Back to Journals » Neuropsychiatric Disease and Treatment » Volume 10
Sleep and cognitive problems in patients with attention-deficit hyperactivity disorder
Authors Lee HK, Jeong J, Kim N, Park M, Kim T, Seo H, Lim H, Hong S, Han J
Received 17 June 2014
Accepted for publication 27 July 2014
Published 17 September 2014 Volume 2014:10 Pages 1799—1805
DOI https://doi.org/10.2147/NDT.S69562
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Roger Pinder
Hae Kook Lee, Jong-Hyun Jeong, Na-Young Kim, Min-hyeon Park, Tae-Won Kim, Ho-Jun Seo, Hyun-Kook Lim, Seung-Chul Hong, Jin-Hee Han
Department of Psychiatry, College of Medicine, The Catholic University of Korea, Seoul, Korea
Objectives: Attention-deficit hyperactivity disorder (ADHD) is characterized by inattentive and impulsive behavior. Many ADHD patients reportedly have cognitive dysfunction and sleep problems, including longer sleep latency, lower sleep efficiency, and shorter total sleep time. The purpose of this study was to examine neurocognitive functions and nocturnal sleep parameters in patients with ADHD, using a cognitive function test and actigraphy.
Methods: Subjects included 37 male patients with ADHD and 32 controls (7–12 years of age). For each participant, we determined intelligence quotient (IQ) and administered the Matching Familiar Figures Test (MFFT) and 72-hour actigraphy. The relationships between sleep parameters and cognitive functions were assessed.
Results: ADHD patients significantly differed from controls in several cognitive functions and sleep variables. In the MFFT, response error rate (P<0.001) and error counts (P=0.003) were significantly increased in ADHD patients compared with control children. MFFT response latency was significantly shorter in ADHD patients than in controls (P<0.001). In addition, sleep latency (P=0.01), wake after sleep onset (WASO) (P<0.001), and fragmentation index (P<0.001) were evaluated by actigraphy and found to be significantly increased in patients with ADHD compared with controls. However, no significant differences in total sleep time or sleep efficiency were observed. WASO and response error rates were positively correlated in patients with ADHD (rho =0.52, P=0.012). Furthermore, fragmentation index sleep variables were significantly positively correlated with response error (rho =0.44, P=0.008) and response latency rates (rho =0.4, P=0.018) in the MFFT. Reaction error rate was significantly associated with the fragmentation index (beta =0.94, P=0.024).
Conclusion: Patients with ADHD had more sleep problems, including significantly increased sleep latency, WASO, and fragmentation index, and poorer cognitive function, compared with controls. Some of these sleep problems, including WASO and the fragmentation index, were positively correlated with impulsivity, illustrated by the cognitive function tests in patients with ADHD. However, further studies with large sample sizes and the addition of polysomnography and determination of ADHD subtypes should be performed to confirm our results regarding sleep and cognitive problems in patients with ADHD.
Keywords: ADHD, sleep problems, actigraphy, MFFT
Introduction
Attention-deficit hyperactivity disorder (ADHD), one of the most common mental disorders, is found in 3%–5% of school-age children and is characterized by an inattentiveness and increased impulsivity. These characteristics could lead to cognitive and behavioral dysfunction, such as poor schoolwork, socially maladaptive behaviors, and difficulties in frontal lobe higher functions.1 Another characteristic of ADHD is daytime hyperactivity and uncontrollable impulsivity. In addition to these symptoms, children and adolescents with ADHD are highly sensitive to stimuli and often have unstable responses.2
Approximately 25%–50% of ADHD patients reportedly have sleep problems.3 Earlier studies demonstrated that the variable sleep pattern in these patients is caused by ADHD therapeutic agents, particularly stimulants.4,5 However, it is now generally accepted that the effect of stimulants is just one of the causes of these patients’ sleep problems,6 and behavioral symptoms related to ADHD can cause sleep problems, including a higher level of nocturnal activity,7–9 longer sleep latency,10 lower sleep efficiency,11 more frequent night awakenings,12 and shorter total sleep time.11–13
Conversely, other studies reported that primary sleep disorders, such as obstructive sleep apnea (OSA),14,15 restless leg syndrome (RLS),8,16 and periodic limb movement disorder (PLMD),15,17 can induce daytime attention deficiency, hyperactivity, and cognitive dysfunction in children. Moreover, it has been reported that these ADHD-like symptoms are improved with sleep disorder treatment.18,19
Unfortunately, the findings describing the relationship between ADHD and sleep problems differ depending on the measurement tool used to evaluate sleep problems. For example, studies based on reporting from parents of ADHD patients reveal many sleep problems, but results based on objective measurements have yielded more controversial results. Specifically, findings based on polysomnography and actigraphy are not consistent,20,21 but results based on objective measurements have yielded more controversial results. Specifically, findings based on polysomnography and actigraphy are not consistent,7,20,22–24 except those showing increased activity during sleep7,20,22,24,25 and high night-to-night sleep variability in ADHD patients.23,26 In contrast, results based on the multiple sleep latency test (MSLT) consistently show that ADHD patients have a shorter sleep latency during the daytime.15,25
Polysomnography and MSLT are the most objective and comprehensive tests. However, these tests also have some disadvantages, in that daily activity is limited during the test, the cost is considerable, and the results are limited by test time. To overcome these disadvantages, sleep research using actigraphy, the measurement of body movement with a compact computerized wrist device, is increasing. These results have greater reliability than those based on patient or parent memory. Also, actigraphy-based sleep parameters are closely correlated with polysomnography results.27,28
Sleep restrictions or sleep disturbance in ADHD patients could lead on to neurobehavioral dysfunction, especially vigilance and attention.29–31 Also, topographical electroencephalogram (EEG) study showed neuromaturational delay in ADHD patients.
Our hypothesis is that cognitive and behavioral dysfunction might be associated with sleep problems in ADHD patients. The authors investigated nocturnal sleep parameters using actigraphy, in addition to cognitive dysfunction, in ADHD patients.
Methods
Subjects
Drug-naïve ADHD patients (n=37), as determined by Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR) criteria, were selected from 7- to 12-year-old outpatients that visited St Vincent’s Hospital Child and Adolescent Psychiatry Clinic at the Catholic University of Korea. By means of a mental status examination, actigraphy, and physical examination, patients who had psychiatric disorders other than ADHD, including primary sleep disorders (eg, OSA, RLS, and PLMD), and physical and neurologic disorders were excluded. Age- and sex-matched healthy controls (n=32) were selected to participate in this study. The mean age of the ADHD patients was 8.7±2.1 years (age range 7–12 years), and the control group mean age was 9.3±1.9 (age range 7–12 years). Both the ADHD and control groups comprised males. There were no significant differences between the two groups.
The contents and methods of the study were explained to all subjects and to their parents, and written informed consent was obtained for each participant. Research was carried out in accordance with the rules of the institutional review board at St Vincent’s Hospital, the Catholic University of Korea, and the Declaration of Helsinki.
Cognitive function tests
Cognitive function tests included intelligence quotient (IQ) and Matching Familiar Figures Test (MFFT) to identify executive function. The Korea Wechsler Intelligence Scale for children (K-WISC-III)32 was used to determine IQ. This test is divided into three parts: verbal IQ, performance IQ, and total IQ. Testing items in the verbal IQ include information, digit span, vocabulary, arithmetic, comprehension, and similarity; testing items in the performance IQ include picture completion, picture arrangement, block design, object assembly, coding, maze, and symbol search. From the verbal IQ, we can determine linguistic understanding and concentration ability; from the performance IQ, we can determine cognitive organization and processing speed. These four factors make up the IQ.
The Matching Familiar Figures Test for Korean Children (MFFT-KC)33 was used in this study. This is an objective test comprised of pictures used to determine impulsivity in children and does not require linguistic or numeric ability. Subjects ranging from 7 to 12 years of age complete the test in 15 minutes. Time is measured with a stopwatch from the start of question presentation to the subject until the subject begins to respond. Twelve items in the MFFT book are used for the test, and the subject is instructed to select the picture identical to the sample from the six pictures available. The time of the first response and the number of incorrect answers are recorded, and the result is automatically checked by computer. In this study, the percentile of response error and the percentile of response latency (the shorter the time until the first response, the greater the percentile of response latency) as well as response error counts and response latency time were calculated.
Actigraphy
Following the cognitive function tests, all subjects underwent actigraphy by wearing a device on the wrist of the nondominant arm for 72 continuous weekday hours (Actiwatch; MiniMitter, Bend, OR, USA). To evaluate and compare the sleep patterns of ADHD patients and normal controls using actigraphy data, we assessed total sleep time, sleep latency, sleep efficiency, wake after sleep onset (WASO), and the fragmentation index. Total sleep time is defined as the time from sleep onset to arising, that is, the real sleep time plus wake time (WASO) calculated in minutes. Sleep latency is the period in minutes from lying in bed or turning off the light to the point identified as sleep by actigraphy. Sleep efficiency is defined as the ratio between total sleep time and the entire time spent lying in bed at night. WASO is the total time in minutes of “waking” after sleep has been initiated. Finally, the fragmentation index, which is equivalent to a movement or restlessness index, is calculated as the total sum of movement time against time without movement.
Statistical analysis
Demographic data, IQ, MFFT, and sleep parameters are presented as mean ± standard deviation (SD) and percentage. The differences in demographic data, IQ, MFFT, and sleep parameters between ADHD patients and controls were analyzed using independent t-tests and chi-square tests. The relationships between these variables were analyzed by Pearson’s correlation and multiple linear regression tests. The significance level used for all statistical data was P<0.05. All tests were performed using SPSS 10.0 for Windows (SPSS Inc., Chicago, IL, USA).
Results
IQ and MFFT
The ADHD patient group MFFT response error rate (P<0.001) and response error counts (P=0.003) were significantly higher than those of the control group. In addition, response initiation was significantly shorter in the ADHD group than in the control group (P<0.001). No significant differences were observed between the ADHD and control groups regarding mean total IQ, verbal IQ, and performance IQ (Table 1).
Sleep parameters
The ADHD group had significantly longer sleep latency than the control group (16.2±19.7 minutes vs 6.6±6.2 minutes) (P=0.01). The ADHD group also had a significantly longer WASO than the control group (57.4±23.2 minutes vs 30.7±13.7 minutes) (P<0.001). Moreover, the fragmentation index was significantly higher in the ADHD group compared with the control group (16.7±4.5 vs 10.3±4.4) (P<0.001). In contrast, no significant differences between the ADHD and control groups were found in the total sleep time or sleep efficiency measured by actigraphy (Table 2).
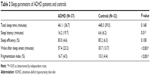  | Table 2 Sleep parameters of ADHD patients and controls |
Relationship between cognitive function and sleep parameters
WASO measured by actigraphy positively correlated with the error rate of response measured by the MFFT in the ADHD patient group (rho =0.52, P=0.012). Moreover, the fragmentation index positively correlated with the response error rate (rho =0.44, P=0.008) and the response latency rate (rho =0.4, P=0.018) in the MFFT (Table 3).
The reaction error rate was significantly associated with the fragmentation index (beta =0.94, P=0.024) (Table 4).
Discussion
In this study, the ADHD patient group did not differ significantly from the control group in total sleep time or sleep efficiency, but they exhibited longer sleep latency, more WASO, and a higher fragmentation index. These results are partially consistent with a large meta-analysis of sleep research in ADHD patients, in which objective sleep measurements using actigraphy showed that ADHD patients have longer sleep latency and shorter total sleep time than controls but are not significantly different from controls with regard to sleep efficiency and WASO.11 The longer sleep latency in this study may explain why the parents of affected children report sleep resistance and sleep initiation problems. However, actigraphy revealed marked differences among studies. For example, in insomnia patients who do not move even when awake, the sleep latency times can be measured as shorter than they actually are. Similarly, sleep apnea patients who have abnormal movements when asleep can have inaccurately measured sleep latency times. Therefore, some studies reported that sleep latency is the most unreliable sleep parameter measured by actigraphy.27,28 Furthermore, a meta-analysis using polysomnography found that the ADHD patient group and the control group were not significantly different in terms of sleep latency. Polysomnography measures the time of sleep latency to sleep stage 2, using the results of EEG, electrooculography, and electromyography, while actigraphy measures sleep latency as the time from lying in bed or turning off the light to the first epoch of inactivity of at least 10 minutes. Thus, sleep latency can be underestimated if patients do not move during the waking state. Because ADHD patients are usually hyperactive, when using actigraphy, sleep latency can be measured to be longer than it actually is.
Sleep efficiency is measured by the rate of the time lying in bed against the total sleep time, determined by actigraphy. In this study, no difference in sleep efficiency between ADHD patients and controls was found. This result corresponds to a previous meta-analysis of results determined by actigraphy.11 However, our findings differ from the previously reported polysomnography results showing that the sleep efficiency rate of ADHD patients is lower than that of controls.11,34 Generally, polysomnography is more efficient than actigraphy for measuring sleep efficiency rate.11 Polysomnography records the time of being awake without movement as nonsleep time, but actigraphy records the time of being awake without movement as sleep time. The fact that ADHD patients have more difficulty in getting up, according to patient and parent reports,13,35 could lead to overestimation of sleep efficacy by actigraphy.
WASO and the fragmentation index were significantly higher in the ADHD patient group. Although no difference in sleep efficiency between ADHD patients and the control group was observed, we predict that ADHD patients’ real sleep time is shorter than that of the control group, on the basis of their significantly longer WASO. This is equivalent to previous actigraphy results showing that ADHD patients’ real sleep time is shorter than that of the control group.11 Moreover, some studies have reported that ADHD patients have more frequent awakenings during sleep.12 Considering all of these results, we suggest that ADHD patients have more and longer sleep fragmentation. A higher fragmentation index indicates that the movement time while sleeping is longer. Previous studies showed that ADHD patients have significantly increased movement during sleep7,36 and periodic limb movement disorder.16,36 Increased movement during sleep does not necessarily mean more frequent awakening during sleep. No research to date has confirmed that increased movement during sleep can cause sleep fragmentation. However, polysomnography studies consistently show that movement and breathing difficulty during sleep exist in a significant number of ADHD patients.7–9 Thus, the relationship between these symptoms and sleep fragmentation should be investigated further.
Because impulsivity is one of the defining characteristics of ADHD, it can be assumed that the ADHD patient group will have a higher response error rate and shorter response latency than the control group in the MFFT, a test designed to measure impulsivity in children. In this study, we found that ADHD patients had a significantly higher response error rate and shorter response latency; these results are consistent with prior research.37
In this study, WASO and the fragmentation index were positively correlated with response error in the MFFT. Also, reaction error rates were associated with fragmentation index. The greater the error rate of response, the more impulsive and less controllable are the children. Just as daytime hyperactivity is accepted as a secondary symptom caused by impulse control difficulty, patients’ hyperactivity also increases nocturnal hyperactivity, resulting in the increased WASO and higher fragmentation index. Similarly, some studies have shown that involuntary movement during sleep positively correlates with impulsivity in combined-type ADHD patients, due to the close relationship between inattention/hyperactivity symptoms and sleep disorders like RLS and PLMD.4,17,38 Nevertheless, the results from other studies on the relationship between ADHD subtype and sleep problems have not been consistent.4,14,38
Some studies have shown that OSA has a strong relationship with hyperactivity symptoms, and chronic night snoring is found more in patients with ADHD, predominant hyperactive-impulsive type.35 However, others have shown that an abnormal breathing pattern is not related to ADHD subtype.38 Furthermore, sleep deprivation can decrease regional cerebral metabolic rate for glucose in the prefrontal cortex,39 resulting in performance disturbance affecting episodic memory encoding, decision making, critical ability, deduction, language, learning, attention, and deep thought.40,41
These dysfunctional areas in sleep deprivation correspond to those of ADHD patients, suggesting that ADHD cognitive problems could be related to sleep problems. Therefore, the correlation between cognitive dysfunction and sleep problems in ADHD is supported by our finding that the increased WASO and fragmentation index are related to the increased response error in the MFFT.
This study has several limitations. We did not perform a structural interview for the diagnosis and not classify with ADHD subtyping. Sample sizes were small, and they consisted of only male patients.
In summary, patients with ADHD had more sleep problems, including increased sleep latency and WASO and fragmentation index compared with controls. Some of these sleep problems, such as increased WASO and the fragmentation index, were related to poor executive function in patients with ADHD. However, further studies with large sample sizes with female patients, classification of ADHD subtypes, and the addition of polysomnographic findings should be performed to confirm our results regarding sleep problems in boys with ADHD.
Disclosure
The authors report no conflict of interest in this work.
References
Frazier TW, Demaree HA, Youngstrom EA. Meta-analysis of intellectual and neuropsychological test performance in attention-deficit/hyperactivity disorder. Neuropsychology. 2004;18(3):543–555. | ||
Greenhill LL, Hechtman LI. Attention-deficit/hyperactivity disorder. In: Sadock BJ, Sadock VA, Ruiz P, editors. Kaplan and Sadock’s Comprehensive Textbook of Psychiatry. 9th ed. Philadelphia, PA: Lippincott Williams and Wilkins; 2009:3560–3571. | ||
Owens JA. The ADHD and sleep conundrum redux: moving forward. Sleep Med Rev. 2006;10(6):377–379. | ||
Corkum P, Moldofsky H, Hogg-Johnson S, Humphries T, Tannock R. Sleep problems in children with attention-deficit/hyperactivity disorder: impact of subtype, comorbidity, and stimulant medication. J Am Acad Child Adolesc Psychiatry. 1999;38(10):1285–1293. | ||
Mick E, Biederman J, Jetton J, Faraone SV. Sleep disturbances associated with attention deficit hyperactivity disorder: the impact of psychiatric comorbidity and pharmacotherapy. J Child Adolesc Psychopharmacol. 2000;10(3):223–231. | ||
Tsai MH, Huang YS. Attention-deficit/hyperactivity disorder and sleep disorders in children. Med Clin North Am. 2010;94(3):615–632. | ||
Konofal E, Lecendreux M, Bouvard MP, Mouren-Simeoni MC. High levels of nocturnal activity in children with attention-deficit hyperactivity disorder: a video analysis. Psychiatry Clin Neurosci. 2001; 55(2):97–103. | ||
Cortese S, Konofal E, Lecendreux M, et al. Restless legs syndrome and attention-deficit/hyperactivity disorder: a review of the literature. Sleep. 2005;28(8):1007–1013. | ||
Gruber R. Sleep characteristics of children and adolescents with attention deficit-hyperactivity disorder. Child Adolesc Psychiatr Clin N Am. 2009;18(4):863–876. | ||
Hvolby A, Jørgensen J, Bilenberg N. Actigraphic and parental reports of sleep difficulties in children with attention-deficit/hyperactivity disorder. Arch Pediatr Adolesc Med. 2008;162(4):323–329. | ||
Cortese S, Faraone SV, Konofal E, Lecendreux M. Sleep in children with attention-deficit/hyperactivity disorder: meta-analysis of subjective and objective studies. J Am Acad Child Adolesc Psychiatry. 2009;48(9): 894–908. | ||
Owens JA, Maxim R, Nobile C, McGuinn M, Msall M. Parental and self-report of sleep in children with attention-deficit/hyperactivity disorder. Arch Pediatr Adolesc Med. 2000;154(6):549–555. | ||
Owens J, Sangal RB, Sutton VK, Bakken R, Allen AJ, Kelsey D. Subjective and objective measures of sleep in children with attention-deficit/hyperactivity disorder. Sleep Med. 2009;10(4):446–456. | ||
O’Brien LM, Holbrook CR, Mervis CB, et al. Sleep and neurobehavioral characteristics of 5- to 7-year-old children with parentally reported symptoms of attention-deficit/hyperactivity disorder. Pediatrics. 2003; 111(3):554–563. | ||
Prihodova I, Paclt I, Kemlink D, Skibova J, Ptacek R, Nevsimalova S. Sleep disorders and daytime sleepiness in children with attention-deficit/hyperactivity disorder: a two-night polysomnographic study with a multiple sleep latency test. Sleep Med. 2010;11(9): 922–928. | ||
Wagner ML, Walters AS, Fisher BC. Symptoms of attention-deficit/hyperactivity disorder in adults with restless legs syndrome. Sleep. 2004;27(8):1499–1504. | ||
Chervin RD, Archbold KH, Dillon JE, et al. Associations between symptoms of inattention, hyperactivity, restless legs, and periodic leg movements. Sleep. 2002;25(2):213–218. | ||
Dillon JE, Blunden S, Ruzicka DL, et al. DSM-IV diagnoses and obstructive sleep apnea in children before and 1 year after adenotonsillectomy. J Am Acad Child Adolesc Psychiatry. 2007;46(11):1425–1436. | ||
Konofal E, Arnulf I, Lecendreux M, Mouren MC. Ropinirole in a child with attention-deficit hyperactivity disorder and restless legs syndrome. Pediatr Neurol. 2005;32(5):350–351. | ||
Corkum P, Tannock R, Moldofsky H. Sleep disturbances in children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 1998;37(6):637–646. | ||
Shur-Fen Gau S, Shur-Fen GS. Prevalence of sleep problems and their association with inattention/hyperactivity among children aged 6–15 in Taiwan. J Sleep Res. 2006;15(4):403–414. | ||
Busby K, Firestone P, Pivik RT. Sleep patterns in hyperkinetic and normal children. Sleep. 1981;4(4):366–383. | ||
Palm L, Persson E, Bjerre I, Elmqvist D, Blennow G. Sleep and wakefulness in preadolescent children with deficits in attention, motor control and perception. Acta Paediatr. 1992;81(8):618–624. | ||
O’Brien LM, Ivanenko A, Crabtree VM, et al. Sleep disturbances in children with attention deficit hyperactivity disorder. Pediatr Res. 2003;54(2):237–243. | ||
Wiebe S, Carrier J, Frenette S, Gruber R. Sleep and sleepiness in children with attention deficit/hyperactivity disorder and controls. J Sleep Res. 2013;22(1):41–49. | ||
Crabtree VM, Ivanenko A, Gozal D. Clinical and parental assessment of sleep in children with attention-deficit/hyperactivity disorder referred to a pediatric sleep medicine center. Clin Pediatr (Phila). 2003;42(9): 807–813. | ||
Sadeh A, Hauri PJ, Kripke DF, Lavie P. The role of actigraphy in the evaluation of sleep disorders. Sleep. 1995;18(4):288–302. | ||
Ustinov Y, Lichstein KL. Actigraphy reliability with normal sleepers. Behav Sleep Med. 2013;11(5):313–320. | ||
Kirov R, Brand S. Sleep problems and their effect in ADHD. Expert Rev Neurother. 2014;14(3):287–299. | ||
Gruber R, Wiebe S, Montecalvo L, Brunetti B, Amsel R, Carrier J. Impact of sleep restriction on neurobehavioral functioning of children with attention deficit hyperactivity disorder. Sleep. 2011;34(3):315–323. | ||
Gruber R, Grizenko N, Schwartz G, Bellingham J, Guzman R, Joober R. Performance on the continuous performance test in children with ADHD is associated with sleep efficiency. Sleep. 2007;30(8):1003–1009. | ||
Kwak KJ, Park HW, Kim CT. [K-WISC-III (Korean Wechsler Intelligence Scale for Children)]. Seoul: Special Education; 2001. | ||
Oh HY. [Standardization of Matching Familiar Figures Test for Korea Children]. Seoul: Sungkyunkwan University Press; 2002. | ||
Miano S, Donfrancesco R, Bruni O, et al. NREM sleep instability is reduced in children with attention-deficit/hyperactivity disorder. Sleep. 2006;29(6):797–803. | ||
LeBourgeois MK, Avis K, Mixon M, Olmi J, Harsh J. Snoring, sleep quality, and sleepiness across attention-deficit/hyperactivity disorder subtypes. Sleep. 2004;27(3):520–525. | ||
Kirov R, Kinkelbur J, Heipke S, et al. Is there a specific polysomnographic sleep pattern in children with attention deficit/hyperactivity disorder? J Sleep Res. 2004;13(1):87–93. | ||
Barkley R. Attention deficit hyperactivity disorder: A handbook for diagnosis and treatment. New York: The Guildford Press; 1990. | ||
Wiggs L, Montgomery P, Stores G. Actigraphic and parent reports of sleep patterns and sleep disorders in children with subtypes of attention-deficit hyperactivity disorder. Sleep. 2005;28(11):1437–1445. | ||
Thomas M, Sing H, Belenky G, et al. Neural basis of alertness and cognitive performance impairments during sleepiness. I. Effects of 24 hours of sleep deprivation on waking human regional brain activity. J Sleep Res. 2000;9(4):335–352. | ||
Curcio G, Ferrara M, De Gennaro L. Sleep loss, learning capacity and academic performance. Sleep Med Rev. 2006;10(5):323–337. | ||
Jones K, Harrison Y. Frontal lobe function, sleep loss and fragmented sleep. Sleep Med Rev. 2001;5(6):463–475. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



