Back to Journals » Drug Design, Development and Therapy » Volume 16
Salidroside Protects Acute Kidney Injury in Septic Rats by Inhibiting Inflammation and Apoptosis
Authors Fan H, Su B, Le J, Zhu J
Received 10 February 2022
Accepted for publication 17 March 2022
Published 30 March 2022 Volume 2022:16 Pages 899—907
DOI https://doi.org/10.2147/DDDT.S361972
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Tuo Deng
Heng Fan,1,* Bin-jie Su,2,* Jian-wei Le,1 Jian-hua Zhu1
1Department of Intensive Care Unit, Ningbo First Hospital, Ningbo, Zhejiang Province, People’s Republic of China; 2Department of Intensive Care Unit, Ningbo First Hospital Haishu Branch, Ningbo, Zhejiang Province, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Jian-hua Zhu, Department of Intensive Care Unit, Ningbo First Hospital, No.59 Liuting Road, Ningbo, Zhejiang Province, 315010, People’s Republic of China, Tel/Fax +86-574-87084625, Email [email protected]
Purpose: To clarify the protective effect and mechanism of salidroside (SLDS) on acute kidney injury (AKI) in septic rats.
Methods: We pretreated rats with different doses of SLDS and analyzed the impact of SLDS on the survival of septic rats. We evaluated the levels of inflammatory factors in rats, the expression of NF-ƙB p65 in the kidney, and the apoptosis of kidney tubular epithelial cells (KTECs).
Results: SLDS significantly decreased the mortality of septic rats, and it reduced the levels of TNF-α, IL-1β, and IL-17A in plasma and kidneys and decreased the levels of serum creatinine, plasma renal injury molecule-1 and plasma neutrophil gelatin-associated lipocalin. Moreover, SLDS could significantly decrease the expression of NF-ƙB p65 in kidney tissues and the apoptotic number of KETCs, while reducing the mRNA levels of Caspase-3 and Bax mRNA, and increasing the level of Bcl-2 mRNA.
Conclusion: SLDS pretreatment protects against AKI in septic rats by inhibiting the inflammation of kidney and the apoptosis of KTECs.
Keywords: salidroside, acute kidney injury, sepsis, apoptosis, inflammation
Introduction
Septic acute kidney injury (SAKI) is an acute injury of renal function caused by severe infection, which rapidly decreases glomerular filtration rate in a short period of time, leading to accumulation of nitrogen metabolites, and the disorders of electrolyte and acid-base balance.1 Significant increase in serum creatinine (Scr), plasma renal injury molecule 1 (pKIM-1) and plasma neutrophil gelatin-associated lipocalin (pNGAL) is an important basis for the diagnosis of SAKI.2 It is widely known that sepsis is an important risk factor for acute kidney injury (AKI) and an independent risk factor for death.3 However, the current treatment of SAKI is still based on renal replacement therapy, and there is a lack of corresponding effective therapeutic drugs.4 Therefore, the research and development of new drugs to prevent and treat SAKI will effectively reduce the mortality of critical illness patients.
Rhodiola is an herbal medicine of the Rosaceae Crassulaceae family in traditional Chinese medicine, and it is distributed in western, northern and subarctic regions of Asia.5 Salidroside (SLDS) is a tyrosol glycoside extracted from Rhodiola, and it is considered to be one of the effective components of Rhodiola for antidepressant and anti-anxiety effects.6 Studies indicated that SLDS has a variety of pharmacological activities such as anti-inflammatory, anti-aging, anti-fatigue and anti-oxidation.7,8 In addition, SLDS also has therapeutic effects in malignant tumors such as lung cancer, colorectal cancer and kidney cancer, so it is expected to become a new drug for adjuvant treatment of cancer.9 However, there is still no evidence for the treatment of SAKI with SLDS, and it has not been widely used and promoted in clinical practice.
Modern medicine has made significant progress in the treatment of SAKI, but the fatality rate is still high and the prognosis is poor. Traditional Chinese medicine is a “huge treasure house”, and it plays an important role in the treatment of critically ill patients. Therefore, exploring the treatment of SAKI with traditional Chinese medicine through animal experiments is extremely important for improving patients’ short-term and long-term survival rates, and reducing medical expenses. In this study, we established a rat model of sepsis by cecal ligation and perforation (CLP) to explore the protective effect of SLDS on SAKI and its mechanism, and provide new ideas for the clinical treatment of SAKI.
Materials and Methods
Experimental Animals and Grouping
60 male Sprague Dawley (SD) rats, specific pathogen free, 10–12 weeks old, 250–300g body weight, were purchased from the Experimental Animal Center of Jiangsu University. The rats were bred in the Animal Experiment Center of Ningbo University, and they were adaptively fed for 2 weeks, during which they were free to eat and drink. We randomly divided the rats into 6 groups, namely: the Control group, the Sham group, the CLP group, the CLP+SLDS (50mg/kg) group, the CLP+SLDS (100mg/kg) group and the CLP+SLDS (150mg/kg) group, 10 of each group.
We used the CLP method to construct a sepsis model based on our previous experience.10 Briefly, we made a 1–2cm incision in the midline of the abdomen, found the cecum and ligated it at the proximal end, pierced the end of the cecum with a 21-gauge needle, returned the cecum to the abdomen, and sutured the incision. For the control group, we did not do any processing. In the Sham group, we opened the abdomen to find the cecum, but did not ligate or pierce the cecum. In the CLP+SLDS group, three different doses (50 mg/kg, 100 mg/kg, or 150 mg/kg) of SLDS were injected via the tail vein of the rats for 3 consecutive days, and CLP was performed 12h after the last injection.
All rats after CLP were put back into their original cages, water and fasting were allowed. Rats were sacrificed after 24h of CLP, blood was collected by cardiac puncture, centrifuged at 2000g for 5min, placed it in −80°C refrigerator for subsequent use. We took both kidneys, the left kidney was fixed in 4% paraformaldehyde for 24h, and the right kidney was placed in −80°C refrigerator. All our operations were carried out in accordance with the experimental animal ethics standards and had been approved by the Experimental Animal Ethics Committee of Ningbo University.
H&E Staining
The left upper kidney was taken and fixed in 4% paraformaldehyde for 24h. The kidney tissue specimens were routinely dehydrated, transparent, paraffin embedded, sectioned, and H&E staining to make histopathological sections of kidneys. We observed the pathological changes under light microscope. Based on previous experience, we performed a semi-quantitative score based on the severity of kidney tubular epithelial cells (KTECs) damage.10 KTECs are normal and regular in shape, 0 points; KTECs damage range < 25%, 1 point; 25% ≤ KTECs damage range < 50%, 2 point; 50% ≤ KTECs damage range < 75%, 3 point; 75% ≤ KTECs damage range, 4 point.
Determination of Inflammatory Factors
We took fresh rat plasma and right upper kidney, and used ELISA (Xitang Biological Technology Co., Ltd., Shanghai, China) to detect the levels of TNF-α, IL-1β and IL-17A in plasma and kidney tissues. We carried out the specific operation in accordance with the kit instructions.
Immunohistochemistry
We took the left lower kidney and fixed it in 4% paraformaldehyde for 24h. We made kidney tissue sections according to the routine. Briefly, after deparaffinization and transparency, the primary antibody (NF-ƙB p65, Zhenhai Biological Technology Co., Ningbo, China) was added and incubated overnight at 4°C. After washing the sections, the secondary antibody was added. We used Image J software (NIH Co., Bethesda, US) to analyze the IOD value and relative expression of NF-ƙB p65.
TUNEL
The left lower kidney was taken, and paraffin sections were routinely made. After transparent and gradient dehydration, the tissue was treated with Proteinase K (Zhenhai Biological Technology Co., Ningbo, China) working solution for 15–30min, and 50μL TdT+450μL fluorescein-labeled dUTP solution (Boster Biological Technology Co., Wuhan, China) was added. After the slides were dry, 50μL TUNEL reaction mixture was added. On the specimen, added a cover glass or parafilm to react at 37°C×60min in a dark humidified box. We counted apoptotic cells under a fluorescence microscope.
RT-qPCR
We took 50mg of fresh right lower kidney tissue, homogenized it in liquid nitrogen and extracted total RNA (Takala co., Tokyo, Japan). We used an ultraviolet spectrophotometer to identify the purity and concentration of total RNA in kidney tissue. We followed the reverse transcription kit (Takala co., Tokyo, Japan) and SYBR PremixEx Taq TM II kit (Takala co., Tokyo, Japan) instructions for gene amplification. Caspase-3 upstream primer: 5’-CCA CGG TAG TCT CTA GCG AGC CGT CA-3’; downstream primer: 5’-CAG TCG GCC TCA GGC CGA ACG TA-3’; Bax upstream primer: 5’-CCG ATG CAG ACT CAC TAG TCC CGA CG-3’; downstream primer: 5’-GTG GTC CCC ACT GCT CCT TCG CA-3’; Bcl-2 upstream primer: 5’-CCT GCC CTG ACA GTA CCA TGC CAT CA-3’; downstream primer: 5’-AAT GAG CAC ACT CCC CGT TCG CC-3’; fragment length 450–500bp. Reaction conditions: 95°C pre-denaturation 30s: PCR reaction (40 cycles): 95°C 5s; 60°C 30s; melting curve analysis: 95°C 15s; 60°C 30s; 95°C 15s. We used the 2-ΔΔCt method to calculate the relative level of the target gene.
Survival Rate
We randomly divide the rats into 6 groups, namely: the Control group, the Sham group, the CLP group, the CLP+SLDS (50mg/kg) group, the CLP+SLDS (100mg/kg) group and the CLP+SLDS (150mg/kg) group, 20 of each group. According to the above experimental protocol, after pretreatment with above different doses of SLDS, we used the CLP method to construct a sepsis model and record the 7-day survival of the rats.
Statistical Analysis
We used GraphPad 8.4 Software to analyze the data, and expressed the data as the mean±standard deviation. We compared the differences between the six groups by the one-way analysis of variance, and compared the differences between the two groups by Tukey’s post hoc test. We used Kaplan-Meier survival analysis to compare the mortality of rats in different groups. P<0.05 was considered statistically different.
Results
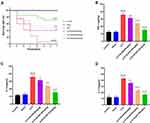
SLDS Reduces Mortality and Plasma Inflammation Levels in Septic Rats
We successfully constructed a rat model of sepsis by the CLP method. We found that CLP caused the 7-day mortality rate of septic rats to increase to 85%, and the death time of the rats was concentrated within 4 days. We pretreated the rats with different doses of SLDS, and found that three doses of SLDS (50mg/kg, 100mg/kg, 150mg/kg) all decreased the 7-day mortality of septic rats, of which 50mg/kg of SLDS reduced the mortality to 60%, 100mg/kg of SLDS reduced to 55%, and 150mg/kg of SLDS reduced to 45% (Figure 1A). Then, we detected the levels of TNF-α, IL-1β and IL-17A in the plasma, and found that three doses of SLDS pretreatment significantly reduced the plasma TNF-α, IL-1β and IL-17A levels in septic rats, respectively, in a dose-dependent manner (Figure 1B–D). These results indicated that SLDS decreased the mortality of septic rats, and effectively inhibited the inflammatory factors release.
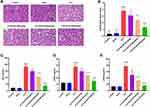
SLDS Improves Kidney Function in Septic Rats
To explore the protective effect of SLDS on SAKI, we assessed the kidney pathological damage and renal function changes of rats, and found that the kidney tissues of the Control group and Sham group were clear and complete. In the CLP group, the glomeruli were ruptured, renal tubular epithelial cells fell off, some tissues were necrotic, and the renal interstitium was full of inflammatory cells. After pretreatment with SLDS, the kidney pathological damage of rat was reduced, the pathological changes of glomerulus and renal tubule tissue were improved, and the renal interstitial inflammatory cells were reduced (Figure 2A). Through semi-quantitative analysis, our results showed that SLDS significantly decreased the pathological damage score of KETCs in a dose-dependent manner (Figure 2B). Furthermore, we evaluated the levels of Scr, pKIM-1 and pNGAL in each group of rats, and found that after pretreatment with different doses of SLDS, the levels of Scr, pKIM-1 and pNGAL were significantly reduced, of which 150mg/kg of SLDS improved the above three renal function indexes with the greatest effect (Figure 2C–E). Our results suggested that SLDS could improve the renal function in septic rats.
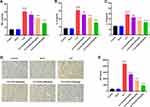
SLDS Alleviates Inflammatory Activity of Kidney in Septic Rats
Studies showed that SLDS has a significant anti-inflammatory effect on sepsis.11,12 To clarify the effect of SLDS on CLP-induced inflammation of AKI, we detected the the levels of inflammatory factors in kidney of septic rats. We found that after pretreatment with SLDS, the levels of TNF-α, IL-1β and IL-17A were significantly down-regulated, and 150mg/kg of SLDS pretreatment had the greatest down-regulation effect of inflammation indexes (Figure 3A–C). Moreover, we detected the expression of NF-ƙB p65 in kidney tissues by immunohistochemistry, and found that SLDS pretreatment significantly decreased the expression of NF-ƙB p65 (Figure 3D and E). Our above results indicated that SLDS down-regulated CLP-induced inflammation response in kidney, and the specific mechanism might be related to the NF-ƙB p65 signaling pathway.
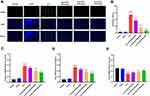
SLDS Inhibits Apoptosis of KTECs in Septic Rats
It is widely known that apoptosis is an important molecular biological mechanism leading to the occurrence and development of SAKI.13 To investigate the effect of SLDS on the apoptosis of kidney cortex induced by CLP, we used TUNEL to detect the apoptosis of KTECs in rats. We found that CLP caused a significant increase in apoptosis of KTECs, while SLDS pretreatment significantly inhibited cell apoptosis, and dose-dependently reduced the number of KTECs apoptosis caused by CLP (Figure 4A and B). Moreover, we detected the levels of Caspase-3, Bax and Bcl-2 mRNA in the KTECs of rats, and found that SLDS pretreatment significantly reduced the levels of Caspase-3 and Bax mRNA, while significantly increasing the level of Bcl-2 mRNA (Figure 4C–E). These results suggested that SLDS could significantly inhibit the number of KTECs apoptosis induced by CLP, and had an important protective effect on SAKI.
Discussion
Current studies believe that in the pathogenesis of sepsis, multiple mechanisms work together to cause AKI, and the inflammatory response plays an important role.2,14 In the circulating blood, a large number of inflammatory mediators cause the degeneration and necrosis of KTECs, which are the main pathogenesis of SAKI.15,16 In the initial stage of sepsis, cytokines activate neutrophils, endothelial cells and epithelial cells, leading to organ microcirculation disorders, tissue hypoxia and damage, causing neutrophils to accumulate in the kidney, leading to AKI.2
Modern medicine has made achievements in various fields, but the curative effect for critical ill diseases (such as SAKI) is not ideal. Nevertheless, the clinical treatment for patients with SAKI is still based on renal replacement therapy, but the effect is not significant, and the mortality rate is still high. In recent years, relevant studies found that traditional Chinese medicine shows special advantages for the treatment of critically ill patients, but the specific mechanism is unknown, and it has not been widely used and promoted.6,7 Therefore, it is of great significance to explore the clinical efficacy of traditional Chinese medicine in the treatment of SAKI.
SLDS is one of the effective ingredients of Rhodiola crassiflora, which is widely used in the adjuvant treatment of anti-aging, anti-tumor, cardiovascular and cerebrovascular diseases.6 In the treatment of adriamycin nephropathy with SLDS, the anti-renal fiber effect is obvious, and the curative effect on diabetic nephropathy is also definite.8 Modern pharmacological experiments confirmed that SLDS has obvious anti-fibrosis effects in diabetic nephropathy, and its mechanism of action is to inhibit the expression of TGF-β1, thereby improving fibrosis.17 Yin et al18 found that SLDS can significantly inhibit the proliferation of renal mesangial cells induced by high glucose, and reduce the production of cellular ROS. In addition, under hypoxia, SLDS can significantly inhibit the transdifferentiation of KTECs induced by cobalt chloride to myofibroblasts, and the mechanism of reducing ECM production is related to the inhibition of the expression of TGF-β1.19 Lan et al20 found that SLDS treats septic acute lung injury by inhibiting SIRT1-mediated HMGB1 nucleocytoplasmic translocation and NF-κB activation. However, the therapeutic effect of SLDS on SAKI is still unclear, and in-depth research is urgently needed.
In this study, by constructing a sepsis rat model, we first explored the effect of SLDS on the survival of septic rats. We found that SLDS could significantly improve the survival rate of septic rats, and significantly reduce the plasma TNF-α, IL-1β, and IL-17A levels in septic rats, preliminarily confirming the protective effect of SLDS on septic rats. These findings are consistent with Liu et al,21 and they also clearly show that SLDS attenuates pro-inflammatory responses by enhancing bacterial clearance and may be a promising treatment for sepsis. Furthermore, we assessed the renal function and kidney pathological damage in rats, and found that SLDS significantly improved the renal function of septic rats, and effectively reduced the kidney pathological damage. In a recent study, You et al22 found that SLDS protected LPS-induced endothelial cell injury by enhancing autophagy and inhibiting inflammation. The above studies fully prove that SLDS can effectively treat sepsis.
Studies found that the excitement of the renin-angiotensin-aldosterone system (RASS) in septic patients can lead to violent contraction of the renal arteries, resulting in insufficient renal arterial filling and perfusion, renal blood flow and glomerular filtration rate decrease.23,24 Meanwhile, under the action of endotoxins or endotoxin-like substances released by bacteria, the neutrophils, monocytes, macrophages, and vascular endothelial cells undergo a complex immune network reaction, and release a large amount of endogenous inflammatory mediators into the blood circulation, leading to AKI.25,26 In order to verify the inflammatory regulation effect of SLDS on SAKI, we measured the inflammatory factors levels in the kidney of septic rats. We found that SLDS decreased the levels of TNF-α, IL-1β and IL-17A in kidney, and through immunohistochemistry, it was found that SLDS also reduced the expression of NF-ƙB p65 in kidney. It can be seen that SLDS can significantly inhibit SAKI inflammation, and the specific mechanism may be related to the regulation of NF-ƙB p65 signaling pathway.
Apoptosis is an important mechanism for the occurrence and development of SAKI.27 Studies showed that the increased apoptosis of KTECs is closely related to inflammation and hypoxia.28–30 In present study, to investigate the effect of SLDS on the apoptosis of kidney, we tested the apoptosis of KTECs. We found that SLDS significantly reduced the number of KTECs apoptosis, and inhibited the expression of pro-apoptotic genes Caspase-3 and Bax mRNA, and increased the expression of apoptosis-suppressing gene Bcl-2 mRNA. Therefore, we proposed that SLDS could significantly inhibit the apoptosis level of KTECs induced by CLP.
Conclusion
SLDS can reduce the mortality of septic rats, and has a significant protective effect on renal function and kidney pathological damage, and the specific mechanism is closely related to its anti-inflammatory and anti-apoptotic. However, the pharmacological mechanism of SLDS is complex and exerts many different functions. Moreover, it is necessary to thoroughly explore the specific molecular mechanism of SLDS in the treatment of SAKI at the cellular and animal levels to provide more evidence for clinical trials.
Acknowledgment
This research was supported by the Zhejiang Provincial Medical and Health Science Fund of China under Grant No. 2021KY998, and Natural Science Foundation of Ningbo under Grant No. 202003N4237.
Disclosure
The authors declare that they no potential conflicts of interest.
References
1. Bellomo R, Kellum JA, Ronco C, et al. Acute kidney injury in sepsis. Intensive Care Med. 2017;43(6):816–828. doi:10.1007/s00134-017-4755-7
2. Fan H, Zhao Y, Chen GD, Sun M, Zhu JH. Health insurance status and risk factors of mortality in patients with septic acute kidney injury in Ningbo, China. J Int Med Res. 2019;47(1):370–376. doi:10.1177/0300060518802526
3. Gómez H, Kellum JA. Sepsis-induced acute kidney injury. Curr Opin Crit Care. 2016;22(6):546–553. doi:10.1097/MCC.0000000000000356
4. Petejova N, Martinek A, Zadrazil J, et al. Acute kidney injury in septic patients treated by selected nephrotoxic antibiotic agents-pathophysiology and biomarkers-a review. Int J Mol Sci. 2020;21(19):7115. doi:10.3390/ijms21197115
5. Bai XL, Deng XL, Wu GJ, Li WJ, Jin S. Rhodiola and salidroside in the treatment of metabolic disorders. Mini Rev Med Chem. 2019;19(19):1611–1626. doi:10.2174/1389557519666190903115424
6. Xu F, Xu J, Xiong X, Deng Y. Salidroside inhibits MAPK, NF-κB, and STAT3 pathways in psoriasis-associated oxidative stress via SIRT1 activation. Redox Rep. 2019;24(1):70–74. doi:10.1080/13510002.2019.1658377
7. Zhong Z, Han J, Zhang J, Xiao Q, Hu J, Chen L. Pharmacological activities, mechanisms of action, and safety of salidroside in the central nervous system. Drug Des Devel Ther. 2018;12:1479–1489. doi:10.2147/DDDT.S160776
8. Huang X, Xue H, Ma J, et al. Salidroside ameliorates Adriamycin nephropathy in mice by inhibiting β-catenin activity. J Cell Mol Med. 2019;23(6):4443–4453. doi:10.1111/jcmm.14340
9. Sun AQ, Ju XL. Advances in research on anticancer properties of salidroside. Chin J Integr Med. 2021;27(2):153–160. doi:10.1007/s11655-020-3190-8
10. Fan H, Le JW, Zhu JH. Protective effect of N-acetylcysteine pretreatment on acute kidney injury in septic rats. J Surg Res. 2020;254:125–134. doi:10.1016/j.jss.2020.04.017
11. Ma S, Evans RG, Iguchi N, et al. Sepsis-induced acute kidney injury: a disease of the microcirculation. Microcirculation. 2019;26(2):e12483. doi:10.1111/micc.12483
12. Kellum JA, Wen X, de Caestecker MP, Hukriede NA. Sepsis-associated acute kidney injury: a problem deserving of new solutions. Nephron. 2019;143(3):174–178. doi:10.1159/000500167
13. Fani F, Regolisti G, Delsante M, et al. Recent advances in the pathogenetic mechanisms of sepsis-associated acute kidney injury. J Nephrol. 2018;31(3):351–359. doi:10.1007/s40620-017-0452-4
14. Stanski NL, Stenson EK, Cvijanovich NZ, et al. PERSEVERE biomarkers predict severe acute kidney injury and renal recovery in pediatric septic shock. Am J Respir Crit Care Med. 2020;201(7):848–855. doi:10.1164/rccm.201911-2187OC
15. Lankadeva YR, Okazaki N, Evans RG, Bellomo R, May CN. Renal medullary hypoxia: a new therapeutic target for septic acute kidney injury? Semin Nephrol. 2019;39(6):543–553. doi:10.1016/j.semnephrol.2019.10.004
16. Kosaka J, Lankadeva YR, May CN, Bellomo R. Histopathology of septic acute kidney injury: a systematic review of experimental data. Crit Care Med. 2016;44(9):e897–e903. doi:10.1097/CCM.0000000000001735
17. Shati AA, Alfaifi MY. Salidroside protects against diabetes mellitus-induced kidney injury and renal fibrosis by attenuating TGF-β1 and Wnt1/3a/β-catenin signalling. Clin Exp Pharmacol Physiol. 2020;47(10):1692–1704. doi:10.1111/1440-1681.13355
18. Yin D, Yao W, Chen S, Hu R, Gao X. Salidroside, the main active compound of Rhodiola plants, inhibits high glucose-induced mesangial cell proliferation. Planta Med. 2009;75(11):1191–1195. doi:10.1055/s-0029-1185717
19. Sun M, Lu Z, Cai P, Zheng L, Zhao J. Salidroside enhances proliferation and maintains phenotype of articular chondrocytes for autologous chondrocyte implantation (ACI) via TGF-β/Smad3 Signal. Biomed Pharmacother. 2020;122:109388. doi:10.1016/j.biopha.2019.109388
20. Lan KC, Chao SC, Wu HY, et al. Salidroside ameliorates sepsis-induced acute lung injury and mortality via downregulating NF-κB and HMGB1 pathways through the upregulation of SIRT1. Sci Rep. 2017;7(1):12026. doi:10.1038/s41598-017-12285-8
21. Liu S, Yu X, Hu B, et al. Salidroside rescued mice from experimental sepsis through anti-inflammatory and anti-apoptosis effects. J Surg Res. 2015;195(1):277–283. doi:10.1016/j.jss.2015.01.021
22. You L, Zhang D, Geng H, Sun F, Lei M. Salidroside protects endothelial cells against LPS-induced inflammatory injury by inhibiting NLRP3 and enhancing autophagy. BMC Complement Med Ther. 2021;21(1):146. doi:10.1186/s12906-021-03307-0
23. Starr MC, Banks R, Reeder RW, et al. Severe acute kidney injury is associated with increased risk of death and new morbidity after pediatric septic shock. Pediatr Crit Care Med. 2020;21(9):e686–e695. doi:10.1097/PCC.0000000000002418
24. Morrell ED, Kellum JA, Pastor-Soler NM, Hallows KR. Septic acute kidney injury: molecular mechanisms and the importance of stratification and targeting therapy. Crit Care. 2014;18(5):501. doi:10.1186/s13054-014-0501-5
25. Zarbock A, Gomez H, Kellum JA. Sepsis-induced acute kidney injury revisited: pathophysiology, prevention and future therapies. Curr Opin Crit Care. 2014;20(6):588–595. doi:10.1097/MCC.0000000000000153
26. Manrique-Caballero CL, Del Rio-Pertuz G, Gomez H. Sepsis-associated acute kidney injury. Crit Care Clin. 2021;37(2):279–301. doi:10.1016/j.ccc.2020.11.010
27. Fan H, Le JW, Sun M, Zhu JH. Sirtuin 3 deficiency promotes acute kidney injury induced by sepsis via mitochondrial dysfunction and apoptosis. Iran J Basic Med Sci. 2021;24(5):675–681. doi:10.22038/ijbms.2021.54905.12312
28. Romanovsky A, Morgan C, Bagshaw SM. Pathophysiology and management of septic acute kidney injury. Pediatr Nephrol. 2014;29(1):1–12. doi:10.1007/s00467-013-2427-6
29. Fan H, Le JW, Sun M, Zhu JH. Pretreatment with S-nitrosoglutathione attenuates septic acute kidney injury in rats by inhibiting inflammation, oxidation, and apoptosis. Biomed Res Int. 2021;2021:6678165. doi:10.1155/2021/6678165
30. Fan H, Zhao Y, Zhu JH. S-nitrosoglutathione protects lipopolysaccharide-induced acute kidney injury by inhibiting toll-like receptor 4-nuclear factor-κB signal pathway. J Pharm Pharmacol. 2019;71(8):1255–1261. doi:10.1111/jphp.13103
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.