Back to Journals » International Journal of General Medicine » Volume 13
Spinal versus General Anesthesia for Patients with Parkinson’s Disease
Authors Bani Hani DA , Aleshawi A J, Al Shalakhti MH, Alhowary A, Al-Jararahih O , Al-Mistarehi AH, Yassin A
Received 28 September 2019
Accepted for publication 16 January 2020
Published 30 January 2020 Volume 2020:13 Pages 9—15
DOI https://doi.org/10.2147/IJGM.S232770
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Diab A Bani Hani, 1 Abdelwahab J Aleshawi, 2 Majd H Al Shalakhti, 2 Alaa’’a Alhowary, 1 Osama Al-Jararahih, 3 Abdel-Hameed Al-Mistarehi, 2 Ahmed Yassin 4
1Department of Anesthesia and Recovery, Faculty of Medicine, Jordan University of Science and Technology, Irbid 21110, Jordan; 2Faculty of Medicine, Jordan University of Science and Technology, Irbid 21110, Jordan; 3Division of Orthopedics, Department of Special Surgery, Faculty of Medicine, Jordan University of Science and Technology, Irbid 22110, Jordan; 4Division of Neurology, Department of Neuroscience, Faculty of Medicine, Jordan University of Science and Technology, Irbid 22110, Jordan
Correspondence: Diab A Bani Hani
Department of Anesthesiology, Faculty of Medicine, Jordan University of Science and Technology, P. O. Box: 3030, Irbid 22110, Jordan
Tel +962798661232
Fax +962 2 7201064
Email [email protected]
Background: Anesthesiologists prefer using general anesthesia (GA) in Parkinson’s disease (PD). However, GA may mask neurological symptoms in the intraoperative period and exacerbate them postoperatively. Furthermore, the anesthetics used in GA have clear interactions with the drugs used to control PD. On the other hand, drugs used in spinal anesthesia (SA) might be safer for patients with PD. The aim of this study is to evaluate the effect of SA and GA in patients with PD who underwent hip fracture repairs.
Methods: Retrospectively, we identified those patients with PD who were admitted due to hip joint fracture. The following information were obtained: demographics, preoperative assessment information of the patients, type of anesthesia, and types of fractures and orthopedic procedures. In addition, intraoperative and postoperative complications were studied. The patients were divided based on the type of anesthesia received and were compared.
Results: Ten (8 males) patients with PD who underwent hip fracture surgery included in the study. Six patients received SA and 4 patients received GA. The mean age was 73.2 years. The preoperative assessment was not significant for all patients. Postoperatively, within the inpatient period, 3 out of 4 patients received GA developed complications (two atelectasis and urinary tract infection) while no patient developed complication from the SA group. Postoperative outpatient complications within one-month included 3 out of 4 cases in the GA group and only one complication in the SA group. The mean hospitalization period was 9 days for patients received GA and 5.8 days for patients received SA.
Conclusion: This study reported less perioperative complications in the SA. Accordingly, further investigations and rp-randomized controlled trials evaluating various anesthetic techniques or drugs are needed.
Keywords: Parkinson’s, spinal anesthesia, postoperative, propofol
Introduction
Parkinson’s disease (PD) is considered one of the most common neurodegenerative diseases, characterized by dopamine deficiency.1 It affects around 1% of individuals over 60 years of age.1 It is a progressive disease that causes physical and mental disabilities.1,2 Furthermore, the progression of the disease cannot be stopped but its symptoms can be managed using mediations. The main pathological findings in PD are loss of pigmented dopaminergic neurons of the substantia nigra pars compacta and the presence of Lewy bodies and Lewy neurites.2,3 As a result of the lack of imbalance of neurotransmitter in caudate nucleus and putamen, patients will complain of excessive saliva, forgetfulness, urinary urgency, hyposmia and constipation.2,3 As well as other physically disabling problems; resting tremor, rigidity, bradykinesia and postural instability with many autonomic dysfunctions that will lead to postural hypotension, dysphagia and diaphragmatic spasms. In addition to that, patients will suffer from severe mental disorders; dementia and depression.3,4
Anesthesiologists prefer using general anesthesia (GA) given that PD is a neurological disease.5 However, GA may mask neurological symptoms in the intraoperative period and exacerbate them postoperatively.5 Furthermore, the anesthetics used in GA have clear interactions with the drugs used to control PD.6–9 On the other hand, drugs used in spinal anesthesia (SA) might be beneficial to the patients with established PD.10 Because of that, we have conducted this study to evaluate the effect of SA on PD in patients who underwent hip fracture repairs.
Methods
This study was conducted at King Abdullah University Hospital, a tertiary care center that is affiliated with the Jordan University of Science and Technology, located in northern Jordan. After obtaining the Institutional Review Board approval, we retrospectively identified those patients with PD who were admitted due to hip joint fracture between January 2015 and December 2018 and were classified as I-III according to the American Society of Anesthesiologists. The following information were obtained: demographics (age at fracture, sex, date of admission), preoperative assessment information of the patients, type of anesthesia, and types of fractures and orthopedic procedures. In addition, intraoperative and postoperative complications were studied and retrieved. These patients had stages 3 and 4 of PD according to the Modified Hoehn and Yahr Scale.
The preoperative assessment information that were retrieved included: duration of PD, co-morbidities, history of falling, chest X-ray findings, echocardiography report, electrocardiography (ECG) report, and history of swallowing dysfunction.
The patients were dichotomized into two groups. The first group comprised patients who received SA and the second group included patients who received GA. The postoperative complications included those complications that developed during the inpatient admission period and complications up to 30 days-postoperatively. Moreover, intensive care unit (ICU) admission was noted.
Anesthetic and Surgical Settings
ECG, non-invasive arterial blood pressure, heart rate, and peripheral blood oxygen saturation (SpO2) levels were all monitored while patients were in the operating theater. Just before the surgery, every patient received the required levodopa. On arrival to the operating theater, two intravenous access sites were secured. For all participants in the study, standard monitoring of blood pressure, three-lead electrocardiogram and oxygen saturation were conducted and continuously monitored during the intraoperative period in the operating theater and during the postoperative period in the post-anesthesia care unit.
SA was administered under aseptic conditions and with local lidocaine 5.9% 3cc, at the level of L3-L4 or L4-L5 in the lateral decubitus position due to the fractures. After assuring clear cerebrospinal fluid, SA was performed with 2.5 cc of 0.5% heavy bupivacaine using 25-gauge spinal needles; 100% O2 was administered through a simple face mask with a flow of 4 L per minute.
For patients receiving GA, after breathing oxygen for 3–5 mins via a face mask, anesthesia was induced with 1.5 μg/kg fentanyl and 1–1.25 mg/kg propofol and 1.2 mg/kg rocuronium to facilitate tracheal intubation, and with rapid sequence intubation using a regular 7.5 mm ID endotracheal tube. Anesthesia was maintained with a propofol infusion at a rate of 0.75–1.25 mg/hour/kg and the inhaled anesthetic agents were discontinued. Endotracheal CO2 (ETCO2) was maintained between 30 mmHg and 40 mmHg throughout the surgery. At the end of the surgery, anesthesia was discontinued, and reversal of the neuromuscular blockade using 2.5 mg of neostigmine and 1 mg of atropine intravenously (IV) was done. Extubation was complete when the patient was able to breathe spontaneously with a good tidal volume, fully awake and could sustain head elevation for more than 5 s.
Upon arrival to the operating theater, both groups received 750 mg of IV cefuroxime, IV 8 mg of dexamethasone, and IV 50 mg of ranitidine.
All the anesthetic procedures were performed by a single anesthesiologist. The orthopedic operations were done by consultant orthopedic surgeons who follow the same surgical guidelines. The diagnosis of PD was conducted by consultant neurologists in the same institution (King Abdullah University Hospital).
Limited statistical analysis tests were used due to the small sample size.
Results
Ten (8 males) patients with PD who underwent hip fracture surgery were identified and included in the study. The mean age was 73.2 years (range: 57–90 years old). The duration of PD ranged from one year to 20 years. Only two patients were not known to have any medical illness at the time of fracture while hypertension was the most common co-morbidity in the rest. However, at the time of admission, all patients achieved normal blood pressure except for one patient who had elevated blood pressure that was later controlled before the surgery. One patient complained of orthostatic hypotension. All patients except for one had a previous history of falling and fractures. The history of swallowing dysfunction and recurrent aspirations was evident in three patients. As a preoperative assessment, the chest X-ray was performed for all patients and revealed clear lungs in all patients. Also, the ECG was done and disclosed only one case of right bundle branch bock. In addition, echocardiography indicated normal ejection fraction in all patients. Table 1 summarizes patients’ characteristics and results of preoperative assessment.
 |
Table 1 Patients Characteristics and Results of Preoperative Assessment |
All patients sustained neck of femur fracture. The left femur was fractured in 7 cases, while the right femur was fractured in 3 cases. Transcervical neck of femur fracture constituted the majority of fractures (6 cases), followed by basocervical neck of femur fracture (3 cases), and only one case of intertrochanteric fracture was included. The SA was conducted in six patients while four patients received GA. No intraoperative complications occurred in any patient. Three out of the 4 patients who received GA developed postoperative inpatient complications (two had atelectasis and were admitted to ICU and one had urinary tract infection). On the other hand, none of the 6 patients who received SA developed any postoperative complication. Regarding postoperative, only one of the 6 patients who received SA developed at one-month post-discharge from the hospital (this is the patient who had swallowing dysfunction at baseline). On the other hand, three out of the 4 patients who received GA developed post-discharge sequalae as summarized in Table 2. Moreover, the mean duration of admission for the SA group was 5.8 days while 9 days for the GA as they required longer duration of monitoring. Furthermore, all patients received GA necessitated more than 24 hrs from admission to operation as a preoperative assessment for fitness for GA while one patient out of 6 received SA needed more than 24 hrs. Finally, Table 3 compares the important points between the SA and GA.
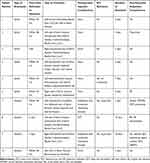 |
Table 2 Operative and Postoperative Complication |
 |
Table 3 Spinal versus General Anesthesia |
Discussion
Upon literature review, we found few case reports were published about the use of SA in patients with PD. Oğuz et al reported a successful SA for female patients with PD who was scheduled for elective surgery for fracture of the left distal tibia.5 In addition, Gautam and Baral published a successful case of SA for laparoscopic cholecystectomy in an elderly patient with PD who had pulmonary dysfunction and was anticipated to have difficult airway management.11 To the best of our knowledge, this is the first observational case series study that compares the effect of SA with GA on patients with PD who underwent hip fracture surgery. Our results revealed that SA poses less risk on such patients compared with GA. This was mainly demonstrated in the postoperative course since both groups had similar preoperative baseline characteristics and no intraoperative complications were detected. Two patients who received the GA complained of postoperative hypoxia and atelectasis that were managed by O2 therapy, chest physiotherapy and close monitoring (patient 7 and 9). After discharge, the respiratory complaints progressed and further investigations revealed aspiration pneumonia, which progressed into multi-organ failure and death (in patient 9). Moreover, the patients who received GA were admitted for longer duration of admission.
Whether they need for hospitalization and surgery is related to PD or not, these patients are at an increased risk for nosocomial morbidity as most of the affected people are above 65 years, and the risk is also augmented by the presence of PD. Swallowing difficulty develops in 50% to 80% of patients during the disease course, leading to increased risk of aspiration and malnutrition.12,13 Accordingly, aspiration pneumonia is considered a leading cause of death in these patients.12 Also, autonomic dysfunction can produce diverse complications, such as orthostatic hypotension, sialorrhea, constipation, incontinence, frequency, excessive sweating, and seborrhea.14 In addition, patients with PD have decreased respiratory function, likely secondary to bradykinesia and insufficient respiratory muscle movement, putting them at greater risk for developing pneumonia.15,16 An obstructive ventilatory pattern has been observed in up to one-third of patients with PD but may also be due to co-existing chronic obstructive pulmonary disease.17,18 Patients with PD also are at an increased risk for falls and urinary tract infections.19,20 Common gastrointestinal symptoms also include loss of appetite, nausea, vomiting, loss of weight, gastric stasis and gastroesophageal reflux.21 In addition, PD can be complicated by neuropsychiatric symptoms like changes in mood (depression), cognition and behavior, delusions and hallucinations. These symptoms with associated tremors and rigidity can lead to postoperative difficult extubation.14,21 Most of these complications get exacerbated by GA.5,10,22,23
Furthermore, the half-life of levodopa is short (one to three hours) and cessation of therapy can result in severe muscle rigidity which interferes with ventilation.23 Moreover, avoidance of drug interactions (between anti-Parkinson and anesthetic drugs) is difficult. The use of arrhythmogenic inhalation anesthetics such as halothane should be avoided as patients who are prone to developing cardiac arrhythmias.22 Even though it has been stated that sevoflurane might be safer, the fact that sevoflurane might cause electrocardiographic alterations that could progress to malignant arrhythmia is a critical point.22,24 Among intravenous anesthetics, the effect of ketamine leading to tachycardia and hypertension should be taken into consideration in patients with cardiac disease.5 Thiopentone decreases dopamine release and PD episodes have been described in patients receiving thiopental.25 Propofol is commonly used in patients with PD and it has anti-parkinsonian effects. It may also aggravate dyskinesias (a common side effect of levodopa) which may interfere with the procedure.26,27 When treating perioperative pain, opioids, particularly fentanyl, may aggravate rigidity and thus better be avoided in larger doses.26
Regional anesthesia (as spinal) has obvious advantages over GA. First, in regional anesthesia we can avoid the effects of general anesthetics and neuromuscular blocking drugs, which may mask tremor. Second, patients can continue to take oral levodopa preoperatively, during surgery, if required, and early in the postoperative period. Third, with regional anesthesia, postoperative nausea and vomiting are also avoided. Fourth and most important, the probability of aspiration pneumonia, respiratory weakness, urinary tract infection, deep vein thrombosis, postural hypotension, and psychiatric flares are avoided and minimized with regional anesthesia. If sedation is required, diphenhydramine may be utilized due to its central anticholinergic activity.22
It is worth to mention that Patients with PD are more likely to have a lower bone mineral density than the general population and are more prone to falls and hip fractures.28–30 Short- and long-term outcomes in patients with PD following hip fracture are generally considered to be worse than patients without PD. One study by Mueller et al, they retrospectively compared 51 patients with PD with 51 controls over 13 years. They found that postoperative falls were more common (P < 0.03), and discharge to home occurred less often secondary to a need for ambulatory rehabilitation (P < 0.03) compared with controls.19 Eventov et al revealed that patients with PD undergoing hip fracture surgery had double the 3-month mortality rate of that for non-Parkinson’s patients.31 GA can make such complications more frequent compared with the SA due to the aforementioned factors.
On the other hand, with regional anesthesia positioning the patient may be difficult, tremor can interfere with some monitoring devices and makes it more difficult to interpret, and if the surgery is careful and accurate, the surgeon may want the patient to be absolutely still. Also, under regional anesthesia, the patients are awake, so, special attention should be given to verify that baseline pharyngeal reflexes are intact and allow for communication of the subjective feelings accompanying PD attacks, thereby prompting earlier treatment. Because of the aforementioned reasons, it is preferred by the most anesthesiologists to utilize the GA instead of SA. Also, this is why a few cases were reported in the literature regarding the successful use of the SA.
In this study, even with proper preoperative assessment and optimization, patients who received GA developed several complications. This was not the case with SA.
Our study is not without limitations. It is a retrospective study that depends on the documented material, not a controlled prospective trial. Also, the sample size is not sufficient to build up a clinical and practical recommendation. A lot of the postoperative complications could not be precisely attributed to the GA effect as those patients had multiple comorbidities and had poly-pharmacy.
Conclusion
As patients with PD suffer from recurrent hip fractures and in light of the co-morbidities and complications related to the disease and its medications, it is important to find a safe and appropriate anesthetic procedure. Our study reported less perioperative complications in the SA. However, this is not conclusive and randomized controlled trials evaluating various anesthetic techniques or drugs are needed.
Abbreviations
PD, Parkinson’s disease; GA, general anesthesia; SA, spinal anesthesia; ECG, electrocardiography; ICU, intensive care unit; IV, intravenously.
Ethics Approval and Consent to Participate
This study was reviewed and approved by the Institutional Board Review of King Abdullah University Hospital and Jordan University of Science and Technology. This study was conducted in accordance with the Declaration of Helsinki. The consent was waived due to the retrospective nature of the study. We confirm that the privacy of the participants was saved, and the data were anonymized and maintained with confidentiality.
Data Sharing Statement
The datasets generated and analyzed during the current study are available from the corresponding author.
Disclosure
The authors declare that they have no competing interests in this work.
References
1. Wirdefeldt K, Adami HO, Cole P, Trichopoulos D, Mandel J. Epidemiology and etiology of Parkinson’s disease: a review of the evidence. Eur J Epidemiol. 2011;26(Suppl 1):S1–58.
2. Moghal S, Rajput AH, D’Arcy C, Rajput R. Prevalence of movement disorders in elderly community residents. Neuroepidemiology. 1994;13:175–178. doi:10.1159/000110376
3. Lang AE, Lozano AM. Parkinson’s disease: second of two parts. N Engl J Med. 1998;339:1130–1143. doi:10.1056/NEJM199810153391607
4. Khoo TK, Yarnall AJ, Duncan GW, et al. The spectrum of nonmotor symptoms in early Parkinson disease. Neurology. 2013;80:276–281. doi:10.1212/WNL.0b013e31827deb74
5. Oğuz E, Öztürk İ, Özkan D, Ergil J, Aydın GB. Parkinson’s disease and spinal anaesthesia. Turk J Anaesthesiol Reanim. 2014;42:280–282. doi:10.5152/TJAR.2014.47135
6. Christian CM, Waller JL, Moldenhauer CC. Postoperative rigidity following fentanyl anesthesia. Anesthesiology. 1983;58:275–277. doi:10.1097/00000542-198303000-00015
7. Krauss JK, Akeyson EW, Giam P, Jankovic J. Propofol-induced dyskinesias in Parkinson’s disease. Anesth Analg. 1996;83:420–422. doi:10.1097/00000539-199608000-00037
8. Zesiewicz TA, Hauser RA, Freeman A, Sullivan KL, Miller AM, Halim T. Fentanyl-induced bradykinesia and rigidity after deep brain stimulation in a patient with Parkinson disease. Clin Neuropharmacol. 2009;32:48–50. doi:10.1097/WNF.0b013e31817e23e3
9. Zornberg GL, Bodkin JA, Cohen BM. Severe adverse interaction between pethidine and selegiline. Lancet. 1991;337:246. doi:10.1016/0140-6736(91)92219-R
10. Shaikh SI, Verma H. Parkinson’s disease and anaesthesia. Indian J Anaesth. 2011;55:228–234. doi:10.4103/0019-5049.82658
11. Gautam B, Baral B. Spinal anaesthesia for laparoscopic cholecystectomy in Parkinson’s disease. JNMA J Nepal Med Assoc. 2018;56:701–704. doi:10.31729/jnma.3611
12. Mu L, Sobotka S, Chen J, et al. Altered pharyngeal muscles in Parkinson disease. J Neuropathol Exp Neurol. 2012;71:520–530. doi:10.1097/NEN.0b013e318258381b
13. Pfeiffer RF. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 2003;2:107–116. doi:10.1016/S1474-4422(03)00307-7
14. DeLong MR, Juncos JL. Parkinson’s disease and other movement disorders. In: Kasper DL, Fauci AS, Longo DL, Braunwald E, Hauser SL, Jameson JL, editors. Harrison’s Principles of Internal Medicine.
15. Monteiro L, Souza-Machado A, Valderramas S, Melo A. The effect of levodopa on pulmonary function in Parkinson’s disease: a systematic review and meta-analysis. Clin Ther. 2012;34:1049–1055. doi:10.1016/j.clinthera.2012.03.001
16. Galvez-Jimenez N, Lang AE. The perioperative management of Parkinson’s disease revisited. Neurol Clin. 2004;22:367–377. doi:10.1016/j.ncl.2004.01.004
17. Neu HC, Connolly JJ, Schwertley FW, Ladwig HA, Brody AW. Obstructive respiratory dysfunction in parkinsonian patients. Am Rev Respir Dis. 1967;95:33–47. doi:10.1164/arrd.1967.95.1.33
18. Obenour WH, Stevens PM, Cohen AA, McCutchen JJ. The causes of abnormal pulmonary function in Parkinson’s disease. Am Rev Respir Dis. 1972;105:382–387. doi:10.1164/arrd.1972.105.3.382
19. Mueller MC, Juptner U, Wuellner U, et al. Parkinson’s disease influences the perioperative risk profile in surgery. Langenbecks Arch Surg. 2009;394:511–515. doi:10.1007/s00423-008-0404-5
20. Pepper PV, Goldstein MK. Postoperative complications in Parkinson’s disease. J Am Geriatr Soc. 1999;47:967–972. doi:10.1111/jgs.1999.47.issue-8
21. Roizen MF, Fleisher LA. Anesthetic implications of concurrent diseases. In: RD M, editor. Miller’s Anaesthesia.
22. Nicholson G
23. Rudra A, Rudra P, Chatterjee S, Das T, Ray M, Kumar P. Parkinson’s disease and anaesthesia. Indian J Anaesth. 2007;51:382.
24. Kleinsasser A, Kuenszberg E, Loeckinger A, et al. Sevoflurane, but not propofol, significantly prolongs the Q-T interval. Anesth Analg. 2000;90:25–27. doi:10.1097/00000539-200001000-00006
25. Muravchick S, Smith DS. Parkinsonian symptoms during emergence from general anaesthesia. Anesthesiology. 1995;82:305–307. doi:10.1097/00000542-199501000-00039
26. Burton DA, Nicholson G, Hall GM. Anaesthesia in elderly patients with neurodegenerative disorders: special considerations. Drugs Aging. 2004;21:229–242. doi:10.2165/00002512-200421040-00002
27. Brennan KA, Genever RW. Managing Parkinson’s disease during surgery. BMJ. 2010;341:c5718. doi:10.1136/bmj.c5718
28. Abou-Raya S, Helmii M, Abou-Raya A. Bone and mineral metabolism in older adults with Parkinson’s disease. Age Ageing. 2009;38:675–680. doi:10.1093/ageing/afp137
29. Yiannopoulou KG, Papageorgiou SG, Anastasiou IP, et al. Prevalence of parkinsonism in older patients with hip fracture. Hip Int. 2011;21:351–355. doi:10.5301/hip.2011.8402
30. Invernizzi M, Carda S, Viscontini GS, et al. Osteoporosis in Parkinson’s disease. Parkinsonism Relat Disord. 2009;15:339–346. doi:10.1016/j.parkreldis.2009.02.009
31. Eventov I, Moreno M, Geller E, Tardiman R, Salama R. Hip fractures in patients with Parkinson’s syndrome. J Trauma. 1983;23:98–101. doi:10.1097/00005373-198302000-00004
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
