Back to Journals » Veterinary Medicine: Research and Reports » Volume 5
Successful medical and surgical treatment of dermal arteritis of the nasal philtrum in a Saint Bernard dog
Authors Vuolo S, Peters L, Licari L
Received 10 May 2014
Accepted for publication 25 June 2014
Published 9 September 2014 Volume 2014:5 Pages 115—118
DOI https://doi.org/10.2147/VMRR.S67182
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Sarah Vuolo, Lisa Peters, Louis Licari
Department of Emergency and Critical Care, Fox Valley Animal Referral Center, Appleton, WI, USA
Abstract: A four-year-old neutered male Saint Bernard dog presented with a 2 year history of nasal philtrum ulceration and recent recurrent arterial hemorrhage from this region. Dermal arteritis of the nasal philtrum was diagnosed and the patient was started on immunosuppressive medications. Clinical signs continued to recur until surgical resection of the lesions was performed. Dermal arteritis of the nasal philtrum is a rare phenomenon but should be considered in certain breeds. Surgical removal of affected tissue should be considered when medical treatment fails. Long-term management with cyclosporine was initiated which has not been previously described in the literature.
Keywords: dermal arteritis, immunology, nasal resection, immunosuppression
Introduction
Dermal arteritis of the nasal philtrum is a rarely described disease process which has been documented in a Giant Schnauzer,1 Saint Bernard,1 Newfoundland,2 and a Basset hound.3 The typical lesion is a solitary, well-circumscribed circular ulcer which is non-pruritic and most commonly non-painful on the nasal philtrum (Figure 1). The nasal planum is unaffected in these patients.1 Histopathologic findings are also characteristic including subendothelial spindle cell proliferation within deep dermal arteries and arterioles around the ulcer along with marked extracellular matrix deposition containing mucin and collagen. Superficially, changes include neutrophilic and lymphoplasmacytic inflammation in the dermis.3 Chronically this disease results in intimal thickening and stenosis of arteries and arterioles. Commonly, life-long medical management is required to treat this condition. In this case, surgical management was required in addition to long-term medical management.
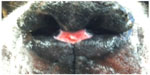  | Figure 1 Classic dermal arteritis of the nasal philtrum lesion at this patient’s first presentation to the hospital. |
Case report
A four-year-old neutered male Saint Bernard dog weighing 54.9 kg presented for evaluation to the Fox Valley Animal Referral Center emergency service with the chief complaint of severe, recurrent epistaxis of 1 week duration. The patient had been treated by the primary care veterinarian with cold compressing and amoxicillin trihydrate/clavulanate potassium (unknown amount) (Clavamox®; Pfizer Animal Health, New York, NY, USA), which was unsuccessful. The patient was up-to-date with vaccinations. Laboratory evaluation performed by the primary care veterinarian revealed a normal complete blood cell count, normal biochemistry panel, and negative heartworm enzyme-linked immunoassay (ELISA) (SNAP® 4Dx® Plus Test, Idexx Laboratories, Westbrook, ME, USA). Blastomycosis urine antigen testing (MiraVista Laboratory, Indianapolis, IN, USA) was performed due to the endemic region and was negative. No abnormalities were noted on three view thoracic radiographs. Prothrombin and partial thromboplastin were within the reference range.
Physical examination at the time of presentation was unremarkable except for bilateral linear nasal philtrum ulcerations. Upon further questioning the owners reported that these lesions had been present since the patient was 1 to 2 years of age and the hemorrhage came from these sites, not from inside the nostril itself.
The patient was admitted to the hospital and started on prednisone (0.5 mg/kg per os (PO) every 12 hours) (West-Ward Pharmaceutical Corp, Eatontown, NY, USA) and tacrolimus 0.1% (small amount applied topically to the nose every 12 hours) (Protopic®; Astellas Pharma, LLC, Northbrook, IL, USA) for systemic and topical immunosuppression. Additional laboratory evaluation included a buccal mucosal bleeding time which was within the normal range. No further hemorrhage was noted and the patient was discharged.
Three severe arterial hemorrhages occurred at home following discharge and a weight loss of 2.7 kg was noted even though the patient was reported to be eating well. The owners also reported lethargy and a change in attitude at home as the patient was less interactive and playful. The patient represented 1 week later to the emergency service. At presentation the patient was administered acepromazine (0.02 mg/kg intravenously once) (PromAce®; Boehringer Ingelheim, Ridgefield, CT, USA) and a cold water/dilute epinephrine (Epinephrine injection, USP; Hospira Inc., Verna, India) compress was applied with a towel to control severe hemorrhage.
Laboratory evaluation was performed. Serum biochemistry analysis revealed a high alanine aminotransferase (ALT) (371 U/L; reference range 10–100 U/L), elevated gamma-glutamyl transferase (GGT) (20 U/L; reference range 0–7 U/L), elevated blood urea nitrogen (BUN) (43 mg/dL; reference range 7–27 mg/dL), and anemia (hematocrit [HCT] 25.7%; reference range 37.1%–61.7%). The remainder of the serum biochemistry and electrolyte panel was unremarkable.
Abdominal ultrasound was performed at the owner’s request due to weight loss. Results were unremarkable except for a solitary, small (5 mm) nodule on the spleen suspected to be a benign process (extramedullary hematopoiesis).
The patient was hospitalized, and topical and oral therapy was continued as previously described. The patient continued to have severe hemorrhage from his nasal philtrum intermittently. Surgery was suggested for a definitive treatment in this patient as the owners were considering humane euthanasia due to continued hemorrhage, weight loss, anemia, and change in attitude. Four days after his second admission to the hospital, the patient was anesthetized and taken to surgery.
The ulcerated part of the nasal philtrum and floor of the nares were sharply excised with a margin of healthy tissue 1 cm deep. Hemorrhage was controlled with cautery. The lower remaining philtrum and lip tissues were undermined and closed in two layers to create a new philtrum area by apposing the dorsal and ventral margins (Figure 2). The excised portion was submitted for histopathology. Anesthetic recovery was routine.
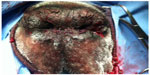  | Figure 2 Postoperative view of the nasal philtrum in our patient. |
The topical tacrolimus was discontinued for 1 week and the prednisone was decreased to 0.25 mg/kg PO every 12 hours due to adverse clinical signs including polydypsia, polyuria, and muscle wasting. An Elizabethan collar was used to prevent self-trauma to the surgery site. Tramadol (Amneal Pharmaceuticals, Glasgow, KY, USA) was started at 4 mg/kg PO every 12 hours for pain control and amoxicillin trihydrate/clavulanate potassium (19.6 mg/kg PO every 12 hours) was started for infection prevention. Cyclosporine (Atopica®; Novartis Animal Health, Greensboro, NC, USA) was started at 4 mg/kg PO every 24 hours for further immunosuppression and as an alternative to prednisone for immunosuppression in this patient. Pentoxifylline (Teva Pharmaceuticals, Sellersville, PA, USA) was started at 9.8 mg/kg PO every 12 hours to improve red blood cell deformability and reduce blood viscosity decreasing platelet aggregation and thrombus formation.
The patient was hospitalized for an additional 5 days prior to discharge (Figure 3). No further hemorrhage was noted from the nasal philtrum.
  | Figure 3 Nasal philtrum at discharge from the hospital postoperatively. |
Histopathology revealed large arterioles and arteries in the submucosa with infiltrates of plasma cells, macrophages, and cellular debris. Within the arterioles there was spindle cell proliferation with an extracellular matrix deposition resulting in intimal thickening and stenosis of the arteries and arterioles consistent with dermal arteritis of the nasal philtrum.
The patient was rechecked 10 days post-discharge and topical tacrolimus was resumed. Prednisone was weaned and discontinued. Fish oil (AllerG-3; Vétoquinol SA, Lure Cedex, France) supplementation was started at 3.9 mg/kg PO every 24 hours and Vitamin E (Nature Made®; Pharmavite, Mission Hills, CA, USA) was started at 7.8 mg/kg PO every 24 hours for anti-oxidant properties and has been previously noted in the literature to be useful in these patients.2 The cyclosporine was continued.
The patient remained clinically normal and was re-evaluated at 3 months postoperatively. The patient had returned to his normal attitude. The ulcerated areas of his nasal philtrum remained absent but there was mild depigmentation laterally on the right side. This was suspected to be scar tissue based on the location of the surgical incision. Laboratory evaluation with complete blood count, serum biochemistry panel, and total thyroxine were all within normal limits (Marshfield Laboratories, Marshfield, WI, USA). Cyclosporine was decreased to 2.9 mg/kg PO every 24 hours.
The patient was evaluated over the phone, email, and through the referring veterinarian (Figure 4). Cyclosporine wean was continued. At the time of this publication, the patient is on pentoxifylline, vitamin E, fish oil, and topical tacrolimus. The cyclosporine dose is 0.98 mg/kg PO every 24 hours currently and the plan is for a recheck at the 1 year postoperative date. The cyclosporine will be continued.
  | Figure 4 The nasal philtrum 3 months postoperatively in our patient. |
Discussion
Dermal arteritis of the nasal philtrum is a rare condition noted in veterinary medicine in certain large breed dogs.1–3 Other ulcerative diseases of the nasal philtrum include neoplasia (squamous cell carcinoma and cutaneous lymphoma), chronic inflammatory processes (aspergillosis, foreign body, deep infections, leishmaniosis), chronic trauma and nasal cutaneous lupus. These diseases tend to affect the planum and philtrum whereas dermal arteritis of the nasal philtrum is confined to the philtrum. Ulceration of the nasal philtrum due to inflammation of the arterioles may lead to dramatic arterial hemorrhage. The typical lesion is a characteristic solitary, well-circumscribed circular ulcer which is non-pruritic and most commonly non-painful on the nasal philtrum. The rest of the nose (nasal planum) is unaffected in these patients.1 Patients can also present with bilateral linear ulcers on the nasal philtrum.2 These lesions can be pathognomonic in previously described breeds, especially when associated with hemorrhage. Histopathologic findings are also classic with involvement of the subendotherlial spindle cell proliferation within deep dermal arteries and arterioles around the ulcer along with marked extracellular matrix deposition containing mucin and collagen. Chronically this results in intimal thickening and stenosis of arteries and arterioles. Changes include neutrophilic and lymphoplasmacytic inflammation in the dermis.3
Various medical treatment options have been described in the literature.3 Treatment has included parenteral and topical medications with immunosuppressant and/or anti-inflammatory therapy administered on a long-term (possibly life-long) basis. Medications used in this condition have included: prednisolone, tetracycline, niacinamide, and fish oil.1,4 These medications are utilized to decrease vascular inflammation in this condition. Medical therapy of dermal arteritis of the nasal philtrum has inconsistent success in the literature with reports ranging from complete control to ongoing hemorrhage. In all documented cases, discontinuation of medical treatment has led to recurrence.3 Although some sources have suggested cyclosporine may be used successfully in this disease, to the author’s knowledge this is the first case report to document this drug treatment modality.
A novel surgical approach has been reported once in the veterinary literature. Pratchske and Hill previously described a surgical option for two patients. The anatomic location of the lesions in dermal arteritis suggests that specific targeting of the lateral nasal branches of the infraorbital arteries, maxillary arteries, and the rostal septal branches of the palatine arteries allows for control hemorrhage in these dogs.5 The goal of surgery in Pratchske and Hill was to resect the vulnerable portion of the nose and ligate arteries supplying this site. In one of these two patients clinical signs recurred postoperatively when the patient was taken off medications. The surgical approach in our patient was similar and consisted of deep local resection and vascular cautery. Surgery was used early in the disease course in this patient as the owners were looking for a quicker resolution of clinical signs of severe arterial hemorrhage and they had been considering humane euthanasia if signs were not controlled. It was decided to continue medical management in this patient long-term due to the risk of recurrence. According to the author’s knowledge, this is the first documented case of successful cyclosporine use as the only immunosuppressive drug in this condition in the literature.
Conclusion
Dermal arteritis of the nasal philtrum is a rare but possibly life-threatening condition affecting large breed dogs. Very few reports are present on this condition in the veterinary literature. This rare phenomenon can be diagnosed based on pathognomonic signs on physical examination with or without clinically significant hemorrhage. It is recommended that medical management be started on this patients and be continued life-long to prevent recurrence of clinical signs. If medical management is insufficient to control clinical signs surgery can be performed as an adjunctive treatment early or late in the disease. In this patient, clinical signs were controlled with a combination of surgery and ongoing medical management with success for 9 months.
Acknowledgments
The authors would like to thank Joseph Frank of the Surgical Service at The Fox Valley Animal Referral Center, and Andrew Lowe of the Dermatology Service at Alta Vista Animal Hospital.
Disclosure
The author reports no conflicts of interest in this work.
References
Torres SM, Brein TO, Scott DW. Dermal arteritis of the nasal philtrum in a Giant Schnauzer and three Saint Bernard dogs. Vet Dermatol. 2002;13(5):275–281. | |
Nuttal T, Harvey RG, McKeever P. Skin Diseases of the Dog and Cat. 2nd ed. London, UK: Mason; 2009. | |
Pratschke KM, Hill PB. Dermal arteritis of the nasal philtrum: surgery as an alternative to long-term medical therapy in two dogs. J Small Anim Pract. 2009;50:99–103. | |
White SD, Rosychuk RA, Reinke SI, Paradis M. Use of tetracycline and niacinamide for treatment of auto-immune skin disease in 31 dogs. JAVMA. 1992;200(10):1497–1500. | |
Evans HE, DeLahunta A. Miller’s Guide to the Dissection of the Dog. 3rd ed. Philadelphia: Saunders; 1988. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
