Back to Journals » OncoTargets and Therapy » Volume 14
Secondary Nasopharyngeal Mixed Adenoneuroendocrine Carcinoma After Radical Radiotherapy for Nasopharyngeal Carcinoma: A Rare Case and Literature Review
Authors Huang D, Huang C, Wang H, He L, Zhang J , Zhong X, Guo Z, Yuan Y, Zhang W
Received 23 September 2020
Accepted for publication 28 December 2020
Published 25 February 2021 Volume 2021:14 Pages 1367—1376
DOI https://doi.org/10.2147/OTT.S283071
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Jianmin Xu
Donglan Huang, 1,* Chunyue Huang, 1,* Hongmei Wang, 1,* Lizhen He, 1, 2,* Jian Zhang, 1,* Xi Zhong, 1, 3 Ziqian Guo, 1, 4 Yawei Yuan, 1 Weijun Zhang 1
1Department of Radiation Oncology, Affiliated Cancer Hospital & Institute of Guangzhou Medical University, Guangzhou, People’s Republic of China; 2Department of Pathology, Affiliated Cancer Hospital & Institute of Guangzhou Medical University, Guangzhou, People’s Republic of China; 3Department of Radiology, Affiliated Cancer Hospital & Institute of Guangzhou Medical University, Guangzhou, People’s Republic of China; 4Endoscopy Center, Affiliated Cancer Hospital & Institute of Guangzhou Medical University, Guangzhou, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Weijun Zhang; Yawei Yuan
Department of Radiation Oncology, Affiliated Cancer Hospital & Institute of Guangzhou Medical University, 78 Hengzhigang Road, Guangzhou, 510095, People’s Republic of China
Tel + 86 20 66673662; +86 20 62931358
Email [email protected]; [email protected]
Abstract: The incidence of primary mixed adenoneuroendocrine carcinoma (MANEC) is rapidly increasing. MANEC mainly arises from the gastrointestinal tract, but occasionally it occurs as a pathological type of second primary malignancy (SPM). These SPMs can occur in the nasopharynx. Herein we describe the case of a first secondary nasopharyngeal MANEC that was detected 20 years after radical radiotherapy for nasopharyngeal carcinoma. The patient was a 50-year-old man who was admitted to our hospital after experiencing 1 month of left nasal congestion and ipsilateral tinnitus caused by a nasopharyngeal mass that was detected via physical examination and magnetic resonance imaging. A biopsy specimen from this nasopharyngeal lesion led to a histopathological diagnosis of recurrent nasopharyngeal carcinoma. He underwent high-dose palliative radiotherapy, followed by a course of gemcitabine–cisplatin-based adjuvant chemotherapy. These treatments failed to achieve local control of the tumor, and progressive left earache emerged. Another two forceps biopsies of the external auditory canal mass were conducted, and immunohistochemical testing for adenocarcinoma and neuroendocrine carcinoma markers including CK7, CK8, CK18, carcinoembryonic antigen, synaptophysin, chromogranin A, and CD56 was conducted. The diagnosis of MANEC was ultimately confirmed 5 months after the first visit, and one additional cycle of chemotherapy was subsequently performed. The patient died of hepatic metastases 8 months after the final diagnosis. Knowledge of this rare case will raise awareness of MANEC as a new pathological type of SPM originating in the nasopharynx, which will reduce delays and promote early diagnosis.
Keywords: adenocarcinoma, head and neck, neuroendocrine carcinoma, second primary malignancy
Corrigendum for this paper has been published
Introduction
Mixed adenoneuroendocrine carcinoma (MANEC) is a blended carcinoma with adenocarcinoma and neuroendocrine carcinoma (NEC) components, in which the percentage of each component is greater than 30%.1 It is commonly located in the digestive system, including in the gastrointestinal tract, pancreas, and gallbladder.2–5 Primary MANEC in the head and neck is rare, with only four reported cases to date,6–9 located in the tongue, sinonasal tract, and soft palate (Table 1). No cases of primary MANEC in the nasopharynx have previously been reported.
 |
Table 1 Case Reports of Mixed Adenoneuroendocrine Carcinomas of the Head and Neck |
Nasopharyngeal carcinoma (NPC) is the most common malignant tumor of the nasopharynx. Population studies conducted in Taiwan and the USA indicate that patients with NPC have a high risk of second primary malignancy (SPM).10,11 Squamous cell carcinoma is considered to be a general pathological type of SPM in patients with a previous history of NPC treated with radiotherapy,12 but SPM with the pathological type of MANEC has not been previously reported. Herein we provide the first report of a case of SPM located in the nasopharynx with the pathological type of MANEC, after successful curative radiotherapy for NPC. In this case, the preliminary diagnosis was undifferentiated nonkeratinized carcinoma, and the definitive diagnosis was only confirmed after additional immunohistochemical staining for adenocarcinoma and NEC markers in additional two biopsy specimens.
Case Report
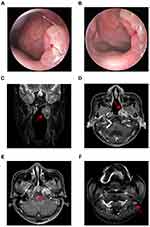
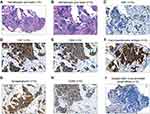
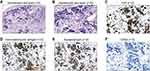
A 50-year-old Cantonese man was admitted to our hospital with a 1-month history of left-sided nasal congestion and ipsilateral tinnitus in July 2016. Physical examination revealed a mass occupying the left wall of the nasopharynx and left torus tubarius. Magnetic resonance imaging (MRI) depicted a 26 × 16 × 27 mm3 contrast-enhanced nasopharyngeal mass extending into the left fossa of Rosenmüller, para-pharyngeal space and skull base (Figure 1C–E), with the involvement of a left cervical lymph node (level II, 12 × 6 × 14 mm3) (Figure 1F). A biopsy of the nasopharyngeal mass was performed with direct nasopharyngoscopy (Figure 1A and B), and the microscopic pathology was interpreted as undifferentiated nonkeratinizing carcinoma (Figure 2A and B).
Neither chest x-ray nor abdominal sonography depicted any signs of lung or liver metastases. There were no gastrointestinal signs, and a stool occult blood test was negative. The patient declined positron emission tomography and further pathological immunohistochemical examination because his basic health insurance was not yet in place. A detailed patient history was obtained, including 4th edition American Joint Committee on Cancer (AJCC) stage III, and single conventional external radiotherapy (70 Gy/35 fractions) without chemotherapy for undifferentiated NPC in 1995. Based on these findings, a diagnosis of NPC recurring after radiotherapy with stage III (rT3N1M0) (AJCC 7th edition) was suggested.
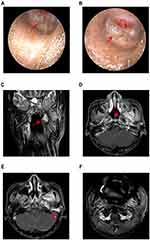
The patient declined surgery and neoadjuvant and concurrent chemotherapy but did undergo nasopharyngeal re-radiotherapy at doses of 66 Gy to nasopharynx gross tumor volume, 60 Gy to gross tumor volume of lymph node and high-risk clinical target volume, and 54 Gy to the low-risk clinical target volume, delivered in 33 fractions with a radiotherapy treatment duration of 50 days. After re-radiotherapy for 2 months, the therapeutic effects on the progressive disease were assessed via MRI at another hospital. He returned to our hospital again and started a course of gemcitabine-cisplatin-based first-line chemotherapy. After chemotherapy, a left external auditory canal mass was detected via a physical examination prompted by a progressive left earache. Another MRI scan verified that parapharyngeal tumor had extended to the middle left ear and the external auditory canal (Figure 3E), although a partial reduction in nasopharyngeal tumor size (Figure 3C and D) and a complete response in the metastatic cervical lymph node (Figure 3F) were evident.
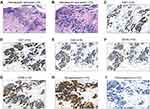
A forceps biopsy of the mass in the external ear was performed under electro-otoscopy (Figure 3A and B). Direct microscopic examination revealed that most of the tumor cells were arranged in a tubular pattern, although some were visibly squeezed (Figure 4A and B). An immunohistochemical test was then conducted because the patient’s insurance had come into effect by that time. In that test, the Ki-67 index value at the hotspot was >90%, and CK7, CK8, and CK18 were strongly positive (Figure 4C–F). Accordingly, a pathological diagnosis of adenocarcinoma was made at that time. Considering that the squeezing of tumor cells might have affected the diagnosis, and that nasopharyngeal adenocarcinoma is rare, a biopsy of the lesion in the external ear was performed again to confirm the diagnosis. Morphology and immunophenotyping analysis of the second biopsy specimen from the external ear were similar to those of the first biopsy specimen from the external ear (Figure 5A–D), and the cells lacked CDX2 expression (Figure 5F). To further characterize the carcinoma the two biopsy specimens were stained for other tumor markers, and it was determined that most of the adenocarcinoma cells simultaneously expressed NEC markers including synaptophysin, chromogranin A, and CD56 (Figures 4G–I and 5E). Adenocarcinoma and NEC markers were simultaneously expressed in >50% of the tumor cells, and the adenocarcinoma cells were not of intestinal type. Thus, a pathological diagnosis of MANEC was clear.
To investigate the relationship between the external auditory canal tumor and the nasopharyngeal tumor, and confirm that the MANEC had originated in the nasopharynx, adenocarcinoma and NEC markers were added to the immunohistochemical analysis of the previously obtained nasopharyngeal biopsy tissue. The tumor cells of the previous nasopharyngeal mass had parallel morphological and immunohistochemical profiles, i.e., >90% exhibited simultaneous expression of the adenocarcinoma markers CK7, CK8, and carcinoembryonic antigen, and the NEC markers CD56 and synaptophysin (Figure 2C–H), and in situ Epstein-Barr virus-encoded small RNA hybridization was negative (Figure 2I). Repeated fecal occult blood tests were also negative, and the patient declined gastrointestinal endoscopy. In December 2016 he was ultimately diagnosed with nasopharyngeal MANEC, which was consistent with the levels of the serum tumor markers neuron-specific enolase 22.81 ng/mL (reference range 0.00–16.30 ng/mL) and carcinoembryonic antigen 8.90 ng/mL (reference range 0.00–5.00 ng/mL). Due to poor nutritional status and moderate anemia, he subsequently underwent one additional cycle of chemotherapy (paclitaxel liposomes 150 mg/m2 and cisplatin 70 mg/m2) when his left earache failed to be relieved. He received no oncotherapy thereafter, and was switched to conservative care. The patient died of multiple liver metastases as determined via contrast-enhanced computed tomography of the abdomen, having survived for 8 months after the definitive diagnosis.
Discussion
Most MANECs are located in the gastro-entero-pancreatic tract, where complete surgical specimens can be easily obtained, and there is usually a clear boundary between adenocarcinoma and NEC components. Given the lack of reports of MANECs in the nasopharynx, or of any SPM of this pathological type, the diagnosis of the case presented herein was a challenge for pathologists. Regardless of medical history, imaging features, and conventional pathology, the initial putative diagnosis was nasopharyngeal undifferentiated nonkeratinizing carcinoma rather than MANEC. The diagnosis of MANEC was made 5 months later, after immunohistochemical assessment of the markers CK7, CK8, CK18, CD56, chromogranin A, and synaptophysin. This indicates that supplemental staining of adenocarcinoma and NEC markers is required when nasopharyngeal epithelial malignancy cells exhibit poor differentiation.
The precise pathogenic mechanisms involved in MANEC are not yet understood.13,14 In previous studies, molecular analysis suggested that the adenocarcinoma and NEC components of MANEC had a common clonal origin.15,16 This was putatively confirmed in subsequent studies that indicated the early separation of two components during malignant transformation, with subsequent independent mutational evolution.17,18 Similar indications were apparent in the current MANEC case in which there was no boundary between the adenocarcinoma, NEC cells were present, and CK18, carcinoembryonic antigen, synaptophysin, and chromogranin A were co-expressed in most of the tumor cells.
In the present patient, the diagnosis of MANEC was confirmed by three biopsies from different sites. These biopsies suggested that the pathological manifestations of MANEC are relatively heterogeneous, and that the accuracy of its diagnosis is related to biopsy depth and biopsy site. When the two components (adenocarcinoma and NEC) are longitudinally distributed in gastric tissue, the pathological manifestations may be inconsistent with those of tumor tissues from different biopsy depths.19 Moreover, more than 60% of gastro-entero-pancreatic MANEC patients are initially diagnosed with a single adenocarcinoma or NEC based on the initial biopsy tissue sample.20 Thus, horizontal and vertical multipoint biopsy should be performed to ensure the accuracy of diagnosis in inoperable carcinoma patients.
The therapeutic options for MANEC remain limited. In a systematic review summarizing 18 retrospective studies (n = 571), more than 90% of patients with gastro-entero-pancreatic MANEC were treated with surgery.20 In another systematic review, surgical resection was the preferred treatment for MANEC in the gallbladder.5 In a population-based study, surgery could significantly improve prognosis in colorectal MANEC, whereas radiation could not.21 There are four previous case reports describing patients with head and neck MANEC who underwent surgical treatment (Table 1).6–9 In the current patient the nasopharyngeal tumor surrounded the carotid sheath and invaded the skull base, where surgery is difficult due to complex anatomy. The patient declined this high-risk surgery and underwent radiotherapy, which ultimately failed to control the primary tumor. If he had undergone the operation at the beginning, his diagnosis may have been confirmed earlier and this may have resulted in a more beneficial clinical outcome. In most MANEC patients there is a characteristic tendency for metastasis to ensue, and MANEC in the head and neck is expected to be no exception. A recent study indicates that neoadjuvant chemotherapy can prolong the overall survival of MANEC patients and does not increase the risk of postoperative complications.22 In the current patient, there was no obvious response to platinum-based adjuvant chemotherapy, and he ultimately died of liver metastases. The effective use of systemic therapy to reduce the risk of distant metastasis of MANEC remains a challenge. In recent studies, colonic MANEC has responded favorably to streptozocin monotherapy and anti-programmed death 1 receptor monoclonal antibody pembrolizumab therapy.23,24 Therefore, we speculate that chemoradiotherapy with a combination of streptozocin and immune-checkpoint inhibitors may be a superior treatment option for inoperable MANEC.
In the present case of MANEC, some information that may have facilitated an earlier diagnosis was lacking. The patient’s initial MRI images depicted the presence of cervical lymph node metastasis. This meant that there was a higher risk of distant migration, but he declined whole-body positron emission tomography/computed tomography scanning. When a definite diagnosis of MANEC was made, he declined gastroenteroscopic examination. Strictly speaking, there was a lack of hard evidence to exclude the tumor having originated in the gastrointestinal tract, but it is notable in this regard that the fecal occult blood test was negative.
Conclusion
Herein, we have presented the first report of a patient diagnosed with secondary nasopharyngeal MANEC after radical radiotherapy for NPC. Its rarity may have delayed the diagnosis, and performing surgery was problematic due to the complex anatomy involved and a previous history of definitive radiotherapy for carcinoma of the nasopharynx. A likely tendency to metastasize also had to be considered, and there was a limited response to platinum-based chemotherapy. Multipoint biopsy at different depths and sites is recommended in cases of poorly differentiated carcinomas, and further research to develop more efficient therapies for nasopharyngeal MANEC is warranted.
Ethics Approval and Consent for Publication
This study was approved by the research ethics committee of Affiliated Cancer Hospital & Institute of Guangzhou Medical University and written informed consent for publication of the clinical details and images was obtained from the patient’s wife.
Informed Consent Statement
Informed written consent was obtained from the patient’s wife for publication of this report and any accompanying images.
Acknowledgments
We thank the head and neck cancer multidisciplinary board and Dr. Min Hou for the assistance and helpful advice.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
This work was financially supported by the Fund of Key Clinical Technique of Guangzhou (No. 2019ZD17).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Rindi G, Arnold R, Bosman FT. Nomenclature and classification of neuroendocrine neoplasms of the digestive system. In: Bosman FT, Carneiro F, Hruban RH, Theise ND, editors. WHO Classification of Tumours of the Digestive System. Lyon: LARC Press; 2010:13–14.
2. Xie JW, Lu J, Wang JB, et al. Prognostic factors for survival after curative resection of gastric mixed adenoneuroendocrine carcinoma: a series of 80 patients. BMC Cancer. 2018;18(1):1021. doi:10.1186/s12885-018-4943-z
3. Wang J, He A, Feng Q, et al. Gastrointestinal mixed adenoneuroendocrine carcinoma: a population level analysis of epidemiological trends. J Transl Med. 2020;18(1):128. doi:10.1186/s12967-020-02293-0
4. Tian F, Dai MH, Jia CW, Liu ZW, Li BL. Retrospective analysis of seven cases of pancreatic mixed adenoneuroendocrine carcinoma from a high-volume center and review of the literature. BMC Surg. 2019;19(1):89.
5. Machairas N, Paspala A, Frountzas M, et al. Mixed Adenoneuroendocrine Carcinoma (MANEC) of the gallbladder: a systematic review of outcomes following surgical management. In Vivo. 2019;33(6):1721–1726. doi:10.21873/invivo.11662
6. La Rosa S, Furlan D, Franzi F, et al. Mixed exocrine-neuroendocrine carcinoma of the nasal cavity: clinico-pathologic and molecular study of a case and review of the literature. Head Neck Pathol. 2013;7(1):76–84. doi:10.1007/s12105-012-0379-y
7. Ligthelm LJ, Bunn BK, Raubenheimer EJ, van Heerden WFP. Mixed adenoneuroendocrine carcinoma of the tongue arising within a congenital enteric cyst. Head Neck. 2018;40(5):E53–E57. doi:10.1002/hed.25117
8. Issa K, Madden J, Jang D, Zomorodi AR, Hachem RA. Sinonasal adenocarcinoma with neuroendocrine differentiation: case report and review of literature. J Neurol Surg B Skull Base. 2020;81(Supplement):1.
9. Hubale BS, Bal M, Patil A, Mittal N, Rane S. A mixed adeno-neuroendocrine carcinoma ex-pleomorphic adenoma of soft palate: a heretofore undescribed occurrence. Indian J Pathol Microbiol. 2019;62(5):S91.
10. Lin C, Lin SW, Weng SF, Lin YS. Risk of second primary malignancies after nasopharyngeal carcinoma: a population-based cohort study in Taiwan. Head Neck. 2014;36(2):209–214. doi:10.1002/hed.23281
11. Chan JY, Gooi Z, Mydlarz WK, Agrawal N. Risk of second primary malignancy after nasopharyngeal carcinoma in the United States: a population-based study. Head Neck. 2016;38(Suppl 1):E1130–E1136. doi:10.1002/hed.24173
12. Wang X, Li Y, Gao Y, Ouyang D, Guo Z. Second primary squamous cell carcinoma in patients with nasopharyngeal carcinoma after radiotherapy-predictors and prognostic factors. Head Neck. 2018;40(7):1434–1442. doi:10.1002/hed.25130
13. Zheng SL, Yip VS, Pedica F, Prachalias A, Quaglia A. Intrahepatic bile duct mixed adenoneuroendocrine carcinoma: a case report and review of the literature. Diagn Pathol. 2015;10:204. doi:10.1186/s13000-015-0439-1
14. Zhang D, Li P, Szankasi P, Liao X. Mixed adenoneuroendocrine carcinoma of the gallbladder, amphicrine type: case report and review of literature. Pathol Res Pract. 2020;216(7):152997. doi:10.1016/j.prp.2020.152997
15. Reu S, Neumann J, Kirchner T. Gastrointestinal mixed adenoneuroendocrine carcinomas. An attempt at classification of mixed cancers. Pathologe. 2012;33(1):31–38. doi:10.1007/s00292-011-1552-x
16. La Rosa S, Marando A, Sessa F, Capella C. Mixed Adenoneuroendocrine Carcinomas (MANECs) of the gastrointestinal tract: an update. Cancers. 2012;4(1):11–30. doi:10.3390/cancers4010011
17. Woischke C, Schaaf CW, Yang HM, et al. In-depth mutational analyses of colorectal neuroendocrine carcinomas with adenoma or adenocarcinoma components. Mod Pathol. 2017;30(1):95–103. doi:10.1038/modpathol.2016.150
18. Koh J, Nam SK, Kwak Y, et al. Comprehensive genetic features of gastric mixed adenoneuroendocrine carcinomas and pure neuroendocrine carcinomas. J Pathol. 2020:e5556. doi:10.1002/path.5556
19. Jeong J, Jung K, Kim JH, et al. Mixed neuroendocrine-non-neuroendocrine neoplasm of the stomach that is distributed in depth on the same tumor: inconsistent with the definition of mixed adenoneuroendocrine carcinoma in the 2010 World Health Organization Classification of tumors of the digestive system. Korean J Gastroenterol. 2019;74(6):349–355. doi:10.4166/kjg.2019.74.6.349
20. Frizziero M, Chakrabarty B, Nagy B, et al. Mixed neuroendocrine non-neuroendocrine neoplasms: a systematic review of a controversial and underestimated diagnosis. J Clin Med. 2020;9(1):273. doi:10.3390/jcm9010273
21. Song LJ, Yuan L. Comparative analysis of colorectal mixed adenoneuroendocrine carcinoma and adenocarcinoma with neuroendocrine differentiation: a population-based study. Int J Clin Exp Pathol. 2019;12(3):922–932.
22. Ma F, Wang B, Xue L, et al. Neoadjuvant chemotherapy improves the survival of patients with neuroendocrine carcinoma and mixed adenoneuroendocrine carcinoma of the stomach. J Cancer Res Clin Oncol. 2020;146(8):2135–2142. doi:10.1007/s00432-020-03214-w
23. Tagai N, Goi T, Morikawa M, et al. Favorable response of colonic mixed adenoneuroendocrine carcinoma to streptozocin monotherapy. Int Cancer Conf J. 2017;6(4):175–179. doi:10.1007/s13691-017-0301-2
24. Stueger A, Winder T, Tinguely M, Petrausch U, Helbling D. Metastatic mixed adenoneuroendocrine carcinoma of the colon with response to immunotherapy with pembrolizumab: a case report. J Immunother. 2019;42(7):274–277. doi:10.1097/CJI.0000000000000279
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.