Back to Journals » OncoTargets and Therapy » Volume 13
Success of 125I-Seed Treatment in Vulvar Squamous-Cell Carcinoma with Aplastic Anemia: A Case Report
Authors Ni Q , Pan C, Guo Q, Wang P, Sun G, Xiao S, Dai S
Received 25 September 2020
Accepted for publication 18 November 2020
Published 7 December 2020 Volume 2020:13 Pages 12561—12566
DOI https://doi.org/10.2147/OTT.S283006
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Arseniy Yuzhalin
Qingtao Ni,1 Chi Pan,2 Qing Guo,1 Peng Wang,1 Guangzhi Sun,1 Shujun Xiao,3 Shengbin Dai1
1Department of Oncology, Jiangsu Taizhou People’s Hospital, Taizhou 225300, People’s Republic of China; 2Department of General Surgery, Jiangsu Taizhou People’s Hospital, Taizhou 225300, People’s Republic of China; 3Department of General Practice, Jiangsu Taizhou People’s Hospital, Taizhou 225300, People’s Republic of China
Correspondence: Shujun Xiao
Department of General Practice, Jiangsu Taizhou People’s Hospital, Taizhou 225300, People’s Republic of China
Tel +86 139-5261-9404
Email [email protected]
Shengbin Dai
Department of Oncology, Jiangsu Taizhou People’s Hospital, Taizhou 225300, People’s Republic of China
Tel +86 135-1515-5717
Email [email protected]
Abstract: Vulvar squamous-cell carcinoma (SCC) is a rare disease that occurs mainly in postmenopausal women. Chemo/radiotherapy with or without surgery is the most important modality for treatment of advanced vulvar cancer. A case of vulvar SCC with aplastic anemia was treated using 125I seeds in our department, because surgery and chemotherapy were not possible due to low platelets, leaving radiotherapy as the lone therapeutic option. 125I seeds present an alternative option for treatment of patients with vulvar SCC and local relapse with lymph-node metastasis following previous radiotherapy.
Keywords: vulvar squamous-cell carcinoma, I-125 seeds, aplastic anemia, 125I
Introduction
Vulvar cancer is the fourth–most frequent type of gynecological cancer, accounting for approximately 5% of all female genital tract malignancies.1 According to data from the American Cancer Society, 6,120 cases of vulvar cancer were newly diagnosed and 1,350 women died from this disease in the US in 2020.2 The most common histological type of vulvar cancer is squamous-cell carcinoma (SCC), which accounts for around 90% of cases.3 Surgery is the main treatment for vulvar cancer,4 with radical resection of the vulva and bilateral inguinal lymphadenectomy commonly applied surgical modalities.5 Current standard surgical treatments frequently lead to unsatisfactory outcomes, particularly in patients with node-positive disease.6 Further, chemo/radiotherapy, with or without surgery is the most important treatment modality for advanced vulvar cancer.7 Advanced vulvar SCC has limited treatment options, and recurrence is often lethal.8 Despite the number of treatment options, patients continue to be afflicted by posttreatment side effects, including substantial morbidity, lymphedema, sexual and psychological dysfunction, and wound-healing disorders.9 Moreover, up to 40% of patients with early-stage vulvar SCC develop recurrence within 10 years.10 Unfortunately, not all patients are suitable for surgery and chemotherapy, such as those with aplastic anemia (AA), a type of acquired bone marrow–failure syndrome.11 AA is an immunomediated disease caused by the destruction of hematopoietic cells, resulting in pancytopenia.12
Implantation of iodine 125 (125I) seeds into tumors is a targeted-radiotherapy method. Under the guidance of ultrasound and other imaging techniques, 125I seeds, which emit low-energy gamma rays, are directly implanted into tumors to kill the malignant tissue. As the dose intensity of 125I is inversely proportional to the square of distance, the cumulative dose in normal tissue around the target tumor is minimized.13 This local treatment is safe and effective, and can provide long-term local control of tumors.14 Adjunctive radiotherapy, such as 125I seeds, significantly lowers the risk of recurrence at the site of the primary tumor relative to patients treated without radiotherapy.15 For some patients with recurrence or localized metastasis after surgery, the use of 125I seeds has obvious advantages. 125I seeds combined with chemotherapy and other systemic therapy can also kill distant metastatic lesions, with clear effects on tumor treatment. In addition, this approach can also be used to supplement conventional radiotherapy and as a means of coordinated treatment to achieve better therapeutic effects.16
Here, we report a case of vulvar SCC in a patient with AA who was treated using 125I seeds in our department in recent years.
Case Report
Here, we present the case of a 50-year-old premenopausal woman who had developed painless, slowly growing masses accompanied by irregular bleeding under the skin of the vulva in August 2016. The masses were located in the left labium majus of the vulva, without palpable inguinal lymph nodes bilaterally. One was 3×3 cm and the other 2×2 cm. No ulceration was observed. Laboratory tests (hormone panel, and gynecological and breast cancer markers) were within normal limits. Pathological examination of a vulva biopsy on August 19, 2016 showed moderately and poorly differentiated SCC (Figure 1). A total-body computed-tomography scan and whole-body bone imaging, performed to determine disease stage, were negative for distant metastases.
 |
Figure 1 Histopathologic specimen. (A) H&E staining, 100×. (B) Ki67 staining, 100×. (C) p16 staining, 100×. (D) p63 staining, 100×. |
The patient had a medical history of AA of >20 years, and had taken folic acid, androgens, and cyclosporine to control the disease. The specific dose of medication was unknown. However, the drugs were ineffective. Subsequently, she was treated with regular blood transfusions, with platelet counts ≤10×109/L. Further, she had a history of type2 diabetes of >4 years.
Due to the low platelet count of the patient because of AA, surgery and chemotherapy were not possible. Local radiotherapy with DT72GY/36FX was performed from August 24, 2016 using noncoplanar electron-beam perpendicular irradiation with conventional linear accelerator on vulvar masses covered with 0.5 mm wet gauze, and the patient placed in the lithotomy position at a collimator angle of 90º to the linear accelerator. A significant regression of the masses in the vulva was found on subsequent physical examination; however, she had grade II radiation injury of the skin, which is common and unavoidable, despite the use of radioprotective agents. During treatment, platelets and red blood cells were supplemented by occasional component blood transfusion.
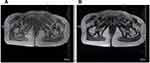
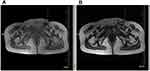
In August 2018, the patient palpated a hard mass in the left inguinal groin, with poor mobility and no tenderness, which had gradually increased in the previous 2 months. On pelvic MRI, a mass of approximately 37×35 mm was observed in the left inguinal region (Figure 2). Due to coagulation disorder resulting from AA, a biopsy was not performed. A diagnosis of metastasis in the left inguinal lymph node was considered, based on MRI. Radiotherapy of the left inguinal groin may cause lymphatic reflux disturbance; therefore, the patient was treated with 35 125I seeds in the left inguinal node under the guidance of ultrasound on October 26, 2018. The biologically effective dose of an 125I seed is almost 100 Gy. The 125I seeds were mainly implanted in the center of the mass at a distance of 1 cm or more from the edge to reduce damage to the skin. On November 18, 2018, the left inguinal mass was approximately 33×33 mm on MRI (Figure 3). Therefore, the patient was again treated with eight 125I seeds in the left inguinal node as a supplementary local dose on January 18, 2019.
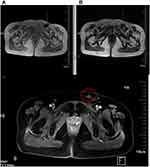
On February 22, 2019, the patient had a mass in the vulva. A 21×16 mm mass and a nodular shadow of approximately 9×8 mm were detected in the left inguinal region on pelvic MRI (Figure 4), indicating that the mass in the groin was receding and the particle implantation was effective. Therefore, 5 days later, 125I seeds were implanted into the base of the new masses in the vulva and left inguinal region. The superficial mass in the vulva gradually subsided soon after seed implantation. At the same time, skin ulceration was still appearing. Unfortunately, 2 months later, the patient developed two more palpable lumps: one of 3×3 cm and the other 2×2 cm. Simultaneously, a 15×6 mm mass was detected in the left inguinal region on ultrasound. Therefore, on May 10, 2019, the patient was treated with implantation of 60 125I seeds in the base of the new lump and left inguinal region. After implantation, the mass completely disappeared again. Although the patient developed bleeding of the vulvar mass, it stopped in response to symptomatic treatment (wet wrap with 0.002% norepinephrine daily and 0.6 g ethylenediamine diaceturate intravenously guttae daily). The ulcers of the epidermis remained.
According to data collected at follow-up examinations performed between May 2019 and June 2019, the patient was able to conduct normal activities, and no new lesions were found. Unfortunately, the patient developed a cough and hemoptysis, and multiple metastatic lesions were found in the lungs on July 12, 2019. In August 2019, the patient received radiotherapy with DT45GY/15FX for the lesions in the lungs; however, the cough and hemoptysis were aggravated following DT27GY/9FX radiotherapy. The patient died on October 31, 2019.
Discussion
This was a unique case of vulvar SCC with AA in a 50-year-old premenopausal woman. Vulvar cancer mainly occurs in older women, primarily those who are postmenopausal.17 The peak age of incidence of vulvar cancer has been reported as >70 years; however, the peak period for vulvar cancer among Chinese women is <60 years.18 The patient reported here was a middle-aged premenopausal woman with a history of AA. Surgical treatment is usually the first choice for patients with early-stage vulvar SCC, including radical vulvectomy with bilateral inguinofemoral lymphadenectomy as a treatment of choice for vulvar SCC;19 however, the substantial loss of tissue involved in such surgery results in a risk of postoperative complications.20 After consultation with several hospitals, it was concluded that surgery and chemotherapy would be intolerable for our patient, because of her history of AA. In this case, radiotherapy was the only option. Radiation therapy has been regarded as a highly effective treatment for patients with noninvasive and inoperable vulvar cancer,21 with recurrence rates of 20% and 35% for patients with stage III and IV disease, respectively.22
Vulvar cancer lesions can directly infiltrate adjacent tissue and organs, such as the vagina, urethral orifice, and anus, or spread directly to the opposite side. Further, vulvar cancer can spread from its original site to locoregional nodes. The 5-year overall survival rate for patients with vulvar cancer without nodal metastasis is >90%, while that for patients with nodal metastasis is <60%.23 Our patient was clinically assessed as lymph-node negative at first diagnosis. Unfortunately, she was unable to sustain prolonged disease-free survival, and experienced local relapse and nodal involvement 2 years after the first radiotherapy. Due to the risk of exceeding normal tissue tolerance, doses are often limited for patients with recurrence after previous radiotherapy. Nevertheless, brachytherapy, such as 125I seeds, allows dose escalation in such situations, which can provide a perfectly targeted and localized boost dose and potentially improve patient outcomes.24
For 125I seeds, a physical dose of 100 Gy delivered at an initial dose rate of approximately 0.05 Gy/hour, is equivalent to 50 Gy of external-beam radiotherapy in fractions of 2 Gy.25 Compared with conventional external-beam radiotherapy, implantation of 125I seeds involves a smaller irradiation dose, longer effect, and more accurate treatment positioning to achieve genuine image-guided radiotherapy and conformal radiotherapy. 125I seeds provide a high local dose effect to tumors, with a small radiation radius and minimal damage to surrounding normal tissue. The radiation dose from 125I seeds is inversely proportional to the square of distance, and the effective diameter of particle radiation is only 17 mm.26 Therefore, 125I seeds represent an excellent local treatment for a well-selected patient population.27
Following treatment with 125I seeds, although the patient developed redness and swelling of the skin after particle implantation, the therapeutic effect was relatively good. To prevent the occurrence of skin symptoms, hyaluronic acid injection can be used to provide space between the tumor and the skin in patients with subdermal tumors, resulting in decreased doses to the skin during brachytherapy.28
Conclusion
125I seeds represent an alternative therapeutic opportunity for patients with vulvar SCC who do not have the option of surgery and chemotherapy and for those with local relapse with lymph-node metastasis after previous radiotherapy.
Ethics Statement
This study was approved by the Human Ethics Review Committee of Jiangsu Taizhou People’s Hospital.
Consent to Publish
Written informed consent to publish this report and the associated medical images was provided by the patient prior to her passing.
Author Contributions
All authors made a significant contribution to the work reported, whether in conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas, took part in drafting, revising, or critically reviewing the article, gave final approval to the version to be published, have agreed on the journal to which the article has been submitted, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Hecking T, Thiesler T, Schiller C, et al. Tumoral PD-L1 expression defines a subgroup of poor-prognosis vulvar carcinomas with non-viral etiology. Oncotarget. 2017;8:92890–92903. doi:10.18632/oncotarget.21641
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7–30. doi:10.3322/caac.21590
3. Anastasi E, Gigli S, Ballesio L, et al. The complementary role of imaging and tumor biomarkers in gynecological cancers: an update of the literature. Asian Pac J Cancer Prev. 2018;19:309–317.
4. Niu Y, Yin R, Wang D, et al. Clinical analysis of neoadjuvant chemotherapy in patients with advanced vulvar cancer: A STROBE-compliant article. Medicine. 2018;97:e11786. doi:10.1097/MD.0000000000011786
5. Saner FA, Schotzau A, Mackay G, et al. Fibrin-thrombin sealant does not reduce lymphocele formation in patients with inguinofemoral lymphadenectomy for vulvar cancer. Cancer Manag Res. 2019;11:3575–3582. doi:10.2147/CMAR.S197143
6. Hockel M, Trott S, Dornhofer N, et al. Vulvar field resection based on ontogenetic cancer field theory for surgical treatment of vulvar carcinoma: a single-centre, single-group, prospective trial. Lancet Oncol. 2018;19:537–548. doi:10.1016/S1470-2045(18)30109-8
7. Zobec LH. Long term results of radiotherapy in vulvar cancer patients in slovenia between 1997–2004. Radiol Oncol. 2017;51:447–454. doi:10.1515/raon-2017-0024
8. Clancy AA, Spaans JN, Weberpals JI. The forgotten woman’s cancer: vulvar squamous cell carcinoma (VSCC) and a targeted approach to therapy. Ann Oncol. 2016;27:1696–1705. doi:10.1093/annonc/mdw242
9. Kortekaas KE, Santegoets SJ, Abdulrahman Z, et al. High numbers of activated helper T cells are associated with better clinical outcome in early stage vulvar cancer, irrespective of HPV or p53 status. J Immunother Cancer. 2019;7:236. doi:10.1186/s40425-019-0712-z
10. G N T, Pouwer AW, de Bock GH, et al. Prognostic factors for local recurrence of squamous cell carcinoma of the vulva: A systematic review. Gynecol Oncol. 2018;148:622–631. doi:10.1016/j.ygyno.2017.11.006
11. Zhang X, Liu L, Dou C, et al. PPAR gamma-regulated microRNA 199a-5p underlies bone marrow adiposity in aplastic anemia. Mol Ther Nucleic Acids. 2019;17:678–687. doi:10.1016/j.omtn.2019.07.005
12. Scheinberg P, Chen J. Aplastic anemia: what have we learned from animal models and from the clinic. Semin Hematol. 2013;50:156–164. doi:10.1053/j.seminhematol.2013.03.028
13. Ma X, Yang Z, Jiang S, et al. Hybrid optimization based on non-coplanar needles for brachytherapy dose planning. J Contemp Brachytherapy. 2019;11:267–279. doi:10.5114/jcb.2019.86167
14. Xiang Z, Li G, Liu Z, et al. 125i brachytherapy in locally advanced nonsmall cell lung cancer after progression of concurrent radiochemotherapy. Medicine. 2015;94:e2249. doi:10.1097/MD.0000000000002249
15. Vora GK, Demirci H, Marr B, et al. Advances in the management of conjunctival melanoma. Surv Ophthalmol. 2017;62:26–42. doi:10.1016/j.survophthal.2016.06.001
16. Song Y, Chan MF, Burman C, et al. Comparison of two treatment approaches for prostate cancer: intensity-modulated radiation therapy combined with 125I seed-implant brachytherapy or 125I seed-implant brachytherapy alone. J Appl Clin Med Phys. 2008;9:2283. doi:10.1120/jacmp.v9i2.2283
17. Peri M, Grassadonia A, Iezzi L, et al. A case of stage i vulvar squamous cell carcinoma with early relapse and rapid disease progression. Case Rep Oncol Med. 2019;2019:1018492.
18. Xiao X, Meng YB, Bai P, et al. Vulvar cancer in china: epidemiological features and risk analysis. J Cancer. 2017;8:2950–2958. doi:10.7150/jca.20496
19. Jiang Y, Tian R, Yu S, et al. Clinical significance of galectin-7 in vulvar squamous cell carcinoma. Oncol Lett. 2015;10:3826–3831. doi:10.3892/ol.2015.3826
20. Dias AR
21. Saito T, Tabata T, Ikushima H, et al. Japan society of gynecologic oncology guidelines 2015 for the treatment of vulvar cancer and vaginal cancer. Int J Clin Oncol. 2018;23:201–234.
22. Sznurkowski JJ, Milczek T, Emerich J. Prognostic factors and a value of 2009 FIGO staging system in vulvar cancer. Arch Gynecol Obstet. 2013;287:1211–1218. doi:10.1007/s00404-012-2683-x
23. Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17:1471–1474. doi:10.1245/s10434-010-0985-4
24. Brun T, Ferron G, Filleron T, et al. Experimental study of pelvic perioperative brachytherapy with iodine 125 seeds (I-125) in an animal model. J Contemp Brachytherapy. 2018;10:463–469. doi:10.5114/jcb.2018.79470
25. Baltas D, Lymperopoulou G, Loffler E, et al. A radiobiological investigation on dose and dose rate for permanent implant brachytherapy of breast using 125I or 103Pd sources. Med Phys. 2010;37:2572–2586. doi:10.1118/1.3426027
26. Wang W, Liu Z, Zhu J, et al. Brachytherapy with iodine 125 seeds for bone metastases. J Cancer Res Ther. 2017;13:742–747. doi:10.4103/jcrt.JCRT_399_17
27. Logghe P, Verlinde R, Bouttens F, et al. Long term outcome and side effects in patients receiving low-dose I125 brachytherapy: a retrospective analysis. Int Braz J Urol. 2016;42:906–917. doi:10.1590/S1677-5538.IBJU.2015.0542
28. Yuan Y, Ying Z, Jianjian D, et al. Subdermal injection of hyaluronic acid to decrease skin toxicity from radiation delivered with low-dose-rate brachytherapy for cancer patients. J Contemp Brachytherapy. 2019;11:14–20. doi:10.5114/jcb.2019.82770
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.