Back to Journals » OncoTargets and Therapy » Volume 15
Spermidine Promotes Nb CAR-T Mediated Cytotoxicity to Lymphoma Cells Through Elevating Proliferation and Memory
Authors Wang H, Jiang D, Liu L, Zhang Y, Qin M, Qu Y, Wang L, Wu S, Zhou H, Xu T, Xu G
Received 15 July 2022
Accepted for publication 5 October 2022
Published 18 October 2022 Volume 2022:15 Pages 1229—1243
DOI https://doi.org/10.2147/OTT.S382540
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Arseniy Yuzhalin
Hongxia Wang,1,2,* Dan Jiang,1,2,* Liyuan Liu,2 Yanting Zhang,2 Miao Qin,2 Yuliang Qu,2 Liyan Wang,2 Shan Wu,2 Haijin Zhou,1 Tao Xu,1 Guangxian Xu1,2
1Guangdong Provincial Key Laboratory of Medical Molecular Diagnostics, School of Medical Technology, The First Dongguan Affiliated Hospital, Guangdong Medical University, Dongguan, People’s Republic of China; 2School of Clinical Medicine, Ningxia Medical University, Yinchuan, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Guangxian Xu, Guangdong Provincial Key Laboratory of Medical Molecular Diagnostics, School of Medical Technology, The First Dongguan Affiliated Hospital, Guangdong Medical University, Dongguan, 523808, People’s Republic of China, Tel +86 13995414482, Email [email protected]; [email protected]
Purpose: Due to the natural advantages of spermidine in immunity, we investigated the effects of spermidine pretreatment on nanobody-based CAR-T cells (Nb CAR-T) mediated cytotoxicity and potential mechanism.
Patients and Methods: The optimal concentration of spermidine was determined by detecting its impact on viability and proliferation of T cells. The phenotypic characteristic of CAR-T cells, which were treated with spermidine for 4 days, was examined by flow cytometry. The expansion ability of CAR-T cells was monitored in being cocultured with tumor cells. Additionally, CAR-T cells were stimulated by lymphoma cells to test its cytotoxicity in vitro, and the supernatant in co-culture models were collected to test the cytokine production. Furthermore, xenograft models were constructed to detect the anti-tumor activity of CAR-T cells in vivo.
Results: The optimal concentration of spermidine acting on T cells was 5μM. The antigen-dependent proliferation of spermidine pretreatment CD19 CAR-T cells or Nb CAR-T cells was increased compared to control. Central memory T cells(TCM) dominated the CAR-T cell population in the presence of spermidine. When spermidine pretreatment CAR-T cells were stimulated with Daudi cells, the secretion of IL-2 and IFN-γ has been significantly enhanced. The ability of CAR-T cells to lysis Daudi cells was enhanced with the help of spermidine, even at higher tumor loads. Pre-treated Nb CAR-T cells with spermidine were able to control tumor cells in vivo, and therefore prolong mice survival.
Conclusion: Our results revealed that spermidine could promote Nb CAR-T mediated cytotoxicity to lymphomas cells through enhancing memory and proliferation, and provided a meaningful approach to strengthen the anti-tumor effect of CAR-T cells.
Keywords: spermidine, nanobody, CAR-T, exhaustion
Introduction
Chimeric antigen receptor T (CAR-T) cell therapy has achieved remarkable success in the treatment of hematologic malignancies,1–3 such as B-cell acute lymphoblastic leukemia(B-ALL), large B-cell lymphoma (DLBCL), relapsed/refractory B-cell lymphoma (r/r LBCL) or relapsed/refractory multiple bone marrow (r/r MM) and other malignant hematologic tumors,4–6 but there are still about 40–50% of patients experience unsatisfied prognosis.7–9 The reasons causing the failure of CAR-T cell therapy are extremely complex,10–12 including the antigen loss or mutation of tumor cells,13–15 and the duration time and differentiated state of CAR-T cells.16–18 Moreover, graft-versus-host disease (GVDH), neurotoxicity and cytokine release syndrome(CRS) also could lead to recurrence after CAR-T cells treatment.19,20 What’s more, unlike hematological tumors, the CAR-T cells in therapy of solid tumor was blocked by the lack of ideal therapeutic targets and complicated tumor microenvironment. What is worse, tumor cells could disturb the metabolic pathway of CAR-T cells via plunder nutrition, resulting in tumor cells escaped.21–23
Furthermore, it is reported that CAR-T cells exhaustion was also a crucial factor to influence therapeutic effect.24,25 Exhausted CAR-T cells exhibited up-regulate expression of numerous inhibitory receptors like programmed cell death protein 1 (PD-1), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and T cell Ig and ITIM domain (TIGIT). So, the proliferation and self-renewal ability of CAR-T cells were limited. Besides, cytolysis molecules like granulomycin B, perforin, TNF-α were decreased.26–28 Recently, several studies claimed that CAR-T cells exhaustion could be rescued via gene editing technologies. Once TOX and NR4A were knockout by shRNA or CRISPR Cas9, the CD19 CAR-T cells exhibited high effective on inhibiting tumor growth and prolonging survival of tumor-bearing mice.29,30 Good et al demonstrated that downmodulation of ID3 and SOX4 expression can improve the efficacy of CAR-T cell therapy in solid tumors.31 Although CAR-T cell exhaustion has been effectively delayed through gene editing, there is still a potential risk of non-target gene mutations. Therefore, it is meaningful to find other novel approaches to prevent this phenomenon.
Spermidine, as a natural compound, participates in the regulation of human metabolism and life span, and it descended with aging process.32–34 Spermidine could promote the generation of lymphocytes, which is known to play a dominant role in immune response.35 Carriche et al demonstrated spermidine could also inhibit the survival of immunosuppressive cells like Treg cells, therefore maintaining the balance of the immune system.36 In addition, various evidence confirmed spermidine possessed function of autophagy regulation and anti-aging.35,37–39 Accordingly, considering spermidine’s powerful potential in leukomonocyte and anti-aging, we hypothesized that spermidine treatment during CAR-T cells expansion might delay CAR-T cells exhaustion and may have a positive effect on anti-tumor function of CAR-T cells.
Thus, in this study, we utilized spermidine as a culture additive in CAR-T cells medium, and analyzed the potential roles and mechanism of spermidine in affecting CAR-T cells survival and cytotoxicity. We observed lower differentiation degree of CAR-T cells with spermidine exist. Meanwhile, spermidine facilitated antigen-dependent proliferation and contributed to production of IL-2 and IFN-γ. Moreover, the cytotoxicity of CAR-T cells to lymphoma cells was also improved in vitro and vivo. Our results proved spermidine pretreatment was benefit for improving CAR-T cells therapeutic efficiency in lymphoma.
Materials and Methods
Cells and Culture Conditions
The chronic myelogenous leukaemia cell line K562, the Burkitt lymphoma cell line Daudi and embryonic kidney cell line 293T were purchased from American Type Culture Collection (ATCC). Daudi cells were then transduced with a lentiviral vector encoding firefly luciferase, followed by puromycin selection of luciferase-positive cells for up to 14 days. All cell lines were maintained in RPMI 1640(BI) supplemented with 10% fetal bovine serum (BI) and 1% penicillin-streptomycin (Solarbio), except 293T cells were cultured in DMEM(BI).
Manufacturing of CAR-T Cells
Construction of Chimeric Antigen Receptors (CAR) Genes
Scfv-based second generation CD19 CAR was generated by linking the single chain variable fragment of CD19 (FMC63 clone), in frame with the CD8α hinge and transmembrane domains of CD8, and the cytoplasmic domains of 4–1BB and CD3ζ. The nanobody based CARs were same as our previously report. Specifically, For CD19 Nb CAR and CD20 Nb CAR, anti-CD19 or anti-CD20 nanobodies was incorporated into basic CARs that were composed of IgG4 hinges, CD8 transmembrane domains, and 4–1BB and CD3ζ signaling domains. For Bispecific Nb CAR, the anti-CD20 and anti-CD19 nanobodies were linked by (EAAAK)3 and then combined with basic CARs. Additionally, the GFP tag on the N-terminus was inserted to track CAR expression. The CAR constructs were cloned into the GV401 lentiviral plasmid backbone under the regulation of a human EF-1α promoter. Then, 293T cells were transiently transfected with the pHelper 1.0 vector and the pHelper 2.0 packaging plasmids. The lentiviral supernatants were collected and stored at −80 °C.
Generation CAR-T Cells
This description was the same as in our previous study. In brief, first, the CD3-positive T cells were enriched from the PBMCs that were separated by density gradient centrifugation (TBD). Next, purified T cells were cultured in X–VIVO15 (Lonza), activated by CD3 and CD28 mAb (PeproTech), and supplied with 10%FBS, 100U/mL penicillin, 100ug/mL streptomycin(solarbio), and 200U/mL IL-2 (Peprotech). Then, 24h later, RetroNectin-coated plates (TaKaRa) were loaded with virus that carrying different CAR genes (CD19 Nb CAR, CD20 Nb CAR, Bispecific Nb CAR, or CD19 CAR) by centrifuging the plates at 2000g for 2h. Once activated, T cells were collected and added to the plates and centrifuged at 1000g for 1h, polybrene(solarbio), IL-2 and antibiotic were also added. Tomorrow, gently replenish constant volume T cell complete medium, and T cells were expanded for 7 days before in vitro and vivo analyze.
Generation Spermidine Pretreat CAR-T Cells
After identified the positive percentage of CAR-T cells, these cells were then cultured under T cell complete medium and in the presence of 0.5μM or 5μM spermidine (Sigma) for 4 days.
Determine the Optimal Concentration of Spermidine
The optimization condition of spermidine was determined by two indexes, that include the effects of spermidine pretreatment on T cell viability and proliferation.
Flow Cytometry Analysis
All data was recorded using a FACSCelestaTM Flow cytometer and analyzed by FlowJo 10 software. For CAR’s expression, T cells were stained with PE-conjugated mouse anti-human CD3, and CAR-T cells were gated on FITC+PE+. For analysis of T cells or CAR-T cell viability, cells were stained with PE Annexin V Apoptosis Detect kit (BD, 559763). For memory phenotype, cells were stained with BB700 conjugated mouse anti-human CCR7, APC conjugated mouse anti-human CD45RA(Biolegend).
Cell Proliferation
T cells or CAR-T cells were first labelled with 1 µM CellTrace Far Red (Life Technologies). To determine spermidine concentration, Labeled T cells were cultured in the presence of different dose of spermidine and expanded to 4 days for analysis. For CAR-T cells proliferation, labeled CAR-T cells were cultured with mitomycin C (MCE) treated target cells at a 10:1 effector/target ratio in X–VIVO15 medium without IL-2 for 4 days, followed by flow cytometric analysis.
ELISA
CAR-T cells and tumor cells were cultured at a ratio of 10:1, 20:1 in X–VIVO medium without additional cytokines in 96-well plates. After 24 h, the supernatant was collected and used for IL-2 and IFN-γ ELISA measurements (Biolegend).
In vitro Cytotoxicity
The LDH assay (Promega) was used to test the cytotoxicity. The experimental setting was designed according to the test instructions. Briefly, tumor cells were incubated with varying amounts of CAR-T cells at a range of E:T (effect cells: target cells) ratios, twenty-four hours later, OD450 values of supernatant were measured by a Thermo Scientific Microplate Reader. The percentage of specific lysis was calculated by the following formula:
Cytolysis%=(Experiment-Effector Spontaneous-Target Spontaneous) /(Target Maximum -Target Spontaneous)×100.
In vivo Xenograft Models
All animal experiments described in this article complied with Guangdong Medical University animal ethics. For the in vivo study of CAR-T cells function, we established intravenously injected lymphoma models. 6–8 weeks old NOD-Prkdcem26Cd52Il2rgem26Cd22/Nju (NCG) mice were administered with 1×106 Daudi-luc by tail vein injection. One week post injection, 2×107 CAR-T cells were injected intravenously. Both ventral and dorsal tumor burden was monitored by IVIS Lumina III imaging system (PerkinElmer) according to mice condition after intraperitoneal injection of luciferin (Promega). The weight and survival were also recorded, and the mice were euthanised when thinning hair and rapid weight loss occurred.
Statistical Analysis
All statistical analyses were performed using SPSS 26.0 and graphs were created using GraphPad Prism version 8. Data variance was analyzed by t-test between the two independent groups and one-way ANOVA between multiple groups. Percent survival was estimated using the Kaplan-Meier method, and statistical significance was calculated by the Log rank test. *p<0.05, **p<0.01, ***p<0.001, ns, not significant (p>0.05). All data are shown as the mean ± SD of three independent replicates.
Results
Determination of the Spermidine Concentration in T Cell Culture
Given the immunomodulation effect of spermidine, we set diverse concentration of spermidine (0μM, 0.5μM, 5μM, 10μM, 20μM, 100μM, and 500μM) to evaluate the optimal dose in T cell culture. Through cell activity detecting, we found that CD8+T cells treated with 0.5μM and 5μM remain regular viability, while T cell in other groups deteriorated as dose increasing (Figure 1A). Next, we observed a boosting proliferation of T cells after spermidine pretreating at the dose of 0.5μM and 5μM, and the proliferative population was 76.5%, 73.2%, respectively (Figure 1B). Meanwhile, absolute numbers of T cells were counted after spermidine treatment. The quantity of T cells elevated significantly at 0.5μM and 5μM of spermidine (p<0.001). In contrast, T cells replication was attenuate at 100μM and 500μM, which was accordant to viability detecting (Figure 1C). Taken together, we choose 0.5μM and 5μM as the preliminary working concentration for its less toxicity on T cells. Considering there is a large gap between 0.5μM and 5μM, it is of great significance to detail the concentration of spermidine hereafter.
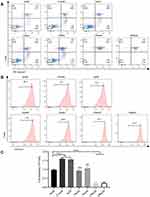
Spermidine Enhanced Proliferative Ability and Memory Phenotype Converting of CAR-T Cells
We constructed scfv-based CD19 CAR-T cells and Nb CAR-T cells via lentivirus (Figure 2A and B) and further assessed proliferative ability and memory phenotype converting of CAR-T cells. CD19 CAR-T cells pretreated with spermidine showed superiority proliferation when co-cultured with Daudi cells. This trend became invalid once K562 cells replaced (which is CD19 negative), whether spermidine existed or not (Figure 2C). Similar results were acquired among Nb CAR-T cells group. Noticeably, the effector of spermidine treated on day 4 were stronger than that normal condition on day 6 (Figure 2D and E). In addition, phenotype of CAR-T cells was identified with the TCM marker (CD45RA and CCR7). As a result, both CD19 CAR-T cells and Nb CAR-T cells evidently shifted to memory-like phenotype with spermidine treated (Figure 3A and B). Above all, spermidine endowed T cells with a higher proliferative potential and a memory-like phenotype transfer.
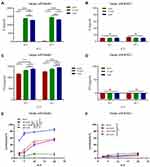
Spermidine Promoted CD19 CAR-T Cells Cytotoxicity Against Daudi Cells in vitro
To investigate the influence of spermidine on CAR-T cells cytotoxicity, we first performed ELISA assay to examine secretion of IL-2 and IFN-γ. After co-cultured with Daudi cells but not K562 cells, we observed elevated cytokine production of CAR-T cells that pretreated by spermidine (Figure 4A–D). Meanwhile, our LDH data demonstrated that spermidine pretreatment CAR-T cells displayed superior anti-tumor activity against Daudi cells. Interestingly, we noted that 5μM treating of spermidine dramatically enhanced CD19 CAR-T cells cytotoxicity to 80% rather than 0.5μM, even at high tumor load (Figure 4E). Nevertheless, we obtained inferior cytotoxicity when K562 cells displaced Daudi cells (Figure 4F). The above results demonstrated that the cytotoxic activity of CD 19 CAR-T cells against lymphoma cells was improved after spermidine treatment in vitro.
Spermidine Enhanced Killing Efficiency of Nanobody Based CAR-T Cells
Our previous findings proved that nanobody based CAR-T cells possessed a highly efficient cytotoxicity to tumor cells, more notably, Bispecific Nb CAR-T cells (target CD19 and CD20 simultaneously) was more effective than single target.60 Therefore, we further explore the killing efficiency of Nb CAR-T cells combined with spermidine. As mentioned, 5μM of spermidine treating brought remarkable cytotoxicity to CD19 CAR-T cells than 0.5μM, thus same dose was utilized to Nb CAR-T cells. As shown in Figures, IL-2 expression of Nb CAR-T cells have been augmented with favor of spermidine (Figure 5A and B). Similar results were observed in IFN-γ detection (Figure 5C and D). Moreover, spermidine endowed all three types Nb CAR-T cells with higher cytotoxicity to Daudi cells compared to control. Besides, Bispecific Nb CAR-T cells show greater advantages in cytokine production and killing function (Figure 5E–H). Together, these results indicated that the spermidine could promote both single and double CAR-T cells effector function.
Spermidine Improved Antitumor Activity of Nanobody Based CAR-T Cells in vivo
Next, we test the function of Nb CAR-T cells that pretreated with spermidine in vivo. NCG mice were injected intravenously (i.v.) with 1×106 Daudi-Luc tumor cells. After one week, a single dose of 2×107 Nb CAR-T cells or the equal volume PBS were transferred in same way, and the tumor burden were measured by bioluminescence (BLI) (Figure 6A). Images showed that both ventral and dorsal tumor load in the Nb CAR-T was significantly decreased compared to the PBS group (Figure 6B and C). Same tendency was confirmed by the total flux (Figure 6D and E). Besides, mice barely suffered from toxic caused by exogenous CAR-T cells for no radically decreasing in weight (Figure 6F and G). As a comparison, the untreated mice died on 23 days post Daudi-Luc cells injection. Although the tumor load was not statistically significant between Bispecific Nb CAR-T and spermidine Bispecific Nb CAR-T cells groups, mice treated with latter have prolonged overall survival to a certain degree (Figure 6H). Eventually, our findings suggested that spermidine was an available enhancer for its improvement in Nb CAR-T cells cytotoxicity both in vitro and vivo.
Discussion
As the conflicts between immune system and tumor cells go more intense, many novel strategies have been proposed to cope with the situation. Tumor immunotherapy, which aims to mobilize the body’s inherent immune system, has dramatically alleviated disease progression and improved patient quality of life compared with conventional treatments, such as surgery, radiation and chemotherapy.40 For instance, immune checkpoint blocking therapy (ICB) that represented by PD-1/ PD-L1 and CTLA-4, restores the effector function of T cells through blocking the immunosuppression effect of tumor cells.41 Adoptive cell therapy, including DC cells, CAR-T cells, CAR-NK cells and TCR-T cells, have already achieved promising clinical feedback. Among these modified cells, TCR-T and CAR-T cells are the most widely used, with therapeutic targets including CD19, CD20, or BCMA.42 Moreover, the combination of multiple tumor therapies has further improved the efficacy of tumor immunotherapy.43–45 Even so, a large number of patients still suffered from tumor relapse after immunotherapy, and the specific mechanisms still remain misty. Current explanations for drug resistance after CAR-T cells therapy in hematologic malignancies mainly contain the quality of prepared CAR-T cells and lack of the desirable tumor antigens. Numerous reports showed improving or reversing CAR-T cell exhaustion was a viable way to overcome barriers above.46–48
Recent study reported that decitabine treatment was able to improve the exhaustion of CAR-T cells. In terms of mechanism, decitabine was a DNA methylase inhibitor and responsible for epigenetic reprogramming, such as regulation of memory related genes (TCF-7) and inhibitory molecules (PD-1 and CTLA-4). At the same time, it played vital roles in T cell proliferation and other biological processes that contribute to CAR-T cell effector function.49,50 In addition, Sukumar M stated that inhibiting glycolysis by 2-deoxy-d-glucose (2DG, a synthetic glucose analogue) could promote the formation of memory T cells.51 More interestingly, rapamycin pretreatment rescued the capacity of CAR-T cells to eliminate the bone marrow AML cell by attenuating mTORC1 activity and increasing the capacity of CAR-T cells to infiltrate bone marrow.52
Through published studies, we found that spermidine might contribute to the cytotoxicity of CAR-T cells due to its superiority in immunoregulation and aging.39,53–55 Eisenberg et al proved that spermidine could prolong lifespan by reducing the activity of autophagy inhibitor EP300, and increasing the stability of autophagy related protein MAP1S.56 Other studies revealed that spermidine might affect the effector function of T cells by maintaining the level of autophagy through eIF5A and TFEB.57 Similarly, spermidine reversed B cell senescence and prolongs B cell survival in the same way.58 In addition, Carriche Guilhermina M found that spermidine participated in the differentiation of CD4+T cells.36 Collectively, spermidine deserved our attention for its roles in regulation of biological activities, like polyamine metabolism, immunosurveillance and autophagy.59
In this study, we first optimized the working concentration of spermidine to evaluate its toxicity in vitro. We observed that 0.5μM spermidine as well as 5μM benefited biological function of T cells for higher proliferation ability and no damage on viability. Given that there was a big gap between 0.5μM and 5μM, we would further optimize the concentration of spermidine to decline the toxic effect as much as possible.
Then, spermidine was added to the medium to explore the biological properties of the CAR-T cells. Similar to T cells, both CD19 CAR-T cells and Nb CAR-T cells proliferated rapidly under spermidine treatment. Furthermore, given the differential status of CAR-T cells related to their anti-tumor activity, we adopted memory marker (CD45RA and CCR7) to distinguish different cell populations. The results showed that CAR-T cells treated with spermidine exhibited relatively lower levels of differentiation than untreated cells, with TCM accounting for more than 55% of the total population. These results encouraged us to further explore the effects of spermidine on CAR-T cells antitumor effects.
Firstly, traditional CD19 CAR-T cells were used to investigate the impact of spermidine. After co-incubated with Daudi cells for 24h, IL-2 and IFN-γ in spermidine pretreated groups were significantly increased (P<0.05). LDH analysis also showed that CD19 CAR-T cells pretreated with spermidine possessed obvious cytotoxicity advantages even at high tumor load (E:T= 5:1), especially when the concentration was 5μM. Compared with the control, about 80% of tumor cells were effectively eliminated by CD19 CAR-T cells pretreated with spermidine (E:T= 5:1), while 60% of tumor cells in untreated group were destroyed, even at E:T of 20:1. Subsequently, previously constructed Nb CAR-T cells were used to explore the effects of spermidine.60 Previous results have shown that nanobody-based CAR-T cells can effectively activation (an activation marker) and proliferation when stimulated by Daudi cells, resulting in lysis of these tumor cells. It was worth mentioning that we found the positive rate had a significant impact on CAR-T cells anti-tumor activity. Through continuous optimization of spermidine dosage, we obtained Nb CAR-T cells that highly expressed Nb CAR gene and their cytotoxicity was indeed increased. Similarly, Nb CAR-T cells treated with spermidine further enhanced their initial effector function, and the TCM population was dominant among Nb CAR-T cells, which may have contributed to their superior cytotoxicity.
Furthermore, we constructed a hematologic tumor model to detect the antitumor potential of CAR-T cells in vivo, and we observed that both tumor load and survival of mice were improved in the Nb CAR-T cell group. The advantages of spermidine were also confirmed in vitro and vivo. Despite there was no significant difference in tumor load between the Bispecific Nb CAR-T cells and spd Bispecific Nb CAR-T cells (Nb CAR-T cells pretreated with spermidine), an increasing survival of mice was observed in spd Bispecific Nb CAR-T cells. Maybe that was owing to the remarkable expansion capacity of CAR-T cells treated with spermidine, which has been demonstrated in vitro. All in all, on account of the unsatisfied results, we plan to further optimize the experimental schemes. For example, the half-life of spermidine in vivo acquired additional determination and high-affinity nanobody sequence needed to be found. Constructing different kinds of CARs with various costimulatory molecules such as 4–1BB, ICOS, CD28 and optimizing the injection dose of CAR-T cells (such as multiple injections in small doses) were both promising ways to improve CAR-T efficiency. Meanwhile, spermidine was supplemented after CAR-T cell infusion.
Overall, spermidine pretreatment could increase the antitumor activity of CAR-T cells, while some deficiencies remain to be improved. Firstly, optimizing the in vivo experimental design to further determine the effect of spermidine. Then, it can be combined with other immunotherapy methods, such as immune checkpoint blockade. And the molecular mechanism by which spermidine acts on CAR-T cells was elucidated. Besides, more tumor models should be constructed (such as liver or lung cancer) to test the applicability of spermidine pretreatment to CAR-T cell therapy.
Conclusion
In this study, we demonstrated that adding proper concentration of spermidine during CAR-T cells generation could enhance a greater propensity of memory-like population, and improve their cytotoxicity potential against tumor cells. In conclusion, these finding provide insight into CAR-T cells study that spermidine can be used to improve their exhaustion.
Abbreviations
CAR-T, chimeric antigen receptor T cells; Nb CAR-T, Nb-based Chimeric antigen receptor T cells; LDH, lactate dehydrogenase; ELISA, enzyme linked immunosorbent assay; B-ALL, B-cell precursor acute lymphoblastic leukaemia; DLBCL, large B-cell lymphoma; r/r LBCL, relapsed/refractory B-cell lymphoma; r/r MM, relapsed/refractory multiple bone marrow; PD-1, programmed cell death protein 1; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; TIGIT, T cell Ig and ITIM domain; TOX, thymocyte selection associated high mobility group box; NR4A, nuclear receptor subfamily 4 group A; ID3, inhibitor of DNA binding 3; SOX4, SRY-box transcription factor 4; ICB, immune checkpoint blocking therapy; BCMA, B-cell maturation antigen; AML, Acute Myelocytic Leukemia; TCF7, transcription factor 7; ICOS, inducible T cell costimulator; eIF5A, eukaryotic translation initiation factor 5A; MAP1S, microtubule-associated protein 1S; TFEB, transcription factor EB; scfv, single chain antibody fragment; TCM, Central memory T cell; TEM, Effector memory T cell; Tn, Naïve T cell.
Data Sharing Statement
The datasets used and analyzed in the current study are available from the corresponding author in response to reasonable requests.
Ethics Approval and Informed Consent
All studies involving human blood cells were approved by the Ethics Committee of the General Hospital of Ningxia Medical University (number: KYLL-2021-216), and consent was obtained from patient. All studies involving animals were approved by laboratory animal ethical committee of Guangdong Medical University (number: GDY2104005).
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This study was supported by the Discipline Construction Project of Guangdong Medical University (No: 4SG21278P) and Scientific Research Start-up Fund for High-end Talents of Guangdong Medical University (4SG21240G).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Brown C, Mackall C. CAR T cell therapy: inroads to response and resistance. Nat Rev Immunol. 2019;19(2):73–74. doi:10.1038/s41577-018-0119-y
2. June C, O’Connor R, Kawalekar O, Ghassemi S, Milone M. CAR T cell immunotherapy for human cancer. Science. 2018;359(6382):1361–1365. doi:10.1126/science.aar6711
3. Larson R, Maus M. Recent advances and discoveries in the mechanisms and functions of CAR T cells. Nat Rev Cancer. 2021;21(3):145–161. doi:10.1038/s41568-020-00323-z
4. Mullard A. FDA approves fourth CAR-T cell therapy. Nat Rev Drug Discov. 2021;20(3):166.
5. Mullard A. FDA approves first BCMA-targeted CAR-T cell therapy. Nat Rev Drug Discov. 2021;20(5):332.
6. Mullard A. FDA approves second BCMA-targeted CAR-T cell therapy. Nat Rev Drug Discov. 2022;21(4):249.
7. Fry T, Shah N, Orentas R, et al. CD22-targeted CAR T cells induce remission in B-ALL that is naive or resistant to CD19-targeted CAR immunotherapy. Nat Med. 2018;24(1):20–28. doi:10.1038/nm.4441
8. Park J, Rivière I, Gonen M, et al. Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N Engl J Med. 2018;378(5):449–459. doi:10.1056/NEJMoa1709919
9. Maude S, Laetsch T, Buechner J, et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N Engl J Med. 2018;378(5):439–448. doi:10.1056/NEJMoa1709866
10. Shah N, Fry T. Mechanisms of resistance to CAR T cell therapy. Nat Rev Clin Oncol. 2019;16(6):372–385. doi:10.1038/s41571-019-0184-6
11. Ho L, Robbins H, Levine A. Assessing workforce needs for the emerging CAR-T cell therapy industry. Nat Biotechnol. 2022;40(2):275–278. doi:10.1038/s41587-022-01212-6
12. Jogalekar M, Rajendran R, Khan F, Dmello C, Gangadaran P, Ahn B. CAR T-cell-based gene therapy for cancers: new perspectives, challenges, and clinical developments. Front Immunol. 2022;13:925985. doi:10.3389/fimmu.2022.925985
13. Lemoine J, Ruella M, Houot R. Overcoming intrinsic resistance of cancer cells to CAR T-cell killing. Clin Cancer Res. 2021;27(23):6298–6306. doi:10.1158/1078-0432.CCR-21-1559
14. Wong D, Roy N, Zhang K, et al. A BAFF ligand-based CAR-T cell targeting three receptors and multiple B cell cancers. Nat Commun. 2022;13(1):217. doi:10.1038/s41467-021-27853-w
15. Hebbar N, Epperly R, Vaidya A, et al. CAR T cells redirected to cell surface GRP78 display robust anti-acute myeloid leukemia activity and do not target hematopoietic progenitor cells. Nat Commun. 2022;13(1):587. doi:10.1038/s41467-022-28243-6
16. Mueller K, Maude S, Porter D, et al. Cellular kinetics of CTL019 in relapsed/refractory B-cell acute lymphoblastic leukemia and chronic lymphocytic leukemia. Blood. 2017;130(21):2317–2325. doi:10.1182/blood-2017-06-786129
17. Maude S, Frey N, Shaw P, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507–1517. doi:10.1056/NEJMoa1407222
18. Meyran D, Terry R, Zhu J, et al. Early-phenotype CAR-T cells for the treatment of pediatric cancers. Ann Oncol. 2021;32(11):1366–1380. doi:10.1016/j.annonc.2021.07.018
19. Cooper M, Choi J, Staser K, et al. An ”off-the-shelf” fratricide-resistant CAR-T for the treatment of T cell hematologic malignancies. Leukemia. 2018;32(9):1970–1983. doi:10.1038/s41375-018-0065-5
20. Morris E, Neelapu S, Giavridis T, Sadelain M. Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy. Nat Rev Immunol. 2022;22(2):85–96. doi:10.1038/s41577-021-00547-6
21. Luo Z, Yao X, Li M, et al. Modulating tumor physical microenvironment for fueling CAR-T cell therapy. Adv Drug Deliv Rev. 2022;185:114301. doi:10.1016/j.addr.2022.114301
22. Akbari P, Katsarou A, Daghighian R, et al. Directing CAR T cells towards the tumor vasculature for the treatment of solid tumors. Biochim Biophys Acta Rev Cancer. 2022;1877(3):188701. doi:10.1016/j.bbcan.2022.188701
23. Zhang Z, Wang T, Wang X, Zhang Y, Song S, Ma C. Improving the ability of CAR-T cells to hit solid tumors: challenges and strategies. Pharmacol Res. 2022;175:106036. doi:10.1016/j.phrs.2021.106036
24. Zebley C, Youngblood B. Mechanisms of T cell exhaustion guiding next-generation immunotherapy. Trends Cancer. 2022;8(9):726–734. doi:10.1016/j.trecan.2022.04.004
25. Gumber D, Wang L. Improving CAR-T immunotherapy: overcoming the challenges of T cell exhaustion. EBioMedicine. 2022;77:103941. doi:10.1016/j.ebiom.2022.103941
26. Delgoffe G, Xu C, Mackall C, et al. The role of exhaustion in CAR T cell therapy. Cancer Cell. 2021;39(7):885–888. doi:10.1016/j.ccell.2021.06.012
27. Verdon D, Mulazzani M, Jenkins M. Cellular and molecular mechanisms of CD8 T cell differentiation, dysfunction and exhaustion. Int J Mol Sci. 2020;21:19. doi:10.3390/ijms21197357
28. Yates K, Tonnerre P, Martin G, et al. Epigenetic scars of CD8 T cell exhaustion persist after cure of chronic infection in humans. Nat Immunol. 2021;22(8):1020–1029. doi:10.1038/s41590-021-00979-1
29. Seo H, Chen J, González-Avalos E, et al. TOX and TOX2 transcription factors cooperate with NR4A transcription factors to impose CD8 T cell exhaustion. Proc Natl Acad Sci U S A. 2019;116(25):12410–12415. doi:10.1073/pnas.1905675116
30. Chen J, López-Moyado I, Seo H, et al. NR4A transcription factors limit CAR T cell function in solid tumours. Nature. 2019;567(7749):530–534. doi:10.1038/s41586-019-0985-x
31. Good C, Aznar M, Kuramitsu S, et al. An NK-like CAR T cell transition in CAR T cell dysfunction. Cell. 2021;184(25):6081–6100.e6026. doi:10.1016/j.cell.2021.11.016
32. Partridge L, Fuentealba M, Kennedy B. The quest to slow ageing through drug discovery. Nat Rev Drug Discov. 2020;19(8):513–532. doi:10.1038/s41573-020-0067-7
33. Madeo F, Eisenberg T, Pietrocola F, Kroemer G. Spermidine in health and disease. Science. 2018;359(6374). doi:10.1126/science.aan2788
34. Casero R, Marton L. Targeting polyamine metabolism and function in cancer and other hyperproliferative diseases. Nat Rev Drug Discov. 2007;6(5):373–390. doi:10.1038/nrd2243
35. Fischer M, Ruhnau J, Schulze J, Obst D, Flöel A, Vogelgesang A. Spermine and spermidine modulate T-cell function in older adults with and without cognitive decline ex vivo. Aging. 2020;12(13):13716–13739. doi:10.18632/aging.103527
36. Carriche G, Almeida L, Stüve P, et al. Regulating T-cell differentiation through the polyamine spermidine. J Allergy Clin Immunol. 2021;147(1):335–348.e311. doi:10.1016/j.jaci.2020.04.037
37. Puleston D, Zhang H, Powell T, et al. Autophagy is a critical regulator of memory CD8(+) T cell formation. eLife. 2014;3:e03706. doi:10.7554/eLife.03706
38. Rubinsztein D, Mariño G, Kroemer G. Autophagy and aging. Cell. 2011;146(5):682–695. doi:10.1016/j.cell.2011.07.030
39. Ren J, Zhang Y. Targeting autophagy in aging and aging-related cardiovascular diseases. Trends Pharmacol Sci. 2018;39(12):1064–1076. doi:10.1016/j.tips.2018.10.005
40. Majzner R, Heitzeneder S, Mackall C. Harnessing the immunotherapy revolution for the treatment of childhood cancers. Cancer Cell. 2017;31(4):476–485. doi:10.1016/j.ccell.2017.03.002
41. Grosser R, Cherkassky L, Chintala N, Adusumilli P. Combination immunotherapy with CAR T cells and checkpoint blockade for the treatment of solid tumors. Cancer Cell. 2019;36(5):471–482. doi:10.1016/j.ccell.2019.09.006
42. Liu E, Marin D, Banerjee P, et al. Use of CAR-transduced natural killer cells in CD19-positive lymphoid tumors. N Engl J Med. 2020;382(6):545–553. doi:10.1056/NEJMoa1910607
43. Johnson L, Lee D, Eacret J, Ye D, June C, Minn A. The immunostimulatory RNA RN7SL1 enables CAR-T cells to enhance autonomous and endogenous immune function. Cell. 2021;184(19):4981–4995.e4914. doi:10.1016/j.cell.2021.08.004
44. Gardner T, Lee J, Bourne C, et al. Engineering CAR-T cells to activate small-molecule drugs in situ. Nat Chem Biol. 2022;18(2):216–225. doi:10.1038/s41589-021-00932-1
45. Baeuerle P, Ding J, Patel E, et al. Synthetic TRuC receptors engaging the complete T cell receptor for potent anti-tumor response. Nat Commun. 2019;10(1):2087. doi:10.1038/s41467-019-10097-0
46. Zhao J, Lin Q, Song Y, Liu D. Universal CARs, universal T cells, and universal CAR T cells. J Hematol Oncol. 2018;11(1):132. doi:10.1186/s13045-018-0677-2
47. Amatya C, Pegues M, Lam N, et al. Development of CAR T cells expressing a suicide gene plus a chimeric antigen receptor targeting signaling lymphocytic-activation molecule F7. Mol Ther. 2021;29(2):702–717. doi:10.1016/j.ymthe.2020.10.008
48. Dimitri A, Herbst F, Fraietta J. Engineering the next-generation of CAR T-cells with CRISPR-Cas9 gene editing. Mol Cancer. 2022;21(1):78. doi:10.1186/s12943-022-01559-z
49. Wang Y, Tong C, Dai H, et al. Low-dose decitabine priming endows CAR T cells with enhanced and persistent antitumour potential via epigenetic reprogramming. Nat Commun. 2021;12(1):409. doi:10.1038/s41467-020-20696-x
50. Greco B, Malacarne V, De Girardi F, et al. Disrupting N-glycan expression on tumor cells boosts chimeric antigen receptor T cell efficacy against solid malignancies. Sci Transl Med. 2022;14(628):eabg3072. doi:10.1126/scitranslmed.abg3072
51. Sukumar M, Liu J, Ji Y, et al. Inhibiting glycolytic metabolism enhances CD8+ T cell memory and antitumor function. J Clin Invest. 2013;123(10):4479–4488. doi:10.1172/JCI69589
52. Nian Z, Zheng X, Dou Y, et al. Rapamycin pretreatment rescues the bone marrow AML cell elimination capacity of CAR-T cells. Clin Cancer Res. 2021;27(21):6026–6038. doi:10.1158/1078-0432.CCR-21-0452
53. Abdellatif M, Sedej S, Carmona-Gutierrez D, Madeo F, Kroemer G. Autophagy in Cardiovascular Aging. Circ Res. 2018;123(7):803–824. doi:10.1161/CIRCRESAHA.118.312208
54. Ni Y, Liu Y. New insights into the roles and mechanisms of spermidine in aging and age-related diseases. Aging Dis. 2021;12(8):1948–1963. doi:10.14336/AD.2021.0603
55. Hofer S, Liang Y, Zimmermann A, et al. Spermidine-induced hypusination preserves mitochondrial and cognitive function during aging. Autophagy. 2021;17(8):2037–2039. doi:10.1080/15548627.2021.1933299
56. Eisenberg T, Knauer H, Schauer A, et al. Induction of autophagy by spermidine promotes longevity. Nat Cell Biol. 2009;11(11):1305–1314. doi:10.1038/ncb1975
57. Alsaleh G, Panse I, Swadling L, et al. Autophagy in T cells from aged donors is maintained by spermidine and correlates with function and vaccine responses. eLife. 2020;9:e57950. doi:10.7554/eLife.57950
58. Zhang H, Alsaleh G, Feltham J, et al. Polyamines control eIF5A hypusination, TFEB translation, and autophagy to reverse B cell senescence. Mol Cell. 2019;76(1):110–125.e119. doi:10.1016/j.molcel.2019.08.005
59. Fan J, Feng Z, Chen N. Spermidine as a target for cancer therapy. Pharmacol Res. 2020;159:104943. doi:10.1016/j.phrs.2020.104943
60. Wang H, Wang L, Li Y, et al. Nanobody-armed T cells endow CAR-T cells with cytotoxicity against lymphoma cells. Cancer Cell Int. 2021;21(1):450. doi:10.1186/s12935-021-02151-z
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.