Back to Journals » Nature and Science of Sleep » Volume 12
Sex-Related Differences in Sleep-Related PSG Parameters and Daytime Complaints in a Clinical Population
Authors Van Eycken S, Neu D , Newell J, Kornreich C , Mairesse O
Received 31 October 2019
Accepted for publication 9 February 2020
Published 19 February 2020 Volume 2020:12 Pages 161—171
DOI https://doi.org/10.2147/NSS.S235642
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Sutapa Mukherjee
Sebastien Van Eycken, 1,* Daniel Neu, 1–3,* Johan Newell, 1 Charles Kornreich, 1, 2 Olivier Mairesse 1, 4, 5
1Sleep Laboratory and Unit for Chronobiology U78, Department of Psychiatry, Brugmann University Hospital, Université Libre de Bruxelles (ULB) and Vrije Universiteit Brussel (VUB), Brussels, Belgium; 2UNI Neuroscience Institute, ULB312 Faculty of Medicine, and ULB388 Faculty of Motor Sciences, Université Libre de Bruxelles (ULB), Brussels, Belgium; 3Center for the Study of Sleep Disorders, Delta Hospital, Neuroscience Pole and Department of Internal Medicine, CHIREC, Brussels, Belgium; 4Department of Brain Body and Cognition (BBCO), Vrije Universiteit Brussel (VUB), Brussels, Belgium; 5Department LIFE, Royal Military Academy, Brussels, Belgium
*These authors contributed equally to this work
Correspondence: Daniel Neu; Olivier Mairesse
Sleep Laboratory and Unit for Clinical Chronobiology U78, Department of Psychiatry, Brugmann University Hospital, Université Libre de Bruxelles (ULB) and Vrije Universiteit Brussel (VUB), Arthur Van Gehuchten Square, Building Hh, Brussels 1020, Belgium
Tel +32 2 477 21 62
Fax +32 2 477 25 54
Email [email protected]; [email protected]
Background: Recent research suggested that perception of sleep impairments might present sex-related effects (ie, women appear to be more prone to report fatigue rather than sleepiness). The latter has been evidenced in sleep-related breathing disorders (SRBD). Differently, it has been suggested that sleep-related movement disorders may also be associated to fatigue rather than to sleepiness. Whether sex-related differences would be similar irrespective of diagnosis remains unclear.
Methods: During a one-year period, systematic clinical evaluation, by means of structured symptom scales, was performed for a cohort of 921 consecutive patients attending an academic sleep center for polysomnography. The Brugmann Fatigue Scale (BFS), an instrument designed for the assessment of rest propensity was used among other scales (ie, Epworth Sleepiness Scale, ESS). According to inclusion and exclusion criteria, 420 men and 376 women were finally included in the study and retained for data analysis.
Results: While men and women presented with similar age, BMI, total sleep time and sleep efficiency, men presented with higher levels of respiratory events and more periodic limb movements. Irrespective of diagnosis, women presented with significantly higher levels of sleep-associated complaints on all scales. Comparative stratifications of daytime symptoms, per diagnostic groups (SRBD, Movement Disorders (SRMD) and Insomnia), revealed significant main effects for diagnosis alongside with main effects of biological sex. Associations between common markers of disease severity for SRBD or SRMD and sleep or rest propensity, respectively, only showed significant correlation between periodic limb movements and rest propensity. The strength of association was similarly significant for both sexes.
Conclusion: While men displayed more objective impairment on polysomnography (PSG) and lower symptom levels, the opposite was true in women. However, both men and women present with statistically significant associations between SRMD severity (PLMS index) and physical fatigue.
Keywords: sleep-related sex effects, fatigue, rest propensity, sleepiness, sleep propensity
Brief Summary
Current knowledge: Recent research suggests that differences in subjective sleepiness and fatigue may be related to biological sex. It remains unclear whether these differences are dependent on diagnosis or due to methodological issues such as the lack of instruments sharing a similar conceptual approach to both concepts, such as behavioral impact vs symptom severity.
Study impact: Our results provide evidence for differences in symptom-expression based on sex and diagnosis. They also suggest that movement disorders may be related to fatigue rather than to sleepiness.
Introduction
By definition, sleepiness is typically considered to be a signal for the spontaneous onset of sleep and therefore refers to sleep propensity.1,2 Fatigue on the other hand, as weariness, is described as a condition in which sustaining motor or mental activities degrees gets increasingly difficult in acute, intense and energy-demanding tasks.3,4 In contrast to sleepiness and sleep propensity, which can be reduced to the approximation of a single objective dimension (ie, sleep onset latency), a consensually accepted objective measure of fatigue is still lacking. Consequently, the clinical evaluation of fatigue remains largely based on symptom assessments.
Complaints of fatigue rather than sleepiness present with sex-related effects with respect to the prevalence of sleep disorders5–7 and irrespective of sleep disorder diagnosis.8,9 It has been showed recently that, in comparison to men, women with Obstructive Sleep Apnea (OSA) tend to present more often with fatigue rather than with sleepiness, while the opposite was true for men,9 suggesting that, next to sleepiness, fatigue assessments in order to apprehend the experience of women. Thorough studies on large clinical cohorts, comprising both, a systematic and structured clinical evaluation alongside with a polysomnography for each participant are however mostly lacking.
On the other hand, fatigue related to physical rest propensity, in particular, has been shown to be higher in Sleep-Related Movement Disorder (SRMD) compared to Sleep-Related Breathing Disorders (SRBD).10 Statistically significant correlations between perceived sleep quality and/or daytime fatigue (but not sleepiness) and polysomnographic variables such as the Periodic Limb Movements during Sleep Index (PLMSI) have also been mentioned.10,11 PLMSI >15 are more prevalent in men12 in general population-based samples13 and in comparison to good sleeper controls, patients with sleep bruxism show higher levels of rest propensity (fatigue) but not of sleep propensity (sleepiness).14 Whether suggested higher rest propensity levels in SRMD (than in SRBD) and its association with objectively observable sleep parameters (such as the PLMSI) stand against findings of biological sex effects remains yet to be determined. The above-mentioned considerations are of clinical relevance, given that fatigue and sleepiness are potentially independent consequences of sleep disorders15 and relate to distinct conditions.3,4,16,17
The aims of the present study are thus: (1) to confirm sex-related effects of sleep complaints disregarding diagnostic categories; (2) to confirm higher intensity of fatigue symptoms and rest propensity levels in SRMD in comparison to SRBD disorders; and (3) to further explore within SRMD patients, the former suspected relations between the behavioural impact of fatigue and polysomnography-derived disease severity.
Methods
Participants
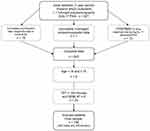
After a period of one year, we sampled 796 hypnotic-free individuals (Mage= 47.66; SDage=12.79) outside a cohort of 921 patients, addressed to the sleep unit of a university hospital (Figure 1). Primary care referred patients, with complaints of sleepiness, fatigue, non-restorative sleep or a combination of these symptoms were included. Sleep disorders such as Sleep-Related Breathing (SRBD), Movement (SRMD) and Insomnia Disorders, (ID) were categorized according to ICSD-3 criteria.18 Four groups were defined: SRBD with a Respiratory Disturbance Index (RDI) larger or equal to 15 and a Periodic Limbic Movement during Sleep index (PLMSI) lower or equal to 15; SRMD with a clinical diagnosis of restless legs (RLS) or PLMSI larger than 15 and an RDI lower than 15; ID with a clinical diagnosis of insomnia, RDI lower than 15 and a PLMSI lower or equal to 15 and unspecified sleep disorder (USD) being the remainders (not fulfilling the criteria of the three aforementioned groups). All patients in our lab completed questionnaires about lifestyle and drinking habits. Naps before PSG and during the hospitalisation stay were not permitted. Preparation of the patients for the PSG recordings was between 22:00 and 23:00. With a view to elude treatment trials (eg, continuous positive airway pressure, etc.), we analysed only first-night PSG (see Figure 1). The protocol was allowed by the ethical committee of Brugmann hospital and was in compliance with the Declaration of Helsinki. Consent was waived due to the retrospective nature of the study, although privacy of the participants was respected, because data were anonymized.
Material
PSG recordings included at least three electroencephalograms recorded from Fp2-Ax, C4-Ax, O2-Ax sites, two electrooculogram, submental and bilateral anterior tibial electromyograms. Oral and nasal airflow were recorded by an oro-nasal cannula (Pro-Flow PlusTM Pro-Tech® Mukilteo, WA, USA), respiratory effort was measured by thoracic and abdominal belts (Pro-Tech® CT2TM, Mukilteo, WA, USA). Capillary oxygen saturation was monitored by photosensitive finger-oxymetry (Nonin® Flexi-Form® II 7000A Nonin Medical Inc, Minneapolis, MN USA and LINOP® Adt Masimo corp. Irvine, CA, USA). All PSG recordings were analyzed on 22’ screens displaying 30-second polysomnograph epochs (Philips Respironics IncTM Alice6®, Philips HealthcareTM, Eindhoven, the Netherlands, European Union) by trained technicians unaware of the aims of the study.
Polysomnography
Sleep records were scored using conventional criteria from the American Academy of Sleep Medicine (AASM) criteria18 with sleep efficiencies (SE) defined by the ratio between TST and TIB (SE1) or between TST and Sleep Period Time (SPT= TIB–SOL) (SE2).
Clinical Scales
The Epworth Sleepiness Scale (ESS),1,19 the Brugmann Fatigue Scale (BFS),10 the Pittsburgh Sleep Quality (PSQI)20 and Hospital Anxiety and Depression rating scale (HADRS)21 were assessed in order to evaluate intensity of clinical complaints.
Statistics
Group differences for nominal variables were computed using Χ2 tests. Violations of normality for continuous variables were assessed by means of Kolmogorov–Smirnov tests.
Biological sex comparisons were carried out by means of independent sample t-tests. Comparisons between sexes and diagnostic groups were performed by means of multivariate analyses of variance (MANOVA – Pillai’s Trace). Capitalization on chance was addressed by applying Bonferroni corrections. Associations between continuous variables were computed using Pearson’s moment r. Hypotheses tests were performed 2-sided at the 5% significance level. Analyses were performed by means IBM SPSS 25® (Industrial Business Machines, SPSSTM Inc., Armonk, NY, USA) and Fisher r-to-z transformations were calculated via Vassarstats (VassarStats® Statistical Computation).
Results
Biological Sex Differences in Demographics, Polysomnography and Daytime Symptoms (Table 1)
Irrespective from diagnostic group, age and BMI are similar for both sexes. Sleep fragmentation (ArI), respiratory disturbance (Apnea–Hypopnea Index (AHI), RDI, Oxygen Desaturation Index (ODI)) and limb movement (PLMSI) indices are significantly lower in female than in male patients (p<0.001 for ArI, AHI, RDI and ODI and p<0.05 for PLMSI, respectively). In contrast, female patients show significantly higher levels of slow-wave sleep (N3) and significantly longer REM sleep latencies (REMLAT) (p<0.05 in both cases). Females also report significantly higher symptom intensities on all instruments, namely regarding affective symptoms (HADA: p<0.001; HADD: p<0.05), sleep propensity (ESS: p<0.05), mental (BFSψ: p<0.001) and physical (BFSφ: p<0.001) rest propensities and sleep quality alteration (PSQI: p<0.001).
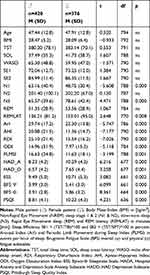 |
Table 1 Descriptive Variables Comparing Male and Female Patients (PSG and Symptom Scales) |
Biological Sex Proportions and Symptom Intensities Across Diagnostic Groups (Figures 2 and 3)
Biological sex proportions significantly differ between diagnostic groups (χ2 (3, N= 792)= 34.992, p<0.001). Within our sample, the prevalence of ID is significantly higher in female patients (66.1%; (χ2 (1, N= 792) = 21.723), p<0.001) and the prevalence of SRBD is significantly higher in male patients (65.8%; (χ2 (1, N= 792)= 22.192), p<0.001). All other diagnostic group proportions are similar for both sexes (Figure 2). Both age and BMI significantly differ between sexes across diagnostic groups (F(6,1562)= 4.370, p<0.001). In SRBD and ID, females are significantly older than males (respectively, F(1,227)= 9.307, p<0.005 and F(1,125)= 10.553, p<0.005). Males with SRBD, present with a significantly higher BMI (F(1,227)= 5.292, p<0.05) (Figure 2). Consequently, age and BMI are included as covariates in all following comparisons.
For all symptom scales (Figure 3), we observe a significant main effect of biological sex (all p’s<0.005, except for the HADD [p<0.05]), with higher symptom intensities observed in women. Except for the BFSψ (p= 0.531), a main effect of diagnostic group is observed for all other symptom scales (all p’s<0.05). Differences in symptom intensities between sexes are largely independent of diagnostic group, as all omnibus interaction tests turned out non-significant (all p’s > 0.05). Conform to Wei et al,22 post hoc analyses were performed and revealed that anxiety symptoms (HADA), are significantly higher in female patients in all diagnostic groups (all p’s<0.01), except for ID. Patients with SRMD present with comparable anxiety symptom intensity individuals with SRBD or ID, but higher than in unspecified sleep-related conditions (p<0.05). However, females with USD report higher depressive symptom intensities compared to males (p<0.01). Depressive symptom intensities are also higher in SRMD compared to SRBD and USD (respectively, p<0.05 and p<0.001). Yet, on average, both depressive symptom intensities remain below clinical thresholds, irrespective of biological sex or diagnostic group. Females presenting with SRBD or SRMD report significantly worse perceived sleep quality (PSQI) compared to males within the same respective groups (respectively, p<0.015 and p<0.05). Patients with ID in general present with higher sleep quality complaints well above clinical thresholds on average and significantly higher than all other diagnostic groups (all p’s<0.001). Additionally, PSQI scores are significantly higher in patients with SRMD compared to the USD group (p<0.001). Regarding the ESS, females in the USD group report higher levels of sleep propensity compared to males (p<0.05). Patients with ID on the other hand report significantly less sleep propensity compared to patients with SRBD or SRMD (respectively, p<0.05 and p<0.001). The latter group presents also with higher sleep propensity than patients with unspecified sleep disorders (p<0.01). Biological sex effects are observed for mental rest propensity (BFSΨ), being significantly higher in females for USD (p<0.001), SRBD (p<0.05), and SRMD (p<0.01). Physical rest propensity (BFSΦ) is systematically significantly larger in females than in males, regardless of diagnostic groups (all p’s<0.001 except for SRBD [p<0.05]). Additionally, physical sleep propensity is higher in SRMD compared to SRBD and USD (respectively, p<0.001 and p<0.05).
Associations Between Indices of SRBD and SRMD and Sleep and Rest Propensity Across Sexes
Pearson’s product-moment correlations revealed only a significant association between the PLMSI and physical rest propensity (BFSΦ; r= 0.117, p<. 005). The strength of association is similar for both sexes (z= 0.26; pr-to-z= 0.397) (see Figure 4).
Discussion
Sex-related differences with respect to sleep are a very contemporary concern. A recent meta-analysis provided evidence about polysomnographic parameters in healthy adult men and women, also considering the effects of age.23 Biological sex differences with respect to prevalence of sleep disorders24 and with respect to sleep-related daytime symptoms in OSA9 have also been suggested by previous reports. Likewise, Chervin et al24 also suggested in an untreated OSAS sample that women report higher levels of sleepiness, fatigue, tiredness and “lack of energy”. The former clinical studies, investigating sex-related effects in daytime symptoms, do often not provide full polysomnography-derived data9 and are mainly, if not solely, focused on SRBDs such as OSA.9,24 Systematic and structured symptom assessments in larger patient cohorts, covering several diagnostic categories, remain sparse. Consequently, the primary aim of our study was to extend and confirm sex-related differences with respect to sleep parameters and daytime complaints in a clinical sample addressed to a general sleep center. Overall, the most striking result here is that, irrespective from post hoc diagnosis and controlling for age and BMI, women with sleep-related complaints report higher symptom intensities (on any scale) than males. These differences are particularly pronounced for fatigue (ie, physical and mental rest propensity) and for anxiety symptoms.
Given the phenomenological differences between fatigue and sleepiness and the prior focus on sex differences with respect to fatigue severity and sleepiness, we assessed sleep and rest propensities for the first time, by means of similarly constructed instruments (rest and sleep propensities, respectively). In addition, recent research suggested that like ID25 and in contrast to SRBD, SRMD may also predominantly present with daytime fatigue rather than with sleepiness. Whether the formerly suspected fatigue is a main daytime symptom of SRMD that would stand against suggested sex effects (as observable in SRBD) was unknown. Therefore, the secondary objective of the present study was to compare the differences of daytime symptoms between sexes with respect to diagnosis and PSG-based markers of disease severity, while considering possibly interfering prevalence disparity and eventually controlling for age and BMI. In the absence of an unequivocal and unanimously accepted objective PSG-derived marker for ID, comparisons of associations between diagnostic category defining sleep parameters and sleep or rest propensity, respectively, had to be limited to SRMD and SRBD (Figure 4).
When comparing sleep parameters between male and female patients of similar age and BMI, irrespective of diagnosis, our results confirm previously suggested biological sex effects with respect to sleep and sleep disorder prevalence. In our sample, male patients present with significantly higher levels of sleep fragmentation, significantly greater severity of respiratory disturbance and significantly more limb movements. Hypotheses about sex differences in SRBD prevalence or severity mainly proposed hormonal causes and average anatomical differences26 Although our finding of higher PLMSI in men also corroborates previous reports12 clear-cut explaining hypotheses (as for SRBD) are generally lacking. Likewise here, biological sex differences have also previously been reported for increased slow-wave sleep7,27 and increased REM latencies23 in women of similar age than men.
In contrast to what has been suggested before,9 women appear to not only complain of more fatigue. When comparing symptom intensities, women tend to systematically show statistically significantly higher levels on all administered instruments. However, clinical interpretations and adjustments are required for the description and contextual understanding of several statistical findings within symptom level comparisons. The numerical difference of sleepiness (ESS) is of no clinical significance and is on average at best borderline for both sexes with respect to any common threshold. Similarly, mean depressive symptom levels remain below clinical thresholds for both male and female patients. Anxiety symptoms, while also being of borderline clinical significance in women, show however numerical differences of potential relevance here. The ‘gonadic hypothesis’ suggested that oestrogen fluctuations may affect several brain regions involved in mood regulation. This may make women more susceptible to stress and more prone to present affective symptoms in general.28 Although women showed significantly greater global daytime impairment, perceived sleep quality was clinically altered (with respect to PSQI thresholds) for both sexes among our sample. The most outstanding clinical difference was though observed with respect to fatigue and rest propensity levels. Only female patients systematically presented with a clinically significant behavioural impact (need and tendency for rest) of daytime fatigue. The latter may suggest a tendency of women to present symptomatic daytime fatigue irrespective of diagnosis that is different than for men.
Hence, etiological hypotheses about these sex-related differences (in sleep and symptoms) mainly ground on immunological responses,29,30 neurobiological differences and anatomical variation.29,31 Whether female patients over-report sleep-related complaints or male patients under-report them remains, of course, to be determined in the light of diagnosis-defining PSG parameters being above thresholds in both sexes.
In order to further understand the potential interplay of biological sex and sleep disorder, we compared symptom levels between sex for each diagnostic subgroup. The biological sex prevalence across diagnostic categories is in line with the available literature. Respective male-to-female ratios of approximatively 2:1 for SRBD and 1:2 for ID are similar to common references.25,32 While controlling for age and BMI, we observed statistically significant main effects for sex (excepted for depression symptoms) and diagnostic group (excepted for mental rest propensity). Hence, mental fatigue, in particular, underlines an exclusively sex-related effect (irrespective of diagnostic group). None of the diagnostic groups showed higher symptom intensities for men. Even statistically similar levels between sexes did not show numerically higher levels for any assessed daytime symptom in male patients. Given that all interaction tests displayed non-significant results, it is confirmed that differences of daytime symptom intensities are hence largely independent of diagnosis. Diagnostic group differences (of symptoms) emerge in largely expected ways, such as severely impaired sleep quality in insomnia (eg, in comparison to SRBD or SRMD). Males and females with insomnia present generally with similar symptom intensities, except for physical rest propensity with males falling on average below clinical thresholds and females presenting elevated scores.
Although physical rest propensity is higher in females across all diagnostic categories, it also is significantly higher in SRMD in comparison to SRBD or USD, for both sexes. While fatigue severity has previously been found to be associated to PLMSI disregarding sex distribution, the latter is underlined here by a similarly significant association between disease severity (PLMSI) and physical rest propensity for both male and female patients.
Limitations of the present study mainly rely on three issues. (1) The setting of the study. Real-life constraints implied here the use of first night derived polysomnographic data. Conducting repeated PSG on several consecutive nights are preferable, however, building a similarly large cohort would not only be very cost intense but also require a (very) long study time period. (2) Absence of unequivocal markers for ID. Likewise for SRBD and SRMD, classical explorations of associations between diagnosis-defined PSG variables and daytime symptoms could not be performed for ID. (3) High levels of comorbidity (combined sleep disorders) on one hand and low prevalence of other specific sleep disorders on the other hand led to the remainder group of USD here. However, USD had similar sex proportions and similar demographics (age, BMI) for both sexes. In addition, with respect to sex-related symptom level distribution, USD reflected similar patterns than the other diagnostic groups.
Conclusion
At last, our results show that in a clinical sample of male and female of similar total sleep time, sleep efficiency ratios, age and BMI, markers of disease severity for SRBD and SRMB tend to be on average significantly higher in men than in women. In contrast to these objective PSG-derived parameters, sex comparison of daytime symptoms show a totally opposite pattern. Women systematically display significantly higher levels on all clinical scales. However, the clinical relevance of the latter emerges essentially for fatigue and rest propensity. In addition, neither sleep nor mental rest propensity did show significant correlation associations to the severity of SRBD (RDI) or SRMD (PLMSI). Only physical rest propensity was significantly correlated to one marker (PLMSI) of disease (SRMD). This association was similarly strong in both men and women.
Abbreviations
AHI, Apnea–Hypopnea Index; ArI, Arousal Index; BFS, Brugmann Fatigue Scale; BFS Ψ, Brugmann Fatigue Scale (mental rest propensity subscale); BFS Φ, Brugmann Fatigue Scale (physical rest propensity subscale); BMI, Body Mass Index; ESS, Epworth Sleepiness Scale; FSS, Fatigue Severity Scale; HADRS, Hospital Anxiety and Depression Rating Scale; HADA, Hospital Anxiety and Depression – Anxiety subscale; HADD, Hospital Anxiety and Depression – Depression subscale; ID, Insomnia Disorders; ODI, Oxygen Desaturation Index; OSA, obstructive sleep apnea; PLM, Periodic Limb Movements; PLMSI, Periodic Limb Movements during Sleep Index; PSG, Polysomnography; PSQI, Pittsburgh Sleep Quality Index; RDI, Respiratory Disturbance Index; REM, Rapid Eye Movement; REMLAT, REM sleep latencies; RERA, respiratory effort-related arousal; RLS, restless legs syndrome; SE, sleep efficiency; SOL, sleep onset latency; SRBD, sleep-related breathing disorders; SRMD, sleep-related movement disorders; SWS, slow-wave sleep; TIB, time in bed; TST, total sleep time; USD, unspecified sleep disorder; WASO, wake after sleep onset.
Acknowledgments
An adapted version of the abstract of this paper has been presented at the 15th World Sleep Congress (Vancouver, Canada, September 20–25, 2019) as a poster presentation with interim findings. The poster’s abstract was published in “Poster Abstracts” in Volume 64, supplement 1 of Sleep medicine: https://doi.org/10.1016/j.sleep.2019.11.773.
Author Contributions
DN, SVE, OM: script writing and data analyses. SVE, JN: data collection and review. DN, OM and CK: supervision, logistics and review. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Johns MW. Sleep propensity varies with behaviour and the situation in which it is measured: the concept of somnificity. J Sleep Res. 2002;2:3–15.
2. Mairesse O, De Valck E, Quanten S, et al. Sleepiness phenomics: modeling individual differences in subjective sleepiness profiles. Int J Psychophysiol. 2014;93:150–161. doi:10.1016/j.ijpsycho.2013.03.021
3. Shen J, Barbera J, Shapiro CM. Distinguishing sleepiness and fatigue: focus on definition and measurement. Sleep Med Rev. 2006;10:63–76. doi:10.1016/j.smrv.2005.05.004
4. Neu D, Linkowski P, Le Bon O. Clinical complaints of daytime sleepiness and fatigue: how to distinguish and treat them, especially when they are ‘excessive’ or ‘chronic’?. Acta Neurol Belg. 2010;110:15–25.
5. Mallampalli MP, Carter CL. Exploring sex and gender differences in sleep health: a Society for Women’s Health Research Report. J Womens’ Health. 2014;23(7):553–562. doi:10.1089/jwh.2014.4816
6. Krishnan V, Collop NA. Gender differences in sleep disorders. Cur Opin Pulm Med. 2006;12(6):383–389. doi:10.1097/01.mcp.0000245705.69440.6a
7. Neu D, Mairesse O, Verbanck P, Linkowski P, Le Bon O. Non-REM sleep EEG power distribution in fatigue and sleepiness. J Psychosom Res. 2014;76:286–291. doi:10.1016/j.jpsychores.2014.02.002
8. Hinz A, Glaesmer H, Brähler E, et al. Sleep quality in the general population: psychometric properties of the Pittsburgh Sleep Quality Index, derived from a German community sample of 9284 people. Sleep Med. 2017;30:57–63. doi:10.1016/j.sleep.2016.03.008
9. Eliasson AH, Kashani MD, Howard RS, Vernalis MN, Modlin RE. Fatigued on venus, sleepy on Mars-gender and racial differences in symptoms of sleep apnea. Sleep Breath. 2015;19(1):99–107. doi:10.1007/s11325-014-0968-y
10. Mairesse O, Damen V, Newell J, Kornreich C, Verbanck P, Neu D. The Brugmann Fatigue Scale: an Analogue to the Epworth Sleepiness Scale to measure behavioral rest propensity. Behav Sleep Med. 2017;24:1–22.
11. Hardy De Buisseret FX, Mairesse O, Newell J, Verbanck P, Neu D. While isolated periodic limb movement disorder significantly impacts sleep depth and efficiency, co-morbid restless leg syndrome mainly exacerbates perceived sleep quality. Eur Neurol. 2017;77(5–6):272–280. doi:10.1159/000471920
12. Haba-Rubio J, Marti-Soler H, Marques-Vidal P, et al. Prevalence and determinants of periodic limb movements in the general population. Ann Neurol. 2016;79(3):464–474. doi:10.1002/ana.24593
13. Szentkirályi A, Stefani A, Hackner H, et al. Prevalence and associated risk factors of periodic limb movement in sleep in two German population-based studies. Sleep. 2018. doi:10.1093/sleep/zsy237
14. Neu D, Baniasadi N, Newell J, Styczen D, Glineur R, Mairesse O. Effect of sleep bruxism duration on perceived sleep quality in middle-aged subjects. European J Oral Sci. 2018;126(5):411–416. doi:10.1111/eos.12564
15. Hossain JL, Ahmad P, Reinish LW, Kayumov L, Hossain NK, Shapiro CM. Subjective fatigue and subjective sleepiness: two independent consequences of sleep disorders? J Sleep Res. 2005;14(3):245–253. doi:10.1111/jsr.2005.14.issue-3
16. Mairesse O, Neu D. Tired of blunt tools? Sharpening the clinical assessment of fatigue and sleepiness. Psychiatry Res. 2016;238:100–108. doi:10.1016/j.psychres.2016.02.005
17. Pigeon WR, Sateia MJ, Ferguson RJ. Distinguishing between excessive daytime sleepiness and fatigue: towards improved detection and treatment. J Psychosom Res. 2003;54:61–69. doi:10.1016/S0022-3999(02)00542-1
18. Berry RB, Brooks R, Gamaldo CE, et al. AASM Scoring Manual Updates for 2017 (Version 2.4). Darien, IL: American Academy of Sleep Medicine; 2017.
19. Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540–545. doi:10.1093/sleep/14.6.540
20. Buysse DJ, Reynolds CF, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28:213–293. doi:10.1016/0165-1781(89)90047-4
21. Zigmand AS, Snaith RP. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983;67(6):361–370. doi:10.1111/j.1600-0447.1983.tb09716.x
22. Wei J, Carroll RJ, Harden KK, Wu G. Comparisons of treatment means when factors do not interact in two-factorial studies. Amino Acids. 2012;42(5):2031–2035. doi:10.1007/s00726-011-0924-0
23. Boulos MI, Jairam T, Kendzerska T, Im J, Mekhael A, Murray BJ. Normal polysomnography parameters in healthy adults: a systematic review and meta-analysis. Lancet Respir Med. 2019;7(6):533–543. doi:10.1016/S2213-2600(19)30057-8
24. Chervin RD. Sleepiness, fatigue, tiredness, and lack of energy in obstructive sleep apnea. Chest. 2000;118:372–379. doi:10.1378/chest.118.2.372
25. Neu D, Mairesse O, Le Bon O. What about sleep homeostasis in insomnia? Comment on the European guideline for the diagnosis and treatment of insomnia. J Sleep Res. 2017;26(6):701. doi:10.1111/jsr.2017.26.issue-6
26. Meino Y, Hayashi M, Sakakibara H, et al. Gender differences in clinical features of sleep apnea syndrome. Internal Med. 2018;57:2157–2163. doi:10.2169/internalmedicine.7570-16
27. Redline S, Kirchner HL, Quan SF, Gottlieb DJ, Kapur V, Newman A. The effects of age, sex, ethnicity and sleep-disordered breathing on sleep architecture. Arch Intern Med. 2004;164:406–418. doi:10.1001/archinte.164.4.406
28. Faravelli C, Scarpato MA, Castellini G, Lo Sauro C. Gender differences in depression and anxiety: the role of age. Psychiatry Res. 2013;210:1301–1303. doi:10.1016/j.psychres.2013.09.027
29. Karshikoff B, Sundelin T, Lasselin J. Role of inflammation in human fatigue: relevance of multidimensional assessments and potential neuronal mechanisms. Front Immunol. 2017;8:21. doi:10.3389/fimmu.2017.00021
30. Lasselin J, Lekander M, Axelsson J, Karshikoff B. Sex differences in how inflammation affects behavior: what we can learn from experimental inflammatory models in humans. Front Neuroendocrinol. 2018;50:91–106. doi:10.1016/j.yfrne.2018.06.005
31. Lei X, Han Z, Chen C, Bai L, Xue G, Dong Q. Sex differences in fiber connection between striatum and subcortical and cortical regions. Front Comput Neurosci. 2016;23, 10:100.
32. Basoglu OK, Tasbakan MS. Gender differences in clinical and polysomnographic features of obstructive sleep apnea: a clinical study of 2827 patients. Sleep Breath. 2014;22:241–249. doi:10.1007/s11325-017-1482-9
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.