Back to Journals » Orthopedic Research and Reviews » Volume 11
Slipped capital femoral epiphysis: current management strategies
Authors Aprato A , Conti A, Bertolo F, Massè A
Received 5 November 2018
Accepted for publication 15 January 2019
Published 29 March 2019 Volume 2019:11 Pages 47—54
DOI https://doi.org/10.2147/ORR.S166735
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Clark Hung
Alessandro Aprato,1 Andrea Conti,2 Federico Bertolo,2 Alessandro Massè2
1Department of Orthopaedics and Traumatology, AOU Città della Salute e della Scienza di Torino – Ospedale CTO, Torino, Italy; 2Department of Orthopaedic and Traumatology, University of Torino, Torino, Italy
Abstract: In orthopedic literature, there is little consensus regarding the best management of slipped capital femoral epiphysis (SCFE). Controversies and disparate trends derive from differences in clinical presentation, various classifications, and a variety of surgical procedures that have been described. Currently, there are no evidence-based recommendations. Surgical procedures vary, and they can be divided into fixation in situ, compensatory osteotomies, and direct corrections of the deformity at the head–neck junction. The first and second group of procedures have so far not gained optimal control over the risk of avascular necrosis or cannot achieve an anatomically aligned epiphysis with normal blood supply. On the other hand, the third technique can achieve this target and prevent residual deformity and the development of early hip arthritis, but it is not widely accepted, because of its surgical complexity. The purpose of this work is to present an overview of current knowledge and provide an orientation on clinical and surgical management of the patient suffering from SCFE.
Keywords: slipped capital femoral epiphysis, Dunn procedure, femoral osteotomy, pinning in situ, surgical hip dislocation, avascular necrosis
Introduction
Slipped capital femoral epiphysis (SCFE) is a common hip disorder in children and adolescents, and it consists in posteroinferior migration of the epiphysis in metaphysis through the physis in proximal femur. The current incidence of SCFE ranges from 0.33 in 100,000 to 24.58 in 100,000 children 8–15 years of age, depending upon sex and ethnicity. There is significant variability within racial groups, and the relative frequency (Caucasians at 1.0) is 5.6 for Polynesians, 3.9 for blacks, and 2.5 for Hispanics. The average age is 12.0 years for boys and 11.2 years for girls, and obese children present earlier than lightweight children.1 With regard to sex, males are more affected than females (13.35 vs 8.07 cases in 100,000).2 Recently, a study by Loder and Schneble suggested an overall peak of presentation in mid-August. As the average temperature increases, a less prominent double peak has been noticed. These seasonal variations are thought to be linked to differences in vitamin D production and levels at different times of the year. The prevalence of vitamin D insufficiency/deficiency in children and adolescents is higher in blacks and/or obese children than in Caucasian and/or nonobese ones.3 First described in the 16th century,4 SCFE was deeply investigated over the last century, increasing understanding of the anatomy and vascularity of the proximal femur. Despite the volume of research on SCFE, its etiology remains unclear, but it seems to involve both mechanical and metabolic factors.5
In this review, an overview of current knowledge of management and treatment of SCFE is presented. Due to the lack of consensus regarding classification, different surgical techniques, and outcome scores, it is hard to establish applicable guidelines or algorithms. With this work, we aim to provide a tool to orient the surgeon among the variety of clinical presentations and surgical procedures.
Etiology and histopathology
Antecedents include greater retroversion of the femoral neck or a coxa profunda5 related to major weakness of the growth plate during the period of rapid growth.6 Gebhart et al examined the differences in two common anatomic measurements – pelvic incidence (PI) and acetabular retroversion – and their associations with post-SCFE deformity. They claimed that in a patient with a small PI, the pelvis will often tilt forward to maintain normal lumbar lordosis and balance sagittal alignment of the spine. Such anterior tilt would load the anterior aspect of the hip joint and deliver the stress across the physis of the proximal femur. This increased stress, along with other mechanical insults, such as obesity, physeal sloping angle, femoral retroversion, and size of the epiphyseal tubercle, could potentially result in the development of an SCFE. Their study demonstrated that specimens with SCFE deformity have a smaller PI than a large cohort of normal control specimens. On the contrary, they did not find significant differences between acetabular versions of specimens with and without SCFE deformity. The unaffected acetabulum of SCFE specimens was not more retroverted than the affected side.7
From a histological point of view, physeal cellular columnar height and organization are significantly altered in SCFE. Since the perichondral ring is thin, the wide surface area of the undulating, interlocking mammillary processes guarantee most of the internal support of the normal physis. On the other hand, SCFE is characterized by physeal widening (as much as 12 mm, with normal range 2–6 mm), a widened hypertrophic zone comprising 60%–80% of physeal height, enlargement of chondrocytes, cellular column disorganization, higher proteoglycan and extracellular matrix concentrations in the physis, and widespread disruption in chondrocyte differentiation and endochondral ossification.8 Radiographic physeal widening underlines a mechanically weakening of the physis that is susceptible to unlocking mammillary processes, resulting in further destabilization. The epiphyseal tubercle has gained increasing attention over the years. It measures around 4 mm in height and is located among the mammillary processes of the posterosuperior quadrant of the epiphysis. Anatomically, it always stands below the foramina for the lateral epiphyseal vessels, and is postulated to confer mechanical strength to the physeal plate. For this reason, it is theorized to be crucial for physeal stability, but it decreases in size and surface area during childhood and adolescence as peripheral physeal cupping increases.8 Liu et al9 postulated that the epiphysis rotates internally on the epiphyseal tubercle and that a widened physis could contribute to epiphyseal dislodgement. Because the lateral epiphyseal arteries are immediately adjacent to and above the epiphyseal tubercle, this could explain the low rate of osteonecrosis in chronic, stable slips (ie, minimal displacement of the lateral epiphyseal vessels).
On the other hand, metabolic causes implicated in SCFE are obesity,10 some endocrinological diseases, such as hypothyroidism and kidney failure, and treatment with growth hormone.5 As previously reported, the onset of SCFE usually occurs during the period of maximum growth, but the age of onset is continuously changing, as there has recently been a tendency for this period to occur earlier.11 Most case series have reported bilateral involvement in as many as 63% of patients,2 underlining the importance of metabolic factors. Kohno et al found that ~70% of contralateral hips in unilateral SCFE patients had a subclinical posterior inclination of the capital femoral epiphysis, indicating the possibility of bilateral involvement. The contralateral posterior sloping angle was a reliable predictor of a contralateral slip, and an angle of 19° was the cutoff value for developing SCFE.12
Clinical presentation, diagnosis, and classification
Patients with SCFE show a great variety of presentations and symptoms. Even if a common presentation is that of an obese, hypogonadal boy during adolescent growth, most SCFE cases show no associated endocrinal disorder.13,14 The most commonly adopted classification divides SCFE into “stable” or “unstable” cases and is based on the ability of the patient to walk. A patient with stable SCFE is usually an obese teen with a brief history of pain that is poorly localized, and it can affect the hip, groin, thigh, and knee. History of a traumatic event occurring in the area is rare. The patient may also present a slight or mild limp, gait with external rotation of the foot, limitation of internal rotation of the hip, or with fixed position in external rotation and flexion of the hip (Drehmann sign).
A patient with unstable SCFE, however, often has severe hip pain that does not allow gait. Medical history is often positive for hip, thigh, and knee pain and previous trauma (of a minor entity that does not justify the condition). The patient lying on the couch has an attitude in external rotation of the side affected and counteracts any passive movement of the hip. Obligatory external rotation of the hip is noted when it is passively flexed to 90°. Although it is complicated to assess the rate of unstable SCFE, it is estimated to be around 10%–35% of patients presenting with such a condition.
The disease can also be divided into acute, acute-on-chronic, and chronic SCFE. Acute SCFE is characterized by sudden epiphyseal displacement and the presence of symptoms for <3 weeks. Chronic SCFE represents the major part of the disease (around 85%): symptoms are present for >3 weeks, with remission and relapse. Acute-on-chronic SCFE is diagnosed when symptoms occur abruptly with exacerbation of pain and inability to walk, with lower-limb pain for >3 weeks.
Radiography is needed when patients 8–15 years of age complain of new-onset limping and lower-limb pain. When SCFE is suspected, radiography should include anteroposterior and frog-leg views of both hips. In unstable SCFE, such imaging should be compared to the unaffected side. Several radiographic signs are suggestive of SCFE, such as widening of the physis, relative decreased height of the epiphysis, loss of intersection of the epiphysis by a lateral cortical line along the femoral neck (Klein’s line) and double density detected at the metaphysis (Steel sign, which is caused by posterior slip of the epiphysis).15–17
Another useful classification is based on Southwick’s method to assess the magnitude of the sliding by measuring the angle between the head and femoral diaphysis on X-ray anteroposterior and axial projections.18 The angle is then compared with the unaffected side for one-side lesion or with normal values for bilateral involvement (145° in anteroposterior projection and 18° in axial projection). Sliding is defined slight when the angle differs by <30°, mild if the angle is 30°–60°, and severe if >60°.5 The Wilson method measures the relative displacement of epiphysis on metaphysis in a frog-leg view, defining degrees of severity in relation to the slip.19
Magnetic resonance imaging (MRI), computed tomography, and bone scintigraphy have a significant role in evaluation of the disease. They can be used at the diagnostic stage, but they are even more useful in appraisal of severity, in surgical planning, and in assessment of prognosis. MRI can detect avascular necrosis (AVN), chondrolabral defects, and periarticular and bone edema. Computed tomography provides a three-dimensional view of the hip, estimating with accuracy the entity of the dislocation. Bone scintigraphy is also a precious aid for diagnosis, with 100% negative predictive value for SCFE.20
Accurate knowledge of vascular anatomy is crucial for correct treatment of SCFE. AVN is sadly quite a common and often unavoidable complication in SCFE. Several studies have focused on this issue, reporting AVN rates of 6%–58% in treated unstable SCFE, while the rate has been reported to be close to zero in treated stable SCFE. Factors related to the development of AVN include unstable hips with separation of the epiphysis from the metaphysis, delay from presentation to surgery, severity of the slip, younger age, short duration of prodromal symptoms, increased intracapsular pressure, and choice of surgical technique. The etiology of AVN is well understood, multifactorial, and includes several causes for an interruption in blood supply to the femoral head. It goes from an increase in capsular pressure leading to reduced blood flow to the epiphysis to compression, overstretch, kinking, or tearing of posterior retinacular blood vessels at the time of injury or during reduction and fixation of the dislocated epiphysis.20
Older studies carried out by Boyer et al21 and Carney et al22 reported that patients presenting acutely within 3 weeks from symptom onset suffered from a higher risk of AVN development. Chronic SCFE accounts for a lower rate of AVN, as the slowly evolving slip of the epiphysis allow a gentle stretch of the posterior retinacular vessels, reducing the risk of kinking, tearing, or disruption. However, Loder and Dietz demonstrated later that AVN is correlated more intimately with stability or instability of the hip than with progression of the disease. They found physeal stability to be predictive of osteonecrosis rate, with 47% of unstable and no stable SCFE developing AVN within 6–18 months.23 More recent data show a decrease in the rate of AVN in unstable SCFE to as low as 23.9%.24
In 2012, Ziebarth et al produced an intraoperative classification of the stability of the physis because of a scarcity of accurate clinical classifications: they classified physeal integrity as intact or disrupted. The physis was considered stable if the periosteum was intact and if several cuts were needed to separate the epiphysis during a modified Dunn procedure. On the other hand, the physis was considered unstable when the epiphysis was completely mobile without the need to free the physis. They also noticed that an intracapsular hematoma was not always present, but still not crucial for classification. Integrity of the retinaculum and of its attachment on the epiphysis was evaluated empirically at the time of surgical dislocation and presentation of the femoral head–neck junction.25
Furthermore, the clinical history of the disease is sometimes misleading: pain and limping may be discontinuous, and the actual onset of symptoms is difficult to assess. This concept is crucial, because treatment decisions and prognosis are based on duration of symptoms and stability of the physis. Such imaging as MRI could be decisive in evaluation of stability in SCFE, as it targets joint effusion, synovitis, and bone-marrow edema as indirect measures of epiphyseal stability.
Treatment
Most authors agree that once SCFE is diagnosed, surgical treatment is indicated, but significant controversies remain regarding the best treatment.26 The goals of treatment are to prevent further slippage and correct the deformity, avoiding osteonecrosis and chondrolysis.
In situ fixation
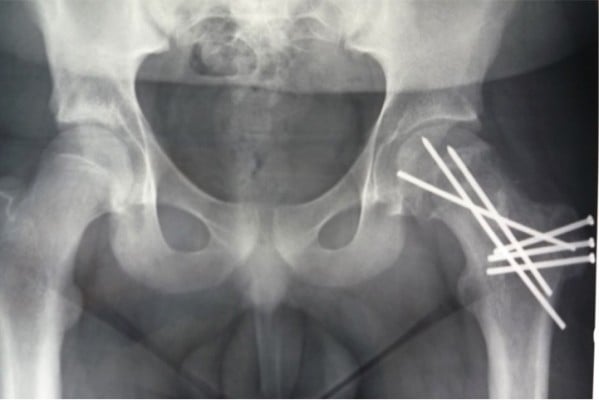
Classical treatment of SCFE consists in percutaneous fixation of the epiphysis. This technique aims to prevent the increase of displacement until eventual closure of the growth plate, and is widely used for both stable and unstable SCFE, regardless of the degree of deformity.23 Several methods have been described, from the implant of one cannulated screw to three or four Kirschner wires across the growth plate. These procedures are minimally invasive, simple to carry out, and continue to be widely used.
The ideal position of the single screw is in the center of the neck and perpendicular to the growth plate. The fovea should be taken into consideration, as the threads should achieve stable fixation, avoiding joint penetration. Considering that the increase in slippage locates the epiphysis more posteriorly relative to the femoral neck, a more anterior starting point is required onto the femoral neck to cross the physis perpendicularly.27,28 It has been reported that several attempts at guide-wire placement creating holes in the proximal femur may increase the risk of postoperative fractures,29,30 yet incorrect screw positioning can bring severe complications, such as further slippage, chondrolysis, joint penetration, loosening of the screw, future subtrochanteric fractures, and AVN. Brodetti31 demonstrated that placing a screw in the posterosuperior quadrant of the femoral neck results in high AVN incidence, as it can affect the perfusion coming from the lateral epiphyseal vessels. As such, when a two-screw design is required, the first should be placed in the ideal position, with the other in the inferolateral quadrant. Also, there is no evidence of biomechanical or clinical advantage between the use of multiple screws vs a single screw.27,32 Cannulated titanium screws scored highly on removal failure. This is explained by the tendency of bone growth over the head of the screw that can complicate the success of the surgery. For this reason, several authors have recommended fully threaded (no-cutback mechanism) stainless-steel screws. Such screws are meant to be removed as soon as the physis is closed.33,34
Some surgeons prefer using Kirschner wires, as they claim that screws can increase the risk of premature physis closure and can interfere with proximal femur remodeling. In younger patients, premature closure of the physis later results in growth disorder, including coxa vara, coxa breva, and overgrowth of the great trochanter, which can cause alteration in the biomechanics of the hip, eventually causing secondary osteoarthritis.35,36
In a study on pinning in situ, Castañeda et al37 reported a high incidence of unsatisfactory results because of technical difficulties in achieving an anatomic reduction. Boyer et al emphasized that in long-term follow-up, 12% of patients had to undergo further surgery, and reported a prevalence of early arthritis in 15% of cases.21 Based on these data, pinning in situ does not reach the main goal of early arthritis prevention in mild or severe cases.5 Furthermore, osteonecrosis rates vary between 10% and 40% with this technique,26 reaching 33% for unstable SCFE in a recent systematic review.38
Prophylactic pinning
Bilateral involvement in SCFE ranges from 14% to 63%, depending on the studies considered.2,39 This risk can increase to up to 80% when diagnosed at a very young age and up to 100% when endocrinopathies are associated.40 Hence, prophylactic pinning of a radiographically and clinically normal hip should be reserved to a selected cohort of patients, such as very young children, presence of endocrinopathy, obese patients, and those whose follow-up is thought to be difficult. Nowadays, consensus about routine prophylactic pinning is increasing, since the complication rate associated with the procedure is considered lower than that associated with the development of contralateral disease.41 In fact, as described by Hägglund in a long-term study, a risk of early development of osteoarthritis in the contralateral hip was detected in 25% of patients who did not undergo prophylactic pinning. The same long-term follow-up did not show any early development of osteoarthritis in patients stabilized prophylactically.42 Such surgical treatment remains controversial.
Compensatory osteotomies
Compensatory osteotomies are not intended to achieve an anatomically aligned epiphysis, since the correction at the site of deformity is reported to risk the blood supply to the epiphysis and thus has not found wide acceptance.43 Such osteotomies include trochanteric osteotomies and direct correction of the deformity at the head -neck junction.44 The most commonly used are intertrochanteric osteotomies and cuneiform osteotomies at the base of the neck. Intertrochanteric osteotomy limits are the distance between the osteotomy and the joint, inability to treat the impingement that is created by the sliding of the head, and inability to restore correct hip anatomy. They create an unwanted deformity that may complicate further hip-joint prosthesis. Many authors have reported poor results, eg, Kartenbender et al45 reported 23% poor clinical results and 33% poor radiographic results in 13.7 years of follow-up. Cuneiform osteotomies also have poor results: Velasco et al46 found at 16 years of follow-up osteonecrosis rates of 11%, chondrolysis rates of 12%, and early arthritis rates of 40%. Nowadays, better understanding of femoroacetabular impingement,47 a pathomechanical process of which SCFE can be an initiator,48 has renewed interest in direct correction of the deformity following SCFE.
Surgical hip dislocation (SHD)
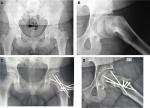
An important role is played by SHD, also called the modified Dunn procedure. Dunn49 introduced a posterolateral dissection of the retinaculum to allow some trimming of the callus formation at the posterior neck, adding a trochanteric osteotomy to facilitate the procedure. The space created, reaching from the head–neck junction to the axilla with the greater trochanter is rather narrow for perfect control of retinacular tension during callus removal, and this may explain why necrosis of the epiphysis could not sufficiently be eliminated with this procedure.49,50 With current and detailed knowledge of the vascular supply of the femoral head and its topography,51 SHD is a safe procedure and has become a well-accepted technique for the treatment of SCFE52 (Figure 1). This technique allows an anatomic reorientation of the epiphysis protecting retinacular vessels and epiphyseal vascularization.53,54 While recognizing the role of screw fixation in slight cases, Leunig et al proposed SHD as a method of open reduction and alignment of proximal femoral epiphysis.55 Although stable slip angles <30° can be treated successfully with in situ fixation, higher slips require subcapital reorientation with a retinacular flap.55,56 This is a modification of Dunn’s procedure in which resection of the posterior callus is performed with a short retinacular release, creating a longer flap that reduces the risk for perioperative overstretch of the vessels.57 This extended retinacular flap is three times longer than the retinacular release obtained in Dunn’s procedure. Such a flap permits distribution of the tension over a larger distance, which decreases the risk of negative effects on epiphysis perfusion.58 Posterior neck-callus formation has been described in the literature in patients with high slips treated with open surgery. Such calluses can interfere with valid epiphyseal perfusion, mostly if not resected before realignment: femoral head perfusion can be evaluated intraoperatively using laser Doppler flowmetry.59
SHD is a procedure with a steep learning curve, and should be performed only in a referral center. Any close reduction of a high slip or aiming for anatomical reduction without expertise in the technique should be avoided, as they increase the rate of interruption of the blood supply. If necessary, temporary in situ fixation could avoid the worsening of the slip until the patient can be referred to the closest center for SHD. This technique permits the surgeon to treat associated lesions, such as early acetabular labrum and cartilage damage or the metaphyseal bump that limits internal rotation, leading to femoroacetabular impingement, which can occur even in mild slips.48,60
An increasing number of articles have appeared in the literature on the treatment of SCFE with surgical dislocation2,5,43,55,61–66 demonstrating that this technique is effective. Novais et al67 compared children with severe stable SCFE treated with the modified Dunn procedure or in situ pinning in terms of proximal femoral radiographic deformity, Heyman and Herndon clinical outcome, complication rate, and number of reoperations performed after the initial procedure. In a total of 30 hips treated for severe stable slip (15 with the modified Dunn procedure and 15 with pinning in situ, statistically comparable regarding age at surgery, sex, affected side, and duration of follow-up), they found that the modified Dunn subcapital realignment procedure using an SHD approach allows for better radiographic correction of the femoral head and neck deformity, better clinical Heyman and Herndon outcome classification, and lower reoperation rate when compared with in situ pinning for treatment of severe stable SCFE at an average of 2 years of follow-up. They affirmed that despite historically better long-term outcomes after in situ pinning compared with reorientation procedures for mild and moderate SCFE, the residual deformity in severe SCFE is recognized to affect its prognosis negatively. In their study they concluded that because of the learning curve associated with the technically demanding modified Dunn procedure, the decision between its use vs in situ pinning is often based on surgeon experience rather than available evidence. Their data suggest that the modified Dunn procedure is to be considered a safer treatment in terms of definitive treatment.
However, SHD shares risks of serious complications with almost all other surgical procedures: slip progression, instability, growth arrest, residual deformity, chondrolysis, and osteonecrosis of the femoral epiphysis.68 Instability after modified Dunn procedure is not a common event, but it could occur for several reasons. Some are related to the primary deformity (SCFE-related) such as acetabular, labral, or cartilage damage, flattening of the acetabular roof, or a bell-shaped epiphysis. Others are related to the surgical procedure, like capsulotomy and ligament teres sacrifice (along with other associated factors), shortening of the neck causing infolding of periarticular tissue, pelvitrochanteric impingement, former femoral osteotomy, and bad leg positioning in the postoperative period. Also, some factors not related to SCFE are retroverted, with deep acetabulum and soft-tissue disorders possibly related to endocrinopathies.69
AVN has been reported after surgical dislocation with pinning in situ, and its rate has been estimated up to 47% in unstable slips.8 In fact, according to Loder’s classification system, it is assumed that none of the stable hips developed AVN,19 but most recent reports have discussed the incidence of that only in unstable SCFE. In a recent literature review, Loder suggested several causes of AVN not related to surgical treatment: kinking of the retinacular vessels, increased intracapsular joint pressure, and a complete tear of the vessel were the most valid hypotheses to explain AVN development.70 Furthermore, Kitano et al suggested that preoperative traction, decompression by arthrocentesis, timing from onset to reduction, sex, body mass index, and age at onset could not influence AVN development and that the only significantly predisposing factor was closed reduction, either purposefully or inadvertently, in acute, acute-on-chronic, and unstable SCFE.71 Also, chondrolysis,8 a loss of the cartilaginous surface of the femoral head and acetabulum, has been reported following all methods of treatment of SCFE, but the highest rates occur following nonoperative treatment, especially high-grade slips and in 1.5% of patients treated with percutaneous in situ fixation.
Conclusion
Even though orthopedic literature has been focused on SCFE over the last few centuries, some aspects of its etiology, presentation, best management, and treatment remain unclear. Careful anamnesis and clinical and radiographic examination at admission are essential, as the disease’s presentation can be subtle and bring catastrophic consequences if not diagnosed promptly. In situ fixation (pins or screws through the physis) has a relevant role in stable/unstable slips with <30° of deformity and in contralateral (normal) hips, prophylactically. Greater slips necessitate an anatomical realignment to restore hip anatomy and to reduce the risk of AVN. The largest recent systematic review of the treatment of unstable slips confirmed that instability was an important risk factor for AVN. It also suggested that open reduction and internal fixation seem to be associated with less AVN than other interventions (scoring similar rates in moderate and severe slips). Surgery should be performed ideally within 24 hours from presentation, and if this is not possible, delaying the operation to >72 hours may be associated with less AVN.38 Anatomic realignment is no longer performed as a closed reduction, because it does not allow direct control of the retinacular vessels. In agreement with recent studies, it may be affirmed that the modified Dunn procedure is safe, efficient, and reproducible, but it has a long learning curve and should be learned in a specialized center before use in clinical practice.
Disclosure
The authors report no conflicts of interest in this work.
References
Loder RT, Skopelja EN. The epidemiology and demographics of slipped capital femoral epiphysis. ISRN Orthop. 2011;2011:486512. | ||
Lehmann CL, Arons RR, Loder RT, Vitale MG. The epidemiology of slipped capital femoral epiphysis: an update. J Pediatr Orthop. 2006;26(3):286–290. | ||
Loder RT, Schneble CA. Seasonal variation in slipped capital femoral epiphysis: new findings using a national children’s hospital database. J Pediatr Orthop. 2019;39(1):e44–e49. | ||
Howorth B. Slipping of the capital femoral epiphysis. History. Clin Orthop Relat Res. 1966;48:11–32. | ||
Massè A, Aprato A, Favuto M, Atzori F, Nicodemo A, Governale G. Epifisiolisi. Lo Scal. 2010;24(3):177–183. | ||
Iwinski HJ. Slipped capital femoral epiphysis. Curr Opin Orthop. 2006;17(6):511–516. | ||
Gebhart JJ, Bohl MS, Weinberg DS, Cooperman DR, Liu RW. Pelvic incidence and acetabular version in slipped capital femoral epiphysis. J Pediatr Orthop. 2015;35(6):565–570. | ||
Georgiadis AG, Zaltz I. Slipped capital femoral epiphysis: how to evaluate with a review and update of treatment. Pediatr Clin North Am. 2014;61(6):1119–1135. | ||
Liu RW, Armstrong DG, Levine AD, Gilmore A, Thompson GH, Cooperman DR. An anatomic study of the epiphyseal tubercle and its importance in the pathogenesis of slipped capital femoral epiphysis. J Bone Joint Surg Am. 2013;95(6):e34–38. | ||
Bhatia NN, Pirpiris M, Otsuka NY. Body mass index in patients with slipped capital femoral epiphysis. J Pediatr Orthop. 2006;26(2):197–199. | ||
Loder RT, Starnes T, Dikos G. The narrow window of bone age in children with slipped capital femoral epiphysis: a reassessment one decade later. J Pediatr Orthop. 2006;26(3):300–306. | ||
Kohno Y, Nakashima Y, Kitano T, et al. Subclinical bilateral involvement of the hip in patients with slipped capital femoral epiphysis: a multicentre study. Int Orthop. 2014;38(3):477–482. | ||
Mann DC, Weddington J, Richton S. Hormonal studies in patients with slipped capital femoral epiphysis without evidence of endocrinopathy. J Pediatr Orthop. 1988;8(5):543–545. | ||
Razzano CD, Nelson C, Eversman J. Growth hormone levels in slipped capital femoral epiphysis. J Bone Joint Surg Am. 1972;54(6):1224–1226. | ||
Fahey JJ, O’Brien ET. Acute slipped capital femoral epiphysis: review of the literature and report of ten cases. J Bone Joint Surg Am. 1965;47:1105–1127. | ||
Peck D. Slipped capital femoral epiphysis: diagnosis and management. Am Fam Physician. 2010;82(3):258–262. | ||
Herman MJ, Martinek M. The limping child. Pediatr Rev. 2015;36(5):184–197. | ||
Southwick WO. Osteotomy through the lesser trochanter for slipped capital femoral epiphysis. J Bone Joint Surg Am. 1967;49(5):807–835. | ||
Loder RT, Richards BS, Shapiro PS, Reznick LR, Aronson DD. Acute slipped capital femoral epiphysis: the importance of physeal stability. J Bone Joint Surg Am. 1993;75(8):1134–1140. | ||
Bittersohl B, Hosalkar HS, Zilkens C, Krauspe R. Current concepts in management of slipped capital femoral epiphysis. Hip Int. 2015;25(2):104–114. | ||
Boyer DW, Mickelson MR, Ponseti IV. Slipped capital femoral epiphysis. Long-term follow-up study of one hundred and twenty-one patients. J Bone Joint Surg Am. 1981;63(1):85–95. | ||
Carney BT, Weinstein SL, Noble J. Long-term follow-up of slipped capital femoral epiphysis. J Bone Joint Surg Am. 1991;73(5):667–674. | ||
Loder RT, Dietz FR. What is the best evidence for the treatment of slipped capital femoral epiphysis? J Pediatr Orthop. 2012;32(Suppl 2):S158–S165. | ||
Zaltz I, Baca G, Clohisy JC. Unstable SCFE: review of treatment modalities and prevalence of osteonecrosis. Clin Orthop Relat Res. 2013;471(7):2192–2198. | ||
Ziebarth K, Domayer S, Slongo T, Kim YJ, Ganz R. Clinical stability of slipped capital femoral epiphysis does not correlate with intraoperative stability. Clin Orthop Relat Res. 2012;470(8):2274–2279. | ||
Loder RT. Unstable slipped capital femoral epiphysis. J Pediatr Orthop. 2001;21(5):694–699. | ||
Kibiloski LJ, Doane RM, Karol LA, Haut RC, Loder RT. Biomechanical analysis of single- versus double-screw fixation in slipped capital femoral epiphysis at physiological load levels. J Pediatr Orthop. 1994;14(5):627–630. | ||
Gourineni P. Oblique in situ screw fixation of stable slipped capital femoral epiphysis. J Pediatr Orthop. 2013;33(2):135–138. | ||
Lehman WB, Menche D, Grant A. The problem of evaluating in situ pinning of slipped capital femoral epiphysis: an experimental model and a review of 63 consecutive cases. J Pediatr Orthop. 1984;4(3):297–303. | ||
Walters R, Simon SR. Joint destruction: a sequel of unrecognized pin penetration in patients with slipped capital femoral epiphyses. In: The hip: proceedings of the eighth open scientific meeting of the hip Society. St. Louis: CV Mosby; 1980:145–164. | ||
Brodetti A. The blood supply of the femoral neck and head in relation to the damaging effects of nails and screws. J Bone Joint Surg Br. 1960;42-B(4):794–801. | ||
Karol LA, Doane RM, Cornicelli SF, Zak PA, Haut RC, Manoli A. Single versus double screw fixation for treatment of slipped capital femoral epiphysis: a biomechanical analysis. J Pediatr Orthop. 1992;12(6):741–745. | ||
Dragoni M, Heiner AD, Costa S, Gabrielli A, Weinstein SL. Biomechanical study of 16-mm threaded, 32-mm threaded, and fully threaded SCFE screw fixation. J Pediatr Orthop. 2012;32(1):70–74. | ||
Ilchmann T, Parsch K. Complications at screw removal in slipped capital femoral epiphysis treated by cannulated titanium screws. Arch Orthop Trauma Surg. 2006;126(6):359–363. | ||
Morscher E, Staubli A, Meyer S, Imhoff A. [5.) 10-year results after epiphyseolysis capitis femoris. a) 10-year results with nails and screws in epiphyseolysis capitis femoris]. Orthopade. 1979;8(1):60–64. German | ||
Chen CE, Ko JY, Wang CJ. Premature closure of the physeal plate after treatment of a slipped capital femoral epiphysis. Chang Gung Med J. 2002;25(12):811–818. | ||
Castañeda P, Macías C, Rocha A, Harfush A, Cassis N. Functional outcome of stable grade III slipped capital femoral epiphysis treated with in situ pinning. J Pediatr Orthop. 2009;29(5):454–458. | ||
Alshryda S, Tsang K, Chytas A, et al. Evidence based treatment for unstable slipped upper femoral epiphysis: systematic review and exploratory patient level analysis. Surgeon. 2018;16(1):46–54. | ||
Kocher MS, Bishop JA, Hresko MT, Millis MB, Kim YJ, Kasser JR. Prophylactic pinning of the contralateral hip after unilateral slipped capital femoral epiphysis. J Bone Joint Surg Am. 2004;86-A(12):2658–2665. | ||
Jerre R, Billing L, Hansson G, Karlsson J, Wallin J. Bilaterality in slipped capital femoral epiphysis: importance of a reliable radiographic method. J Pediatr Orthop B. 1996;5(2):80–84. | ||
Woelfle JV, Fraitzl CR, Reichel H, Nelitz M. The asymptomatic contralateral hip in unilateral slipped capital femoral epiphysis: morbidity of prophylactic fixation. J Pediatr Orthop B. 2012;21(3):226–229. | ||
Hägglund G. The contralateral hip in slipped capital femoral epiphysis. J Pediatr Orthop B. 1996;5(3):158–161. | ||
Massè A, Aprato A, Grappiolo G, Turchetto L, Campacci A, Ganz R. Surgical hip dislocation for anatomic reorientation of slipped capital femoral epiphysis: preliminary results. Hip Int. 2012;22(2):137–144. | ||
Gholve PA, Cameron DB, Millis MB. Slipped capital femoral epiphysis update. Curr Opin Pediatr. 2009;21(1):39–45. | ||
Kartenbender K, Cordier W, Katthagen BD. Long-term follow-up study after corrective Imhäuser osteotomy for severe slipped capital femoral epiphysis. J Pediatr Orthop. 2000;20(6):749–756. | ||
Velasco R, Schai PA, Exner GU. Slipped capital femoral epiphysis: a long-term follow-up study after open reduction of the femoral head combined with subcapital wedge resection. J Pediatr Orthop B. 1998;7(1):43–52. | ||
Ganz R, Parvizi J, Beck M. Femoroacetabular impingement: a cause of osteoarthritis of the hip. Clin Orthop Relat Res. 2003;417:112–120. | ||
Leunig M, Casillas MM, Hamlet M, et al. Slipped capital femoral epiphysis: early mechanical damage to the acetabular cartilage by a prominent femoral metaphysis. Acta Orthop Scand. 2000;71(4):370–375. | ||
Dunn DM. The treatment of adolescent slipping of the upper femoral epiphysis. J Bone Joint Surg Br. 1964;46-B(4):621–629. | ||
Fron D, Forgues D, Mayrargue E, Halimi P, Herbaux B. Follow-up study of severe slipped capital femoral epiphysis treated with Dunn’s osteotomy. J Pediatr Orthop. 2000;20(3):320–325. | ||
Kalhor M, Beck M, Huff TW, Ganz R. Capsular and pericapsular contributions to acetabular and femoral head perfusion. J Bone Joint Surg Am. 2009;91(2):409–418. | ||
Ganz R, Gill TJ, Gautier E, Ganz K, Krügel N, Berlemann U. Surgical dislocation of the adult hip a technique with full access to the femoral head and acetabulum without the risk of avascular necrosis. J Bone Joint Surg Br. 2001;83(8):1119–1124. | ||
Sucato DJ, De La Rocha A. High-grade SCFE: the role of surgical hip dislocation and reduction. J Pediatr Orthop. 2014;34(Suppl 1):S18–24. | ||
Ricciardi BF, Sink EL. Surgical hip dislocation: techniques for success. J Pediatr Orthop. 2014;34(Suppl 1):S25–31. | ||
Leunig M, Slongo T, Kleinschmidt M, Ganz R. Subcapital correction osteotomy in slipped capital femoral epiphysis by means of surgical hip dislocation. Oper Orthop Traumatol. 2007;19(4):389–410. | ||
Leunig M, Horowitz K, Manner H, Ganz R. In situ pinning with arthroscopic osteoplasty for mild SCFE: a preliminary technical report. Clin Orthop Relat Res. 2010;468(12):3160–3167. | ||
Dunn DM, Angel JC. Replacement of the femoral head by open operation in severe adolescent slipping of the upper femoral epiphysis. J Bone Joint Surg Br. 1978;60-B(3):394–403. | ||
Ganz R, Huff TW, Leunig M. Extended retinacular soft-tissue flap for intra-articular hip surgery: surgical technique, indications, and results of application. Instr Course Lect. 2009;58:241–255. | ||
Hempfing AaNotzli HP, Siebenrock KA. Perfusion of the femoral head during surgical dislocation of the hip: monitoring by laser Doppler flowmetry. J Bone Joint Surg. 2002;84-B:300–304. | ||
Rab GT. The geometry of slipped capital femoral epiphysis: implications for movement, impingement, and corrective osteotomy. J Pediatr Orthop. 1999;19(4):419–424. | ||
Spencer S, Millis MB, Kim YJ. Early results of treatment of hip impingement syndrome in slipped capital femoral epiphysis and pistol grip deformity of the femoral head-neck junction using the surgical dislocation technique. J Pediatr Orthop. 2006;26(3):281–285. | ||
Ziebarth K, Zilkens C, Spencer S, Leunig M, Ganz R, Kim YJ. Capital realignment for moderate and severe SCFE using a modified Dunn procedure. Clin Orthop Relat Res. 2009;467(3):704–716. | ||
Rebello G, Spencer S, Millis MB, Kim YJ. Surgical dislocation in the management of pediatric and adolescent hip deformity. Clin Orthop Relat Res. 2009;467(3):724–731. | ||
Tannast M, Jost LM, Lerch TD, Schmaranzer F, Ziebarth K, Siebenrock KA. The modified Dunn procedure for slipped capital femoral epiphysis: the Bernese experience. J Child Orthop. 2017;11(2):138–146. | ||
Ziebarth K, Milosevic M, Lerch TD, Steppacher SD, Slongo T, Siebenrock KA. High survivorship and little osteoarthritis at 10-year followup in SCFE patients treated with a modified Dunn procedure. Clin Orthop Relat Res. 2017;475(4):1212–1228. | ||
Novais EN, Maranho DA, Heare T, Sink E, Carry PM, O’Donnel C. The modified Dunn procedure provides superior short-term outcomes in the treatment of the unstable slipped capital femoral epiphysis as compared to the inadvertent closed reduction and percutaneous pinning: a comparative clinical study. Int Orthop. 2018;43(3):669–67. | ||
Novais EN, Hill MK, Carry PM, Heare TC, Sink EL. Modified Dunn procedure is superior to in situ pinning for short-term clinical and radiographic improvement in severe stable SCFE. Clin Orthop Relat Res. 2015;473(6):2108–2117. | ||
Roaten J, Spence DD. Complications related to the treatment of slipped capital femoral epiphysis. Orthop Clin North Am. 2016;47(2):405–413. | ||
Aprato A, Leunig M, Massé A, Slongo T, Ganz R. Instability of the hip after anatomical re-alignment in patients with a slipped capital femoral epiphysis. Bone Joint J. 2017;99-B(1):16–21. | ||
Loder RT. What is the cause of avascular necrosis in unstable slipped capital femoral epiphysis and what can be done to lower the rate? J Pediatr Orthop. 2013;33(Suppl 1):S88–S91. | ||
Kitano T, Nakagawa K, Wada M, Moriyama M. Closed reduction of slipped capital femoral epiphysis: high-risk factor for avascular necrosis. J Pediatr Orthop B. 2015;24(4):281–285. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.