weight. Am J Epidemiol. 1982;115:223–230. | ||
Muthén LK, Muthén BO. Mplus User’s Guide. 5th ed. Los Angeles: Muthén and Muthén; 1998-2007. | ||
David A. Kenny [website on the Internet]. Identification; 2012. Available from: http://davidakenny.net/cm/identify_formal.htm#B3b. Accessed September 3, 2016. | ||
Christensen H, Jorm AF, Mackinnon AJ, et al. Age differences in depression and anxiety symptoms: a structural equation modelling analysis of data from a general population sample. Psychol Med. 1999;29:325–339. | ||
Grayson DA, Mackinnon A, Jorm AF, Creasey H, Broe GA. Item bias in the Center for Epidemiological Studies Depression scale: effects of physical disorders and disability in an elderly community sample. J Gerontol. 2000;55B(5):273–282. | ||
Radloff LS. The CES-D scale: a self-report depression scale for research in the general population. Appl Psychol Meas. 1977;1:385–401. | ||
Black SA, Markides KS, Ray LA. Depression predicts increases incidence of adverse health outcomes in older Mexican Americans with type 2 diabetes. Diabetes Care. 2003;26(10):2822–2828. | ||
Pan A, Keum N, Okereke OI, et al. Bidirectional association between depression and metabolic syndrome: a systematic review and meta-analysis of epidemiological studies. Diabetes Care. 2012;35(5):1171–1180. | ||
Luppino FS, de Wit LM, Bouvy PF, et al. Overweight, obesity, and depression: a systematic review and meta-analysis of longitudinal studies. Arch Gen Psychiatry. 2010;67(3):220–229. | ||
Katon W, Von Korff M, Ciechanowski P, et al. Behavioral and clinical factors associated with depression among individuals with diabetes. Diabetes Care. 2004;27:914–920. | ||
Eaton WW, Armenian H, Gallo J, Pratt L, Ford D. Depression and risk for onset of type II diabetes: a prospective population-based study. Diabetes Care. 1996;19(4):1097–1102. | ||
de Groot M, Anderson R, Freedland KE, Clouse RE, Lustman PJ. Association of depression and diabetes complications: a meta-analysis. Psychosom Med. 2001;63(4):619–630. | ||
Lustman PJ, Anderson RJ, Freedland KE, de Groot M, Carney RM, Clouse RE. Depression and poor glycemic control: a meta-analytic review of the literature. Diabetes Care. 2000;23(7):934–942. | ||
McKellar JD, Humphreys K, Piette JD. Depression increases diabetes symptoms by complicating patients’ self-care adherence. Diabetes Educ. 2004;30(3):485–492. | ||
Turk BR, Gschwandtner ME, Mauerhofer M, Löffler-Stastka H. Can we clinically recognize a vascular depression? Medicine. 2015;94(18):e743. | ||
Judd LL, Akiskal HS, Paulus MP. The role and clinical significance of subsyndromal depressive symptoms (SSD) in unipolar major depressive disorder. J Affect Disord. 1997;45(1–2):5–18. | ||
Judd LL, Rapaport MH, Paulus MP, Brown JL. Subsyndromal symptomatic depression: a new mood disorder? J Clin Psychiatry. 1994;55 Suppl:18–28. | ||
Babyak MA. What you see may not be what you get: a brief, nontechnical introduction to overfitting in regression-type models. Psychosom Med. 2004;66(3):411–421. | ||
Freedland KE, Carney RM. Depression and medical illness. In: Gotlib IH, Hammen CL, editors. Handbook of Depression. 3rd ed. New York, NY: Guilford; 2014:5–22. | ||
Franse LV, Di Bari M, Shorr RI, et al. Type 2 diabetes in older well-functioning people: who is undiagnosed? Data from the health, aging, and body composition study. Diabetes Care. 2001;24(12):2065–2070. | ||
Ciechanowski P, Katon W, Russo J. Depression and diabetes: impact of depressive symptoms on adherence, function and costs. Arch Intern Med. 2000;160(21):3278–3285. | ||
Marcus M, Wing R, Guare J, Blair EH, Jawad A. Lifetime prevalence of depression and its effect on treatment outcome in obese type 2 diabetic patients. Diabetes Care. 1992;15(2):253–255. | ||
Helmers KF, Mente A. Alexithymia and health behaviors in healthy male volunteers. J Psychosom Res. 1999;47(6):635–645. |
Supplementary material
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.