Back to Journals » Neuropsychiatric Disease and Treatment » Volume 11
Severe arrhythmia induced by orally disintegrating aripiprazole tablets (Bosiqing®): a case report
Authors Shao Q, Quan W, Jia X, Chen J, Ma S, Zhang X
Received 4 July 2015
Accepted for publication 18 September 2015
Published 9 December 2015 Volume 2015:11 Pages 3019—3021
DOI https://doi.org/10.2147/NDT.S91771
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Roger Pinder
Qing Shao,1,2,* Wei Quan,1,2,* Xiaoni Jia,1 Jianbo Chen,1 Shanbo Ma,3 Xiaohong Zhang1
1Xi’an Mental Health Center, Institute of Mental Health, Xi’an Medical University, Xi’an, Shaanxi, People’s Republic of China; 2Institute of Materia Medica, School of Pharmacy, Fourth Military Medical University, Xi’an, Shaanxi, People’s Republic of China; 3Department of Pharmacy, Xijing Hospital, Fourth Military Medical University, Xi’an, Shaanxi, People’s Republic of China
*These authors contributed equally to this work
Abstract: Psychotropic medications have been known to cause cardiac conduction disturbances. Not much is known about the cardiovascular side effects of newer atypical antipsychotics such as aripiprazole. A case of a 13-year-old girl with schizophrenia is presented. An analysis of the presented patient’s clinical history indicates the need for a detailed analysis of the severe arrhythmia induced by aripiprazole. This presented case report contains valuable guidelines that can be of assistance in the treatment of patients with aripiprazole.
Keywords: schizophrenia, aripiprazole, arrhythmia, antipsychotics
Introduction
Schizophrenia is a serious mental disorder often characterized by abnormal social behavior and failure to recognize what is real. Common symptoms include false beliefs, unclear or confused thinking, reduced social engagement and emotional expression, and lack of motivation. Treatment helps relieve many symptoms of schizophrenia, but most people with the disorder need to cope with symptoms throughout their lives. Researchers are developing more effective medications to treat schizophrenia.1
Aripiprazole was developed by Otsuka in Japan. It is a partial dopamine agonist. It is worth noting that aripiprazole is an efficacious antipsychotic for both the positive and negative symptoms of schizophrenia and causes fewer adverse events (such as extrapyramidal side effects) than typical antipsychotics.2 Other uses of aripiprazole include its use as an add-on treatment in major depressive disorder, tic disorders, and irritability associated with autism.
We report a case in which severe arrhythmia was induced by orally disintegrating aripiprazole tablets. This is probably the first such reported case, to our knowledge, in a young schizophrenia patient. Written informed consent was obtained from the patient’s family for publication of this report, and ethical approval was obtained from the research ethics board at the Xi’an Mental Health Center.
Case report
A 13-year-old girl was admitted on May 26, 2014, presenting with idioglossia and unprovoked smile. On admission, she was conscious but had impaired concentration, bizarre thinking, trouble with emotional expression, lack of insight, and poor sleep. She was therefore diagnosed with schizophrenia.
Physical examination revealed a normal body temperature of 36.7°C, pulse rate of 78 beats/min, respiratory rate of 19 times/min, and blood pressure of 90/60 mmHg. An ambulatory electrocardiogram (ECG) examination taken at Xi’an Children’s Hospital indicated negative results, and normal results were obtained from all other examinations, including blood biochemistry, prolactin levels, thyroid function, and head computed tomography. Thus, the possibility of physical illnesses was ruled out.
Orally disintegrating aripiprazole tablets (Bosiqing®, Kanghong Sagent [Chengdu] Pharmaceutical Co., Ltd., Chengdu, People’s Republic of China, 5 mg/d) in combination with the traditional Chinese medicine, Annaowan® (Harbin Dandelion Pharmaceutical Co., Ltd., Harbin, People’s Republic of China) were administered to the patient for management of her psychiatric symptoms. On May 28, the patient presented with a chief complaint of lightheadedness and dizziness. In light of these symptoms, she had undergone routine auscultation and ECG follow-up. Arrhythmia with premature heart beats (approximately 20 beats/min) was observed during auscultation, and abnormal results were obtained by sequential ECG. Frequent, premature ventricular contractions (trigeminy) occurred, which did not improve after repeated auscultations. Oral metoprolol (Toprol-XL®; AstraZeneca Pharmaceutical Co. Ltd., Wuxi, People’s Republic of China, 50 mg) was administered, and dynamic ECG examinations were initiated that afternoon. Since the severe arrhythmia occurred after administration of Bosiqing®, we speculated that this drug induced the attack. As a result, all antipsychotic medications were stopped on May 29, and metoprolol (25 mg/d) was administered with a simultaneous venous infusion of polarized solution. The patient’s symptoms then improved.
To verify that the arrhythmia resulted from aripiprazole administration, we readministered Bosiqing® after obtaining approval from the patient and her relatives. If the arrhythmia was relieved by aripiprazole withdrawal and reappeared after remedication, a correlation between Bosiqing® use and the incidence of arrhythmia could be identified. Thus, after a 7-day withdrawal period, we readministered Bosiqing® to the patient. Three days after remedication, (June 8), the patient’s premature heartbeats increased with occasional trigeminy. After consultation with cardiologists, the arrhythmia was confirmed to be an adverse reaction caused by Bosiqing®.
We thus informed the patient’s relatives of the diagnosis and replaced aripiprazole with Risperidone (Jiangsu Nhwa Pharmaceutical Co., Ltd., Xuzhou, Jiangsu, People’s Republic of China, 4 mg/d), since it causes less cardiovascular damage. We further increased the use of metoprolol to 50 mg/d. On June 12, no premature beats were observed on auscultation, although bradycardia occurred. Later, we reduced the use of metoprolol to 25 mg/d. The patient’s psychotic symptoms were well controlled, with a normal ECG; therefore, we decided to continue the new therapeutic regimen. ECG monitoring of this case is illustrated in Figure 1.
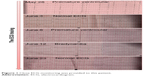  | Figure 1 Close ECG monitoring was provided to this patient. |
Discussion
Aripiprazole, an atypical antipsychotic drug, acts as a partial agonist of the dopamine D2 and the serotonin 5-HT1A receptors. Although it has a good side effect profile, it may cause marked orthostatic hypotension due to its α-1 adrenergic blocking agents. Orthostatic hypotension and postural dizziness were all reported in patients in placebo-controlled trials. However, aripiprazole has not generally been associated with the cardiovascular effects that characterize other conventional antipsychotics, and it has a low incidence of QTc prolongation.3
In 2011, Suzuki et al4 reported a case of aripiprazole (30 mg/d) dose-dependent QTc interval. Even so, as occurs in our case, it is difficult to establish a direct relation between the use of aripiprazole and the arrhythmia. The appearance of ventricular arrhythmia is probably due to a confluence of several factors: the influence of age and the serum levels of aripiprazole. More studies are needed to characterize the mechanism of antipsychotic drug-induced arrhythmias.5 Recently, it is tempting to hypothesize that an activation of the G-protein function followed by enhanced G-protein-dependent signaling pathway may underlie the action of arrhythmias.6 These findings might be beneficial for further understanding of the molecular mechanisms of aripiprazole-induced arrhythmias.
Taken together, the case report provides evidence that the use of arrhythmia is associated with aripiprazole. In clinical practice, severe injury can occur without rapid treatment. Although most drug-induced arrhythmias have been established in previous studies, rare ones, such as the case described here, require considerable attention. Once an arrhythmia occurs, medication should be stopped, symptomatic treatment should be initiated, and close ECG monitoring should be provided to the patient.
Author contributions
QS and WQ participated in drafting of the manuscript. QS, WQ, XJ, JC and SM collected the clinical data. QS, WQ, XJ, JC, SM and XZ participated in the design of the study and revised the article. All authors read and approved the final manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
Holder SD, Wayhs A. Schizophrenia. Am Fam Physician. 2014;90(11):775–782. | ||
Shirley M, Perry CM. Aripiprazole (ABILIFY MAINTENA(R)): a review of its use as maintenance treatment for adult patients with schizophrenia. Drugs. 2014;74(10):1097–1110. | ||
Torgovnick J, Sethi NK, Arsura E. Aripiprazole-induced orthostatic hypotension and cardiac arrhythmia. Psychiatry Clin Neurosci. 2008;62(4):485. | ||
Suzuki Y, Ono S, Fukui N, et al. Dose-dependent increase in the QTc interval in aripiprazole treatment after risperidone. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35(2):643–644. | ||
Kennelly C, Esaian D. Drug-induced cardiovascular adverse events in the intensive care unit. Crit Care Nurs Q. 2013;36(4):323–334. | ||
Ang R, Opel A, Tinker A. The role of inhibitory G proteins and regulators of G protein signaling in the in vivo control of heart rate and predisposition to cardiac arrhythmias. Front Physiol. 2012;3:96. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
