Back to Journals » Journal of Pain Research » Volume 13
Spinal N-Cadherin/CREB Signaling Contributes to Chronic Alcohol Consumption-Enhanced Postsurgical Pain
Authors Ma Y, Zhang X, Li C , Liu S, Xing Y, Tao F
Received 15 June 2020
Accepted for publication 31 July 2020
Published 12 August 2020 Volume 2020:13 Pages 2065—2072
DOI https://doi.org/10.2147/JPR.S267778
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Robert B. Raffa
Yajing Ma,1,2 Xinye Zhang,1 Changsheng Li,3 Sufang Liu,1,2 Ying Xing,1 Feng Tao2
1Department of Physiology and Neurobiology, Zhengzhou University School of Basic Medical Sciences, Zhengzhou, Henan, People’s Republic of China; 2Department of Biomedical Sciences, Texas A&M University College of Dentistry, Dallas, Texas, USA; 3Department of Anesthesiology, Zhengzhou University School of Medicine, Zhengzhou, People’s Republic of China
Correspondence: Ying Xing
Department of Physiology and Neurobiology, Zhengzhou University School of Basic Medical Sciences, 100 Ke Xue Ave, Zhengzhou, Henan 450001, People’s Republic of China
Tel +86-371-67780665
Email [email protected]
Feng Tao
Department of Biomedical Sciences, Texas A&M University College of Dentistry, 3302 Gaston Ave, Dallas, TX 75246, USA
Tel +1-214-828-8272
Email [email protected]
Background: It has been reported that N-cadherin and cAMP response element binding protein (CREB) in the spinal cord are critical for synaptogenesis and regulation of excitatory synapse function, which could underlie chronic pain development. The aim of the present study was to investigate the role of spinal N-cadherin/CREB signaling in postsurgical pain chronicity following chronic alcohol consumption.
Methods: C57BL/6 male mice were randomly assigned into different groups. Plantar incision was used to induce postsurgical pain. Chronic alcohol consumption was conducted by giving mice unlimited access to different concentrations of ethanol for five weeks. We measured paw withdrawal thresholds to test postsurgical pain. Using Western blotting, we examined the expression of N-Cadherin and CREB in the spinal dorsal horn. We further performed intrathecal injection of specific N-cadherin and CREB inhibitors to assess the role of spinal N-cadherin/CREB signaling in chronic alcohol consumption-enhanced postsurgical pain.
Results: We observed that the chronic alcohol consumption significantly prolonged postsurgical pain and enhanced plantar incision-increased N-cadherin expression and CREB phosphorylation at the Ser133 in the spinal cord. Intrathecal injection of specific N-cadherin and CREB inhibitors attenuated chronic alcohol consumption-prolonged postsurgical pain.
Conclusion: Our results suggest that spinal N-cadherin/CREB signaling is involved in chronic alcohol consumption-caused postsurgical pain chronicity.
Keywords: incisional pain, spinal cord, intrathecal injection, ethanol
Introduction
Previous studies have shown that moderate alcohol consumption can reduce pain in chronic pain patients,1 but heavy alcohol drinking and alcohol dependence are associated with chronic pain development.2 Moreover, clinical and preclinical studies have reported that excessive chronic alcohol exposure and withdrawal can increase sensitivity to noxious stimuli.3–5 Approximately one-fifth of people with alcohol abuse or dependence are accompanied by prolonged pain.6 However, the underlying mechanisms for the association between chronic alcohol consumption and pain chronicity are not fully understood.
N-cadherin, a trans-synaptic cell adhesion molecule, is well known to be expressed at glutamatergic synapses.7 This molecule not only plays a crucial role in synapse formation, but also controls excitatory synapse function and stabilizes dendritic spine structures.8–11 It has been demonstrated that synaptic plasticity of primary afferent nociceptive circuitry in the spinal dorsal horn is tightly related to mechanical allodynia.12,13 On the other hand, α-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid (AMPA) receptors are ionotropic glutamate receptors that mediate fast synaptic potential.14 It has been reported that hyperalgesia and allodynia are associated with AMPA receptor-mediated excitatory synaptic transmission.15 Our previous study showed that chronic alcohol consumption prolongs postsurgical pain by enhancing AMPA receptor phosphorylation in the spinal cord.16 N-cadherin can interact with AMPA receptor GluA2 and result in decreased desensitization of the AMPA receptor, thereby facilitating long-term potentiation (LTP) induction.17 Moreover, cAMP response element binding protein (CREB), which can be activated by N-cadherin, is involved in the induction and maintenance of LTP18 and contributes to pain modulation in the spinal cord during the transition from acute to chronic pain.19 Therefore, these previous studies suggest that spinal N-cadherin/CREB signaling could play a role in chronic alcohol consumption-enhanced postsurgical pain.
In the present study, we analyzed N-cadherin expression and CREB phosphorylation in the spinal cord after chronic alcohol consumption and/or plantar incision, and further examined the effects of spinal N-cadherin and CREB inhibition on chronic alcohol consumption-prolonged postsurgical pain.
Materials and Methods
Animals
C57BL/6 male mice (20–25g) were obtained from Animal Center of Zhengzhou University. Mice were housed under standard conditions with 12 h light/dark cycle and allowed access to water and food ad libitum. All behavioral tests were performed by an investigator blinded to the assignment of animal groups, and mice were habituated 30 min per day for three days before behavioral testing. All efforts were made to minimize pain or discomfort and to reduce the number of animals used. All procedures were carried out in accordance with the National Institutes of Health Guide for Care and Use of Laboratory Animals and were approved by the Animal Care and Use Committee at Zhengzhou University. The number of animals for each experiment is indicated in the respective figure legend.
Chronic Alcohol Consumption and Plantar Incision
Chronic alcohol consumption was conducted as described in our previous study with minor modification.16 Briefly, mice received 10% (w/v) ethanol for first two days, 15% (w/v) ethanol for next five days, and 20% (w/v) ethanol for the following four weeks. The mice in the non-alcohol drinking group had free access to water. The consumed amount of ethanol per day was similar in all the mice (4.11 ± 0.05). All ethanol-drinking mice gained weight normally compared with the non-alcohol drinking group.
On the day following five-week ethanol drinking, a 5-mm longitudinal plantar incision was made through the skin and fascia in the left hindpaw under isoflurane anesthesia according to previous studies.20,21 The incision began 2 mm from the proximal edge of the heel and extended toward the toes. The underlying muscle was elevated with a curved forceps, leaving the muscle origin and insertion intact. The skin was apposed with a single suture of 6–0 nylon, and the wound site was covered with antibiotic ointment to prevent infection. The sham-operated mice underwent the same procedure except that the incision was not carried out. All the mice did not show locomotor dysfunction after ethanol drinking and/or plantar incision.
Assessment of Paw Mechanical Hypersensitivity
Pain behaviors were measured one day before incision as baseline and days 1, 3, 5, 7, 10, 20, 30, 40, 50, 60, and 70 after incision. The calibrated von Frey filaments were used to assess mechanical hypersensitivity according to our previous study.20 In brief, mice were placed on an elevated stainless wire mesh floor and were covered with a clear Plexiglas chamber (5 × 5 × 8 cm). Following acclimation for 30 min, the filaments were applied to paws from least to greatest forces (0.08, 0.15, 0.25, 0.41, 0.7, 1.2, and 2.0 g) to determine the paw withdrawal responses to mechanical stimuli. Each monofilament was applied five times to the plantar side of the hindpaw for approximately 1–2 s with a 10 s interval, starting with a lowest force of filament (0.08 g) and continuing in ascending order. A positive response was defined as a sharp withdrawal of hindpaw upon stimulation. The paw withdrawal threshold was calculated as the force at which the positive response occurred in three out of five stimuli.
Drugs Preparation and Intrathecal Injection
The CREB inhibitor KG-501 (Cat. # 70485, Sigma, USA) and N-cadherin inhibitor ADH-1 (Cat. # HY-13541, MedChemExpress, USA) were dissolved in dimethyl sulfoxide (DMSO) and diluted with 0.9% saline. Intrathecal injection of the drugs in a volume of 6 µL was performed using a 10 µL Hamilton microsyringe with a 27-gauge needle on day 20 after plantar incision as described previously.22,23 The accurate placement of the needle was confirmed by a quick “flick” of the mouse’s tail. Pain behavior testing was carried out before and 30 min after the intrathecal injection.
Western Blotting
The mice were sacrificed under isoflurane anesthesia on day 20 after incision and/or alcohol treatment and the ipsilateral L4-L6 lumbar spinal cord tissues were harvested. Proteins from the lumbar spinal cord tissues were extracted as described previously.20,24 Nuclear fraction of the extracted protein was used for detecting CREB and its phosphorylation.24 β-actin and histone H3 were used as loading controls for N-cadherin and CREB, respectively. Protein concentration was determined using the bicinchoninic acid method. The following affinity-purified antibodies were used: anti-N-cadherin (1:1000, Cat. # ab76057, Abcam, USA), anti-CREB (1:1000, Cat. # ab32515, Abcam, USA), anti-phospho-CREB-Ser133 (1:5000, Cat. # ab32096, Abcam, USA), anti-β-actin (1:200000, Cat. # A5316, Sigma, USA), and anti-histone H3 (1:3000, Cat. # 17168-1-AP, Proteintech, USA). The intensities of bands were quantified with densitometry using Image J software (NIH, USA). The intensity values of N-cadherin bands were normalized with β-actin and expressed as a ratio of N-cadherin/β-actin, and the intensity values of the phospho-CREB-Ser133 (p-CREB) were normalized with total CREB and expressed as a ratio of p-CREB/CREB. The specificity of anti-N-cadherin, anti-CREB anti-phospho-CREB-Ser133 antibodies has been validated previously.25–27
Statistical Analysis
Data are expressed as the mean ± SEM. All statistical analyses were performed using GraphPad Prism 8.0.1. Behavioral data was analyzed by two-way analysis of variance (ANOVA) with repeated measures. Western blotting data were analyzed by one-way ANOVA. All ANOVAs were followed by the Tukey’s post hoc test. The level of significance was set at p < 0.05.
Results
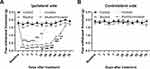
Chronic Alcohol Consumption Prolongs Plantar Incision-Induced Postsurgical Pain
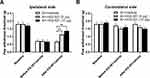
In the plantar incision alone group, paw withdrawal thresholds on the ipsilateral side were significantly decreased from day 1 to day 10 following the incision and did not returned to basal level until day 20 post-incision (Figure 1A). We further observed that 5-week ethanol consumption markedly extended the plantar incision-decreased paw withdrawal thresholds on the ipsilateral side (Figure 1A), though the alcohol drinking alone did not significantly alter paw withdrawal thresholds (Figure 1A). We also observed that paw withdrawal thresholds on the contralateral side were not affected by plantar incision and/or 5-week ethanol consumption (Figure 1B).
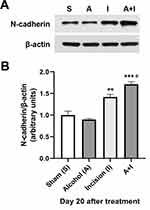
Chronic Alcohol Consumption Enhances Plantar Incision-Increased the Expression of N-Cadherin in the Spinal Cord
To investigate the involvement of spinal N-cadherin in chronic alcohol consumption-prolonged postsurgical pain, we analyzed the expression of N-cadherin in the ipsilateral L4-L6 lumbar spinal cord on day 20 after plantar incision and/or 5-week ethanol consumption. Using quantitative Western blotting, we found that plantar incision increased the expression of N-cadherin in the spinal cord compared to the sham control group (Figure 2), and that 5-week ethanol consumption further enhanced the plantar incision-increased spinal N-cadherin expression (Figure 2), though the alcohol drinking alone had no effect on the N-cadherin expression in the spinal cord (Figure 2).
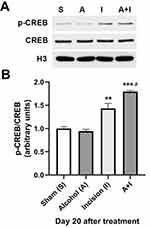
Chronic Alcohol Consumption Enhances Plantar Incision-Increased CREB Phosphorylation in the Spinal Cord
To reveal whether N-cadherin-activated CREB signaling is involved in chronic alcohol consumption-prolonged postsurgical pain, we examined CREB phosphorylation in the ipsilateral L4-L6 lumbar spinal cord on day 20 after plantar incision and/or 5-week ethanol consumption. Using quantitative Western blotting, we found that plantar incision increased the level of phosphorylated CREB at the Serine 133 in the spinal cord compared to the sham control group (Figure 3), and that 5-week ethanol consumption further enhanced the plantar incision-increased spinal CREB phosphorylation (Figure 3), though the alcohol drinking alone had no effect on the CREB phosphorylation in the spinal cord (Figure 3). We also observed that total CREB expression did not show significant change after plantar incision and/or chronic alcohol consumption (Figure 3).
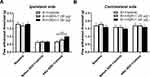
Inhibition of Spinal N-Cadherin/CREB Signaling Attenuates Chronic Alcohol Consumption-Produced Prolongation of Postsurgical Pain
To further determine whether spinal N-cadherin/CREB signaling contributes to the effect of chronic alcohol consumption on plantar incision-induced postsurgical pain, we performed intrathecal injection of ADH-1 (a specific N-cadherin inhibitor) and KG501 (a specific CREB inhibitor) on day 20 after plantar incision and 5-week ethanol consumption. We observed that treatment with 40 μg of ADH-1 significantly increased the decreased paw withdrawal threshold on the ipsilateral side compared to the vehicle control group (Figure 4A) but had no effect on the contralateral paw withdrawal threshold (Figure 4B). We also observed that treatment with KG501 dose-dependently increased the decreased paw withdrawal threshold on the ipsilateral side compared to the vehicle control group (Figure 5A) but did not alter the contralateral paw withdrawal threshold (Figure 5B).
Discussion
In the present study, we investigated the role of spinal N-cadherin/CREB signaling in chronic alcohol consumption-prolonged postsurgical pain. Our results showed that chronic alcohol consumption enhances plantar incision-increased N-cadherin expression and CREB phosphorylation in the spinal cord and that inhibition of spinal N-cadherin or CREB attenuates chronic alcohol consumption-produced prolongation of postsurgical pain. These results suggest that spinal N-cadherin/CREB signaling contributes to the effect of chronic alcohol consumption on plantar incision-induced postsurgical pain.
Our previous studies have demonstrated that AMPA receptor phosphorylation is critical for postsurgical pain chronicity caused by stress or chronic alcohol consumption.16,20 AMPA receptors include four subunits: GluA1–4.28–30 The extracellular N-terminal domain of GluA2 can directly interact with the extracellular domain of N-cadherin, which is a neuron-specific cell adhesion molecule involved in the regulation of synaptic plasticity and many neurological diseases.11,17,31,32 Thus, the interaction between N-cadherin and AMPA receptor GluA2 could be involved in chronic alcohol consumption-enhanced postsurgical pain. It has been reported that the association of N-cadherin with GluA2 serves to stabilize AMPA receptors at synapses as immobilized N-cadherin can decrease lateral diffusion of GluA2.11 In addition, proteomic analysis showed that N-cadherin physically associates with N-methyl-d-aspartic acid (NMDA) receptors in large multi-protein complexes.33 Thus, by coupling to both NMDA receptor and AMPA receptor signaling pathways, N-cadherin in the spinal cord could play an important role in the prolongation of postsurgical pain following chronic alcohol consumption. In this study, we observed that 5-week ethanol treatment further upregulated the expression of spinal N-cadherin after plantar incision and that intrathecal injection of specific N-cadherin inhibitor attenuated chronic alcohol consumption-enhanced postsurgical pain. Our results indicate that spinal N-cadherin contributes to the central mechanism by which chronic alcohol consumption prolongs postsurgical pain. Here, a significant but modest effect produced by the specific N-cadherin inhibitor suggests that besides N-cadherin signaling, other mechanisms may be also involved in chronic alcohol consumption-caused acute-to-chronic pain transition after surgery. Intrathecal injection of this inhibitor in a higher dose could increase the inhibitory effect on such pain chronicity.
Moreover, N-cadherin can interact with CREB signaling molecules to lead to transcriptional regulation.34 CREB-dependent gene expression is critical for the function and plasticity of the nervous system including long-term memory and learning in both vertebrates and invertebrates.35,36 Stimulation of CREB-mediated transcription requires its phosphorylation at the Ser133 site.37–39 It is highly likely that CREB phosphorylation is involved in the modulation of pain-mediating neuropeptide expression, including substance P and calcitonin gene-related peptide, in the dorsal root ganglion neurons.40 In this study, we observed that 5-week ethanol treatment further enhanced the phosphorylation of CREB at the Ser133 site after plantar incision and that intrathecal injection of specific CREB inhibitor attenuated chronic alcohol consumption-enhanced postsurgical pain. Our results suggest that spinal CREB and its phosphorylation participate in the central mechanism that underlies chronic alcohol consumption-produced postsurgical pain prolongation. Taken together, our current study provides experimental evidence to show that N-cadherin/CREB signaling in the spinal cord is involved in chronic alcohol consumption-enhanced postsurgical pain, thus we may target this signaling to develop a new therapy for such pain.
Conclusions
Our data suggest that spinal N-cadherin/CREB signaling plays an important role in chronic alcohol consumption-produced postsurgical pain prolongation and that inhibition of this signaling can be used to treat postsurgical pain chronicity.
Abbreviations
CREB, cAMP response element binding protein; AMPA, α-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid; ANOVA, analysis of variance; NMDA, N-methyl-d-aspartic acid; LTP, long-term potentiation.
Data Sharing Statement
The datasets used and analyzed during the current study are available from the corresponding authors on reasonable request.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed on the journal to which the article will be submitted; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Scott JR, Hassett AL, Schrepf AD, et al. Moderate alcohol consumption is associated with reduced pain and fibromyalgia symptoms in chronic pain patients. Pain Med. 2018;19(12):2515–2527. doi:10.1093/pm/pny032
2. Egli M, Koob GF, Edwards S. Alcohol dependence as a chronic pain disorder. Neurosci Biobeh Rev. 2012;36(10):2179–2192. doi:10.1016/j.neubiorev.2012.07.010
3. Stewart SH, Finn PR, Pihl ROJP. A dose-response study of the effects of alcohol on the perceptions of pain and discomfort due to electric shock in men at high familial-genetic risk for alcoholism. Psychopharmacology. 1995;119(3):261–267.
4. Gatch MBJAC, Research E. Effects of benzodiazepines on acute and chronic ethanol‐induced nociception in rats. Alcoholism. 1999;23(11):1736–1743.
5. Edwards S, Vendruscolo LF, Schlosburg JE, et al. Development of mechanical hypersensitivity in rats during heroin and ethanol dependence: alleviation by CRF1 receptor antagonism. Neuropharmacology. 2012;62(2):1142–1151.
6. Smith ML, Li J, Ryabinin AE. Increased alcohol consumption in urocortin 3 knockout mice is unaffected by chronic inflammatory pain. Alcohol and Alcoholism. 2014;50(2):132–139. doi:10.1093/alcalc/agu084
7. Pielarski KN, van Stegen B, Andreyeva A, et al. Asymmetric N-cadherin expression results in synapse dysfunction, synapse elimination, and axon retraction in cultured mouse neurons. PLoS One. 2013;8:1.
8. Zhou Z, Hu J, Passafaro M, Xie W, Jia Z. GluA2 (GluR2) regulates metabotropic glutamate receptor-dependent long-term depression through N-cadherin-dependent and cofilin-mediated actin reorganization. J Neurosci. 2011;31(3):819–833. doi:10.1523/JNEUROSCI.3869-10.2011
9. Takeichi M, Abe K. Synaptic contact dynamics controlled by cadherin and catenins. Trends Cell Biol. 2005;15(4):216–221. doi:10.1016/j.tcb.2005.02.002
10. Brusés JL. N-cadherin signaling in synapse formation and neuronal physiology. Mol Neurobiol. 2006;33(3):237–252. doi:10.1385/MN:33:3:237
11. Saglietti L, Dequidt C, Kamieniarz K, et al. Extracellular interactions between GluR2 and N-cadherin in spine regulation. Neuron. 2007;54(3):461–477. doi:10.1016/j.neuron.2007.04.012
12. Tan AM, Stamboulian S, Chang Y-W, et al. Neuropathic pain memory is maintained by Rac1-regulated dendritic spine remodeling after spinal cord injury. J Neurosci. 2008;28(49):13173–13183. doi:10.1523/JNEUROSCI.3142-08.2008
13. Hu J, Mata M, Hao S, Zhang G, Fink DJ. Central sprouting of uninjured small fiber afferents in the adult rat spinal cord following spinal nerve ligation. Eur J Neurosci. 2004;20(7):1705–1712. doi:10.1111/j.1460-9568.2004.03652.x
14. Platt SR. The role of glutamate in central nervous system health and disease–a review. Vet J. 2007;173(2):278–286. doi:10.1016/j.tvjl.2005.11.007
15. Kopach O, Viatchenko-Karpinski V, Belan P, Voitenko N. Development of inflammation-induced hyperalgesia and allodynia is associated with the upregulation of extrasynaptic AMPA receptors in tonically firing lamina II dorsal horn neurons. Front Physiol. 2012;3:391. doi:10.3389/fphys.2012.00391
16. Liu S, Zhao Z, Guo Y, et al. Spinal AMPA receptor GluA1 Ser831 phosphorylation controls chronic alcohol consumption-produced prolongation of postsurgical pain. Mol Neurobiol. 2018;55(5):4090–4097. doi:10.1007/s12035-017-0639-7
17. Bozdagi O, Shan W, Tanaka H, Benson DL, Huntley GW. Increasing numbers of synaptic puncta during late-phase LTP: N-cadherin is synthesized, recruited to synaptic sites, and required for potentiation. Neuron. 2000;28(1):245–259. doi:10.1016/S0896-6273(00)00100-8
18. Wu H, Zhou Y, Xiong ZQ. Transducer of regulated CREB and late phase long‐term synaptic potentiation. FEBS J. 2007;274(13):3218–3223. doi:10.1111/j.1742-4658.2007.05891.x
19. Ferrari LF, Bogen O, Reichling DB, Levine JD. Accounting for the delay in the transition from acute to chronic pain: axonal and nuclear mechanisms. J Neurosci. 2015;35(2):495–507. doi:10.1523/JNEUROSCI.5147-13.2015
20. Li C, Yang Y, Liu S, et al. Stress induces pain transition by potentiation of AMPA receptor phosphorylation. J Neurosci. 2014;34(41):13737–13746. doi:10.1523/JNEUROSCI.2130-14.2014
21. Pogatzki EM, Raja SN. A mouse model of incisional pain. Anesthesiology. 2003;99(4):1023–1027. doi:10.1097/00000542-200310000-00041
22. Tao F, Skinner J, Yang Y, Johns RA. Effect of PSD-95/SAP90 and/or PSD-93/chapsyn-110 deficiency on the minimum alveolar anesthetic concentration of halothane in mice. Anesthesiology. 2010;112(6):1444–1451. doi:10.1097/ALN.0b013e3181dcd3dc
23. Tao F, Su Q, Johns RA. Cell-permeable peptide Tat-PSD-95 PDZ2 inhibits chronic inflammatory pain behaviors in mice. Mol Ther. 2008;16(11):1776–1782. doi:10.1038/mt.2008.192
24. Li -D-D, Xie H, Du Y-F, et al. Antidepressant-like effect of zileuton is accompanied by hippocampal neuroinflammation reduction and CREB/BDNF upregulation in lipopolysaccharide-challenged mice. J affect dis. 2018;227:672–680.
25. Abdalla Z, Walsh T, Thakker N, Ward CM. Loss of epithelial markers is an early event in oral dysplasia and is observed within the safety margin of dysplastic and T1 OSCC biopsies. PLoS One. 2017;12(12):e0187449. doi:10.1371/journal.pone.0187449
26. Yu L, Guo X, Zhang P, Qi R, Li Z, Zhang S. Cyclic adenosine monophosphate-responsive element-binding protein activation predicts an unfavorable prognosis in patients with hepatocellular carcinoma. Onco Targets Ther. 2014;7:873–879. doi:10.2147/OTT.S63594
27. Mindos T, Dun XP, North K, et al. Merlin controls the repair capacity of Schwann cells after injury by regulating Hippo/YAP activity. J Cell Biol. 2017;216(2):495–510. doi:10.1083/jcb.201606052
28. Collingridge GL, Olsen RW, Peters J, Spedding M. A nomenclature for ligand-gated ion channels. Neuropharmacology. 2009;56(1):2–5. doi:10.1016/j.neuropharm.2008.06.063
29. Hollmann M, Heinemann S. Cloned glutamate receptors. Annu Rev Neurosci. 1994;17(1):31–108. doi:10.1146/annurev.ne.17.030194.000335
30. Lu W, Shi Y, Jackson AC, et al. Subunit composition of synaptic AMPA receptors revealed by a single-cell genetic approach. Neuron. 2009;62(2):254–268. doi:10.1016/j.neuron.2009.02.027
31. Arikkath J, Reichardt LF. Cadherins and catenins at synapses: roles in synaptogenesis and synaptic plasticity. Trends Neurosci. 2008;31(9):487–494. doi:10.1016/j.tins.2008.07.001
32. Tai CY, Kim SA, Schuman EM. Cadherins and synaptic plasticity. Curr Opin Cell Biol. 2008;20(5):567–575. doi:10.1016/j.ceb.2008.06.003
33. Husi H, Ward MA, Choudhary JS, Blackstock WP, Grant SG. Proteomic analysis of NMDA receptor-adhesion protein signaling complexes. Nat Neurosci. 2000;3(7):661–669. doi:10.1038/76615
34. McCusker CD, Alfandari D. Life after proteolysis: exploring the signaling capabilities of classical cadherin cleavage fragments. Commun Integr Biol. 2009;2(2):155–157. doi:10.4161/cib.7700
35. Kandel ER. The molecular biology of memory storage: a dialogue between genes and synapses. Science. 2001;294(5544):1030–1038. doi:10.1126/science.1067020
36. Lonze BE, Ginty DD. Function and regulation of CREB family transcription factors in the nervous system. Neuron. 2002;35(4):605–623. doi:10.1016/S0896-6273(02)00828-0
37. Arias J, Alberts AS, Brindle P, et al. Activation of cAMP and mitogen responsive genes relies on a common nuclear factor. Nature. 1994;370(6486):226–229. doi:10.1038/370226a0
38. Chrivia JC, Kwok RP, Lamb N, Hagiwara M, Montminy MR, Goodman RH. Phosphorylated CREB binds specifically to the nuclear protein CBP. Nature. 1993;365(6449):855–859. doi:10.1038/365855a0
39. Kwok RP, Lundblad JR, Chrivia JC, et al. Nuclear protein CBP is a coactivator for the transcription factor CREB. Nature. 1994;370(6486):223–226. doi:10.1038/370223a0
40. Ma W, Zheng WH, Powell K, Jhamandas K, Quirion R. Chronic morphine exposure increases the phosphorylation of MAP kinases and the transcription factor CREB in dorsal root ganglion neurons: an in vitro and in vivo study. Eur J Neurosci. 2001;14(7):1091–1104. doi:10.1046/j.0953-816x.2001.01731.x
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.