Back to Journals » Clinical Epidemiology » Volume 12
Statistical Challenges in Development of Prognostic Models in Diffuse Large B-Cell Lymphoma: Comparison Between Existing Models – A Systematic Review
Authors Jelicic J, Larsen TS, Frederiksen H , Andjelic B , Maksimovic M, Bukumiric Z
Received 31 December 2019
Accepted for publication 8 April 2020
Published 27 May 2020 Volume 2020:12 Pages 537—555
DOI https://doi.org/10.2147/CLEP.S244294
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 8
Editor who approved publication: Professor Irene Petersen
Jelena Jelicic,1 Thomas Stauffer Larsen,1,2 Henrik Frederiksen,1,2 Bosko Andjelic,3 Milos Maksimovic,4 Zoran Bukumiric5
1Department of Hematology, Odense University Hospital, Odense, Denmark; 2Department of Clinical Research, University of Southern Denmark, Odense, Denmark; 3Department of Haematology, Blackpool Victoria Hospital, Lancashire Haematology Centre, Blackpool, UK; 4Department of Ophthalmology, Aalborg University Hospital, Aalborg, Denmark; 5Department of Statistics, Faculty of Medicine, University of Belgrade, Belgrade, Serbia
Correspondence: Thomas Stauffer Larsen
Department of Hematology, Odense University Hospital, Odense, Denmark
Tel +45 2145 0236
Fax +45 6541 3035
Email [email protected]
Background and Aim: Based on advances in the diagnosis, classification, and management of diffuse large B-cell lymphoma (DLBCL), a number of new prognostic models have been proposed. The aim of this study was to review and compare different prognostic models of DLBCL based on the statistical methods used to evaluate the performance of each model, as well as to analyze the possible limitations of the methods.
Methods and Results: A literature search identified 46 articles that proposed 55 different prognostic models for DLBCL by combining different clinical, laboratory, and other parameters of prognostic significance. In addition, six studies used nomograms, which avoid risk categorization, to create prognostic models. Only a minority of studies assessed discrimination and/or calibration to compare existing models built upon different statistical methods in the process of development of a new prognostic model. All models based on nomograms reported the c-index as a measure of discrimination. There was no uniform evaluation of the performance in other prognostic models. We compared these models of DLBCL by calculating differences and ratios of 3-year overall survival probabilities between the high- and the low-risk groups. We found that the highest and lowest ratio between low- and high-risk groups was 6 and 1.31, respectively, while the difference between these groups was 18.9% and 100%, respectively. However, these studies had limited duration of follow-up and the number of patients ranged from 71 to 335.
Conclusion: There is no universal statistical instrument that could facilitate a comparison of prognostic models in DLBCL. However, when developing a prognostic model, it is recommended to report its discrimination and calibration in order to facilitate comparisons between different models. Furthermore, prognostic models based on nomograms are becoming more appealing owing to individualized disease-related risk estimations. However, they have not been validated yet in other study populations.
Keywords: diffuse large B-cell lymphoma, discrimination, calibration, prognosis, models, nomograms
Introduction
The most commonly used prognostic index for risk stratification of patients with diffuse large B-cell lymphoma (DLBCL) is the International Prognostic Index (IPI).1,2 Since the introduction of rituximab in DLBCL therapy, the discriminative abilities of the IPI have been challenged in many studies.2–6 Predominantly, based on the new insights into the pathobiology of disease, the predictive power of different biomarkers for prognosis has been extensively investigated and this has led to numerous attempts at incorporating clinical, biological, laboratory, immunohistochemical, and genetic markers in prognostic scores.7–15 However, none of the newly developed models has been as widely used as the IPI.
Despite the standard treatment with chemoimmunotherapy, approximately 30% of patients experience disease relapse or are refractory to therapy.16 Therefore, the utilization of selected group of patients’ characteristics through a statistical model is important to identify high-risk patients who could potentially benefit from more intensive immunochemotherapy and/or molecular-targeted agents.17 Prognostic risk models, which are a mathematically precise way to summarize properties of measurements and their associations, are created to predict events.18 When developing a model, it is important to provide some evaluation of its performance in comparison to an existing model.19 The usefulness of a prognostic test in clinical practice depends mainly on the ability of the test to stratify patients into different risk groups and to provide accurate predictions about their future outcome. However, when biomarkers are included in statistical models for predicting a clinical outcome, two problems can occur, namely “error in discrimination” and “error in calibration”.20 To achieve accurate risk prediction, validation of the specific prediction model is needed. For that task, various statistical methods have been suggested in the literature.20–22
In our previous systematic review, we described the studies that reported different prognostic models for newly diagnosed DLBCL with a focus on clinical, laboratory, molecular, and/or imaging parameters included in each model. However, the analysis of statistical methods used to compare the novel models in DLBCL with the previously reported models, to the best of our knowledge, has never been performed. Therefore, this study aims to investigate the statistical instruments that have been used to develop, compare, and evaluate the performance of prognostic models for DLBCL. Another aim is to analyze possible limitations of each statistical method that can restrict the wide usage of the derived model.
Materials and Methods
Search Strategy
Similarly to our previous research, this review was conducted in accordance with the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-analysis.23 A comprehensive search was conducted in PubMed and Embase to identify original publications that proposed prognostic scores for newly diagnosed DLBCL.24 Only studies in English published between 1993 and 15 July 2019 were considered.
The relevant Medical Subject Headings (MeSH) were used where possible and the following search terms were used in our analysis: lymphoma, large B-cell, diffuse/DLBCL, index/indices, model, score, prognosis/prognostic, outcome, survival, and comparison. The additional eligible studies were retrieved through secondary references.24 The search results were independently double-screened by the research team (JJ, MM, BA) according to inclusion/exclusion criteria at the abstract and the full-text screening. All the studies included in the final list were checked for data availability by BA and underwent full statistical analysis, performed by ZB.
Selection Criteria and Data Extraction
This analysis included studies that developed a new risk model for newly diagnosed DLBCL patients by combining at least two parameters, of which one must be a clinical or a laboratory parameter with or without imaging or a molecular prognostic marker. The studies evaluated overall survival (OS) using the Kaplan–Meier method and/or expressed the outcome as the percentage of surviving patients. The exclusion criteria referred to all articles that did not provide a new prognostic model (eg case reports, commentaries, meeting reports, reviews), as well as those that analyzed only one subtype of DLBCL, proposed an index based exclusively on histopathological or molecular data, and/or lacked sufficient data for estimating OS. Moreover, we excluded studies that only tested scores previously developed on patient populations other than newly diagnosed DLBCL.24
After exclusion of the irrelevant studies based on the titles and abstracts, the full texts of the selected articles were analyzed. We also reviewed the full text of the studies from which a decision could not be made on the abstract alone. All studies included in the final list were reviewed for their data accuracy.24 The data extracted from each study were all of the following whenever reported: author and publication year, index name, risk categories, number of patients per group of new index and previously reported index (eg IPI, revised IPI), model performance, and OS expressed as the 3-year percentage of surviving patients. If missing, the 3-year OS for newly created models and previously reported models (used to compare with the newly developed model) was estimated from the survival curves using GetData Graph Digitizer 2.26. Three-year OS was chosen because the majority of studies report this endpoint.
For the studies that compared performance of a new score and previously tested scores, the statistical methods of comparison were recorded. Additional statistics included c-index, Bayesian information criterion (BIC), concordance probability estimate (CPE), Akaike’s information criterion (AIC), Hosmer–Lemeshow goodness of fit, and receiver operating characteristics (ROC) curves. The difference between c-index was calculated in the studies that reported the c-index. For each new score, the difference and proportion between high- and low-risk groups were calculated. We first extracted 3-year OS measures, then these values were compared between high-risk and low-risk groups using calculations of differences and proportions. Proportion was assessed by dividing 3-year OS estimates in high-risk patients by those in low-risk patients, while the difference was calculated by subtraction of 3-year OS of the low-risk group from the high-risk group. This value was expressed in percentages. Furthermore, similar calculations were performed if the studies reported 3-year OS survival for previously reported indices (eg IPI). If the 3-year OS was not reached because of censored patients, or if the OS was 0%, the proportion was labeled as not applicable (NA) for mathematical reasons. The scores with the highest values of the difference and proportion were regarded as those with better differentiation between risk categories.
Results
The initial search strategy identified 5239 articles, but after the inclusion/exclusion criteria were used, of 418 potentially relevant articles, 46 studies were included in the final list. Among these, seven additional articles were retrieved through the references included in the eligible studies and relevant reviews (Figure 1). Although the IPI was developed based on a broad cohort of patients with aggressive lymphoma subtypes, this model was included in the current study because it is widely used for prognostication in DLBCL.1,24 Furthermore, the search strategy identified six additional studies that used nomograms to develop prognostic models.25–30 Although these studies did not meet the inclusion criteria regarding OS and risk stratification, they are briefly discussed later owing to their individualized approach for risk prediction. In total, 52 studies were analyzed.
 |
Figure 1 Flowchart representing the selection process of including studies published between 1993 and July 2019. |
In 46 studies, a total of 55 prognostic models were proposed for DLBCL patients. From 1993 until 2019, a significant increase in the number of proposed prognostic models for DLBCL patients has been observed (Figure 2). In all, 40 studies proposed one prognostic model, while four studies proposed two models,10,31-33 one proposed three,1 and one study proposed four prognostic models.11
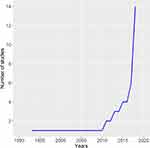 |
Figure 2 Graph showing an increasing number of reported studies over the past two decades. |
In total, 26 prognostic models used a four-category risk stratification with recognition of low-, low–intermediate-, high–intermediate-, and high-risk groups (Tables 1–4). Three-category risk stratification (low-, intermediate-, and high-risk groups) was proposed in 20 studies. Two-category risk stratification with high- and low-risk groups was reported in six studies, while five-category risk stratification was proposed in three studies.
 |
Table 4 Summary of Characteristics (Number of Patients, Additional Statistics Used to Compare Novel Model and Previously Developed Models) of Studies that Used Nomograms |
Each of six studies that developed nomograms proposed just one prognostic model based on the individual risk prognostication. Additional data regarding each study are provided in the supplementary table.
Statistical Analysis
Twelve of 46 studies analyzed populations with more than 400 patients (range 403–2031), while 32 studies included fewer patients (range 45–391). Among studies based on nomograms, three of six included more than 400 patients.
The majority of studies lacked the splitting of the analyzed population into training and validation sets as well as a comparison to previously proposed models.24 Seven studies of 46 used training and validation sets to develop a prognostic index and to validate it.1,2,16,31,34-36 Among the studies using nomograms, three used both internal and external validation,26–28 one used internal validation only,25 and two did not assess the performance through validation.29,30
Model performance was evaluated using different methods, which are discussed in the following subsections.
C-Statistics
As a measure of discrimination, the area under the receiver operating characteristics (ROC) curve for survival outcomes (the c-index) was used in seven of 46 studies.7,16,32,34,37-39 Higher values of the c-index indicated better discrimination. The value of the c-index for the novel prognostic models ranged from 0.708 for the comorbidity National Comprehensive Cancer Network-IPI (cNCCN-IPI) to 0.830 for the lipoprotein prognostic index (Lipo-PI). The c-index was also calculated for the indices that were previously reported, such as IPI (range 0.67–0.76), revised IPI (c-index 0.642), and NCCN-IPI (range 0.6950.791). Two studies pointed to the use of Harrell’s c-index/modified method as a discrimination method,16,32 while one reported the use of the c-index according to Uno et al.22
Concordance Probability Estimate (CPE)
The discrimination of the newly developed prognostic models and previously developed models was measured by CPE along with its 95% confidence intervals (CI) in four of 46 studies.2,3,10,40 A higher CPE indicated better discrimination. The CPE value was 0.66 for E-IPI,3 0.781 for a new risk model,40 0.783 for modified NCCN-IPI,10 and 0.8 for NCCN-IPI. For the compared models, the lowest CPE value was 0.59 for R-IPI,3 while the highest was 0.76 for NCCN-IPI.40
Bayesian Information Criterion (BIC)
A global measure of fit, the BIC was assessed in two studies, where lower values of BIC indicate a better fit.7,32
Akaike’s Information Criterion (AIC)
The performance of indices was compared by a measure of global fit (AIC) in five studies, in which a lower value indicated a better fit.2,3,9,31,40 The lowest AIC of 662.5 was observed in a new risk model,40 while the highest value was 4566 in NCCN-IPI.2 Of the compared models, the highest AIC value was observed in the IPI2 and the lowest in NCCN-IPI.40
Hosmer–Lemeshow Goodness of Fit
One study used Hosmer–Lemeshow goodness of fit to assess calibration, by comparing the proportions of patients whose estimated risk shifted in the correct and wrong directions on the basis of a χ2-test within reclassified categories for each score separately.36
Relative Brier Score Reduction (RBSR)
One study reported RBSR, which represents a measure of the overall model performance. The lower the Brier score for a set of predictions, the better the predictions are calibrated.34
Receiver Operating Characteristics (ROC) Curves
In 11 of 46 studies, the cut-off of a continuously distributed measurement for OS prediction was defined as the point at which the sensitivity plus the specificity were maximized in the ROC curves.15,35,37,40-47 One study each used ROC curves to select the best cut-off of beta-2 microglobulin (B2M),40 C-reactive protein (CRP),44 lymphocyte to monocyte ratio,44 platelet to lymphocyte ratio (PLR),45 tumor-infiltrating T-lymphocyteproportion and ratio between CD4-positive and CD8-positive T-lymphocytes,48 age,35 maximum standardized uptake value,47 and ratio Deauville score.46 In two studies, ROC was used to assess the albumin level,35,44 and in two the absolute monocyte count and absolute lymphocyte count.42,43 One study used X-tile software to calculate the optimal cut-off value for the albumin to globulin ratio.41
Regarding the absolute monocyte count, absolute lymphocyte count, and platelet level, previously reported cut-off points were used in five studies.9,33,49-51 However, the majority of studies used institutional upper/lower limits of normal (ULN/LLN) for the continuously distributed measurements, including hemoglobin levels, platelet counts,52 absolute monocyte count, absolute neutrophil count, B2M, LDH,37 B2M,36,45,53,54 and lipid levels.16
Three studies used other statistical techniques (eg percentile value, medians) for testing different cut-off points of continuous measurements.8,33,55
Difference and Ratio in 3-Year Overall Survival Between Risk Groups
Fifteen of 46 studies reported 3-year OS for novel prognostic models, while OS was calculated from Kaplan–Meier curves for the rest3,9,10,16,31,34,35,39,40,44,52,56-59 (Tables 1–3). Only the minority of studies (six of 46) reported 3-year OS for compared indices.9,10,31,39,44,52 Regarding 3-year OS of the novel models, the highest ratio between the low- and high-risk groups was 6,53 while the lowest was 1.31.41 These studies included 71 and 335 patients, respectively. In six articles the ratio in 3-year OS between low-risk and high-risk groups was not applicable owing to the short follow-up of high-risk patients or there being no surviving patients at 36 months.35,45,47,56,60,61 The difference in 3-year OS ranged from 18.9%41 to 100%56 with335 and 105 patients, respectively, being analyzed.
Regarding the IPI, the ratio between risk groups ranged from 1.38 to 7.73, and the difference from 22.9% to 68.8%. The lowest age-adjusted IPI (aaIPI) ratio score between the low-risk and high-risk groups was 2.05 and the highest was 2.48, while the lowest difference was 42.0% and the highest was 53.4%. Regarding NCCN-IPI, the ratio ranged from 1.75 to 3.70, and the difference from 40.9% to 72.9%. Additional information regarding differences and the ratios of other indices are provided in Tables 1–4.
Figure 3A and B shows the graphical presentation of the ratio and difference, with preferable position close to the upper right point, which indicates better model power. Both parts of the figure allow the visual comparison of different models owing to their position with the identical range on the Xand Y axes. The studies with a larger number of patients had lower variability than the studies with a limited number of patients (Figure 3A, B). Furthermore, it was observed that the IPI in different studies tends to have a lower difference and ratio (Figure 3B).
Prognostic Models Based on Nomograms
A total of six studies proposed prognostic models for DLBCL patients based on calculated individual risk using nomograms (Table 4).25–27,29,30 In all studies, the predictive accuracy of the nomogram was evaluated using discrimination and/or calibration. Three studies used splitting of the analyzed population into training and validation sets.26–28 One study combined the machine learning method, known as a stacking algorithm, with clinical data obtained from nationwide lymphoma registries in order to develop a stacking-based prognostic model, which was superior to both IPI and NCCN-IPI.28
All nomogram-based studies reported the c-index25–30 and one study also applied integrated Brier score (IBS).28 Two studies reported the c-index for estimating progression-free survival.25,30 The nomogram and other prognostic indices were compared with the 2 log likelihood (2LL) and the AIC in terms of goodness of fit in one study.29 Calibration plots were reported in three nomogram studies26,27,29,while one study applied Hosmer–Lemeshow goodness of fit to assess calibration .25 New models were typically compared with established indices such as IPI, NCCN-IPI, and R-IPI.
Discussion
Our review shows that a large number of new prognostic models for patients with DLBCL have been proposed, aiming at improving the discriminative power compared to the IPI. However, general application of the majority of these new prognostic models has been prevented because of a lack of validation, limited number of analyzed patients, and other statistical difficulties regarding model development. The most commonly validated models used for prognostication as well as for the comparison with other novel models were IPI, R-IPI, and NCCN-IPI.24 Among these, NCCN-IPI had the highest performance values, although the performance of models based on nomograms was superior to the NCCN-IPI. However, these models are relatively new and have not been validated in other study populations.
The traditional splitting of data into the training and validation data sets when developing a new model was used in only 19% of studies. Using the training data set, it is possible to construct an initial prediction model, the accuracy of which is then assessed using the validation set. However, the usefulness of any model actually depends on its accuracy, ie the ability of the model to correctly identify each patient’s outcome.17 To report the discrimination (accuracy) value of the model, seven studies plus all studies based on nomograms used the concordance (c) statistic, which is conceptually similar to the ROC curve.17 The c-index scores between 0.5 (no discrimination) and 1.0 (perfect discrimination).20 The value of the c-index for novel prognostic models ranged from 0.708, indicating a good model, to 0.830, indicating a strong model. However, the c-index for the widely applied indices such as IPI, R-IPI, and NCCN-IPI had the lowest value of only approximately 0.6, which defines low discrimination powers. In the analyzed studies the c-index refers to time-to-event outcome (OS). In such analyses, the c-index and its interpretation are less well established than a binary outcome, since some patients had not experienced an event at the time of analysis.17 There is no consensus on how to handle time-to-event data, because this requires exclusion of censored patients or using indirect estimates of survival, based on the regression model, with a tendency to give lower c-statistics than other methods.17,62 Another issue with the c-statistic is that it measures discrimination and not calibration, which is the agreement between observed outcomes and predictions.17 The c-statistic is not a good measure of the actual probability of events if only a small number of patients experience the event of interest.17 In addition, four studies reported CPE in order to evaluate the discriminatory power and the predictive accuracy of nonlinear statistical models.2,3,10,40 An early estimate of the concordance probability, which was the predominant discrimination statistic adapted for survival analysis, was Harrell’s c-index, used in three studies.16,25,32,63 However, Harrell’s c-index was influenced by the rate of patient accrual and the length of the study. To avoid this limitation, Uno et al introduced inverse probability censoring weights to the c-index. This method was used in one study.22,33,39
In six studies, the performance of indices was compared using the AIC.2,3,9,29,40,64 To compare the quality of a set of statistical models (to perform model comparisons) with each other, two studies used another criterion for model selection – the BIC, which measures the trade-off between model fit and complexity of the model.7,18,32 Both BIC and AIC balance the level of fit (based on the log-likelihood, a basic estimate of fit) with model complexity. Furthermore, the formula for the BIC is similar to the formula for the AIC, but with a different penalty for the number of parameters.18 Lower AIC and BIC values are preferred, and indicate a more explanatory and informative model.64 The AIC is the commonly used model selection tool for choosing between alternative models and has a preference for more complex models compared to the BIC, owing to its ability to eliminate unnecessarily complicated models, which contain too many parameters for accurate estimation on a given data set.64 However, the AIC takes into account each model and ranks them from the best to worst, then it chooses between the models based on the goodness of fit using the lowest number of variables that explains the outcome best. This means that if all models are poor, it will choose the best of these, because quality is not taken into account.65 Furthermore, when the sample size is small, there is a high probability that the AIC will select models that have too many parameters, leading to overfitting. To address such potential overfitting, alternative versions of the AIC have been proposed to make it easier to compare models estimated on different data sets of varying sizes.66
Two studies used the Hosmer–Lemeshow test goodness of fit to assess calibration.25,36,67 Models are well calibrated if expected and observed event rates in subgroups of risk models are similar. The main limitations of this test are that it is based on an arbitrary grouping of observations, it has poor power in small data sets, and the result is expressed only in a p-value.68 In addition, three models used a calibration plot, described as a graphical illustration of the Hosmer–Lemeshow test, which is another method to assess calibration in which the observed event status is plotted against the predicted risk estimates. The plot is often supplied with a calibration curve to help to diagnose a lack of fit, and will lie on the 45-degree line in a well-calibrated model.68,69 Some authors have suggested the use of the calibration slope, which, in addition to the p-value, provides a confidence interval and a measure of size of effect, since the estimated slope is obtained from the regression model and does not require the patients to be grouped. Therefore, the calibration slope does not suffer from the limitations of the Hosmer–Lemeshow test.68
To determine the optimal cut-off point for continuous measurements for predicting OS, 11 of 46 studies used the ROC curve, which maximizes the likelihood ratio.15,35,37,40–47 The ROC curve analysis has earned a place in biomedical studies when interpreting the results of diagnostic accuracy of a continuous marker. The performance of a marker is evaluated by the area under the ROC curve (AUC), an indicator of overall “accuracy”, in which a higher AUC value indicates a better performance. However, there is variation in the sensitivity and specificity from point to point along the ROC curve and therefore it is important to consider the aims of diagnostic tests with respect to the significance and costs of a false-positive or negative interpretation.70 Regarding the cut-off points of continuous variables used in prognostic models in DLBCL, it is obvious that different thresholds based on ROC results were incorporated in the models, where an additional five studies used cut-off points that had already been reported.9,33,49–51 However, in ROC, one’s choice of the value based on specificity and sensitivity can largely depend on the data set, meaning that the optimal cut-off value in one population might not be the optimal value in another.
Because of the lack of comparisons between prognostic models and the use of a variety of statistical methods for model comparison, it was not possible to make a universal comparison. Therefore, we have chosen to compare models by using very simple tools: the difference and proportion between scores among low-risk and high-risk groups. The bigger the observed value between the low- and high-risk groups, the better the model’s power. However, the number of patients in the high-risk group has a major impact on the results. Hence, it is not surprising that the highest ratio was observed in a study including just 73 patients, while the highest difference was observed in a study that analyzed 337 patients. However, these tools have pointed to the rather limited power of the IPI, as well as the lower prognostic value of models developed in a limited number of patients. Although these are simple methods of comparison, they can give a false impression of superiority of one model, possibly due to a limited number of patients in high-risk groups or relatively short follow-up. Thus, they should be used as an addition to other performance measures and not as the primary method of comparison owing to these limitations. The studies that used nomograms presented a visual representation of a statistically predictive model that estimates the probability of a clinical event by calculating the cumulative effect of weighted independent variables.71 Although these models cannot be compared by the ratio or difference because they do not use risk categories, they provide the discrimination and/or calibration to compare different prognostic models. Irrespective of the fact that that these complex models did not entirely fulfill our inclusion criteria, their recognition indicates the current trend towards more individualized prognostication. This is mainly due to the fact that grouping of patients into risk categories results in an ineffective use of the data and tends to reduce the predictive accuracy of a prognostic model. In addition, the complexity of nomograms can be offset by using electronic versions.72 Still, there are no guidelines regarding which value could be used in decision making when nomograms are used for prognostication. However, it would advance clinical application if future studies evaluated nomograms in the risk-adapted therapeutic strategies.26 Nevertheless, due to other limitations of nomograms, the focus of future studies, besides developing new models, should be validation of the existing prognostic models.73
In addition, one study developed a prognostic model by combining data from a clinical database with machine learning techniques.28 The stacking algorithm, used in the study, is a way of ensembling multiple regression models to obtain survival curves, eliminating the need for the specification of one prognostic modeling approach.74 Although the machine learning technique is not a new concept, it is gaining more attention in the classification, prognostication, and genetic analysis of DLBCL.28,75,76 It is evident that the clinical and genetic heterogeneity of DLBCL, as well as overlapping of DLBCL subgroup classifications, represent significant challenges for accurate outcomes prediction.74 To provide a prediction of clinically relevant outcomes for patients with DLBCL, future studies will likely have to combine different factors (clinical, sociodemographic, tumor microenvironment, genetic, etc), possibly with the aid of the machine learning and high-dimensional data analysis or other statistical methods in order to develop comprehensive, multilevel prognostic models that should be easily applicable in clinical care.74
Conclusion
Although an increasing number of prognostic models for DLBCL has emerged in the past two decades, there is no universally accepted statistical method of reporting prognostic models in DLBCL. When reporting a new prognostic model, we would recommend assessing the discrimination and calibration of the prognostic model. Other measures of its performance may be used when adding a novel predictor to an established model.19 In addition, comparisons of different available prognostic models based on the same population should be provided. This is needed in order to avoid the vagueness that currently exists in the literature, owing to an inability to compare current prognostic models in DLBCL. Furthermore, to adapt models to a more individualized approach, an increasing number of models based on nomograms has been published. These models generally report the discrimination and calibration in order to compare the novel model with previous models, and tend to have higher predictive accuracy by avoiding potential loss of information due to the omission of risk categorization. However, future prognostication studies that integrate advances in statistics with growing knowledge on the diagnostics, pathology, and therapy of DLBCL are necessary.
Acknowledgment
This work is dedicated to our teacher and dear friend Professor Goran Trajkovic, who died too early.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Shipp MA. A predictive model for aggressive non-hodgkin’s lymphoma. the international non-hodgkin’s lymphoma prognostic factors project. N Engl J Med. 1993;329:987–994.
2. Zhou Z, Sehn LH, Rademaker AW, et al. An enhanced international prognostic index (NCCN-IPI) for patients with diffuse large B-cell lymphoma treated in the rituximab era. Blood. 2014;123(6):837–842. doi:10.1182/blood-2013-09-524108
3. Advani RH, Chen H, Habermann TM, et al. Comparison of conventional prognostic indices in patients older than 60 years with diffuse large B-cell lymphoma treated with R-CHOP in the US Intergroup Study (ECOG 4494, CALGB 9793): consideration of age greater than 70 years in an elderly prognostic index (E-IPI). Br J Haematol. 2010;151(2):143–151. doi:10.1111/j.1365-2141.2010.08331.x
4. Sehn LH, Berry B, Chhanabhai M, et al. The revised international prognostic index (R-IPI) is a better predictor of outcome than the standard IPI for patients with diffuse large B-cell lymphoma treated with R-CHOP. Blood. 2007;109(5):1857–1861. doi:10.1182/blood-2006-08-038257
5. Hong J, Kim SJ, Chang MH, et al. Improved prognostic stratification using NCCN- and GELTAMO-international prognostic index in patients with diffuse large B-cell lymphoma. Oncotarget. 2017;8(54):92171–92182. doi:10.18632/oncotarget.20988
6. Saygin C, Jia X, Hill B, et al. Impact of comorbidities on outcomes of elderly patients with diffuse large B-cell lymphoma. Am J Hematol. 2017;92:989–996.
7. Salles G, de Jong D, Xie W, et al. Prognostic significance of immunohistochemical biomarkers in diffuse large B-cell lymphoma: a study from the lunenburg lymphoma biomarker consortium. Blood. 2011;117:7070–7078.
8. Tomita N, Sakai R, Fujisawa S, et al. SIL index, comprising stage, soluble interleukin-2 receptor, and lactate dehydrogenase, is a useful prognostic predictor in diffuse large B-cell lymphoma. Cancer Sci. 2012;103:1518–1523.
9. Huang Y-C, Liu C-Y, Lu H-J, et al. Comparison of prognostic models for patients with diffuse large B-cell lymphoma in the rituximab era. Ann Hematol. 2013;92:1513–1520.
10. Melchardt T, Troppan K, Weiss L, et al. A modified scoring of the NCCN-IPI is more accurate in the elderly and is improved by albumin and β 2 -microglobulin. Br J Haematol. 2015;168(2):239–245. doi:10.1111/bjh.13116
11. Xu-Monette ZY, Li L, Byrd JC, et al. Assessment of CD37 B-cell antigen and cell-of-origin significantly improves risk prediction in diffuse large B-cell lymphoma. Blood. 2016;128(26):3083–3100. doi:10.1182/blood-2016-05-715094
12. Lanic H, Mareschal S, Mechken F, et al. Interim positron emission tomography scan associated with international prognostic index and germinal center B cell-like signature as prognostic index in diffuse large B-cell lymphoma. Leuk Lymphoma. 2012;53(1):34–42. doi:10.3109/10428194.2011.600482
13. Barrans SL, Carter I, Owen RG, et al. Germinal center phenotype and bcl-2 expression combined with the International Prognostic Index improves patient risk stratification in diffuse large B-cell lymphoma. Blood. 2002;99(4):1136–1143. doi:10.1182/blood.V99.4.1136
14. Jung S-H, Yang D-H, Ahn J-S, Kim Y-K, Kim H-J, Lee -J-J. Serum lactate dehydrogenase with a systemic inflammation score is useful for predicting response and survival in patients with newly diagnosed diffuse large B-cell lymphoma. Acta Haematol. 2015;133(1):10–17. doi:10.1159/000360068
15. Kim S-H, Go S-I, Seo J, et al. Prognostic impact of pretreatment albumin to globulin ratio in patients with diffuse large B-cell lymphoma treated with R-CHOP. Leuk Res. 2018;71:100–105. doi:10.1016/j.leukres.2018.07.014
16. Gao R, Liang J-H, Wang L, et al. Low serum cholesterol levels predict inferior prognosis and improve NCCN-IPI scoring in diffuse large B cell lymphoma. Int J Cancer. 2018;143(8):1884–1895. doi:10.1002/ijc.31590
17. Caetano SJ, Sonpavde G, Pond GR. C-statistic: a brief explanation of its construction, interpretation and limitations. Eur J Cancer. 2018;90:130–132. doi:10.1016/j.ejca.2017.10.027
18. Vrieze SI. Model selection and psychological theory: a discussion of the differences between the Akaike information criterion (AIC) and the Bayesian information criterion (BIC). Psychol Methods. 2012;17(2):228–243. doi:10.1037/a0027127
19. Steyerberg EW, Vickers AJ, Cook NR, et al. Assessing the performance of prediction models: a framework for traditional and novel measures. Epidemiology. 2010;21(1):128–138. doi:10.1097/EDE.0b013e3181c30fb2
20. Tripepi G, Jager KJ, Dekker FW, Zoccali C. Statistical methods for the assessment of prognostic biomarkers (Part I): discrimination. Nephrol Dial Transplant. 2010;25(5):1399–1401. doi:10.1093/ndt/gfq018
21. Tripepi G, Jager KJ, Dekker FW, Zoccali C. Testing for causality and prognosis: etiological and prognostic models. Kidney Int. 2008;74(12):1512–1515. doi:10.1038/ki.2008.416
22. Uno H, Cai T, Pencina MJ, D’Agostino RB, Wei LJ. On the C-statistics for evaluating overall adequacy of risk prediction procedures with censored survival data. Stat Med. 2011;30(10):1105–1117. doi:10.1002/sim.4154
23. Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6(7):e1000100. doi:10.1371/journal.pmed.1000100
24. Jelicic J, Larsen TS, Maksimovic M, Trajkovic G. Available prognostic models for risk stratification of diffuse large B cell lymphoma patients: a systematic review. Crit Rev Oncol Hematol. 2019;133:1–16. doi:10.1016/j.critrevonc.2018.10.006
25. Chow A, Phillips M, Siew T, et al. Prognostic nomogram for diffuse large B-cell lymphoma incorporating the international prognostic index with interim-positron emission tomography findings. Intern Med J. 2013;43(8):932–939. doi:10.1111/imj.12194
26. Zhong H, Chen J, Cheng S, et al. Prognostic nomogram incorporating inflammatory cytokines for overall survival in patients with aggressive non-Hodgkin’s lymphoma. EBioMedicine. 2019;41:167–174. doi:10.1016/j.ebiom.2019.02.048
27. Han Y, Yang J, Liu P, et al. Prognostic nomogram for overall survival in patients with diffuse large B-cell lymphoma. Oncologist. 2019;24(11):e1251–e1261. doi:10.1634/theoncologist.2018-0361
28. Biccler JL, Eloranta S, de Nully Brown P, et al. Optimizing outcome prediction in diffuse large B-cell lymphoma by use of machine learning and nationwide lymphoma registries: a nordic lymphoma group study. JCO Clin Cancer Informatics. 2018;2(2):1–13. doi:10.1200/CCI.18.00025
29. Go S-I, Park MJ, Song H-N, et al. Prognostic impact of sarcopenia in patients with diffuse large B-cell lymphoma treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. J Cachexia Sarcopenia Muscle. 2016;7(5):567–576. doi:10.1002/jcsm.12115
30. Go S-I, Park S, Kim JH, et al. A new prognostic model using the NCCN-IPI and neutrophil-to-lymphocyte ratio in diffuse large B-cell lymphoma. Tumori J. 2018;104(4):292–299. doi:10.5301/tj.5000694
31. Procházka V, Pytlík R, Janíková A, et al. A new prognostic score for elderly patients with diffuse large B-cell lymphoma treated with R-CHOP: the prognostic role of blood monocyte and lymphocyte counts is absent. PLoS One. 2014;9(7):e102594. doi:10.1371/journal.pone.0102594
32. Gang AO, Pedersen M, d’Amore F, et al. A clinically based prognostic index for diffuse large B-cell lymphoma with a cut-off at 70 years of age significantly improves prognostic stratification: population-based analysis from the danish lymphoma registry. Leuk Lymphoma. 2015;56(9):2556–2562. doi:10.3109/10428194.2015.1010078
33. Keane C, Tobin J, Talaulikar D, et al. A high LDH to absolute lymphocyte count ratio in patients with DLBCL predicts for a poor intratumoral immune response and inferior survival. Oncotarget. 2018;9(34):23620–23627. doi:10.18632/oncotarget.25306
34. Kobayashi T, Kuroda J, Yokota I, et al. The kyoto prognostic index for patients with diffuse large B-cell lymphoma in the rituximab era. Blood Cancer J. 2016;6(1):e383–e383. doi:10.1038/bcj.2015.111
35. Miura K, Konishi J, Miyake T, et al. A host-dependent prognostic model for elderly patients with diffuse large B-cell lymphoma. Oncologist. 2017;22(5):554–560. doi:10.1634/theoncologist.2016-0260
36. Montalbán C, Díaz-López A, Dlouhy I, et al. Validation of the NCCN-IPI for diffuse large B-cell lymphoma (DLBCL): the addition of β 2 -microglobulin yields a more accurate GELTAMO-IPI. Br J Haematol. 2017;176(6):918–928. doi:10.1111/bjh.14489
37. Chen Y, Neelapu S, Feng L, et al. Prognostic significance of baseline peripheral absolute neutrophil, monocyte and serum β2-microglobulin level in patients with diffuse large b-cell lymphoma: a new prognostic model. Br J Haematol. 2016;175(2):290–299. doi:10.1111/bjh.14237
38. Kang J, Yoon S, Suh C. Relevance of prognostic index with β2-microglobulin for patients with diffuse large B-cell lymphoma in the rituximab era. Blood Res. 2017;52(4):276. doi:10.5045/br.2017.52.4.276
39. Antic D, Jelicic J, Trajkovic G, et al. Is it possible to improve prognostic value of NCCN-IPI in patients with diffuse large B cell lymphoma? The prognostic significance of comorbidities. Ann Hematol. 2018;97(2):267–276. doi:10.1007/s00277-017-3170-z
40. Kanemasa Y, Shimoyama T, Sasaki Y, et al. Beta-2 microglobulin as a significant prognostic factor and a new risk model for patients with diffuse large B-cell lymphoma. Hematol Oncol. 2017;35(4):440–446. doi:10.1002/hon.2312
41. Yue W, Liu B, Gao L, et al. The pretreatment albumin to globulin ratio as a significant predictor in patients with diffuse large B cell lymphoma. Clin Chim Acta. 2018;485:316–322. doi:10.1016/j.cca.2018.07.015
42. Wilcox RA, Ristow K, Habermann TM, et al. The absolute monocyte and lymphocyte prognostic score predicts survival and identifies high-risk patients in diffuse large-B-cell lymphoma. Leukemia. 2011;25(9):1502–1509. doi:10.1038/leu.2011.112
43. Ho C-L, Lu C-S, Chen J-H, Chen Y-G, Huang T-C, Wu -Y-Y. Neutrophil/lymphocyte ratio, lymphocyte/monocyte ratio, and absolute lymphocyte count/absolute monocyte count prognostic score in diffuse large B-cell lymphoma. Medicine (Baltimore). 2015;94(24):e993. doi:10.1097/MD.0000000000000993
44. Sun F, Zhu J, Lu S, et al. An inflammation-based cumulative prognostic score system in patients with diffuse large B cell lymphoma in rituximab era. BMC Cancer. 2018;18:5.
45. Zhao P, Zang L, Zhang X, et al. Novel prognostic scoring system for diffuse large B-cell lymphoma. Oncol Lett. 2018;15(4):5325–5332. doi:10.3892/ol.2018.7966
46. Toledano MN, Vera P, Tilly H, Jardin F, Becker S. Comparison of therapeutic evaluation criteria in FDG-PET/CT in patients with diffuse large-cell B-cell lymphoma: prognostic impact of tumor/liver ratio. PLoS One. 2019;14(2):e0211649. doi:10.1371/journal.pone.0211649
47. Li X, Sun X, Li J, et al. Interim PET/CT based on visual and semiquantitative analysis predicts survival in patients with diffuse large B-cell lymphoma. Cancer Med. 2019;8;
48. Chen Z, Deng X, Ye Y, et al. Novel risk stratification of de novo diffuse large B cell lymphoma based on tumour-infiltrating T lymphocytes evaluated by flow cytometry. Ann Hematol. 2019;98:391–399.
49. Aoki K, Tabata S, Yonetani N, Matsushita A, Ishikawa T. The prognostic impact of absolute lymphocyte and monocyte counts at diagnosis of diffuse large B-cell lymphoma in the rituximab era. Acta Haematol. 2013;130(4):242–246. doi:10.1159/000350484
50. Batty N, Ghonimi E, Feng L, et al. The absolute monocyte and lymphocyte prognostic index for patients with diffuse large B-cell lymphoma who receive R-CHOP. Clin Lymphoma Myeloma Leuk. 2013;13(1):15–18. doi:10.1016/j.clml.2012.09.009
51. Ochi Y, Kazuma Y, Hiramoto N, et al. Utility of a simple prognostic stratification based on platelet counts and serum albumin levels in elderly patients with diffuse large B cell lymphoma. Ann Hematol. 2017;96(1):1–8. doi:10.1007/s00277-016-2819-3
52. Nakayama S, Matsuda M, Adachi T, et al. Novel prognostic index based on hemoglobin level and platelet count for diffuse large B-cell lymphoma, not otherwise specified in the R-CHOP era. Platelets. 2019;30:637–645.
53. Conconi A, Zucca E, Roggero E, et al. Prognostic models for diffuse large B-cell lymphoma. Hematol Oncol. 2000;18:61–73.
54. Candelaria M, Reynoso-Noverón N, Ponce M, Castillo-Llanos R, Nolasco-Medina D, Cantú-De-Leon D. A prognostic score for survival in patients older than 65 years with diffuse large B-cell lymphoma. Rev Invest Clin. 2018;70:46–52.
55. Cox MC, Nofroni I, Ruco L, et al. Low absolute lymphocyte count is a poor prognostic factor in diffuse-large-B-cell-lymphoma. Leuk Lymphoma. 2008;49:1745–1751.
56. Kong Y, Qu L, Li Y, Liu D, Lv X, Han J. Predictive significance of a new prognostic score for patients with diffuse large B-cell lymphoma in the interim-positron emission tomography findings. Medicine (Baltimore). 2016;95:e2808.
57. Pardal E, Díez Baeza E, Salas Q, et al. A new prognostic model identifies patients aged 80 years and older with diffuse large B-cell lymphoma who may benefit from curative treatment: a multicenter, retrospective analysis by the Spanish GELTAMO group. Am J Hematol. 2018;93:867–873.
58. Matsumoto K, Fujisawa S, Ando T, et al. Anemia associated with worse outcome in diffuse large B-cell lymphoma patients: a single-center retrospective study. Turkish J Haematol off J Turkish Soc Haematol. 2018;35:181–184.
59. Gao R, Liang J-H, Man T-S, et al. Diabetes mellitus predicts inferior survival in diffuse large B-cell lymphoma: a propensity score-matched analysis. Cancer Manag Res. 2019;11:2849–2870.
60. Nols N, Mounier N, Bouazza S, et al. Quantitative and qualitative analysis of metabolic response at interim positron emission tomography scan combined with International Prognostic Index is highly predictive of outcome in diffuse large B-cell lymphoma. Leuk Lymphoma. 2014;55:773–780.
61. Liu H, Zhang C-L, Feng R, Li J-T, Tian Y, Wang T. Validation and refinement of the age, comorbidities, and albumin index in elderly patients with diffuse large b-cell lymphoma: an effective tool for comprehensive geriatric assessment. Oncologist. 2018;23:722–729.
62. Wang M, Long Q. Addressing issues associated with evaluating prediction models for survival endpoints based on the concordance statistic. Biometrics. 2016;72:897–906.
63. Harrell FE, Califf RM, Pryor DB, Lee KL, Rosati RA. Evaluating the yield of medical tests. JAMA. 1982;247:2543–2546.
64. Akaike H Information theory and an extension of the maximum likelihood principle.
65. Burnham KP, Anderson DR. Multimodel inference: understanding AIC and BIC in model selection. Sociol Methods Res. 2004;33:261–304.
66. Pan W. Akaike’s information criterion in generalized estimating equations. Biometrics. 2001;57:120–125.
67. Hosmer DW Jr LS. Applied Logistic Regression. Hoboken, NJ: John Wiley & Sons, Inc; 2004.
68. Crowson CS, Atkinson EJ, Therneau TM. Assessing calibration of prognostic risk scores. Stat Methods Med Res. 2016;25:1692–1706.
69. Gerds TA, Andersen PK, Kattan MW. Calibration plots for risk prediction models in the presence of competing risks. Stat Med. 2014;33:3191–3203.
70. Hoo ZH, Candlish J, Teare D. What is an ROC curve? Emerg Med J. 2017;34:357–359.
71. Iasonos A, Schrag D, Raj GV, Panageas KS. How to build and interpret a nomogram for cancer prognosis. J Clin Oncol. 2008;26:1364–1370.
72. Shariat SF, Karakiewicz PI, Suardi N, Kattan MW. Comparison of nomograms with other methods for predicting outcomes in prostate cancer: a critical analysis of the literature. Clin Cancer Res. 2008;14:4400–4407.
73. He Y, Ong Y, Li X, et al. Performance of prediction models on survival outcomes of colorectal cancer with surgical resection: a systematic review and meta-analysis. Surg Oncol. 2019;29:196–202.
74. Harkins RA, Chang A, Patel SP, et al. Remaining challenges in predicting patient outcomes for diffuse large B-cell lymphoma. Expert Rev Hematol. 2019;12:959–973.
75. Da Costa CBT. Machine learning provides an accurate classification of diffuse large B-cell lymphoma from immunohistochemical data. J Pathol Inform. 2018;9:21.
76. Shipp MA, Ross KN, Tamayo P, et al. Diffuse large B-cell lymphoma outcome prediction by gene-expression profiling and supervised machine learning. Nat Med. 2002;8:68–74.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.







