Back to Journals » Clinical Epidemiology » Volume 7
Surviving severe traumatic brain injury in Denmark: incidence and predictors of highly specialized rehabilitation
Authors Odgaard L, Poulsen I, Kammersgaard LP, Paaske Johnsen S, Feldbæk Nielsen J
Received 25 November 2014
Accepted for publication 22 January 2015
Published 20 March 2015 Volume 2015:7 Pages 225—234
DOI https://doi.org/10.2147/CLEP.S78141
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Henrik Sørensen
Lene Odgaard,1 Ingrid Poulsen,2 Lars Peter Kammersgaard,2 Søren Paaske Johnsen,3 Jørgen Feldbæk Nielsen,1
1Hammel Neurorehabilitation Center and University Research Clinic, Aarhus University, Aarhus, Denmark; 2Department of Neurorehabilitation, TBI and Research Unit on Brain injury rehabilitation (RUBRIC), Glostrup Hospital, Copenhagen University, Copenhagen, Denmark; 3Department of Clinical Epidemiology, Aarhus University Hospital, Aarhus, Denmark
Purpose: To identify all hospitalized patients surviving severe traumatic brain injury (TBI) in Denmark and to compare these patients to TBI patients admitted to highly specialized rehabilitation (HS-rehabilitation).
Patients and methods: Patients surviving severe TBI were identified from The Danish National Patient Registry and The Danish Head Trauma Database. Overall incidence rates of surviving severe TBI and incidence rates of admission to HS-rehabilitation after severe TBI were estimated and compared. Patient-related predictors of no admission to HS-rehabilitation among patients surviving severe TBI were identified using multivariable logistic regression.
Results: The average incidence rate of surviving severe TBI was 2.3 per 100,000 person years. Incidence rates of HS-rehabilitation were generally stable around 2.0 per 100,000 person years. Overall, 84% of all patients surviving severe TBI were admitted to HS-rehabilitation. Female sex, older age, and non-working status pre-injury were independent predictors of no HS-rehabilitation among patients surviving severe TBI.
Conclusion: The incidence rate of hospitalized patients surviving severe TBI was stable in Denmark and the majority of the patients were admitted to HS-rehabilitation. However, potential inequity in access to HS-rehabilitation may still be present despite a health care system based on equal access for all citizens.
Keywords: database, health care disparities, registries, validity
Introduction
Traumatic brain injury (TBI) is a global public health problem associated with high socioeconomic costs and substantial loss of healthy life years due to ill health, disability, and/or early death.1 The incidence rates of severe TBI in European countries are not well known; however, hospitalization rates have been reported to vary from 4.1–20.0 per 100,000 person years.2–7
TBI care and rehabilitation has evolved substantially over the last 20 years and the need for specialized rehabilitation is widely accepted.8 Additionally, it has been acknowledged that the effectiveness of TBI care and rehabilitation on the prognosis in TBI are best explored in large TBI populations.9,10
In general, patients surviving severe TBI have highly complex rehabilitation needs. In Denmark, it is recommended by the National Board of Health that these patients are offered highly specialized rehabilitation (HS-rehabilitation).11 HS-rehabilitation is a centralized, intensive rehabilitation specifically targeting patients with highly complex needs after TBI.11,12
Theoretically, all patients surviving severe TBI should be admitted to HS-rehabilitation as the Danish health care system is based on equal access to health care services for all citizens. However, indications of inequities in the access to health care in Denmark and other welfare states have previously been reported for other areas of health care, although the available data remain sparse.13
To further explore and characterize potential inequities in the access to HS-rehabilitation, we aimed to identify all patients in Denmark surviving severe TBI and to compare these patients to severe TBI patients admitted to HS-rehabilitation between 2010 and 2012. Furthermore, we aimed to identify all patients admitted to HS-rehabilitation after severe TBI between 2004 and 2012.
Materials and methods
Denmark has 5.6 million inhabitants and a tax-financed health care system. The organization of hospital services and rehabilitation after TBI is defined by The Danish National Board of Health and includes free access to acute care, neurosurgery, and rehabilitation after severe TBI. Neurosurgery has historically been centralized to five hospitals, and since 2010 to four hospitals. The primary inpatient rehabilitation for patients with highly complex needs after TBI has been centralized to two hospitals since the year 2000: Glostrup Hospital, which covers the eastern part of Denmark, and Hammel Neurorehabilitation Centre and University Research Clinic, which covers the western part of Denmark, with each unit covering half of the country as uptake area.12 HS-rehabilitation is usually followed by rehabilitation at decentralized rehabilitation units.
All Danish registries may be linked using a ten-digit personal number encoding age and sex, which is assigned to all citizens by the Civil Registration System.14
Danish National Patient Registry
The Danish National Patient Registry (DNPR) was established in 1977 as a national administrative register. DNPR contains data on all hospital admissions and outpatient contacts including diagnosis codes according to the International Classification of Diseases, version 10 (ICD-10).15 The registry serves as basis for hospital payments.16
Danish Head Trauma Database
The Danish Head Trauma Database (DHD) was established in 2004 on the recommendation of the Danish National Board of Health as a clinical registry focused on the rehabilitation of patients with severe TBI.
Inclusion criteria are:
- Glasgow Coma Scale (GCS) ≤8 (lowest, nonsedated GCS within 48 hours after injury).
- Admission to one of the two national hospitals that offer HS-rehabilitation after severe TBI.
Data are collected at admission, during rehabilitation, at discharge, and at 1-year follow-up. After discharge, data are entered into a central database using a web-based interface.
The database primarily includes rehabilitation data such as clinical rehabilitation scores and complications following TBI. Information on cause and date of injury, preinjury status, and neurosurgical treatment are also included. See “Supplementary materials” for details.
Population
All patients surviving severe TBI between 2010 and 2012
We identified all patients with severe TBI between 2010 and 2012, residing in Denmark, aged 15 years or older, who survived primary hospitalization. The time period was chosen pragmatically as electronic health records were available from 2010, which facilitates easy access to medical records for review.
Patients admitted to HS-rehabilitation after severe TBI identified from DNPR and DHD as described below. Patients who were not admitted to HS-rehabilitation were identified from DNPR by defining patients with a total length of hospital stay of at least 50 days after TBI as patients surviving possible severe TBI. The medical records of these patients were retrieved (n=359) to identify patients who were not admitted to HS-rehabilitation despite severe TBI, ie, GCS ≤8, or if GCS was missing, posttraumatic amnesia (PTA) ≥15 days.17
During the medical review, we excluded patients for the following reasons: non-TBI (n=48); injury before 2010/after 2012 (n=26); GCS >8 (n=230); living abroad at injury (n=2); and died (n=2).
The number of patients with severe TBI between 2010 and 2012 identified from DNPR and from the cohort of HS-patients is illustrated in Figure 1.
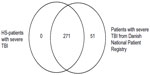  | Figure 1 Number of patients with severe TBI between 2010 and 2012. |
The total length of stay was calculated as the number of days from the first hospital admission to last discharge in consecutive hospitalizations following TBI. Consecutive hospitalizations were defined as hospital stays after TBI including interruptions of 30 days or fewer (eg, leave from hospital). Days of interruption and nested hospitalizations (eg, admission to acute hospitals during rehabilitation) were not included in the total length of stay.
Patients admitted to HS- rehabilitation after severe TBI between 2004 and 2012
We identified patients aged 15 years or older, residing in Denmark, and admitted to HS-rehabilitation up to 6 months after severe TBI between 2004 and 2012 (HS-patients) based on information from DHD, DNPR, and medical records, using the algorithm presented in Figure 2.
In the dataset from DHD, we corrected missing data on injury date, GCS, and length of PTA with available data from the medical records, and assessed the agreement of parameters for injury severity (GCS and PTA) with the medical records (Supplementary materials and Table S1).
In the dataset from DNPR, we identified HS-patients with severe TBI, who were not included initially in the DHD.
After correcting for missing data in DHD and adding patients not included initially, we excluded nonrelevant patients, such as patients where the TBI diagnosis was not confirmed by the review of the medical records, or patients residing outside of Denmark at injury according to the address registered in Civil Registration System.
Due to the problems of missing data in GCS, we decided to use PTA, which is another commonly used measure of severity,1 as an additional criterion to define severe TBI for patients with missing GCS.17 Consequently, we excluded patients we could not verify as having severe TBI (see Figure 2 for details).
Potential predictors of HS-rehabilitation
We assessed the following potential predictors: sex, age, preinjury living status, preinjury vocational status, and Charlson Comorbidity Index (CCI), which is a measure for the level of comorbidity,18 calculated from DNPR data with coding algorithms for defining comorbidities in ICD-10 administrative data.19
The selection of relevant variables was based on clinically suspected referral practice together with previously reported indications of inequities in the access to health care services in Denmark.13 Only variables where valid information was available were considered. No formal statistical criteria were used to decide whether the covariate should be included.
Age was categorized using the typical age of retirement during the study period (65 years) as the cutoff point. Allowing for a reasonable sample size in the age groups, we further dichotomized the working age population into a younger and an older group. Additionally, we analyzed age as a continuous variable.
Medical records review
All reviews were performed based on the clinical interpretation recorded by clinicians treating the patients. The first 40 reviews to identify HS-patients not included initially in DHD were performed by a research nurse and a physician in conjunction to reach consensus on the TBI diagnosis and injury severity (GCS and PTA) (LO and JFN in West Denmark, IP and LPK in East Denmark).
The subsequent reviews were performed by the first and second author (LO in West Denmark and IP in East Denmark).
Approvals for the study were obtained from the Danish Data Protection Agency (journal number 2007-58-0010) and The Danish National Board of Health (journal number 3-3013-301/1).
Data analysis
We computed the overall incidence rate of patients surviving severe TBI between 2010 and 2012 and the incidence rate of HS-rehabilitation after severe TBI in the period between 2004 and 2012. The incidence rates were calculated using the annual total adult Danish population as the denominator, ie, the number of persons aged 15 years or older in each year.20
In addition, we estimated the number of HS-patients with severe TBI relative to all patients with severe TBI in Denmark. The proportion was calculated using the number of HS-patients injured between 2010 and 2012 as the numerator. The denominator was the total number of patients with severe TBI between 2010 and 2012.
Finally, we used multivariable logistic regression to identify predictors of no HS-rehabilitation among the potential predictors described above.
Data management and analyses were performed using STATA software, version 11 (StataCorp LP, College Station, TX, USA).
Results
Characteristics of the identified HS-patients are presented in Table 1.
The incidence rates of patients surviving severe TBI ranged from 2.0 to 2.7 per 100,000 person years. The average incidence rate was 2.3 per 100,000 person years. The incidence rates of HS-rehabilitation after severe TBI were generally stable around 2.0 per 100,000 person years across calendar years. The incidence rates of patients surviving severe TBI and the incidence rates of HS-rehabilitation fluctuated accordingly (Figure 3).
The proportion of patients with severe TBI between 2010 and 2012 who received HS-rehabilitation was 84% (95% confidence interval [CI], 79.7–88.0), with no significant difference between the two hospitals offering HS-rehabilitation (proportion in West Denmark, 82.2% [95% CI, 75.6–87.7]; proportion in East Denmark 86.3% [95% CI, 79.8–91.3]; difference, −4.0% [95% CI, −12.0 to 3.9]).
The associations between the potential predictors and no HS-rehabilitation among all patients surviving severe TBI between 2010 and 2012 are presented in Table 2.
Females and patients aged 40 years or older were less likely to be admitted to HS-rehabilitation compared to males and patients aged 15–39 years in the multivariable analysis. In addition, patients, who were unemployed, homemakers, or on sick leave were less likely to be admitted to HS-rehabilitation compared to patients who were working or studying preinjury.
In contrast, comorbidity as measured by CCI and preinjury living status were not significantly associated with no HS-rehabilitation after severe TBI.
The analysis with age as a continuous variable (data not shown) resulted in minor changes of the estimates, but did not alter the identified predictors of no HS-rehabilitation.
Discussion
Using national medical registries and medical records, we identified patients surviving severe TBI in Denmark and estimated an average incidence rate of patients surviving severe TBI of 2.3/100,000 person years. An estimated 84% of all patients surviving severe TBI were admitted to HS-rehabilitation. Female sex, older age, and no preinjury working status were identified as predictors of no HS-rehabilitation. The estimated incidence rates of surviving severe TBI were low compared to previous estimated incidence rates of severe TBI in European countries.2–7 However, direct comparisons between the studies are difficult due to different scales used to classify severity, different definitions of TBI cases, and estimates usually including fatal cases.
Our incidence rate is well below the estimated incidence rate of patients surviving severe TBI in 1988 in Denmark (9.5/100,000).3 The difference may be explained by a general decrease in TBI incidence,21 but the previous study also classified severe TBI as PTA ≥7 days, ie, classified more patients as having severe TBI.
Compared to studies using GCS ≤8 to classify severe TBI, our incidence rates are only slightly lower than recent incidence rates of patients surviving severe TBI between 2009 and 2010 in Norway (2.9–3.6/100,000),5 and somewhat lower than incidence rates of patients surviving severe TBI in 1996 in France (4.1 per 100,000).7 The differences may again be explained by a general decrease in TBI incidence,21 but different incidence rates of severe TBI across European countries, with the Nordic countries presenting the lowest rates at present, may also explain the differences.
The stable incidence rates of HS-rehabilitation since 2004 indicate that the need for HS-rehabilitation after severe TBI has not decreased over time.
The estimated high proportion of patients admitted to HS-rehabilitation after surviving severe TBI corresponds to the principle of equal access to health care services in Denmark. Despite this, 16% of patients surviving severe TBI were not admitted to HS-rehabilitation. We do not know whether these patients had highly complex rehabilitation needs. However, the identified predictors suggest that referred patients are selected according to sex, age, and vocational status, reflecting potential inequity in access to HS-rehabilitation.
We speculate that factors associated with both male sex and complex rehabilitation needs explain the observed prediction by sex. Men are often more severely injured than women22 and are more often abusers of drugs and alcohol.23 Furthermore, it is a widely held notion that men are more violent and agitated during recovery after TBI, although this is not supported by research according to our knowledge.24 This may lead to men being provided easier access to HS-rehabilitation, which, contrary to the immediate interpretation of inequity, points to vertical equity, ie, those with greater needs are provided easier access.25
Our results that older age and no working status predict no referral to HS-rehabilitation agree with reports of selection into other rehabilitation areas (vocational rehabilitation in Sweden26,27 and cancer rehabilitation in Denmark28,29) which point to inequity in rehabilitation access.
Living alone was neither an independent predictor in our study nor in other Danish studies29 and may reflect a health care system serving the citizens largely independent of interventions from relatives or families.
Finally, comorbidity, contrary to previous reports of selection into rehabilitation,26 was not a predictor of no HS-rehabilitation. It is therefore unlikely that some comorbidity blocks access to HS-rehabilitation. Nevertheless, to detect potential differences, a more detailed operationalization of comorbidity than the dichotomized CCI used in our study would be required.
Our study may add to the attention of potential inequities in health care systems with a principle of equal access to health care services. Considering the estimated high proportion of patients admitted to HS-rehabilitation after severe TBI, selection into HS-rehabilitation appears to be a minor problem.
Our study has some limitations. First, our analyses were performed on national data, but as only a few patients experience severe TBI, caution should be taken when considering the statistical precision of the estimates, as reflected by the width of the 95% CIs.
Second, we defined TBI severity using GCS, which is the most commonly used score for classification of severity in TBI research. Nevertheless, GCS is influenced by a number of medical conditions that often accompany the acute stage of severe TBI, which may contribute to decreased consciousness.30 However, by restricting the definition of patients not admitted to HS-rehabilitation to those who were hospitalized for at least 50 days, we believe we excluded patients with a GCS that deteriorated due to factors other than TBI itself. We probably also excluded those who recovered rapidly despite severe TBI, leading to a potential underestimation of overall incidence rates of surviving severe TBI and potential overestimation of the proportion referred to HS-rehabilitation.
The exclusion of patients hospitalized for less than 50 days might also bias the analyses of potential predictors of HS-rehabilitation. Because increased length of stay in hospital may be associated with lower sociodemographic status and older age due to increased comorbidity, we most probably included all older patients with low sociedemographic status but potentially excluded some younger patients with high sociodemographic status inducing overestimated differences between HS-patients and patients who were not admitted to HS-rehabilitation.
Finally, we believe that potential misclassifications of HS-patients, if any, were random: first and foremost, because the coding of hospitals in DNPR is assumed to be complete;16 second, because a systematic inaccurate and incomplete coding of TBI diagnoses in DNPR is considered unlikely throughout hospitalizations following severe TBI, as it usually includes referrals between several units; and third, because patients with missing GCS were included if they emerged from PTA more than 15 days after injury, which is a high cutoff point compared to conventional use of PTA to classify severe TBI.1 We thereby most likely reduced the risk of falsely classifying patients with mild or moderate TBI as severe TBI.
The presumed unbiased identification of HS-patients facilitates large population-based studies of TBI prognosis and the identified selection into HS-rehabilitation can be used to assess potential selection bias in future studies.
Many large studies of TBI prognosis have been conducted using The Traumatic Brain Injury Model Systems, which is a large TBI rehabilitation database nested in a multidisciplinary system of high-quality rehabilitation centers in the United States.31,32 We assume that access to and quality of TBI rehabilitation is more homogeneous in the Nordic countries compared to the United States, given the tax financed health care system, the relatively small population sizes, and the ethnic homogeneity of the population. From this perspective, the identified HS-patients may constitute a European alternative to The Traumatic Brain Injury Model Systems.
Conclusion
We found stable incidence rates of hospitalized patients surviving severe TBI in Denmark, and the vast majority of these patients were admitted to HS-rehabilitation. The observed sex-, age-, and work-related selection for HS-rehabilitation could reflect inequities in care, despite a health care system based on the principle of equal access for all citizens. The identified HS-patients may be used to study TBI prognosis.
Acknowledgments
Asger R Pedersen, statistician, PhD, Hammel Neurorehabilitation Center and University Research Clinic, is acknowledged for valuable inputs on establishing the cohort of HS-patients. Marianne Eskildsen, DHD Data manager, TBI and Research Unit on Brain injury rehabilitation, Glostrup Hospital, is acknowledged for support in collecting missing data.
Disclosure
The authors report no conflicts of interest in this work.
References
Corrigan JD, Selassie AW, Orman JA. The epidemiology of traumatic brain injury. J Head Trauma Rehabil. 2010;25(2):72–80. | |
Annoni JM, Beer S, Kesselring J. Severe traumatic brain injury – epidemiology and outcome after 3 years. Disabil Rehabil. 1992;14(1):23–26. | |
Engberg A. Severe traumatic brain injury – epidemiology, external causes, prevention, and rehabilitation of mental and physical sequelae. Acta Neurol Scand Suppl. 1995;164:1–151. | |
Masson F, Thicoipe M, Aye P, et al. Epidemiology of severe brain injuries: a prospective population-based study. J Trauma. 2001;51(3):481–489. | |
Andelic N, Anke A, Skandsen T, et al. Incidence of hospital-admitted severe traumatic brain injury and in-hospital fatality in Norway: a national cohort study. Neuroepidemiology. 2012;38(4):259–267. | |
de Kloet AJ, Hilberink SR, Roebroeck ME, et al. Youth with acquired brain injury in The Netherlands: A multi-centre study. Brain Inj. 2013;27(7–8):843–849. | |
Masson F, Thicoipe M, Mokni T, et al. Epidemiology of traumatic comas: a prospective population-based study. Brain Inj. 2003;17(4):279–293. | |
Ashley MJ. Repairing the injured brain: why proper rehabilitation is essential to recovering function. Cerebrum. 2012;2012:8. | |
Maas AI, Menon DK, Lingsma HF, Pineda JA, Sandel ME, Manley GT. Re-orientation of clinical research in traumatic brain injury: report of an international workshop on comparative effectiveness research. J Neurotrauma. 2012;29(1):32–46. | |
Maas AI, Murray GD, Roozenbeek B, et al. Advancing care for traumatic brain injury: findings from the IMPACT studies and perspectives on future research. Lancet Neurol. 2013;12(12):1200–1210. | |
Sundhedsstyrelsen [National Board of Health]. Behandling af traumatiske hjerneskader og tilgrænsende lidelser: Nuværende og fremtidig organisation: Redegørelse. [Treatment of traumatic brain injuries and similar conditions: Current and future organization: Statement.] Copenhagen: National Board of Health; 1997. Danish. | |
Engberg AW. A Danish national strategy for treatment and rehabilitation after acquired brain injury. J Head Trauma Rehabil. 2007;22(4):221–228. | |
OECD. OECD Reviews of Health Care Quality: Denmark 2013: Raising Standards. Paris: OECD Publishing; 2013. Available from: http://www.nsdm.no/filarkiv/File/Eksterne_rapporter/OECD_rapport_Danmark_1_.pdf. Accessed September 10, 2014. | |
Pedersen CB. The Danish Civil Registration System. Scand J Public Health. 2011;39(7 Suppl):22–25. | |
ICD-10 version:2010 [webpage on the Internet]. Geneva: World Health Organization; 2010. Available from: http://apps.who.int/classifications/icd10/browse/2010/en. Accessed June 25, 2014. | |
Lynge E, Sandegaard JL, Rebolj M. The Danish National Patient Register. Scand J Public Health. 2011;39(7 Suppl):30–33. | |
Nakase-Richardson R, Sepehri A, Sherer M, Yablon SA, Evans C, Mani T. Classification schema of posttraumatic amnesia duration-based injury severity relative to 1-year outcome: analysis of individuals with moderate and severe traumatic brain injury. Arch Phys Med Rehabil. 2009;90(1):17–19. | |
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–383. | |
Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130–1139. | |
StatBank.dk/folk1 [webpage on the Internet]. Copenhagen: Statistics Denmark. Available from: http://www.statistikbanken.dk/folk1. Accessed April 7, 2014. | |
Pérez K1, Novoa AM, Santamariña-Rubio E, et al. Incidence trends of traumatic spinal cord injury and traumatic brain injury in Spain, 2000–2009. Accid Anal Prev. 2012;46:37–44. | |
Berry C, Ley EJ, Tillou A, Cryer G, Margulies DR, Salim A. The effect of gender on patients with moderate to severe head injuries. J Trauma. 2009;67(5):950–953. | |
Corrigan JD. Substance abuse as a mediating factor in outcome from traumatic brain injury. Arch Phys Med Rehabil. 1995;76(4):302–309. | |
Kadyan V, Mysiw WJ, Bogner JA, Corrigan JD, Fugate LP, Clinchot DM. Gender differences in agitation after traumatic brain injury. Am J Phys Med Rehabil. 2004;83(10):747–752. | |
Oliver A, Mossialos E. Equity of access to health care: outlining the foundations for action. J Epidemiol Community Health. 2004;58(8):655–658. | |
Lidwall U. [The Swedish social insurance agency and work-targeted rehabilitation – active measures and return to work]. Stockholm: Försäkringsverket; 2006. Swedish. Available from: http://www.forsakringskassan.se/wps/wcm/connect/40709c84-4008-44a6-975f-217c6629f3d0/analyserar_2006_10.pdf?MOD=AJPERES. Accessed. | |
Ahlgren A, Bergroth A, Ekholm J. Work resumption or not after rehabilitation? A descriptive study from six social insurance offices. Int J Rehabil Res. 2004;27(3):171–180. | |
Holm LV, Hansen DG, Johansen C, et al. Participation in cancer rehabilitation and unmet needs: A population-based cohort study. Support Care Cancer. 2012;20(11):2913–2924. | |
Holm LV, Hansen DG, Larsen PV, et al. Social inequality in cancer rehabilitation: a population-based cohort study. Acta Oncol. 2013;52(2):410–422. | |
Grote S, Böcker W, Mutschler W, Bouillon B, Lefering R. Diagnostic value of the Glasgow Coma Scale for traumatic brain injury in 18,002 patients with severe multiple injuries. J Neurotrauma. 2011;28(4):527–534. | |
Bushnik T. Introduction: The Traumatic Brain Injury Model Systems of Care. Arch Phys Med Rehabil. 2003;84(2):151–152. | |
Tbindsc.ord [homepage on the Internet.] Colorado: Traumatic Brain Injury Model Systems National Data and Statistical Center [Updated 2014]. Available from: https://www.tbindsc.org/. Accessed September 10, 2014. |
Supplementary materials
Overview of the included data in Danish Head Trauma Database
The data elements are listed according to the structure proposed by Maas et al for data collection in traumatic brain injury research.1
Subject characteristics:
- Demographics:
- Age and sex
- Social status preinjury and at 1-year follow-up:
- Residence
- Socioeconomic status
- Employment/school status
- Marital status
- Persons living with
Participant and family history:
- Physical limitations preinjury
- Mental limitations preinjury:
- Mental illness
- Drug/alcohol abuse
- Special class for children with learning difficulties
- Brain injury prior to index traumatic brain injury
Injury/disease related events:
- Type of injury based on International Classification of Diseases, version 10 diagnoses
- Injury severity:
- Lowest nonsedated score on Glasgow Coma Scale in the first 48 hours following injury
- Length of posttraumatic amnesia (PTA)
- Cause of injury
- Date of injury
- Date of admission to and discharge from the acute care unit
- Date of admission to and discharge from the highly specialized rehabilitation (HS-rehabilitation) hospital
- Discharge destination
Treatments:
- Type of surgical therapy
- Days on ventilator
Outcome and function:
- Neurological assessment and acute therapy assessment:
- Glasgow Coma Score (GCS) at admission to HS-rehabilitation hospital
- Duration of PTA
- Clinical neurorehabilitation scores on admission to the HS-rehabilitation hospital, at discharge from the HS-rehabilitation hospital, and at 1-year follow-up:
- Functional Independence Measure
- Early Functional Abilities score
- Ranchos Los Amigos Score
- Functional Oral Intake Scale
- Glasgow Outcome Scale Extended at 1-year follow-up
- Complications on admission to the HS-rehabilitation hospital, during rehabilitation, at discharge from the HS-rehabilitation hospital, and at 1-year follow-up:
- Hydrocephalus
- Meningitis
- Epilepsy
- Neuropsychiatric problems such as agitation, depression, and psychosis
- Autonomic dysfunction
- Neuroendocrine dysfunction
- Spasticity and contractions
- Paresis, dystonia, and ataxia
- Loss of taste, smell, and hearing
- Cognitive and communicative problems
- Deep vein thrombosis
- Pneumonia
- Decubitus
- Urinary tract infection
- Weight and height (to monitor decrease or increase of body mass index)
- Quality of life at 1-year follow-up
- Return to work at 1-year follow-up
- Type of rehabilitation at 1-year follow-up.
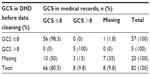
GCS and length of PTA in the Danish Head Trauma Database before data cleaning: agreement with medical records
We compared the GCS and length of PTA with values from medical records using a random sample (10% of the total sample) from the Danish Head Trauma Database before data cleaning.
The analysis of GCS showed agreement between the Danish Head Trauma Database and medical records on the available data dichotomized at GCS ≤8. The comparison revealed that some observations with missing GCS data in the database, had GCS>8 according to the medical records according to the medical records (Table S1).
The analysis of PTA showed poor agreement due to clinicians’ miscalculations before registration, in the data entered by the hospital for HS-rehabilitation in Western Denmark. Hence, PTA data on patients from West Denmark were recollected from the medical records as date of emergence from PTA according to the scales used to monitor PTA. Length of PTA was recalculated as the difference between the date of injury and the date of emergence from PTA.
Reference
Maas AI, Harrison-Felix CL, Menon D, et al. Standardizing data collection in traumatic brain injury. J Neurotrauma. 2011;28(2):177–187. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.