Back to Journals » Cancer Management and Research » Volume 13
Significance of Oncotype DX 21-Gene Test and Expression of Long Non-Coding RNA MALAT1 in Early and Estrogen Receptor-Positive Breast Cancer Patients
Authors Huang Z, Qin Q, Xia L, Lian B, Tan Q, Yu Y, Mo Q
Received 12 August 2020
Accepted for publication 4 November 2020
Published 22 January 2021 Volume 2021:13 Pages 587—593
DOI https://doi.org/10.2147/CMAR.S276795
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Seema Singh
Zhen Huang,* Qinghong Qin,* Longjie Xia,* Bin Lian, Qixing Tan, Yinghua Yu, Qinguo Mo
Department of Breast Surgery, The Affiliated Tumor Hospital of Guangxi Medical University, Nanning, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Qinguo Mo
Department of Breast Surgery, The Affiliated Tumor Hospital of Guangxi Medical University, No. 71 Hedi Road, Nanning 530021 Guangxi Province, People’s Republic of China
Tel +86-771-530859
Email [email protected]
Objective: To investigate the association between the recurrence score (RS) obtained by Oncotype DX 21-gene test and long non-coding RNA (lncRNA) MALAT1 expression in early and estrogen receptor-positive (ER+) breast cancer.
Materials and Methods: The Oncotype DX 21-gene test and MALAT1 expression detection were performed in tumor samples from 76 ER+ and early breast cancer patients with the Surplex liquid chip. The RS value was calculated based on the expression of total 21 genes. The level of MALAT1 was measured in both tumor tissue and para-tumor tissue, and relatively quantified with an internal control gene. Mann–Whitney U-test or Kruskal–Wallis test were used to analyze the association between MALAT1 level and different clinical pathological characteristics, including age, tumor stage, disease grade, lymph node status, Ki-67 expression, and progesterone receptor (PR) status. The association between the RS and different characteristics was analyzed by Wilcoxon rank-sum test. Correlation between two parameters was analyzed by Spearman’s rank correlation analysis.
Results: The expression of MALAT1 was more abundant in tumor tissue (2.992 ± 2.256) than that in adjacent normal tissue (1.641± 1.438, Z=− 2.594, p= 0.009), and it was not correlated with any clinical pathological characteristics. According to the old criteria for RS stratification, 52.7% of patients were in low risk (RS< 18), 36.8% of patients were in medium risk (18≤RS≤ 30), and 10.5% of patients were in high risk (RS> 30). While under the new criteria, 18.4% were in low risk group (RS< 11), 63.2% were in a medium risk group (11≤RS≤ 26), and 18.4% were in a high risk group (RS> 26). The Oncotype DX 21-gene results only correlated with Ki-67 expression under both new and old criteria, and it was not related with other cancer characteristics. The expression of lncRNA MALAT1 was significantly correlated with the Oncotype DX 21-gene results under the old criteria.
Conclusion: MALAT1 is a novel breast cancer biomarker independent of tumor stage, disease grade and lymph node status. MALAT1 level is associated with the Oncotype DX 21-gene RS value. Therefore, combination of MALAT1 and the Oncotype DX 21-gene test may be used to predict prognosis in ER+ and early stage breast cancer.
Keywords: Oncotype DX 21-gene test, MALAT1, long non-coding RNA; lncRNA, breast cancer, risk of recurrence
Introduction
Breast cancer is the second most common and the second leading cause of death in cancer among women.1 Estrogen exposure is considered as the most important risk factor for breast cancer.2 Based on the status of estrogen receptor (ER), breast cancer can be divided into two types, respectively are ER-positive (ER+) and ER-negative (ER−) breast cancer. Due to the highly distinctive etiological and epidemiological characteristics between ER+ and ER− breast cancers, the treatment and prognosis of both types are different. Genetic studies have discovered that the two types of breast cancer have their specific gene expression profiling, and some signature genes associated with tumor metastasis, chemo-resistance, and poor prognosis have been developed to guide clinical treatment.3–7
Oncotype DX 21-gene test is a genomic test that measures 21 cancer-related genes, and it has been shown to have the ability in predicting 10-year distant recurrence in patients with ER+ and axillary lymph node-negative breast cancer. Besides, this genomic test has also been reported to be able to predict tumor response to chemotherapy and endocrine therapy. Under this test, large, prospective and randomized clinical trials are currently underway, and paraffin-embedded blocks instead of previous methods, which require fresh frozen tissue, are used as one of the advantages.7 Many studies have validated the utility of the Oncotype DX 21-gene test in predicting recurrence score (RS) in ER+ breast cancer,8–10 while recent studies have also found that such methods can be applied to predict the outcomes of patients undergoing chemotherapy.11–13 For early breast cancer, the Oncotype DX 21-gene test has been recommended to guide clinical treatment.14
Long non-coding RNAs (lncRNAs) are defined as transcripts longer than 200 nucleotides that have no capability of protein-coding.15 LncRNAs are a potentially important regulator in many biological processes in physiology and disease, especially those relevant to endocrinology, reproduction, metabolism, immunology, neurobiology, muscle biology, and cancer.16 Many lncRNA molecules have been found dysregulated in breast cancer tissue, including metastasis-associated lung adenocarcinoma transcript 1 MALAT1).17 It has been reported that the expression of MALAT1 is increased in numerous types of tumor and it exerts a significant role in regulating tumor cell proliferation, migration, invasion and apoptosis.18 Besides, MALAT1 performs an important role in the pathological alteration of organs which are associated with sex hormones and several reproductive system cancers. MALAT1is considered as a biomarker for the diagnosis and prognosis prediction of cancers, and may also serve as a therapeutic target for treatment of specific tumors.19
In this study, we enrolled 76 ER+ and early breast cancer patients who were first diagnosed in our clinic. Oncotype DX 21-gene test and MALAT1 expression detection in those patients were performed by using their biopsy samples. It was found that MALAT1 level was increased in tumor tissue and its level was independent of age, tumor stage, disease grade, lymph node status and progesterone receptor (PR) status of the patients. Interestingly, the Oncotype DX 21-gene test results were only correlated with Ki-67 expression, while independent of disease stage, tumor grade, and lymph node status. Overall, MALAT1 level is associated with the Oncotype DX 21-gene, and combination of the two may be promising in predicting the prognosis of ER+ and early breast cancer.
Materials and Methods
Study Design
This study was carried out with the approval of the Institutional Review Board of the Affiliated Tumor Hospital of Guangxi Medical University. Breast cancer patients who were initially diagnosed in our hospital from March 1, 2016 to November 31, 2017 were screened for this study, and the patients who met the following criteria were enrolled: 1) ER+ and human epidermal growth factor receptor 2-negative (HER2−) breast cancer; 2) Stage T1-T2 breast cancer; 3) Negative or with one lymph node infiltration (0–1); and 4) no sex and age limitations. Eventually, a total of 76 female patients were enrolled and all the patients had signed Informed Consent.
Oncotype DX 21-Gene Test and MALAT1 lncRNA Expression Examination
The Oncotype DX 21-gene test was performed via the SurPlexTM liquidchip technology by SurExam Institute (Guangzhou, China). Briefly, paraffin-embedded tissue blocks were digested by tissue lysis buffer for mRNA isolation. Target mRNA was captured by a gene-specific probe labeled with magnetic beads. PCR was performed by using the magnetic beads enriched mRNA as template. The level of the target mRNA was calculated based on fluorescence signal by Luminex. The 21 genes are comprised of proliferation-related genes (Ki67, STK15, BIRC5, CCNB1, MYBL2), metastasis-related genes (MMP11, CTSL2), HER2-related genes (GRB7, HER2), sex hormone-related genes (ER, PGR, BCL2, SCUBE2, GSTM1, BAG1, CD68) and internal control genes (ACTB, GAPDH, GUS, RPLPO and TFRC). RS was calculated based on the Oncotype Dx® formula. According to old criteria, RS<18 represents low risk, 18≤RS≤30 represents mediate risk, and RS>30 represents high risk. After modification, a new type of criteria has been developed: RS<11 refers to low risk, 11≤RS≤26 refers to mediate risk, and RS>26 refers to high risk. Since it is not clear which standard is more accurate to represent cancer status, both criteria were referenced in this study.
MALAT1lncRNA expression was measured by quantitative real-time PCR. Total RNA was firstly extracted from tumor and para-tumor samples. Complementary DNA (cDNA) was synthesized by using the SuperScript® Reverse Transcriptase (Invitrogen). The SYBR® Green PCR Master Mix (Applied Biosystems) was employed for the real-time PCR and the reaction was performed with the StepOnePlus™ Real-Time PCR System (Applied Biosystems). The expression of MALAT1 was normalized with RPLPO as the internal control.
Statistical Analysis
The comparisons of MALAT1 expression among different sub-groups were analyzed by Mann–Whitney u-test or Kruskal–Wallis test. Association between two parameters was analyzed by Spearman’s rank correlation analysis. SPSS 16.0 software was used for all the analysis. P<0.05 was considered significant.
Results
General Information of All the Subjects
Breast cancer patients who were initially diagnosed in our hospital from March 1st, 2016 to November 31st, 2017 were screened for this study. Patients who met the following criteria were enrolled: 1) ER+ and HER2− breast cancer; 2) Stage T1-T2 breast cancer; 3) negative or with one lymph node infiltration (0–1); and 4) no sex and age limitations. Totally, 76 female breast cancer patients were selected, with the average age of 49.4-year-old (36- to 69-years-old). Among the subjects, 35.5% (27 of 76) patients were postmenopausal. All the patients were diagnosed as ER+ breast cancer with lymph node infiltration (0 or 1), and received radical mastectomy with axillary lymph node dissections. Based on the Oncotype DX 21-gene test, the patients were treated with ER+ antagonist or chemotherapy after surgery.
Expression of MALAT1 in Breast Cancer Tissue
MALAT1 is a novel cancer biomarker and it is involved in multiple steps of gene regulation in cancer cells. It is established that MALAT1 expression is increased in breast cancer patients, yet whether it is increased in ER+ and early breast cancer remains unclear. Therefore, we examined the expression of MALAT1 in our ER+ breast cancer samples by quantitative real-time PCR. The MALAT1 expression level was normalized using an internal control gene, and presented as relative expression.
In order to find the clinical relevance of MALAT1 to ER+ breast cancer, we did an association analysis between MALAT1 expression and different characteristics of breast cancer, including age, tumor stage, disease grade, lymph node status, Ki-67 expression, and PR status. Surprisingly, MALAT1 level was not related with any of above characteristics. As shown in Table 1, MALAT1 level was independent of age (2.918±2.671 in age<50 vs. 3.101±1.497 in age>50, p=0.124), tumor stage (2.885±2.071 in T1 vs. 3.199±2.608 in T2, p=0.728), lymph node status (2.952±2.285 in N0 vs. 3.293±2.129 in N+, p=0.381), disease grade (2.782±2.209 in Grade Ⅱ vs. 3.939±2.650 in Grade ш, p=0.217), Ki-67 level (2.513±1.829 in cancers with low Ki-67 vs. 3.273±2.446 in cancers with high Ki-67, p=0.258), and PR status (2.990±2.257 in PR+ cancers vs. 3.018±2.468 in PR− cancers, p=0.847).
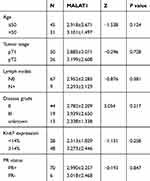 |
Table 1 MALAT1 Expression in Different Subgroups of Breast Cancer Patients |
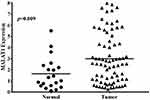
In order to evaluate the expression of MALAT1 in tumor and normal tissues, we isolated RNA from tumor and para-tumor samples of all the patients, and quantitative real-time PCR was used. As shown in Figure 1, MALAT1 level was much higher in tumor tissue than that in para-tumor tissue (2.992±2.256 in tumor tissue vs. 1.641±1.438 in para-tumor tissue, p<0.05). Collectively, the result above indicated that MALAT1 may be an independent biomarker for ER+ and early breast cancer.
Oncotype DX 21-Gene Test and Clinical Characteristics of Breast Cancer
According to the old criteria of the Oncotype DX 21-genetest, 52.7% patients (40 of 76) had RS below 18, which means low risk of recurrence; 36.8% patients (28 of 76 patients) had RS between 18 and 30, which means medium risk of recurrence; 10.5% patients (8 of 76) had RS over 30, which means high risk of recurrence. Additionally, it was noted that the RS value was not related to age, tumor stage, lymph node status, and PR status of patients with breast cancer (Table 2), but it was significantly associated with disease grade (Z=7.628, p=0.022) and Ki-67 expression (Z=7.628, p=0.022).
 |
Table 2 Association Between the RS of the Oncotype DX 21-Gene Test and Breast Cancer Clinical Characteristics |
Recently, the old criteria have been modified into a new one for better prognosis prediction.20 In the new criteria, RS below 11 refers to low risk, RS over 26 refers to high risk, and RS between 11 and 26 refers to medium risk. A re-analysis was conducted according to the new criteria. It turned out that 18.4% patients (14 of 76) were with low risk of recurrence, 63.2% patients (48 of 76) were in medium risk, and 18.4% patients (14 of 76) were in high risk. Moreover, the RS was noted to be not significantly associated with age, lymph node status, disease grade, and PR status, while associated with tumor stage (Z=−2.374, p=0.018) and Ki-67 expression (Z=−3.892, p<0.001).
Although there is a recommendation recently to use the new criteria to predict breast cancer prognosis, it is still unclear which standard is better. Therefore, both criteria were used in our analysis. We found that the RS obtained via the Oncotype DX 21-gene test was shown to be associated with Ki-67 expression under the both two standards, but not related with other pathological characteristics of breast cancer.
Association Between the Oncotype DX 21-Gene Test and MALAT1 Expression
MALAT1 is a very important molecule that regulates gene expression in many types of cancer. As aforementioned, MALAT1 level was significantly increased in breast cancer tissue compared with that in para-tumor tissue. Therefore, we also analyzed the association between the Oncotype DX 21-genetest and MALAT1 expression. Firstly, we used the old criteria for analysis. We found that MALAT1 level was increased along with RS. Specifically, MALAT1 expression was 2.286 ± 2.012 in low risk group, 3.576± 2.111 in medium risk group, and 4.484± 2.840 in high risk group (p=0.006). Secondly, we applied the new criteria for analysis, and we found that MALAT1 level was also increased along with RS, yet no statistically significant difference was noted when comparing 2 groups (p=0.195).
Discussion
Breast cancer is the second largest cancer that mainly affects women in United States and it is also the second leading cause of death in all types of cancer. The incidence of breast cancer in recent ten years has been significantly increasing, while the mortality rate has been decreasing, which is largely due to the wide use of screening procedures for early breast cancer, contributing to more early cancers identified and a better chance to perform surgical resection. Mastectomy as the main treatment procedure for breast cancer can remove a large area of breast tissue, usually including axillary lymph nodes as well, on the basis of tumor stage. Nevertheless, this procedure alone may be detrimental to patients both physiologically and psychologically. Therefore, it is important to minimize the damage of surgery while treating patients.
To achieve this goal, we need to predict the prognosis of breast cancer in each individual patient. The Oncotype DX 21-gene test is a genomic test that analyzes the activity of a group of genes to reflect how a cancer is likely to behave and responds to treatment. The Oncotype DX 21-gene test can be used to evaluate the risk of recurrence in ER+ breast cancer patients, and to assess how likely the patients to benefit from chemotherapy after surgery. The results of the Oncotype DX 21-gene test combined with other features of the cancer will help doctor make a more informed decision about whether or not to apply chemotherapy to treat early-stage, ER+ breast cancer or radiotherapy to treat DCIS.
In this study, we enrolled 76 newly diagnosed ER+ and early breast cancer patients, and we did the Oncotype DX 21-gene test before treatment. Based on the RS values, the risk of recurrence of the patients could be divided into three groups: low risk, medium risk and high risk. When the old criteria were considered, 52.7% patients were in low risk group, 36.8% patients were in medium risk group and 10.5% patients were in high-risk group. The distribution of each group is similar to a previous reported study.21 In that study, there were 74,778 breast cancer patients enrolled and the Oncotype DX 21-gene test found that the percentages of patients in low risk, medium risk and high risk groups were 59.5%, 32.0%, and 8.5%, respectively.21 Given the results, we found that the RS was associated with disease grade and Ki-67 expression, but not associated with other characteristics of the cancer such as age, tumor stage, lymph node status, and PR status. When the new criteria were applied, the RS was noted to be correlated with tumor stage and Ki-67 expression level, but not correlated with other factors. Although it is still unclear which criteria should be used for data analysis, the Oncotype DX 21-gene test result was consistently correlated with Ki-67 expression level under both criteria, suggesting that the Oncotype DX-21 gene test may reflect tumor proliferation in our ER+ early-stage breast cancer patients.
LncRNA MALAT1 is a biomarker for many types of cancer, and it regulates several critical pathways involved in cancer metastasis.22–24 It has been reported that high level of MALAT1 is associated with poor relapse-free survival in breast cancer patients.25 In our study, we measured MALAT1 expression level in our collected breast cancer samples, and found that the expression of MALAT1 was much higher in tumor tissue compared with that in para-tumor tissue. Additionally, it was observed that the MALAT1 expression was independent of age, tumor stage, disease grade, lymph node status, PR status and Ki-67 expression (Table 3), suggesting that MALAT1 is an independent biomarker for breast cancer.
 |
Table 3 Association Between the RS of the Oncotype DX 21-Gene Test and lncRNA MALAT1 Expression |
In order to test whether the combination of MALAT1 and the Oncotype DX21-gene test is of value in predicting prognosis of breast cancer, we firstly analyzed the association between MALAT1 and the Oncotype DX21-gene test result. Interestingly, there was significant association between the two when the old criteria were applied, but no association was observed from the results under the new criteria, suggesting the importance of RS definition. Therefore, further studies shall need to be carried out to clarify the puzzle. Notably, our study only included 76 breast cancer samples, which limits the research on value of such biomarkers. In order to clarify the association of MALAT1 expression to prognosis of early breast cancer in a more correct and meaningful manner, sample size should be enlarged and more indexes should be considered in further research, such as Tailor-Rx or MINDACT.
To sum up, our study identified MALAT1 as a novel biomarker for breast cancer, and discovered that there was an association between MALAT1 expression and RS value of the Oncotype DX 21-gene test. Clinically, MALAT1 expression examination plus the Oncotype DX 21-gene test can be used to predict the prognosis of ER+ and early breast cancer.
Ethical Approval
We that this study was conducted in accordance with the Declaration of Helsinki. This study was approved by hospital ethical committee. All participants were informed consent.
Acknowledgment
Equal contribution/first author: Zhen Huang, Qinghong Qin, and Longjie Xia.
Funding
This study was supported by Nature Science Foundation of Guangxi (2017GXNSFAA198088) and the Self-financing Project of Guangxi Health and Family Planning Commission (Z2015613) and Youth Foundation of Guangxi Medical University (GXMUYSF201405).
Disclosure
The authors report no conflicts of interest for this work.
References
1. McGuire A, Brown JA, Malone C, McLaughlin R, Kerin MJ. Effects of age on the detection and management of breast cancer. Cancers. 2015;7:908–929. doi:10.3390/cancers7020815
2. Kulkoyluoglu-Cotul E, Arca A, Madak-Erdogan Z. Crosstalk between estrogen signaling and breast cancer metabolism. Trends Endocrinol Metab. 2018. doi:10.1016/j.tem.2018.10.006
3. Li Z, Zhang Y, Zhang Z, Zhao Z, Lv Q. A four-gene signature predicts the efficacy of paclitaxel-based neoadjuvant therapy in human epidermal growth factor receptor 2-negative breast cancer. J Cell Biochem. 2018. doi:10.1002/jcb.27891
4. Buechler SA, Gokmen-Polar Y, Badve SS. EarlyR signature predicts response to neoadjuvant chemotherapy in breast cancer. Breast. 2018;43:74–80. doi:10.1016/j.breast.2018.11.006
5. Aalders KC, Kuijer A, Straver ME, et al. Characterisation of multifocal breast cancer using the 70-gene signature in clinical low-risk patients enrolled in the EORTC 10041/BIG 03-04 MINDACT trial. Eur J Cancer. 2017;79:98–105. doi:10.1016/j.ejca.2017.03.034
6. Wallden B, Storhoff J, Nielsen T, et al. Development and verification of the PAM50-based Prosigna breast cancer gene signature assay. BMC Med Genomics. 2015;8:54. doi:10.1186/s12920-015-0129-6
7. Cobleigh MA, Tabesh B, Bitterman P, et al. Tumor gene expression and prognosis in breast cancer patients with 10 or more positive lymph nodes. Clin Cancer Res. 2005;11:8623–8631. doi:10.1158/1078-0432.CCR-05-0735
8. Tang G, Shak S, Paik S, et al. Comparison of the prognostic and predictive utilities of the 21-gene Recurrence Score assay and Adjuvant! For women with node-negative, ER-positive breast cancer: results from NSABP B-14 and NSABP B-20. Breast Cancer Res Treat. 2011;127:133–142. doi:10.1007/s10549-010-1331-z
9. Albanell J, Gonzalez A, Ruiz-Borrego M, et al. Prospective transGEICAM study of the impact of the 21-gene Recurrence Score assay and traditional clinicopathological factors on adjuvant clinical decision making in women with estrogen receptor-positive (ER+) node-negative breast cancer. Ann Oncol. 2012;23:625–631. doi:10.1093/annonc/mdr278
10. Kelly CM, Bernard PS, Krishnamurthy S, et al. Agreement in risk prediction between the 21-gene recurrence score assay (Onco type DX®) and the PAM50 breast cancer intrinsic classifier™ in early-stage estrogen receptor–positive breast cancer. Oncol. 2012;17:492–498. doi:10.1634/theoncologist.2012-0007
11. Chen XH, Zhang WW, Wang J, et al. 21-gene recurrence score and adjuvant chemotherapy decisions in patients with invasive lobular breast cancer. Biomark Med. 2018. doi:10.2217/bmm-2018-0396
12. Khan MA, Henderson L, Clarke D, Harries S, Jones L. The Warwick experience of the Oncotype DX(R) Breast Recurrence Score(R) assay as a predictor of chemotherapy administration. Breast Care. 2018;13:369–372. doi:10.1159/000489131
13. Geyer CE, Tang G, Mamounas EP, et al. 21-gene assay as predictor of chemotherapy benefit in HER2-negative breast cancer. NPJ Breast Cancer. 2018;4:37. doi:10.1038/s41523-018-0090-6
14. Green N, Al-Allak A, Fowler C. Benefits of introduction of Oncotype DX((R)) testing. Ann R Coll Surg Engl. 2019;101:55–59. doi:10.1308/rcsann.2018.0173
15. Perkel JM. Visiting “noncodarnia”. BioTechniques. 2013;54:301, 303–304. doi:10.2144/000114037
16. Sun M, Kraus WL. From discovery to function: the expanding roles of long noncoding RNAs in physiology and disease. Endocr Rev. 2015;36:25–64. doi:10.1210/er.2014-1034
17. Klinge CM. Non-coding RNAs in breast cancer: intracellular and intercellular communication. Non Coding RNA. 2018;4:40. doi:10.3390/ncrna4040040
18. Zhao M, Wang S, Li Q, Ji Q, Guo P, Liu X. MALAT1: a long non-coding RNA highly associated with human cancers. Oncol Lett. 2018;16:19–26. doi:10.3892/ol.2018.8613
19. Li ZX, Zhu Q-N, Zhang H-B, et al. MALAT1: a potential biomarker in cancer. Cancer Manag Res. 2018;10:6757–6768. doi:10.2147/CMAR.S169406
20. Kim I, Choi HJ, Ryu JM, et al. A predictive model for high/low risk group according to oncotype DX recurrence score using machine learning. Eur J Surg Oncol. 2019;45:134–140. doi:10.1016/j.ejso.2018.09.011
21. Jha MK, Lee S, Park DH, et al. Diverse functional roles of lipocalin-2 in the central nervous system. Neurosci Biobehav Rev. 2015;49:135–156. doi:10.1016/j.neubiorev.2014.12.006
22. Ji DG, Guan L-Y, Luo X, et al. Inhibition of MALAT1 sensitizes liver cancer cells to 5-flurouracil by regulating apoptosis through IKKalpha/NF-kappaB pathway. Biochem Biophys Res Commun. 2018;501:33–40. doi:10.1016/j.bbrc.2018.04.116
23. Chen R, Liu Y, Zhuang H, et al. Quantitative proteomics reveals that long non-coding RNA MALAT1 interacts with DBC1 to regulate p53 acetylation. Nucleic Acids Res. 2017;45:9947–9959. doi:10.1093/nar/gkx600
24. Yuan J, Xu XJ, Lin Y, et al. LncRNA MALAT1 expression inhibition suppresses tongue squamous cell carcinoma proliferation, migration and invasion by inactivating PI3K/Akt pathway and downregulating MMP-9 expression. Eur Rev Med Pharmacol Sci. 2019;23:198–206. doi:10.26355/eurrev_201901_16765
25. Wang Z, Katsaros D, Biglia N, et al. High expression of long non-coding RNA MALAT1 in breast cancer is associated with poor relapse-free survival. Breast Cancer Res Treat. 2018;171:261–271. doi:10.1007/s10549-018-4839-2
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.