Back to Journals » Cancer Management and Research » Volume 11
Sequential therapy with bevacizumab and EGFR inhibitors for metastatic colorectal carcinoma: a national registry-based analysis
Authors Buchler T , Chloupkova R, Poprach A, Fiala O , Kiss I, Kopeckova K, Dusek L, Veskrnova V, Slavicek L, Kohoutek M, Finek J, Svoboda M, Petruzelka L, Melichar B
Received 8 August 2018
Accepted for publication 7 December 2018
Published 28 December 2018 Volume 2019:11 Pages 359—368
DOI https://doi.org/10.2147/CMAR.S183093
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Kenan Onel
Tomas Buchler,1 Renata Chloupkova,2 Alexandr Poprach,3 Ondrej Fiala,4,5 Igor Kiss,3 Katerina Kopeckova,6 Ladislav Dusek,2 Veronika Veskrnova,1 Lubomir Slavicek,7 Milan Kohoutek,8 Jindrich Finek,4 Marek Svoboda,3 Lubos Petruzelka,9 Bohuslav Melichar10
1Department of Oncology, First Faculty of Medicine, Charles University and Thomayer University Hospital, 140 59 Prague, Czech Republic; 2Institute of Biostatistics and Analyses, Faculty of Medicine, Masaryk University, 625 00 Brno, Czech Republic; 3Department of Comprehensive Cancer Care and Faculty of Medicine, Masaryk Memorial Cancer Institute and Masaryk University, Brno 656 53, Czech Republic; 4Department of Oncology, University Hospital, 304 60 Pilsen, Czech Republic; 5Biomedical Center, Faculty of Medicine in Pilsen, Charles University, Prague, Czech Republic; 6Department of Oncology, Motol University Hospital and Second Faculty of Medicine, Charles University, 150 00 Prague, Czech Republic; 7Department of Oncology, Jihlava Hospital Comprehensive Cancer Centre, Jihlava, Czech Republic; 8Department of Oncology, T Bata Hospital and Comprehensive Cancer Centre, Zlin, Czech Republic; 9Department of Oncology, General University Hospital and Charles University First Faculty of Medicine, 128 08 Prague, Czech Republic; 10Department of Oncology, Palacky University Medical School and Teaching Hospital, 775 20 Olomouc, Czech Republic
Purpose: Although inhibitors of vascular endothelial growth factor and inhibitors of epidermal growth factor receptor (EGFRi) are commonly used for the treatment of metastatic colorectal cancer (mCRC), the optimal sequencing of these agents is currently unclear.
Methods: A national registry of targeted therapies was used to analyze baseline characteristics and outcomes of patients with mCRC and wild-type KRAS exon 2 status who received bevacizumab and EGFRi (cetuximab or panitumumab) as a part of first- and second-line treatment in either sequence.
Results: The cohort included 490 patients (181 patients treated with first-line EGFRi and second-line bevacizumab and 309 patients treated with first-line bevacizumab and second-line EGFRi). Median overall survival (OS) from the initiation on first-line therapy was similar for patients treated with either sequence, reaching 31.8 (95% CI 27.5–36.1) vs 31.4 months (95% CI 27.8–35.0) for EGFRi → bevacizumab vs bevacizumab → EGFRi cohort, respectively. Time from first-line initiation to progression on the second-line therapy [progression-free survival (PFS)] was 21.1 (95% CI 19.3–23.0) vs 19.3 months (95% CI 17.3–21.3) for bevacizumab → EGFRi vs EGFRi → bevacizumab cohort, respectively (P=0.016).
Conclusion: This retrospective analysis of real-world data of patients with wild-type KRAS exon 2 mCRC showed no differences in OS between cohorts treated with bevacizumab → EGFRi vs the reverse sequence while combined PFS favored the bevacizumab → EGFRi sequence.
Keywords: colorectal carcinoma, bevacizumab, panitumumab, cetuximab, sequence
Introduction
The monoclonal antibody (MoAb) against vascular endothelial growth factor (VEGF) bevacizumab and MoAbs directed against the epidermal growth factor receptor (EGFR) cetuximab and panitumumab are commonly used in the treatment of metastatic colorectal cancer (mCRC).
Three randomized trials have compared first-line treatment with VEGF inhibitors (VEGFi) vs EGFR inhibitors (EGFRi) added to a chemotherapy backbone. In the FIRE-3 trial, patients received 5-fluorouracil, leucovorin, and irinotecan (FOLFIRI) chemotherapy in combination with either cetuximab or bevacizumab. There were no differences in progression-free survival (PFS) or overall response rate (ORR); however, there was a significant difference in overall survival (OS) reflected in a late separation of survival curves favoring the cetuximab arm.1 PEAK trial was a randomized phase II study using 5-fluorouracil, leucovorin, and oxaliplatin (FOLFOX) chemotherapy with either bevacizumab or panitumumab in wild-type KRAS exon 2 mCRC. Similarly to FIRE-3, there was a numerical advantage in OS for the panitumumab cohort, although in the PEAK study, the difference was not statistically significant, probably due to the relatively small number of enrolled patients.2 The largest trial specifically designed to compare between the two types of MoAbs in mCRC was the Cancer and Leukemia Group (CALGB) 80405 study. Patients were treated with either FOLFOX or FOLFIRI and randomized to bevacizumab or cetuximab. No differences in PFS or ORR were reported, and OS was also similar between patients receiving VEGFi and EGFRi therapy, contrasting with results of the FIRE-3 and the PEAK studies.3 Three meta-analyses of these randomized studies have been carried out, all showing superior OS in patients who start with EGFRi as opposed to VEGFi.4–6
In the present retrospective study, a large clinical registry of cancer patients treated with targeted therapies was used to analyze and to compare the outcomes of patients treated with bevacizumab followed by EGFRi therapies including cetuximab or panitumumab vs patients treated with the reverse sequence of the targeted therapies.
Materials and methods
Study design
The CORECT (http://corect.registry.cz) registry was used as a data source for the present analysis. The registry is a prospective, non-interventional post-registration database that contains anonymized data on patients with mCRC treated with targeted therapy. Individual patient data including baseline characteristics, treatment type, response, survival, and adverse events are recorded. The data entries are updated twice a year. The therapy with targeted agents in the Czech Republic is reimbursed only in comprehensive cancer centers, and the registry provides data on ~90% of all patients treated with targeted therapy for mCRC in these centers.
All study procedures performed were in accordance with the ethical standards of the institutional and national research committee and with the Declaration of Helsinki 1964 and its later amendments. The CORECT registry and the use of registry data for analysis were approved by the Multicenter Ethics Committee of the Masaryk Memorial Cancer Institute in Brno, Czech Republic. All patients included in the study signed informed consent with the inclusion and subsequent analysis of their data in the registry.
Patients
The cohort for analysis included all patients with colorectal adenocarcinoma and valid data in the registry as of October 10, 2016 who met the following inclusion criteria: 1) sequential therapy with first-line cetuximab or panitumumab and second-line bevacizumab OR first-line bevacizumab and second-line cetuximab or panitumumab; 2) progression documented in the database between the first and the second line; 3) first-line therapy using FOLFOX or FOLFIRI regimens as chemotherapy backbone; 4) wild-type KRAS exon 2 status; 5) wild-type or unknown KRAS exon 3 and 4; and 6) wild-type or unknown NRAS status. The Consolidated Standards of Reporting Trials (CONSORT) diagram depicting patient selection is shown in Figure 1.
The interval of tumor assessments was not pre-specified. However, reimbursement conditions for targeted therapies stipulate radiological tumor assessment at least every 3 months or three cycles of therapy as applicable. Treatment response was assessed using the Response Evaluation Criteria in Solid Tumors version 1.1.7
Patients with primary tumor in the caecum, the colon ascendens, and transversum were considered to have a right-sided primary, while tumors in the descending and sigmoid colon or the rectum were classified as left-sided primary tumors.
Statistical analysis
Descriptive statistics and frequency tables were used to characterize the sample data set. Statistical significance of differences in categorical parameters was assessed using the Fisher’s exact test or Pearson’s test. For continuous variables, Mann–Whitney test was used. PFS was defined as the time from first-line treatment initiation to the date of documented progression on the second-line therapy or death of any cause. OS was defined as the time from first-line target therapy initiation to the date of death of any cause. PFS and OS were estimated using Kaplan–Meier method and all point estimates include 95% CI. Statistical significance of the differences in Kaplan–Meier estimates was assessed using the log-rank test. Univariate and multivariate Cox tests of proportional risks were used to calculate differences in survival for selected subgroups and select the principal prognostic factors. Point estimates of HR are shown with 95% CI. Statistical significance of HR was calculated using the Wald test. An alpha of 0.05 was used as the cutoff of significance for all statistical tests.
Results
Baseline characteristics
Overall, 490 patients, including 181 patients treated with first-line cetuximab or panitumumab and second-line bevacizumab and 309 patients treated with first-line bevacizumab and second-line cetuximab or panitumumab were included in the analysis (Figure 1).
Baseline patient characteristics are shown in Table 1. Patients treated with bevacizumab → EGFRi sequence were significantly younger at the time of diagnosis and initiation of the first- and second-line treatment compared to patients receiving the reverse sequence, although the difference in median age was only 3 years and the proportion of patients >60 years of age was similar. A lower proportion of patients were tested for NRAS and BRAF mutations in the bevacizumab → EGFRi cohort. This cohort was also more likely to receive FOLFOX as a part of the first-line treatment (71% vs 63%, respectively, P<0.001).
An overview of patient characteristics at the time of second-line therapy initiation is provided in Table 2. Patients treated with the bevacizumab → EGFRi sequence were younger at the start of second-line treatment and tended to receive less intensive second-line backbone chemotherapy and 28.2% only received EGFRi monotherapy.
More than 98% of patients had Eastern Cooperative Oncology Group performance status of 0–1 at the time of initiation of the first and second line, in accordance with reimbursement conditions for targeted therapies.
Median follow-up from the initiation of the first-line therapy was 20.7 and 30.9 months for the EGFRi → bevacizumab and the bevacizumab → EGFRi cohort, respectively. Median follow-up from the initiation of the second-line therapy was 7.7 and 11.8 months, respectively.
Overall survival
Median OS from the initiation on first-line therapy was similar for patients treated with different sequences, reaching 31.4 (95% CI 27.8–35.0) vs 31.8 months (95% CI 27.5–36.1) for the bevacizumab → EGFRi vs the EGFRi → bevacizumab cohort, respectively (P=0.940) (Figure 2).
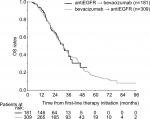  | Figure 2 OS from first-line treatment initiation. Abbreviations: EGFR, epidermal growth factor receptor; OS, overall survival. |
The Cox analysis of proportional risk showed that there was a trend bordering on statistical significance (P=0.050) in patients with wild-type RAS (ie, tested negative for both expanded KRAS and NRAS mutations, n=182) suggesting benefit from the bevacizumab → EGFRi sequence (Figure 3).
Multivariate Cox analysis revealed that male gender, left colon primary, M0 stage at diagnosis, and FOLFOX as first-line chemotherapy regimens were independently associated with favorable OS (Table 3)
PFS
PFS calculated as the time from the initiation of first-line therapy to progression on the second-line therapy was similar for the two sequences. Median PFS was 21.1 (95% CI 19.3–23.0) vs 19.3 months (95% CI 17.3–21.3) for the bevacizumab → EGFRi vs the EGFRi → bevacizumab cohort, respectively (P=0.016) (Figure 4). The time from the discontinuation of the first-line to the initiation of the second-line chemotherapy was 1.1 (95% CI 0.2–12.4) vs 1.7 months (95% CI 0.3–15.6) for the EGFRi → bevacizumab vs the bevacizumab → EGFRi (P=0.003). In multivariate analysis, bevacizumab → EGFRi and left colon primary were associated with better PFS (Table 3).
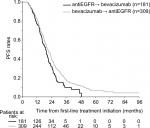  | Figure 4 Time from treatment initiation to progression on second-line treatment. Abbreviations: EGFR, epidermal growth factor receptor; PFS, progression-free survival. |
Discussion
A large cohort of patients with mCRC treated with EGFRi agents and bevacizumab in either sequence in the real-world setting was analyzed in this study. No statistically significant differences in OS were detected between patients treated with the bevacizumab → EGFRi sequence and the reverse sequence for the whole cohort or for subgroups of patients defined by age, chemotherapy backbone, the presence of synchronous/metachronous metastatic disease, anatomic location of the primary tumor, RAS gene status, and body mass index. PFS but not OS was better in the bevacizumab–EGFRi cohort even though less intensive backbone chemotherapy regimens were used with second-line EGFRi therapies and 28% of the patients received second EGFRi treatment only as monotherapy.
Proper sequencing of targeted agents in mCRC is currently a matter of substantial controversy. Two approaches have been used to explain the discrepancies between the results of FIRE3 and PEAK studies on one hand, and CALGB 80405 on the other. First, there are inter-trial differences in off-study second-line therapies, particularly in the exposure to second-line biologicals. In the CALGB trial, almost 90% patients crossed over to the other type of antibody, while this proportion in the FIRE3 trial was approximately 50%.8,9 In the PEAK trial, 50% of patients subsequently received the other type of inhibitor, while approximately one-third continued on the same type of inhibitor.2 In this respect, CALGB 80405 is similar to the present study, which confirms its findings in the real-world setting. Tumor sidedness has been associated with relative benefits of different sequences in post-hoc analyses of FIRE-3 and CALGB 80405. The results suggest that first-line EGFRi are optimal for patients with left-sided primary tumors, while patients with right-sided primary tumor might benefit from the bevacizumab as a part of the first-line regimen.10,11
Another possible explanation postulates an interaction between the two types of therapies administered sequentially. It has been hypothesized that EGFRi therapy given as a part of first-line treatment enhances the effect of post-progression VEGFi therapy.9 This theory has been formulated to explain the late divergence of OS curves in the FIRE-3 trial.1,9 There is also some evidence from preclinical studies indicating that chronic VEGF inhibition produces changes in phenotype of tumor cells that lead to increased invasiveness and metastatic potential.12,13 The biological hypotheses on the potential temporal interaction between EGFRi, VEGFRi and subsequent disease course and response to sequential treatments have been extensively reviewed by Wainberg and Drakaki.14
Several recent retrospective studies also support the hypothesis that the carryover effect of VEGFi therapy negatively influences subsequent treatment efficacy. Derangère et al found difference in PFS on EGFRi depending on previous treatment with bevacizumab, although there was no difference in OS.15 In a study published by Moehler et al, patients treated with EGFRi → VEGFi sequence had better OS than those treated with reverse sequence.16 Sato et al reported that patients not receiving bevacizumab prior to cetuximab had better OS, although there were imbalances in age and treatment intensity, which could have caused or contributed to the findings.17 No patients pretreated with bevacizumab responded to subsequent EGFRi in a small cohort analyzed by Norguet et al.18 Conversely, prior EGFRi therapy had no influence of the efficacy of subsequent bevacizumab-containing regimens in another retrospective analysis.19 The influence of VEGFi on subsequent EGFRi may depend on interval between the two therapies, the effect decreasing over time.20
In contrast, several prospective studies identified no adverse impact of VEGFi therapy on subsequent outcomes. Price et al reported similar survival of patients previously treated vs untreated with bevacizumab-containing therapy in the randomized ASPECCT trial comparing cetuximab to panitumumab.21 There was even a trend to improved OS in patients who continued on VEGFi in the Prodige 18 randomized phase II study assessing the efficacy and safety of crossover chemotherapy plus bevacizumab or cetuximab after progression on bevacizumab-containing regimen.22 Price et al found that outcomes of EGFRi treatment were independent of previous use of VEGFi.23 The efficacy of continued VEGF blockade was confirmed by the results of the ML18147 randomized phase III study.24 An ongoing clinical trial called STRATEGIC-1 is aimed at optimizing the sequential approach to targeted therapies in mCRC, with one of the arms testing a three-line strategy with continued bevacizumab after progression on bevacizumab and a switch to EGFRi only after the second progression and the other arm the EGFRi → VEGFi approach.25
The prognostic and predictive role of the primary tumour location has been recently reported in several studies. It is evident that the different clinical and molecular characteristics of right-sided vs left-sided tumors translate into different clinical outcomes, with right-sided tumors displaying a markedly worse prognosis.26–32 The results of recently published meta-analysis of 13 randomized clinical trials by Holch et al confirmed that right-sided tumors are associated with significantly shorter OS compared to left-sided tumors.33 In our study, the sidedness of primary tumour was not associated with benefit of either sequence as all patients were exposed to both EGFRi and VEGFRi. However, the negative prognostic impact of right-sided primary tumors was confirmed in multivariate analysis (Table 3).
The strength of the present study includes the exposure of all patients to both types of treatment, enabling to test the hypothesis on the possible interactions between EGFRi and VEGFi strategies given sequentially.
The limitations are inherent in the retrospective, registry-based approach with retroactive cohort identification. In a prospective study, certainly less than 100% of patients receiving any first-line therapy would be able to proceed to second-line therapy. The protocol of the CORECT registry prevented direct comparison of individual lines of treatment, as it permits only combined analyses rather than comparisons of individual drugs. Only a small number of patients had BRAF analysis, precluding us from analyzing the impact of this important biomarker.
There are imbalances in baseline characteristics of patients that can be mostly linked to changing preferences and reimbursement for the respective therapies in different time periods, resulting in selection bias. Prior to 2012, a higher proportion of patients treated with bevacizumab outside of clinical studies in the Czech Republic continued to EGFRi than vice versa. The possibility of selection of early patients with favorable disease course in the bevacizumab → EGFRi cohort cannot be excluded, and the inclusion of these patients could have biased the results in favor of the bevacizumab → EGFRi. Many patients in the bevacizumab → EGFRi cohort had been treated before the predictive role of NRAS mutations and exon 3+4 KRAS mutations were recognized.34–36 Full RAS testing could potentially improve the outcomes of the bevacizumab → EGFRi cohort (Figure 3). The fact that RAS wt population appears to derive a benefit from bevacizumab–antiEGFR sequence over the reverse sequence is of interest, although the finding needs to be prospectively verified. There are actually few published data on the efficacy of second-line EGFRi given in combination with fluorouracil-based chemotherapy, with a single trial showing improved PFS on FOLFIRI/panitumumab in patients pretreated with a fluoropyrimide-based regimen.37 Previous therapy with bevacizumab was not allowed in this trial.38 However, a biological explanation is also possible. Correale et al published a study showing that exposure to chemotherapy increased the membrane expression of EGFR on cancer cells and sensitized them to cetuximab-mediated antibody-dependent cell cytotoxicity.39 In a recently published study, we report that limited pretreatment with chemotherapy before the start of EGFRi could be associated with modestly improved outcomes.40
Conclusion
The present retrospective analysis of real-world data of patients with mCRC without KRAS mutation treated with EGFRi agents and bevacizumab in either sequence found no differences in OS between cohorts treated with bevacizumab → EGFRi vs the reverse sequence. There was a modest but statistically significant improvement in PFS for the bevacizumab → EGFRi sequence. Tumor sidedness was not significantly associated with benefit of either sequence.
Acknowledgments
The study was presented as a poster at the 42nd European Society for Medical Oncology (ESMO) 2017 Congress in Madrid, Spain, 8–12 September 2017, with the abstract published in the Abstract Book.
This study was supported by Grant AZV 15-26535A from the Czech Health Research Council. CORECT registry maintenance is supported in part by Roche, Amgen, and Merck.
We would like to thank the following heads of the comprehensive cancer centers for the permission to use data of patients from their respective regional networks: Dr Martina Chodacka, Chomutov Hospital and Masaryk Hospital in Usti nad Labem; Dr Vaclav Janovsky, Ceske Budejovice Hospital; Dr Otakar Bednarik, University Hospital, Brno; Dr Jana Prausova, Motol University Hospital, Prague; Dr David Feltl, University Hospital, Ostrava; Professor Jiri Petera, University Hospital, Hradec Kralove; Dr Jana Katolicka, St Anna University Hospital, Brno; Professor Rostislav Vyzula, Masaryk Memorial Institute of Oncology, Brno; Dr Jiri Bartos, County Hospital, Liberec; Dr Martin Safanda, Na Homolce Hospital, Prague; Dr Renata Soumarova, Novy Jicin Hospital; Professor Jitka Abrahamova, Thomayer Hospital, Prague. We are also indebted to all physicians who provided data for the CORECT registry.
Disclosure
TB, OF, and BM have received research funding, travel grants, and honoraria from Roche, Merck, and Amgen. AP has received honoraria and travel grants from Roche and Amgen. IK has received speakers’ honoraria from Roche, Merck, and Amgen. JF has received lecture honoraria and travel grants from Roche, Merck, Pfizer, Novartis, and Amgen. The authors declare no other conflicts of interest in this work.
References
Heinemann V, von Weikersthal LF, Decker T, et al. FOLFIRI plus cetuximab versus FOLFIRI plus bevacizumab as first-line treatment for patients with metastatic colorectal cancer (FIRE-3): a randomised, open-label, phase 3 trial. Lancet Oncol. 2014;15(10):1065–1075. | ||
Rivera F, Karthaus M, Hecht JR, et al. Final analysis of the randomised PEAK trial: overall survival and tumour responses during first-line treatment with mFOLFOX6 plus either panitumumab or bevacizumab in patients with metastatic colorectal carcinoma. Int J Colorectal Dis. 2017;32(8):1179–1190. | ||
Venook AP, Niedzwiecki D, Lenz HJ, et al. Effect of first-line chemotherapy combined with cetuximab or bevacizumab on overall survival in patients with KRAS wild-type advanced or metastatic colorectal cancer: a randomized clinical trial. JAMA. 2017;317(23):2392–2401. | ||
Khattak MA, Martin H, Davidson A, Phillips M. Role of first-line anti-epidermal growth factor receptor therapy compared with anti-vascular endothelial growth factor therapy in advanced colorectal cancer: a meta-analysis of randomized clinical trials. Clin Colorectal Cancer. 2015;14(2):81–90. | ||
Pietrantonio F, Cremolini C, Petrelli F, et al. First-line anti-EGFR monoclonal antibodies in panRAS wild-type metastatic colorectal cancer: a systematic review and meta-analysis. Crit Rev Oncol Hematol. 2015;96(1):156–166. | ||
Tamburini E, Rudnas B, Gianni L, et al. E02*Anti-EGFR or bevacizumab in first line treatment of RAS wild type metastatic colorectal neoplasm (RwtMCRC): meta-analysis of randomized clincal trials. Ann Oncol. 2015;26(Suppl 6):vi36. | ||
Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–247. | ||
Sclafani F, Cunningham D. Cetuximab or bevacizumab in metastatic colorectal cancer? Lancet Oncol. 2014;15(10):1040–1041. | ||
Modest DP, Stintzing S, von Weikersthal LF, et al. Impact of subsequent therapies on outcome of the FIRE-3/AIO KRK0306 trial: first-line therapy with FOLFIRI plus cetuximab or bevacizumab in patients with KRAS wild-type tumors in metastatic colorectal cancer. J Clin Oncol. 2015;33(32):3718–3726. | ||
Modest DP, Stintzing S, von Weikersthal LF, et al. Exploring the effect of primary tumor sidedness on therapeutic efficacy across treatment lines in patients with metastatic colorectal cancer: analysis of FIRE-3 (AIOKRK0306). Oncotarget. 2017;8(62):105749–105760. | ||
Venook AP, Niedzwiecki D, Innocenti F, et al. Impact of primary (1º) tumor location on overall survival (OS) and progression-free survival (PFS) in patients (pts) with metastatic colorectal cancer (mCRC): analysis of CALGB/SWOG 80405 (Alliance). J Clin Oncol. 2016;343504(15_suppl):3504. | ||
Pàez-Ribes M, Allen E, Hudock J, et al. Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell. 2009;15(3):220–231. | ||
Zhou L, Liu XD, Sun M, et al. Targeting MET and AXL overcomes resistance to sunitinib therapy in renal cell carcinoma. Oncogene. 2016;35(21):2687–2697. | ||
Wainberg ZA, Drakaki A. The importance of optimal drug sequencing in metastatic colorectal cancer: biological rationales for the observed survival benefit conferred by first-line treatment with EGFR inhibitors. Expert Opin Biol Ther. 2015;15(8):1205–1220. | ||
Derangère V, Fumet JD, Boidot R, et al. Does bevacizumab impact anti-EGFR therapy efficacy in metastatic colorectal cancer? Oncotarget. 2016;7(8):9309–9321. | ||
Moehler M, Thomaidis T, Zeifri C, et al. Inclusion of targeted therapies in the standard of care for metastatic colorectal cancer patients in a German cancer center: the more the better?! J Cancer Res Clin Oncol. 2015;141(3):515–522. | ||
Sato Y, Matsusaka S, Suenaga M, Shinozaki E, Mizunuma N. Cetuximab could be more effective without prior bevacizumab treatment in metastatic colorectal cancer patients. Onco Targets Ther. 2015;8:3329–3336. | ||
Norguet E, Dahan L, Gaudart J, Gasmi M, Ouafik L, Seitz JF. Cetuximab after bevacizumab in metastatic colorectal cancer: is it the best sequence? Dig Liver Dis. 2011;43(11):917–919. | ||
Hasegawa H, Taniguchi H, Mitani S, et al. Efficacy of second-line bevacizumab-containing chemotherapy for patients with metastatic colorectal cancer following first-line treatment with an anti-epidermal growth factor receptor antibody. Oncology. 2017;92(4):205–212. | ||
Taniguchi H, Komori A, Narita Y, et al. A short interval between bevacizumab and anti-epithelial growth factor receptor therapy interferes with efficacy of subsequent anti-EGFR therapy for refractory colorectal cancer. Jpn J Clin Oncol. 2016;46(3):228–233. | ||
Price T, Kim TW, Li J, et al. Final results and outcomes by prior bevacizumab exposure, skin toxicity, and hypomagnesaemia from ASPECCT: randomized phase 3 non-inferiority study of panitumumab versus cetuximab in chemorefractory wild-type KRAS exon 2 metastatic colorectal cancer. Eur J Cancer. 2016;68:51–59. | ||
Hiret S, Borg C, Bertaut A, et al. Bevacizumab or cetuximab plus chemotherapy after progression with bevacizumab plus chemotherapy in patients with wtKRAS metastatic colorectal cancer: a randomized phase II study (Prodige 18 –UNICANCER GI). J Clin Oncol. 2016;34(15_suppl):3514–3514. | ||
Price TJ, Hardingham J, Karapetis C. 524PBevacizumab first line and impact on subsequent anti-EGFR activity. Ann Oncol. 2017;28050(suppl_5):mdx393.050. | ||
Bennouna J, Sastre J, Arnold D, et al; ML18147 Study Investigators. Continuation of bevacizumab after first progression in metastatic colorectal cancer (ML18147): a randomised phase 3 trial. Lancet Oncol. 2013;14(1):29–37. | ||
Chibaudel B, Bonnetain F, Tournigand C, et al. STRATEGIC-1: a multiple-lines, randomized, open-label GERCOR phase III study in patients with unresectable wild-type RAS metastatic colorectal cancer. BMC Cancer. 2015;15(1):496. | ||
von Einem JC, Heinemann V, von Weikersthal LF, et al. Left-sided primary tumors are associated with favorable prognosis in patients with KRAS codon 12/13 wild-type metastatic colorectal cancer treated with cetuximab plus chemotherapy: an analysis of the AIO KRK-0104 trial. J Cancer Res Clin Oncol. 2014;140(9):1607–1614. | ||
Loupakis F, Yang D, Yau L, et al. Primary tumor location as a prognostic factor in metastatic colorectal cancer. J Natl Cancer Inst. 2015 Mar; 107(3): dju427. | ||
Tejpar S, Stintzing S, Ciardiello F. Prognostic and predictive relevance of primary tumor location in patients with ras wild-type metastatic colorectal cancer retrospective analyses of the CRYSTAL and FIRE-3 trials. JAMA Oncol. 2016;360(14). | ||
Wang F, Bai L, Liu TS, et al. Right-sided colon cancer and left-sided colorectal cancers respond differently to cetuximab. Chin J Cancer. 2015;34(9):384–393. | ||
Brulé SY, Jonker DJ, Karapetis CS, et al. Location of colon cancer (right-sided versus left-sided) as a prognostic factor and a predictor of benefit from cetuximab in NCIC CO.17. Eur J Cancer. 2015;51(11):1405–1414. | ||
Chen KH, Shao YY, Chen HM, et al. Primary tumor site is a useful predictor of cetuximab efficacy in the third-line or salvage treatment of KRAS wild-type (exon 2 non-mutant) metastatic colorectal cancer: a nationwide cohort study. BMC Cancer. 2016;16:327. | ||
Moretto R, Cremolini C, Rossini D, et al. Location of primary tumor and benefit from anti-epidermal growth factor receptor monoclonal antibodies in patients with RAS and braf wild-type metastatic colorectal cancer. Oncologist. 2016;21(8):988–994. | ||
Holch JW, Ricard I, Stintzing S, Modest DP, Heinemann V. The relevance of primary tumour location in patients with metastatic colorectal cancer: a meta-analysis of first-line clinical trials. Eur J Cancer. 2017;70:87–98. | ||
De Roock W, Claes B, Bernasconi D, et al. Effects of KRAS, BRAF, NRAS, and PIK3CA mutations on the efficacy of cetuximab plus chemotherapy in chemotherapy-refractory metastatic colorectal cancer: a retrospective consortium analysis. Lancet Oncol. 2010;11(8):753–762. | ||
Douillard JY, Oliner KS, Siena S, et al. Panitumumab-FOLFOX4 treatment and RAS mutations in colorectal cancer. N Engl J Med. 2013;369(11):1023–1034. | ||
Van Cutsem E, Lenz HJ, Köhne CH, et al. Fluorouracil, leucovorin, and irinotecan plus cetuximab treatment and RAS mutations in colorectal cancer. J Clin Oncol. 2015;33(7):692–700. | ||
van Helden EJ, Menke-van der Houven van Oordt CW, Heymans MW, Ket JCF, van den Oord R, Verheul HMW. Optimal use of anti-EGFR monoclonal antibodies for patients with advanced colorectal cancer: a meta-analysis. Cancer Metastasis Rev. 2017;36(2):395–406. | ||
Peeters M, Price TJ, Cervantes A, et al. Randomized phase III study of panitumumab with fluorouracil, leucovorin, and irinotecan (FOLFIRI) compared with FOLFIRI alone as second-line treatment in patients with metastatic colorectal cancer. J Clin Oncol. 2010;28(31):4706–4713. | ||
Correale P, Marra M, Remondo C, et al. Cytotoxic drugs up-regulate epidermal growth factor receptor (EGFR) expression in colon cancer cells and enhance their susceptibility to EGFR-targeted antibody-dependent cell-mediated-cytotoxicity (ADCC). Eur J Cancer. 2010;46(9):1703–1711. | ||
Fiala O, Veskrnova V, Chloupkova R, et al. Impact of delayed addition of anti-EGFR monoclonal antibodies on the outcome of first-line therapy in metastatic colorectal cancer patients: a retrospective registry-based analysis. Target Oncol. 2018;13(6):735-743. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.





