Back to Journals » Drug Design, Development and Therapy » Volume 12
Short-term efficacy of intravitreal conbercept in treatment-naive patients with polypoidal choroidal vasculopathy
Authors Peng Y , Zhang X, Li M, Liu B , Mi L, Zuo C, Wen F
Received 29 November 2017
Accepted for publication 27 December 2017
Published 19 February 2018 Volume 2018:12 Pages 339—345
DOI https://doi.org/10.2147/DDDT.S158368
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Tuo Deng
Yuting Peng, Xiongze Zhang, Miaoling Li, Bing Liu, Lan Mi, Chengguo Zuo, Feng Wen
State Key Laboratory of Ophthalmology, Zhongshan Ophthalmic Center, Sun Yat-sen University, Guangzhou, China
Introduction: To evaluate the functional and morphological outcomes of intravitreal conbercept monotherapy in patients with polypoidal choroidal vasculopathy (PCV).
Materials and methods: In this retrospective, observational case series study, we reviewed medical records of 48 eyes (48 patients) with naive PCV that were treated with a series of 3 monthly intravitreal injections of 0.5 mg of conbercept followed by as-needed injections (3+pro re nata). All patients completed at least 6 months of monthly follow-up. Changes in the best-corrected visual acuity, optical coherence tomography, and indocyanine green angiography were retrospectively evaluated.
Results: At 6 months, the mean best-corrected visual acuity significantly improved from 0.89±0.35 (20/160 in Snellen equivalent) at baseline to 0.58±0.26 (Snellen equivalent of 20/80; P<0.001), and 60.42% (29/48) of eyes had an improvement of three lines of vision; the mean central retinal thickness significantly decreased from 333.56±171.04 µm at baseline to 187.65±54.46 µm (P<0.001), and 93.75% (45/48) achieved a dry macula. At 3 months, 6 of 32 eyes (18.75%) showed partial regression of branching vascular network, 14 of 32 (43.75%) patients showed complete resolution of polyps. The mean number of injections was 3.4±0.9 through 6 months. No conbercept-related systemic or ocular adverse effects were observed.
Conclusion: Intravitreal injection of conbercept using “3+pro re nata” regimen significantly improved visual acuity and anatomical outcomes in treatment-naive patients with PCV.
Keywords: conbercept, intravitreal injection, PCV, short-term efficacy, “3+PRN”
Introduction
Age-related macular degeneration (AMD) is the leading cause of irreversible blindness in elderly people worldwide1 and accounts for a large proportion of neovascular AMD in Asians.2–8 Given aging Asian populations, it is estimated even more people will eventually suffer from polypoidal choroidal vasculopathy (PCV). Compared with neovascular AMD, PCV exhibits a reduced prevalence of bilateral disease (range 5.9%–24.1%) and a marked preponderance in men (range 63.0%–78.5%).9 Half of all patients with PCV showed recurrences and eventual visual loss9 indicating the urgent need for effective PCV treatments.
Anti-vascular endothelial growth factor (VEGF) treatment is now the first line of PCV therapy. The most commonly used drugs are bevacizumab, ranibizumab, and aflibercept; bevacizumab and ranibizumab block the same receptor binding domain of all VEGF-A isoforms.10 Conbercept was tested in a Phase III clinical trial and approved to treat neovascular AMD by the China State Food and Drug Administration in December 2013. Conbercept has strong antiangiogenetic effects and binds all isoforms of VEGF-A and VEGF-B as well as placental growth factor with high neutralizing affinity.11,12 Conbercept is similar to aflibercept in structure; while compared with aflibercept, conbercept contains the fourth Ig-like domain of VEGFR-2, which is essential for receptor dimerization and enhances VEGF association with the receptor. Conbercept has a higher affinity for VEGF than aflibercept.13 Early studies reported that both ranibizumab and bevacizumab stabilized vision and reduced exudation in PCV patients, but limited effects on polypoidal lesions or choroidal vascular changes were noted.14–16 The rate of complete polypoidal lesion regression was ~10%14 with intravitreal bevacizumab, ~20%17,18 with intravitreal ranibizumab, and ~54.8%–72.5%19,20 with aflibercept injections.
Bevacizumab has been used in an off-label manner, which is a large limitation. Aflibercept is effective in the eyes for treatment-naive PCV to achieve polypoidal lesion resolution. Our previous study demonstrated that conbercept significantly improved visual and anatomical outcomes in choroidal neovascularization secondary to punctate inner choroidopathy.21 Conbercept produced significant increases in best-corrected visual acuity (BCVA) and reductions in central retinal thickness (CRT) at 3 and 12 months of neovascular AMD.22 In addition to visual improvement and a significant CRT decrease, a relatively high rate of polypoidal lesion resolution and/or branching vascular network (BVN) was observed in PCV patients.23 Few studies have reported about the efficacy of “3+ pro re nata (PRN)” regimen intravitreal conbercept monotherapy for PCV.
In this study, we retrospectively evaluated the efficacy of intravitreal conbercept injection using a “3+PRN” regimen for treatment-naive PCV during the 6-month follow-up period.
Materials and methods
Study design
This study was a retrospective, observational case series. This study was performed at the Zhongshan Ophthalmic Center of Sun Yat-sen University.
Patients and study population
We reviewed the medical records of 48 eyes of 48 consecutive patients, with symptomatic, treatment-naive PCV, who received intravitreal conbercept injections at the Macula Service of Zhongshan Ophthalmic Center of Sun Yat-sen University between November 2014 and June 2016. Thirty-two of 48 patients were reexamined with indocyanine green angiogram (ICGA) after three continuous monthly intravitreal injections.
The study group was composed of those patients who met the inclusion and exclusion criteria. The inclusion criteria included the following: 1) treatment-naive PCV characterized by the presence of polyps with or without BVN in posterior pole on ICGA, with subretinal fluid (SRF) or intraretinal fluid (IRF) observed on optical coherence tomography (OCT) or leakage on fluorescein angiography; 2) received three continuous monthly intravitreal injections of 0.5 mg conbercept (KH902; Chengdu Kanghong Biotech Ltd., Sichuan, China) followed by as-needed injections (3+PRN; reinjection was considered if any intraretinal or subretinal fluid was observed after the third injection); 3) underwent intravitreal conbercept injections as the primary treatment for PCV and completed at least 6 months of monthly follow-up exams after the first treatment. Exclusion criteria included the following: 1) angioid streaks, multifocal choroiditis, punctate inner choroidopathy, pathologic myopia, retinal angiomatous proliferation, or other diseases that can cause choroid neovascularization; 2) the presence of any other ocular diseases that might affect visual acuity; and 3) received any other treatment, including thermal laser photocoagulation, submacular surgery, intravitreal of any other anti-VEGF drugs, and photodynamic therapy.
Ethics approval and informed consent
Ethics approval was obtained from the medical ethics board of Zhongshan Ophthalmic Center, Sun Yat-sen University, and the approval number is 2014MEKY011. The study was in adherence to the tenets of the Declaration of Helsinki, and as deemed exempt from written informed consent by the medical ethics board of Zhongshan Opthalmic Center, because it used only retrospective, de-identified patient data.
Main observation index
Visual outcomes included the mean BCVA change in logarithm of the minimum angle of resolution (logMAR) and the proportion that improved, stabilized, or deteriorated compared with the baseline. An improvement of ≥0.3 in logMAR visual acuity was defined as improved VA, an improvement of <0.3 in logMAR visual acuity was defined as stabilized VA, and a decrease of ≥0.3 logMAR visual acuity was defined as deteriorated VA. A change of 0.1 logMAR was considered a change of one line. Anatomical outcomes were evaluated by OCT and ICGA. SRF or IRF or pigment epithelial detachment (PED) build-up was evaluated based on OCT at first and sixth months’ visit. The regression of polyps or BVN, greatest linear dimension (GLD), total lesion area (TLA), polyp largest diameter (PLD), and polyp lesion area (PLA) were assessed based on ICGA at baseline and the visit after three continuous injections. The regression of polyps or BVN was assessed by ICGA24: 1) complete regression – no polyps or BVN observed on the imaging; 2) partial regression – ≥10% decrease in polyp area or BVN area compared with baseline; 3) no change – <10% change in polyp area or BVN area compared with baseline; and 4) ≥10% increase in polyp area or BVN area compared with baseline. The BCVA and CRT were measured monthly. A “dry macula” was defined as the disappearance of subretinal or intraretinal spaces by OCT.
Statistical analyses
All the data are expressed as the mean ± SD. Statistical analyses were performed with the Statistical Package for the Social Sciences (SPSS Version 20.0, IBM Corporation, Chicago, IL, USA). BCVA was converted to the logMAR equivalent for statistical analysis. To compare the mean BCVA at each time point with the BCVA at baseline, a paired t-test was used. The PLD at baseline and at 3 months were compared using a paired t-test. CRT at baseline and at months 1, 2, 3, 4, 5, and 6 as well as the mean changes in GLD, TLA, and PLA at baseline and at month 3 were compared using Wilcoxon signed-rank test. A P-value of <0.05 was considered statistically significant.
Results
Baseline characteristics
In total, 48 eyes from 48 patients were analyzed. The clinical details of eyes from patients are listed in Table 1. Briefly, the patients had a mean age of 63.33±7.42 (range, 50–83) years and were predominantly male (33 male, 68.75%). Of the 48 treated eyes, 43 (89.58%) exhibited macular PCV, whereas the remaining 5 (10.42%) showed peripapillary PCV. In total, 95.83% (46/48) of patients exhibited BVN. BCVA at baseline was 0.89±0.35. GLD, TLA, PLD, and PLA at baseline were 3.24±1.57 mm, 7.36±7.14 mm2, 301.66±115.04 μm, and 0.39±0.46 mm2, respectively. CRT at baseline was 333.56±171.04 μm. In addition, 97.92% (47/48) of patients had SRF, 31.25% (15/48) had IRF, and 45.83% (22/48) had PED.
Visual outcome changes after intravitreal conbercept monotherapy for PCV
The mean BCVA at 1, 2, 3, 4, 5, and 6 months after the initial injection were 0.75±0.30, 0.64±0.27, 0.59±0.26, 0.58±0.26, 0.58±0.26, and 0.58±0.26, respectively. Vision gain from baseline at each monthly visit was −0.14±0.15, −0.25±0.19, −0.30±0.19, −0.31±0.20, −0.31±0.20, and −0.31±0.20, respectively; the mean BCVA at 1, 2, 3, 4, 5, and 6 months decreased significantly (P<0.001 for all points). Vision gain from baseline is presented in Figure 1.
After assessing BCVA at each follow-up visit, we divided the patients into three groups: improved, stabilized, and deteriorated. The mean BCVA improved in 60.42% (29/48) and stabilized in 39.58% (19/48) of patients at 6 months. No patients showed deteriorated BCVA.
Anatomical changes after intravitreal conbercept monotherapy for PCV
OCT outcomes
The mean CRT values at 1, 2, 3, 4, 5, and 6 months after the initial injection were 253.44±90.44, 206.17±68.30, 192.88±60.73, 192.10±57.60, 191.88±57.77, and 187.65±54.46 μm, respectively. The mean CRT at 1, 2, 3, 4, 5, and 6 months decreased significantly compared with baseline (P<0.001 for all points). The changes in CRT are shown in Figure 2.
The OCT characteristics at baseline and at 6 months are shown in Table 2. At baseline, 97.92% (47/48) of eyes exhibited SRF, 31.25% (15/48) of eyes had IRF, and 45.83% (22/48) of eyes had serous and/or hemorrhagic PED; at 6 months, the values were 6.38% (3/47), 0% (0/15), and 59.09% (13/22), respectively. In other words, the exudate fading rate was 93.62% (44/47) in eyes with SRF. In addition, 100% (15/15) of eyes with IRF and 40.91% (9/22) of eyes with PED resolved completely. In total, 93.75% (45/48) of eyes had a dry macula at 6 months.
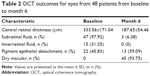  | Table 2 OCT outcomes for eyes from 48 patients from baseline to month 6 |
ICGA outcomes
There were 32 patients with ICGA images available at 3 months. In total, 43.75% (14/32) showed complete polyp regression, 34.38% (11/32) showed partial polyp regression, 15.62% (5/32) demonstrated no change, and 6.25% (2/32) exhibited increased polyps. Thus, 78.13% of eyes with polypoidal lesions had good response to conbercept. All the 32 patients had BVN at baseline, but none of the patients exhibited complete BVN regression. In addition, 18.75% (6/32) exhibited partial regression, 75% (24/32) showed no change, and 6.25% (2/32) increased at 3 months.
The GLD of the 32 patients decreased from 3.17±1.56 to 3.00±1.60 mm (P=0.003). TLA decreased from 6.82±7.37 to 6.55±6.95 mm2 (P=0.035), PLD decreased from 300.84±114.74 to 173.84±181.05 μm (P<0.001), and PLA decreased from 0.47±0.54 to 0.33±0.77 mm2 (P=0.001).
Treatment
The mean number of injections through 6 months was 3.4±0.9 (range, 3–6). Thirty-nine patients (81.25%) did not require additional injections, because no recurrence of exudation was observed after three consecutive monthly injections. A total of 162 injections were administered. In total, nine patients (18.75%) required additional injections. Among them, four eyes required three extra injections, one eye needed two additional injections, the remaining four eyes required one extra injection.
Ocular/systemic complications
No patient developed systemic complications, such as a cerebrovascular episode, because of intravitreal conbercept injections. No ocular complications, including increased intraocular pressure, cataracts, endophthalmitis, retinal detachment, or RPE tears, were detected.
Discussion
It has been reported that conbercept is an effective treatment not only for patients with choroidal neovascularization secondary to punctate inner choroidopathy,21 neovascular AMD,22 macular edema secondary to branch retinal vein occlusion,25 but also for patients with PCV.23 In this retrospective study, treatment-naive PCV patients treated with conbercept using a “3+PRN” method over 6 months of follow-up visits resulted in a significant improvement in visual acuity and anatomical results.
Conbercept has a structure similar to aflibercept but a higher affinity for all isoforms of VEGF-A. Various studies have demonstrated that intravitreal injection of conbercept or aflibercept reduced foveal thickness, improved or stabilized visual acuity in eyes with PCV, and exhibited a relatively high rate of complete or partial regression of polyps.23,26–29 For polyp regression, using drugs with multiple anti-VEGF targets (conbercept and aflibercept) is superior to single-target anti-VEGF drugs (bevacizumab and ranibizumab).28,29
In our study, we analyzed 48 patients with PCV treated with conbercept monotherapy and found that BCVA improved significantly and was maintained for at least 6 months, and 60.42% (29/48) eyes had vision improvement of three lines. The CRT declined from baseline at 333.56±171.04 to 187.65±54.46 μm by the sixth month visit (P<0.001). The results suggested that conbercept significantly decreased the CRT of patients with PCV and are similar to those reported in previous studies.22,23 At the sixth month follow-up visit, the exudation fading rate was 93.62% (44/47) in eyes with SRF. In addition, 100% (15/15) of eyes with IRF and 40.91% (9/22) of eyes with PED resolved completely. In 93.75% (45/48) of eyes, the maculae were dry. Ijiri and Sugiyama26 reported the effect of 3 monthly aflibercept injections in 33 PCV patients, and at the third month visit, 31 eyes (97%) achieved dry macula as evaluated by OCT. These findings indicated that conbercept monotherapy was effective at absorbing subretinal or IRF and driving PED regression.
In addition, the closure of polypoidal lesions has traditionally been considered an important endpoint in PCV treatment because achieving this angiographic endpoint implies the possibility to stop treatment until recurrence occurs. Polyp regression does not necessarily indicate a good vision prognosis.30 Nevertheless, the polypoidal lesion can cause recurrent subretinal hemorrhage and SRF. And these secondary changes can result in visual impairment. Therefore, polyp regression is important for maintaining long-term visual function. In a subgroup analysis of the Aurora Study,23 in the 0.5-mg group, 13 (46.4%) patients showed complete polyp regression and 11 (39.3%) demonstrated partial regression after three monthly injections. In our study, these values were 43.75% and 34.38%, respectively. The rate of polyp regression was similar to that reported in previous studies using conbercept and aflibercept.23,26,27 However, the rate was enhanced compared with that reported for bevacizumab or ranibizumab.31,32 The rate of polyp regression might be related to pharmacological characteristics of conbercept, such as an increased affinity for VEGF and an increased half-life compared with those of ranibizumab or bevacizumab. Furthermore, conbercept binds to VEGF-B and placental growth factor in addition to VEGF-A, whereas ranibizumab binds only to VEGF-A. The simultaneous inhibition of multiple VEGF family molecules might contribute to the enhanced treatment responses to conbercept observed in PCV patients. In the 0.5-mg group in the Aurora Study, 34.4% (11/32) of eyes showed BVN at baseline. BVN partially regressed in 72.7% (8/11) of patients, and only one patient exhibited complete BVN regression at 3 months. In our study, all 32 patients had BVN at baseline. In addition, 18.75% (6/32) of eyes with BVN exhibited partial regression, and no patients experienced complete BVN regression at 3 months. The presence of BVN was an important prognostic factor that determined the success rate of PCV.33 Compared with the Aurora Study, ours included a greater proportion of patients with BVN. This difference might explain the slightly lower regression rate of polyps and BVN, although inconsistent criteria might also explain these differences. Conbercept was a more effective treatment than ranibizumab on polypoidal lesions and BVN.34 Although some differences in results were noted, these studies have confirmed that conbercept contributed to polyp and BVN regression. In our study, the GLD, TLA, PLD, and PLA significantly decreased from baseline to 3 months, and the P-values were 0.003, 0.035, <0.001, and 0.001, respectively. Recently, a study35 with a minimum 3-year follow-up found that the largest polyp diameter at baseline significantly correlated with PCV recurrence. No recurrences were significantly correlated with a better long-term visual outcome.36
The limitations of the current study include short follow-up period and retrospective non-comparative study; some patients had not been reviewed with ICGA at the third month visit. Thus, further prospective, multicenter, and controlled study with long-term follow-up is needed to validate our findings.
Conclusions
Our findings suggest that intravitreal injections of conbercept using 3+PRN can significantly improve visual acuity and anatomical morphology for at least 6 months in eyes with naive PCV. Intravitreal conbercept might increase the possibility of achieving involution of polyps and BVN.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgment
This work was supported by the National Natural Science Foundation of China (grant number 81470647) and the Fundamental Research Funds of State Key Laboratory of Ophthalmology.
Disclosure
The authors report no conflicts of interest in this work.
References
Lim LS, Mitchell P, Seddon JM, Holz FG, Wong TY. Age-related macular degeneration. Lancet. 2012;379(9827):1728–1738. | ||
Ciardella AP, Donsoff IM, Huang SJ, Costa DL, Yannuzzi LA. Polypoidal choroidal vasculopathy. Surv Ophthalmol. 2004;49(1):25–37. | ||
Wen F, Chen C, Wu D, Li H. Polypoidal choroidal vasculopathy in elderly Chinese patients. Graefe’s Archive Clin Experimental Ophthalmol. 2004;242(8):625–629. | ||
Liu Y, Wen F, Huang S, et al. Subtype lesions of neovascular age-related macular degeneration in Chinese patients. Graefes Arch Clin Exp Ophthalmol. 2007;245(10):1441–1445. | ||
Maruko I, Iida T, Saito M, Nagayama D, Saito K. Clinical characteristics of exudative age-related macular degeneration in Japanese patients. Am J Ophthalmol. 2007;144(1):15–22. | ||
Byeon SH, Lee SC, Oh H, et al. Incidence and clinical patterns of polypoidal choroidal vasculopathy in Korean patients. Jpn J Ophthalmol. 2008;52(1):57–62. | ||
Chang Y, Wu W. Polypoidal Choroidal Vasculopathy in Taiwanese Patients. Ophthalmic Surgery Lasers Imaging. 2009;40(6):576–581. | ||
Lim LS, Cheung CMG, Wong TY. Asian age-related macular degeneration. Asia Pac J Ophthalmol (Phila). 2013;2(1):32–41. | ||
Wong CW, Yanagi Y, Lee W, et al. Age-related macular degeneration and polypoidal choroidal vasculopathy in Asians. Prog Retin Eye Res. 2016;53:107–139. | ||
Ferrara N, Damico L, Shams N, Lowman H, Kim R. Development of ranibizumab, an anti–vascular endothelial growth factor antigen binding fragment, as therapy for neovascular age-related macular degeneration. Retina. 2006;26(8):859–870. | ||
Zhang M, Zhang J, Yan M, Li H, Yang C, Yu D. Recombinant anti-vascular endothelial growth factor fusion protein efficiently suppresses choroidal neovasularization in monkeys. Mol Vis. 2008;14:37–49. | ||
Wang Q, Li T, Wu Z, et al. Novel VEGF decoy receptor fusion protein conbercept targeting multiple VEGF isoforms provide remarkable anti-angiogenesis effect in vivo. PLoS One. 2013;8(8):e70544. | ||
Zhang M, Zhang J, Yan M, et al. A phase 1 study of KH902, a vascular endothelial growth factor receptor decoy, for exudative age-related macular degeneration. Ophthalmology. 2011;118(4):672–678. | ||
Gomi F, Sawa M, Sakaguchi H, et al. Efficacy of intravitreal bevacizumab for polypoidal choroidal vasculopathy. Brit J Ophthalmol. 2007;92(1):70–73. | ||
Oishi A, Miyamoto N, Mandai M, et al. LAPTOP study: a 24-month trial of verteporfin versus ranibizumab for polypoidal choroidal vasculopathy. Ophthalmology. 2014;121(5):1151–1152. | ||
Hikichi T, Kitamei H, Shioya S. Prognostic factors of 2-year outcomes of ranibizumab therapy for polypoidal choroidal vasculopathy. Brit J Ophthalmol. 2015;99(6):817–822. | ||
Hikichi T, Ohtsuka H, Higuchi M, et al. Improvement of angiographic findings of polypoidal choroidal vasculopathy after intravitreal injection of ranibizumab monthly for 3 months. Am J Ophthalmol. 2010;150(5):674–682. | ||
Ueno C, Gomi F, Sawa M, Nishida K. Correlation of indocyanine green angiography and optical coherence tomography findings after intravitreal ranibizumab for polypoidal choroidal vasculopathy. Retina. 2012;32(10):2006–2013. | ||
Lee JE, Shin JP, Kim HW, et al. Efficacy of fixed-dosing aflibercept for treating polypoidal choroidal vasculopathy: 1-year results of the VAULT study. Graefes Arch Clin Exp Ophthalmol. 2017;255(3):493–502. | ||
Oshima Y, Kimoto K, Yoshida N, et al. One-year outcomes following intravitreal aflibercept for polypoidal choroidal vasculopathy in Japanese patients: the APOLLO study. Ophthalmologica. 2017;238(3):163–171. | ||
Peng Y, Zhang X, Mi L, et al. Efficacy and safety of conbercept as a primary treatment for choroidal neovascularization secondary to punctate inner choroidopathy. BMC Ophthalmol. 2017;17(1):87. | ||
Li X, Xu G, Wang Y, et al. Safety and Efficacy of Conbercept in Neovascular Age-Related Macular Degeneration: results from a 12-month randomized phase 2 study: AURORA study. Ophthalmology. 2014;121(9):1740–1747. | ||
Qu J, Cheng Y, Li X, Yu L, Ke X; AURORA Study Group. Efficacy of intravitreal injection of conbercept in polypoidal choroidal vasculopathy. Retina. 2016;36(5):926–937. | ||
Koh A, Lee WK, Chen L, et al. EVEREST Study: efficacy and safety of verteporfin photodynamic therapy in combination with ranibizumab or alone versus ranibizumab monotherapy in patients with symptomatic macular polypoidal choroidal vasculopathy. Retina. 2012;32(8):1453–1464. | ||
Li F, Sun M, Guo J, Ma A, Zhao B. Comparison of conbercept with ranibizumab for the treatment of macular edema secondary to branch retinal vein occlusion. Curr Eye Res. 2017;42(8):1174–1178. | ||
Ijiri S, Sugiyama K. Short-term efficacy of intravitreal aflibercept for patients with treatment-naïve polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol. 2015;253(3):351–357. | ||
Koizumi H, Kano M, Yamamoto A, et al. Aflibercept therapy for polypoidal choroidal vasculopathy: short-term results of a multicentre study. Brit J Ophthalmol. 2015;99(9):1284–1288. | ||
Yamamoto A, Okada AA, Kano M, et al. One-year results of intravitreal aflibercept for polypoidal choroidal vasculopathy. Ophthalmology. 2015;122(9):1866–1872. | ||
Hara C, Sawa M, Sayanagi K, Nishida K. One-year results of intravitreal aflibercept for polypoidal choroidal vasculopathy. Retina. 2016;36(1):37–45. | ||
Oishi A, Kojima H, Mandai M, et al. Comparison of the effect of ranibizumab and verteporfin for polypoidal choroidal vasculopathy: 12-month LAPTOP study results. Am J Ophthalmol. 2013;156(4):644–651. | ||
Cho HJ, Baek JS, Lee DW, Kim CG, Kim JW. Short-term effectiveness of intravitreal bevacizumab vs. ranibizumab injections for patients with polypoidal choroidal vasculopathy. Korean J Ophthalmol. 2012;26(3):157–162. | ||
Cho HJ, Kim KM, Kim HS, et al. Intravitreal aflibercept and ranibizumab injections for polypoidal choroidal vasculopathy. Am J Ophthalmol. 2016;165:1–6. | ||
Wong IY, Shi X, Gangwani R, et al. 1-year results of combined half-dose photodynamic therapy and ranibizumab for polypoidal choroidal vasculopathy. BMC Ophthalmol. 2015;15(1):66. | ||
Kokame GT, Yeung L, Lai JC. Continuous anti-VEGF treatment with ranibizumab for polypoidal choroidal vasculopathy: 6-month results. Brit J Ophthalmol. 2010;94(3):297–301. | ||
Kang HM, Koh HJ, Lee SC. Baseline polyp size as a potential predictive factor for recurrence of polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol. 2016;254(8):1519–1527. | ||
Kang HM, Koh HJ. Long-term visual outcome and prognostic factors after intravitreal ranibizumab injections for polypoidal choroidal vasculopathy. Am J Ophthalmol. 2013;156(4):652–660. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



