Back to Journals » International Journal of General Medicine » Volume 13
Seizure Outcomes in Patients with Complete versus Anterior Corpus Callosotomy: Analysis of Outcome
Authors Thohar Arifin M, Muttaqin Z , Bakhtiar Y , Andar E, Priambada D, Kurnia H, Risdianto A, Tsaniadi K , Kusnarto G, Bunyamin J
Received 28 January 2020
Accepted for publication 6 March 2020
Published 31 March 2020 Volume 2020:13 Pages 105—110
DOI https://doi.org/10.2147/IJGM.S247438
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Muhamad Thohar Arifin, Zainal Muttaqin, Yuriz Bakhtiar, Erie Andar, Dody Priambada, Happy Kurnia, Ajid Risdianto, Krisna Tsaniadi, Gunadi Kusnarto, Jacob Bunyamin
Department of Neurosurgery, Faculty of Medicine, Diponegoro University, Kariadi Hospital, Semarang, Indonesia
Correspondence: Muhamad Thohar Arifin Email [email protected]
Introduction: Corpus callosotomy (CCT) is a palliative procedure to treat injurious drop attacks or multifocal/generalized seizures in which resection of the epileptogenic focus is not feasible. We are presenting our experience in treating intractable epilepsy patients by CCT procedures.
Methods: We observed 16 patients who underwent callosotomy (male to female ratio 7:9; adult to pediatric ratio 3:13). Initial seizure frequency was reported ranged from 1 to 2 attacks daily to very often (more than 20 episodes daily).
Results: Our observation showed that among patients with drop attacks, complete and > 90% seizure freedom was reported by 4 and 6 of 13 patients, respectively (76.9% combined).
Conclusion: Our observation showed that corpus callosotomy yielded good outcome in patients with intractable epilepsy in Indonesia. Our observation showed total callosotomy achieved complete seizure freedom better compared to partial callosotomy patients.
Keywords: corpus callosotomy, drop attacks, Indonesia
Introduction
Corpus callosotomy is a palliative procedure to treat injurious drop attacks or multifocal/generalized seizures in which resection of the epileptogenic focus is not feasible. Drop attacks may affect the patients’ quality of life because of repeated injury resulting from falls and the high dose of antiepileptic drugs (AEDs). The hypothesis behind corpus callosotomy lies on the assumed role of corpus callosum as the interhemispheric bridge and the pathophysiology of generalized seizure.1 As the seizure is believed to spread from one hemisphere to the other, severing the hemispheric connection might hamper the generalization, thus confine the seizure activity in one hemisphere and reducing the seizure outcomes.2
We are presenting our experience in treating intractable epilepsy patients by corpus callosotomy procedures in our neurosurgical centres in Semarang, Indonesia. To our best knowledge, this is the first and the most extensive documentation of corpus callosotomy in our country to date.
Methods
Settings
We conducted an observational study based on the medical records of patients who underwent callosotomy in two hospitals serving as comprehensive epilepsy neurosurgical centres in Semarang, Indonesia, between January 2000 to December 2018. Each case was discussed to determine whether the patient would benefit from surgery in Kariadi Hospital which is affiliated with the Faculty of Medicine Diponegoro University. Written informed consent was obtained from all patients for being included in the study, or a parent/legal guardian of patients under the age of 18. This study has been ethically approved by the Ethical Committee of Kariadi Hospital according to the Helsinki declaration.
Preoperative Assessment
Preoperative evaluation included routine interictal scalp electroencephalography (EEG) monitoring and magnetic resonance imaging (MRI). MRI (0.5 T, Tomikon, Bruker, France) including regular axial, coronal, and sagittal planes in T1 and T2 weighted image for the whole brain and fluid-attenuated inversion recovery (FLAIR) sequences. For patients who were operated in 2011 and above, ictal video monitoring was performed. The presence of electro-clinical syndrome was documented. Preoperative neurocognitive ability was assessed by Wechsler Adult Intelligence Scale-IV (WAIS-IV) test for adults and Wechsler Intelligence Scale for Children WISC for children.
Surgical Consideration
As there is no universally accepted indication for callosotomy to date,3 in our institution, corpus callosotomy was indicated for refractory cases related to bilateral cortical dysplasia, generalized tonic-clonic seizures (GTCS) without resectable epileptogenic focus including drop attacks (tonic and atonic seizures), and refractory focal with impaired awareness seizures (FIAS) which are not a candidate for temporal lobectomy. Consideration for total callosotomy was preferred for patients under 10 years old of age and/or with severe cognitive impairment, while partial callosotomy was mostly reserved for patients with older age at surgery and/or with mild cognitive impairment.
Surgical Technique
All patients were operated by a single neurosurgeon (Z.M.). All callosotomy procedures were performed with craniotomy approach from the opening of the coronal suture through interhemispheric fissure below the falx to reach the corpus callosum and divided it at its midline. In partial callosotomy, the corpus callosum was dissected from the callosal genu to the callosal isthmus, while in total callosotomy the corpus callosum was dissected completely.
Postoperative Assessment
The main outcome of this study is the seizure reduction in the latest follow-up visits. Six months was used as a baseline cutoff. Seventeen patients were subjected to corpus callosotomy from January 2000 to December 2018. However, one was excluded from this study because of death secondary of respiratory failure in 1 month after surgery. Seizure reduction rate was classified into 100% seizure freedom; >90%; 50–90%; and <50% reduction from the initial frequency. We transferred the results into Engel surgical outcome scales (1993) for patients with at least 2 years’ follow-up and the ILAE outcome scale (2001) after 1 year’s follow-up. Both scales have very good inter-rater reliability and significant correlation.4
Results
Clinical Profile and Seizure Semiology
The patients’ list is presented in Table 1. We observed 16 patients who underwent callosotomy (male to female ratio 7:9; adult to pediatric ratio 3:13). The average age at surgery was 13.06±8.38 years, average age of onset was 4.65±3.48 years, and the average duration from onset to surgery was 8.78±6.69 years. Initial seizure frequency was reported ranged from 1 to 2 attacks daily to very often (more than 20 episodes daily).
 |
Table 1 Clinical Characteristics of the Study Participant |
From all patients operated in our centers (n=16), 13 patients were presented with drop attacks, 2 with GTCS, and 1 with FIAS. Among patients with drop attacks, 1 had drop attacks plus two other types of seizures and 8 had drop attacks plus one other type of seizures (GTCS, FIAS, and infantile spasm). One patient with GTCS had cerebral palsy. Structural brain abnormality was found in 12 patients, including bilateral atrophy (3), bilateral lissencephaly (2), bilateral cortical heterotopia (1), polymicrogyria (1), hypothalamic hamartoma (1), tuberous sclerosis (1), congenital cerebral dysgenesis with temporal aplasia (1), and tuberous sclerosis with multiple infarcts (1). Four patients had normal-appearing MRI. Three patients had matched the electro-clinical diagnosis of Lennox-Gastaut syndrome (LGS).
Surgical Procedure and Seizure Outcome
The patients were followed up from 12 to 144 months (mean 73.25±46.67 months) to assess and determine the seizure reduction. From 13 patients with drop attacks (atonic seizures), 4 had complete seizure freedom, 6 had >90% seizure reduction, and 3 had 50–90% seizure reduction on the latest visit. Other two patients who were presented with other seizure (GTCS and FIAS) was free from seizure, and only one had less than 50% reduction in frequency (see Figure 1).
 |
Figure 1 Callosotomy outcomes based on the extent of callosotomy. |
We also divided the patients into those who underwent total callosotomy (n=9) and partial callosotomy (n=7). Five of nine patients who were performed total callosotomy was seizure-free, two had >90% seizure reduction, and other two had 50–90% seizure reduction. In patients who underwent partial callosotomy, one had complete seizure freedom, four had >90% reduction, one had 50–90% reduction, and 1 had less than 50% seizure reduction (see Figure 2).
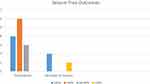 |
Figure 2 Callosotomy outcomes based on the type of seizure. |
We did not observe major neurological or surgical complications requiring second surgery. One patient was excluded from this study due to death from respiratory failure in 1 month after surgery. In general, from 16 patients who underwent corpus callosotomy, we observed that 6 patients fell into ILAE Outcome class I, 6 patients in class III, 3 patients in class IV, and 1 patient in class V.
Discussion
To our best knowledge, this is the first and the most extensive report of corpus callosotomy performed in Indonesia covering both pediatric and adult population. This series also involved patients with drop attacks and other types of seizures (GTCS and FIAS). Our observation showed that among patients with drop attacks, complete and >90% seizure freedom was reported by 4 and 6 of 13 patients, respectively (76.9% combined). Other studies reported that 65% of the patients having >50% seizure reduction5 and 74.1% having >75% seizure reduction6 in mixed-age populations. Total callosotomy yields favorable outcome in drop attacks’ freedom compared to partial callosotomy.7 Our observation showed that 5 of 9 patients with total callosotomy achieved complete seizure freedom compared to 1 of 7 partial callosotomy patients. A study in drop attacks patient demonstrated that the seizure-free rate in total and partial callosotomy was 90% and 54% respectively during a 7-year follow-up.8
Corpus callosotomy might be a more effective treatment for patients with idiopathic intractable generalized epilepsy.9 Compared to other treatment modality, a meta-analysis showed that callosotomy had better rate of seizure freedom and control than vagal nerve stimulation (VNS) for drop attacks.10 Since earlier age at surgery resulted in better outcomes and lower complications, corpus callosotomy should be performed immediately once the epileptic patient is diagnosed as medically intractable.11 The average duration from onset to surgery was 8.78±6.69 years, almost comparable with a study from the US (mean 8.4 years).9 Other known factors affecting seizure freedom besides a shorter duration of epilepsy were the presence of infantile spasm and normal MRI finding.12 In our series, both patients with infantile spasm reached complete and >90% seizure freedom, respectively. Three of four patients with normal imaging finding had >90% to complete seizure freedom while the rest had <50% reduction.
The complications following corpus callosotomy have been described by several authors, including neurological and surgical. Major neurological complications were defined as neurological complications that persist longer than 3 months postoperative. A meta-analysis conducted by Graham et al reviewed that transient (less than 6 weeks) disconnection syndrome was the most frequent neurological complication (7.0%), followed by other minor complications including transient ataxia, apraxia, alien limb, agraphia, paresis, and mutism (13%), in which the rate was higher in patients who underwent total callosotomy compared to partial callosotomy (15.4% vs 6.9%).3 Surgical complications were divided into major (hydrocephalus, deep cerebral infection) and minor complications (CSF leak, intracranial/extracranial infection, aseptic meningitis, DVT/PE, pneumonia, intracranial hematomas, and metabolic disturbances).13 The rate of minor surgical complication was found higher in partial callosotomy patients compared to total callosotomy patients (5.9% vs 2.7%).3 In this series, we did not observe major surgical and neurological complications in our patients. However, one patient was dead secondary to respiratory failure in 1 month postoperative. Another study also reported no complications in a series of 65 patients.5
We also acknowledge several limitations in our study which are (1) relatively small size of subjects, and (2) short duration of follow-up in some subjects. We could not measure any statistical significance among procedures, seizure remission, and affecting factors due to the small number of subjects available during the period of 18 years; however, several reports of CCT procedures also reported a similar amount of subjects given to the infrequent nature of the cases. A future study with longer follow-up duration and larger sample size would give us more insight regarding intractable epilepsy in the Indonesian population. Achieving a shorter period between age of onset and age at surgery remains a challenge since cultural, social, and economic barriers may prevent the patient to receive immediate proper treatment.
Conclusion
Our observation showed that corpus callosotomy yielded good outcome in patients with intractable epilepsy in Indonesia. Our observation showed that total callosotomy achieved complete seizure freedom better compared to partial callosotomy patient. In our series of 16 patients, we observed that 6 patients fell into ILAE Outcome class I, 6 patients in class III, 3 patients in class IV, and 1 patient in class V. This finding might provide additional information to support the global data of corpus callosotomy as a palliative measure for intractable seizures.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Asadi-pooya AA, Sharan A, Nei M, Sperling MR. Epilepsy & behavior corpus callosotomy. Epilepsy Behav. 2008;13(2):271–278. doi:10.1016/j.yebeh.2008.04.020
2. Unterberger I, Bauer R, Walser G, Bauer G. Corpus callosum and epilepsies. Seizure Eur J Epilepsy. 2016;37:55–60. doi:10.1016/j.seizure.2016.02.012
3. Graham D, Tisdall MM, Gill D. Corpus callosotomy outcomes in pediatric patients: a systematic review. Epilepsia. 2016;57(7):1053–1068. doi:10.1111/epi.13408
4. Durnford AJ, Rodgers W, Kirkham FJ, et al. Very good inter-rater reliability of Engel and ILAE epilepsy surgery outcome classifications in a series of 76 patients. Seizure Eur J Epilepsy. 2011;20(10):809–812. doi:10.1016/j.seizure.2011.08.004
5. Passamonti C, Zamponi N, Foschi N, et al. Epilepsy & Behavior Long-term seizure and behavioral outcomes after corpus callosotomy. Epilepsy Behav. 2014;41:23–29. doi:10.1016/j.yebeh.2014.08.130
6. Wagenen V. Long-term seizure outcome after corpus callosotomy: a retrospective analysis of 95 patients. J Neurosurg. 2009;110:332–342. doi:10.3171/2008.3.17570
7. Chan AY, Rolston JD, Lee B, Vadera S, Englot DJ. Rates and predictors of seizure outcome after corpus callosotomy for drug-resistant epilepsy: a meta-analysis. J Neurosurg. 2018:1–10. doi:10.3171/2017.12.JNS172331
8. Sunaga S, Shimizu H, Sugano H, et al. Long-term follow-up of seizure outcomes after corpus callosotomy. Seizure. 2009;18(2):124–128. doi:10.1016/j.seizure.2008.08.001
9. Luat AF, Asano E, Kumar A, et al. Corpus callosotomy for intractable epilepsy revisited: the Children’s Hospital of Michigan series. J Child Neurol. 2018;32(7):624–629. doi:10.1177/0883073817697847
10. Rolston JD, Englot DJ, Wang DD, et al. Corpus callosotomy versus vagus nerve stimulation for atonic seizures and drop attacks: a systematic review. Epilepsy Behav. 2015;51:13–17. doi:10.1016/j.yebeh.2015.06.001.Corpus
11. Bower RS, Wirrell E, Wetjen NM, Marsh WR, Meyer FB. Seizure outcomes after corpus callosotomy for drop attacks. Neurosurgery. 2013;73(6):10–12. doi:10.1227/NEU.0000000000000161
12. Iwasaki M, Uematsu M, Sato Y, et al. Complete remission of seizures after corpus callosotomy. J Neurosurg Pediatr. 2012;10(July):7–13. doi:10.3171/2012.3.PEDS11544
13. Hader WJ, Tellez-zenteno J, Metcalfe A, et al. Complications of epilepsy surgery: a systematic review of focal surgical resections and invasive EEG monitoring. Epilepsia. 2013;54(5):840–847. doi:10.1111/epi.12161
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
