Back to Journals » Cancer Management and Research » Volume 10
Secondhand smoking increases bladder cancer risk in nonsmoking population: a meta-analysis
Authors Yan H , Ying Y , Xie H, Li J, Wang X, He L, Jin K, Tang J, Xu X, Zheng X
Received 23 May 2018
Accepted for publication 13 July 2018
Published 21 September 2018 Volume 2018:10 Pages 3781—3791
DOI https://doi.org/10.2147/CMAR.S175062
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Raphael Catane
Huaqing Yan, Yufan Ying, Haiyun Xie, Jiangfeng Li, Xiao Wang, Liujia He, Ke Jin, Jianer Tang, Xin Xu, Xiangyi Zheng
Department of Urology, The First Affiliated Hospital, College of Medicine, Zhejiang University, Hangzhou 310003, China
Background: Tobacco smoking has been widely acknowledged to be the most important risk factor for bladder cancer. However, whether secondhand smoking (SHS) increases the risk of bladder cancer still remains uncertain. We conducted a meta-analysis about the risk of bladder cancer and lifetime SHS and childhood SHS.
Materials and methods: We searched PubMed, EMBASE, Web of Science, and Chinese National Knowledge Infrastructure (CNKI) up to March 12, 2018, and checked references of the retrieved articles and relevant reviews to include 14 studies. Relative risk (RR) and 95% confidence interval (CI) were used to assess this risk.
Results: The pooled RR of 14 eligible studies based on the retrieved articles and relevant reviews illustrated a significantly increased risk of bladder cancer with RR 1.22, 95% CI 1.06–1.4. No heterogeneity or publication bias was found. But we need more evidence to prove a more reliable association between childhood SHS and bladder cancer.
Conclusion: There was a statistically significant 22% increased risk of bladder cancer for lifetime SHS exposure in nonsmoking patients compared with unexposed nonsmoking population. But the association between childhood SHS exposure compared with unexposed nonsmoking population was unclear. Further research should be conducted to confirm our findings and reveal the potential biological mechanisms.
Keywords: environmental smoking, bladder tumor, nonsmoking adult, childhood exposure
Introduction
Bladder cancer is the ninth most common cancer globally, with an estimated 430,000 new cases and 165,000 bladder cancer deaths in 2012.1 A significant male predominance was observed, with male bladder cancer cases comprising three quarters of the total cases. Europe has among the highest incidence rates of bladder cancer in the world. The highest rate of incidence in men was recorded in Spain with age-standardized rate = 36.7 per 100,000 according to Cancer Registry Data. New bladder cancer diagnoses rose 1.5-fold between 1990 and 2013 with deaths rising 1.3-fold between 1990 and 2013.2
Tobacco smoking has been widely acknowledged to be the most important risk factor for bladder cancer. However, whether secondhand smoking (SHS) increases the risk of bladder cancer still remains uncertain. Although a number of clinical studies were conducted to identify the association between SHS and bladder cancer,3–16 only one of these studies reported a significantly increased risk of SHS and bladder cancer in nonsmoking population. Six studies reported an elevated risk of bladder cancer in lifelong nonsmokers who were exposed to SHS compared with those who had no SHS exposure but without significance, while others reported a null relationship.3,5,9,10,13,16 This may mainly be due to the measurement error of accessing SHS and the lack of nonsmoking bladder cancer cases exposed to tobacco.
Meta-analysis could synthesize the evidence provided by studies mentioned above. Thus, we could not only reach the most extensive study population but also minimize the influence of methodological heterogeneity of each study and exclude some low-quality studies. So, we urgently need a meta-analysis that provides a higher level of evidence to draw a reliable conclusion about SHS and bladder cancer. In 2009, the only meta-analysis published reported no association between SHS and bladder cancer.17 However, this meta-analysis included only eight studies. So, we did this meta-analysis including 14 studies aiming to ascertain the association between SHS and bladder cancer. Additionally, we did another meta-analysis about exposure to SHS in childhood to identify whether the risk between childhood SHS and bladder cancer is higher than lifetime exposure.
Materials and methods
Publication search
This meta-analysis was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines.18 We carried out a search in PubMed, EMBASE, Web of Science, and Chinese National Knowledge Infrastructure (CNKI) databases to cover all the eligible articles published up to March 12, 2018. The search strategy included terms for outcome (bladder neoplasm or bladder cancer or bladder tumor or bladder tumour or carcinoma of bladder) and exposure [Secondhand smok* or ETS*(environmental tobacco smoke) or environmental smok* or SHS or passive smok* or tobacco smoke pollution]. We carefully evaluated every potentially relevant publication by scanning their titles and abstracts. All the studies matching the inclusion criteria were retrieved. The references from retrieved articles and reviews were also thoroughly checked to identify any additional relevant study. No language restrictions were imposed. The search process was done separately by two authors independently. Disagreement was resolved by discussion; if no agreement was reached, a third independent expert acted as an arbiter.
Inclusion criteria
Studies included in this meta-analysis had to meet all the following criteria: 1) They had a cohort design or case–control design; 2) The subjects included in this study contained nonsmokers; 3) The exposure of interest included SHS; 4) The outcome of studied subjects contained incidence of bladder cancer; 5) Studies provide odds ratio (OR), hazard ratio (HR), relative risk (RR), or standardized incidence ratio (SIR) with their 95% CIs or sufficient data to calculate them. Studies concerning mortality rates of bladder cancer were not included because it could be unpredictably confounded by survival-related factors. If multiple studies researching the same population were available, the most recent and detailed study was entitled to be included in this meta-analysis.
Data extraction
Two authors independently extracted data by using a predefined data collection form, with disagreements being resolved by consensus. For each study, the following characteristics were collected: first author’s family name, year of publication, participant characteristics (age and gender), study type, study population, range for follow-up, numbers of events and nonevent subjects, and covariates adjusted for in the analysis. From each of the studies, we optionally extracted the RR or OR estimate that was adjusted for the greatest number of potential confounders. If data were not shown in the full article, we contacted the corresponding author to get the desired data.8
Quality assessment
Quality assessments of searched papers were also undertaken independently by two authors according to the Newcastle–Ottawa scale. The disagreements between authors were resolved by discussion. If no agreement was reached, a third independent expert acted as an arbiter.
Statistical methods
The OR was assumed the same as RR, and the RR was defined as the study outcome to estimate the risk of SHS and bladder cancer. These two measurements of effects can similarly estimate RR because the absolute risk of bladder cancer is low. In the study by Kabat et al,11 adjusted OR or RR was not provided, so we calculated raw OR and its 95% CI using original data. If several approaches of “exposure to SHS” were reported in a publication while overall RR was not provided, the RRs with their 95% CI of each approach exposed to SHS were synthesized using random effects models to generate an RR that estimates the risk of SHS and bladder cancer in the publication (as in Figure S4).
Overall RR estimates with their corresponding 95% CIs were calculated with the random effects models, which explicitly accounts for the heterogeneity of studies through a statistical parameter representing the interstudy variation.19 Heterogeneity among studies was measured by Q-test and quantified by I2, with higher value contributing to greater degree of heterogeneity. Publication bias was assessed using Begg’s test and Egger’s test. P<0.05 was considered to be representative of a significant statistical publication bias. Sensitivity analyses were also performed to assess the stability of obtained results with a single study deleted each time to manifest the influence of the individual dataset to the pooled RR. All the statistical analyses were performed with STATA 14.0 (StataCorp, College Station, TX, USA), using two-sided P-values.
Results
Literature search
Figure 1 outlines our study searching and selecting process. Briefly, after removing publication duplications, our search strategy generated 524 articles. After a first scanning of the titles and abstracts of these articles, the majority were excluded generally because they were reviews, experimental researches, meeting abstracts, and obviously irrelevant to our study. Thirty-six articles seemed relevant to our study, and we tried to identify and screen the full text. We were unable to get one potentially relevant publication by Sandler et al.20 After full-text reviewing of 35 papers, 21 studies were excluded for the reasons as follows: 1) Included population were not secondhand smokers (n=19); 2) Outcomes were not bladder cancer (n=2). Although we checked the reference list of retrieved studies and reviews carefully, no new articles were obtained. Thus, a total of 14 cohort studies or case–control studies,3–16 which met the inclusion criteria, were included in the meta-analysis. Of these studies, six were never included in former meta-analysis.4,8,9,13,14,16
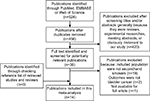  | Figure 1 Flowchart of study assessment and selection. |
Study characteristics
The characteristics of the 14 studies are presented in Table 1. Of these, three were cohort studies and 11 were case–control studies. The study population in 14 studies consisted of both sexes. The included studies were published between 1986 and 2012. The sample size ranged from 1267 to 220,7905. The total population included in this meta-analysis reaches 325,264 with 4,002 cases and 321,262 controls. Adjustments were made for potential confounders of more than two factors in all 14 studies.
Exposure to SHS and risk of bladder cancer
A significantly increased risk of exposure to SHS and bladder cancer was found in our review (Figure 2; RR 1.22, 95% CI 1.06–1.40). There is barely any heterogeneity among these studies with Q=10.04, P=0.691, and I2=0.
When we did meta-analysis about childhood SHS and bladder cancer, an even more higher risk that is statistically significant was detected (Figure S1, RR 1.43, 95% CI 1.03–1.98). But there was significant heterogeneity in these studies with Q=16.05, P=0.03, and I2=56.4%.
Publication bias
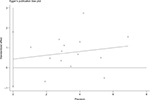
There was no evidence demonstrating significant publication bias either with Begg’s test (P=0.74) or with Egger’s test (Figure S2, P=0.50). In the meta-analysis concerning childhood SHS and risk of bladder cancer, a significant publication bias was detected with Egger’s test (Figure S3, P=0.09).
Sensitivity analysis
Sensitivity analysis of the pooled studies was performed to test the stability of our result. By means of the random effects model, we calculated all integrated RR or OR once again. When omitting each study in the meta-analysis, the pooled result (Table S1) always remained stable. When we analyzed the sensitivity of childhood SHS and bladder cancer, the result changed several times (Table S2).
Discussion
The present meta-analysis synthesized all eligible evidence from 11 case–control studies and three cohort studies and provided a statistically significant 22% increased risk of bladder cancer for lifetime SHS exposure in nonsmoking patients compared with unexposed nonsmoking population. Our result was convincing because there was no heterogeneity detected, which indicated that the combination of these 14 studies was reasonable. Sensitivity analysis showed that when omitting each study, the result remains always consistent, which represents a good stability of this meta-analysis. There was no evidence showing obvious publication bias either.
When we conducted another meta-analysis of childhood exposure to SHS and risk of bladder cancer, we found an even higher increased risk of 43% significantly. But we found obvious heterogeneity and publication bias in this study. Meanwhile, the result of sensitivity analysis was not stable either. It means that the increasing risk between childhood SHS and bladder cancer is not reliable and should be treated with caution. In sensitivity analysis when we delete the study by Dai et al,8 the heterogeneity decreased and became acceptable with I2=11.3% and P=0.343, RR=1.21 and its 95% CI 0.95–1.53. This result suggests that the study by Dai et al contributes most of the heterogeneity in this meta-analysis.8 The source of heterogeneity may mainly stem from methodological diversity and measure error when conducting multicenter case–control study. More studies should be done in future to identify a relationship between childhood SHS exposure and bladder cancer.
Smoking population has a four-fold risk of bladder cancer compared with nonsmokers.21 Chemically, tobacco contains over 60 species of carcinogens including polycyclic aromatic hydrocarbons and aromatic amines, which could excrete via kidney. This process could damage urethra and lead to the incidence of bladder cancer. In a basic research in vitro by Geng et al, exposure to cigarette smoke extract (CSE) induced proliferation in normal human urothelial cells via the mitogen-activated protein kinase-1/activator protein-1 pathway, which causes CSE-related carcinogenesis of bladder cancer.22 In another basic research, nicotine in tobacco induces tumor growth and chemoresistance through activation of the phosphatidylinositol-3-kinase/Akt/mammalian target of rapamycin pathway in bladder cancer.23 Our study suggests that secondhand smoke may contain these carcinogens and activate the pathways mentioned above. This point was proved in part by an experiment demonstrating that cigarette sidestream smoke induced mutagenic acrolein-DNA adducts, inhibited DNA repair, and enhanced anchorage-independent growth cell transformation in mice.24 In the future, more basic experiments should be done to confirm that secondhand smoke contributes to the formation of bladder cancer.
Our study has several strengths. First, we take a transparent and robust approach to examine the evidence base, including adherence to PRISMA guidelines. Second, we obtained some data that were not provided in article through contacting the author8 and included two Chinese studies,8,9 so this meta-analysis included the broadest and most comprehensive data about SHS exposure and risk of bladder cancer. Third, our heterogeneity was inapparent with a low risk of publication bias and good result stability, which enhanced the strength and reliability of our result. Fourth, we were able to analyze the association between childhood SHS and bladder cancer and suggest that more clinical research should be done to address this issue in the future.
Of course, several limitations should be mentioned. First, most of the papers included were case–control studies; therefore, selection biases and recall biases could not be avoided. Second, only published studies were included in this meta-analysis. Even if Begg’s test and Egger’s test did not show evidence of publication bias, some inevitable publication bias may exist because studies with null results tend not to be published. Third, we could not solve the potential confounding factors like diabetes, drug use, or other unreported factors in the included studies.25 In some included studies, only two confounders were adjusted.8,9,15 Finally, we could not give an explicit evidence to prove the association between childhood SHS exposure and bladder cancer risk. Because of these above, more future cohort studies are expected to be carried out to confirm the conclusion in this meta-analysis.
In conclusion, this meta-analysis indicated that there was a statistically significant 22% increased risk of bladder cancer for lifetime SHS exposure in nonsmoking patients compared with unexposed nonsmoking population. But the association between childhood SHS exposure compared with unexposed nonsmoking population was unclear. Further research should be conducted to confirm our findings and clarify the potential biological mechanisms.
Ethics approval
All analyses were based on previous published studies; thus, no ethical approval and patient consent were required.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (Grant No. 81472375 and No. 81702500) and Zhejiang Province Medical Technology Project (2016KYA173).
Disclosure
The authors report no conflicts of interest in this work.
References
Antoni S, Ferlay J, Soerjomataram I, Znaor A, Jemal A, Bray F. Bladder cancer incidence and mortality: a global overview and recent trends. Eur Urol. 2017;71(1):96–108. | ||
Dy GW, Gore JL, Forouzanfar MH, Naghavi M, Fitzmaurice C. Global burden of urologic cancers, 1990-2013. Eur Urol. 2017;71(3):437–446. | ||
Alberg AJ, Kouzis A, Genkinger JM, et al. A prospective cohort study of bladder cancer risk in relation to active cigarette smoking and household exposure to secondhand cigarette smoke. Am J Epidemiol. 2007;165(6):660–666. | ||
Baris D, Karagas MR, Verrill C, et al. A case-control study of smoking and bladder cancer risk: emergent patterns over time. J Natl Cancer Inst. 2009;101(22):1553–1561. | ||
Bjerregaard BK, Raaschou-Nielsen O, Sørensen M, et al. Tobacco smoke and bladder cancer--in the European Prospective Investigation into Cancer and Nutrition. Int J Cancer. 2006;119(10):2412–2416. | ||
Burch JD, Rohan TE, Howe GR, et al. Risk of bladder cancer by source and type of tobacco exposure: a case-control study. Int J Cancer. 1989;44(4):622–628. | ||
Chen YC, Su HJ, Guo YL, Houseman EA, Christiani DC. Interaction between environmental tobacco smoke and arsenic methylation ability on the risk of bladder cancer. Cancer Causes Control. 2005;16(2):75–81. | ||
Dai QS, He HC, Cai C, et al. Multicenter case-control study of the relationship between smoking and bladder cancer in China. Zhonghua Yi Xue Za Zhi. 2011;91(34):2407–2410. | ||
Hu Z, Su Y, Zeng X, et al. Relationship between active smoking and environmental tobacco smoking with the risk of bladder cancer: A hospital-based case-control study in Wuhan. Chinese J Exp Surg. 2010;27:1308–1310. | ||
Jiang X, Yuan JM, Skipper PL, Tannenbaum SR, Yu MC. Environmental tobacco smoke and bladder cancer risk in never smokers of Los Angeles County. Cancer Res. 2007;67(15):7540–7545. | ||
Kabat GC, Dieck GS, Wynder EL. Bladder cancer in nonsmokers. Cancer. 1986;57(2):362–367. | ||
Samanic C, Kogevinas M, Dosemeci M, et al. Smoking and bladder cancer in Spain: effects of tobacco type, timing, environmental tobacco smoke, and gender. Cancer Epidemiol Biomarkers Prev. 2006;15(7):1348–1354. | ||
Tao L, Xiang YB, Wang R, et al. Environmental tobacco smoke in relation to bladder cancer risk--the Shanghai bladder cancer study [corrected]. Cancer Epidemiol Biomarkers Prev. 2010;19(12):3087–3095. | ||
Wei Z, Bing XY, Xia SC, et al. Relationship between smoking and exposure to environmental tobacco smoking with bladder cancer: a case-control study in Shanghai. Tumor. 2006;26:42–47. | ||
Zeegers MP, Goldbohm RA, van den Brandt PA. A prospective study on active and environmental tobacco smoking and bladder cancer risk (The Netherlands). Cancer Causes Control. 2002;13(1):83–90. | ||
Zheng YL, Amr S, Saleh DA, et al. Urinary bladder cancer risk factors in Egypt: a multicenter case-control study. Cancer Epidemiol Biomarkers Prev. 2012;21(3):537–546. | ||
van Hemelrijck MJ, Michaud DS, Connolly GN, Kabir Z. Secondhand smoking, 4-aminobiphenyl, and bladder cancer: two meta-analyses. Cancer Epidemiol Biomarkers Prev. 2009;18(4):1312–1320. | ||
Moher D, Shamseer L, Clarke M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev. 2015;4:1. | ||
Dersimonian R, Kacker R. Random-effects model for meta-analysis of clinical trials: an update. Contemp Clin Trials. 2007;28(2):105–114. | ||
Sandler DP, Everson RB, Wilcox AJ. Passive smoking in adulthood and cancer risk. Am J Epidemiol. 1985;121(1):37–48. | ||
Freedman ND, Silverman DT, Hollenbeck AR, Schatzkin A, Abnet CC. Association between smoking and risk of bladder cancer among men and women. JAMA. 2011;306(7):737–745. | ||
Geng H, Zhao L, Liang Z, et al. Cigarette smoke extract-induced proliferation of normal human urothelial cells via the MAPK/AP-1 pathway. Oncol Lett. 2017;13(1):469–475. | ||
Yuge K, Kikuchi E, Hagiwara M, et al. Nicotine induces tumor growth and chemoresistance through activation of the PI3K/Akt/mTOR pathway in bladder cancer. Mol Cancer Ther. 2015;14(9):2112–2120. | ||
Lee HW, Wang HT, Weng MW, et al. Cigarette side-stream smoke lung and bladder carcinogenesis: inducing mutagenic acrolein-DNA adducts, inhibiting DNA repair and enhancing anchorage-independent-growth cell transformation. Oncotarget. 2015;6(32):33226–33236. | ||
Yan H, Xie H, Ying Y, et al. Pioglitazone use in patients with diabetes and risk of bladder cancer: a systematic review and meta-analysis. Cancer Manag Res. 2018;10:1627–1638. |
Supplementary materials
  | Table S1 Sensitivity analysis of SHS and bladder cancer Abbreviations: RR, relative risk; SHS, secondhand smoking; CI, confidence interval. |
  | Table S2 Sensitivity analysis of childhood SHS and bladder cancer Abbreviations: RR, relative risk; SHS, secondhand smoking; CI, confidence interval. |
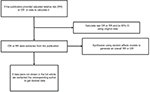  | Figure S4 Flowchart of data extraction and statistical methods. |
References
Kabat GC, Dieck GS, Wynder EL. Bladder cancer in nonsmokers. Cancer 1986;57:362–367. | ||
Burch JD, Rohan TE, Howe GR, et al. Risk of bladder cancer by source and type of tobacco exposure: A case-control study. International Journal of Cancer. 1989;44:622–628. | ||
Zeegers MP, Goldbohm RA, van den Brandt PA. A prospective study on active and environmental tobacco smoking and bladder cancer risk (The Netherlands). Cancer causes & control : CCC. 2002;13:83–90. | ||
Chen YC, Su HJ, Guo YL, Houseman EA, Christiani DC. Interaction between environmental tobacco smoke and arsenic methylation ability on the risk of bladder cancer. Cancer causes & control : CCC. 2005;16:75–81. | ||
Bjerregaard BK, Raaschou-Nielsen O, Sorensen M, et al. Tobacco smoke and bladder cancer--in the European Prospective Investigation into Cancer and Nutrition. International Journal of Cancer. 2006;119:2412–2416. | ||
Samanic C, Kogevinas M, Dosemeci M, et al. Smoking and bladder cancer in Spain: effects of tobacco type, timing, environmental tobacco smoke, and gender. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 2006;15:1348–1354. | ||
Wei Z, Bing XY, Xia SC, et al. Relationship between smoking and exposure to environmental tobacco smoking with bladder cancer..a case-control study in Shanghai. Tumor. 2006;26:42–47. | ||
Alberg AJ, Kouzis A, Genkinger JM, et al. A prospective cohort study of bladder cancer risk in relation to active cigarette smoking and household exposure to secondhand cigarette smoke. American Journal of Epidemiology. 2007;165:660–666. | ||
Jiang X, Yuan JM, Skipper PL, Tannenbaum SR, Yu MC. Environmental tobacco smoke and bladder cancer risk in never smokers of Los Angeles County. Cancer Research. 2007;67:7540–7545. | ||
Baris D, Karagas MR, Verrill C, et al. A Case-Control Study of Smoking and Bladder Cancer Risk: Emergent Patterns Over Time. Journal of the National Cancer Institute. 2009;101:1553–1561. | ||
Hu Z, Su Y, Zeng X, et al. Relationship between active smoking and environmental tobacco smoking with the risk of bladder cancer: A hospital-based case-control study in Wuhan. Chinese Journal of Experimental Surgery. 2010;27:1308–1310. | ||
Tao L, Xiang YB, Wang R, et al. Environmental tobacco smoke in relation to bladder cancer risk--the Shanghai bladder cancer study [corrected]. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2010;19:3087–3095. | ||
Dai QS, He HC, Cai C, et al. Multicenter case-control study of the relationship between smoking and bladder cancer in China. National Medical Journal of China. 2011;91:2407–2410. | ||
Zheng YL, Amr S, Saleh DA, et al. Urinary bladder cancer risk factors in Egypt: a multicenter case-control study. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 2012;21:537–546. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.