Back to Journals » International Journal of General Medicine » Volume 14
Risk Factors of PICC-Related Venous Thrombosis in Breast Cancer Patients Undergoing Chemotherapy
Received 7 December 2020
Accepted for publication 24 February 2021
Published 16 April 2021 Volume 2021:14 Pages 1337—1341
DOI https://doi.org/10.2147/IJGM.S296178
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Bing-Xin Lin, Chun-Sen Xu
Department of Breast Surgery, Union Hospital of Fujian Medical University, Fuzhou, People’s Republic of China
Correspondence: Chun-Sen Xu
Department of Breast Surgery, Union Hospital of Fujian Medical University, No. 29 of Xinquan Road, Gulou District, Fuzhou, 350001, People’s Republic of China
Tel/Fax +86 591 83357896
Email [email protected]
Background: To analyze the risk factors of a peripherally inserted central catheter (PICC)-related venous thrombosis in patients with breast cancer undergoing chemotherapy and explore its preventive measures.
Methods: Data of 780 patients with breast cancer who underwent PICC chemotherapy in our hospital from January 2014 to June 2015 were retrospectively analyzed. The incidence of catheter-related thrombosis was observed, and related factors of venous thrombosis were analyzed.
Results: Among the 780 patients with breast cancer, 36 developed PICC-related venous thrombosis. The incidence of which was 4.62% (36/780). The PICC retention time ranged between 60 and 136 days, and the median time was 92 days. Thrombosis was found to occur within seven days after catheterization in three patients (8.33%), between 7 and 30 days in 18 patients (50%), between 31 and 92 days in 12 patients (33.3%), and ≥ 92 days in three patients (8.33%). Basilic vein puncture-induced thrombosis occurred in 25 patients (3.68%), and median cubital vein and cephalic vein puncture-induced thrombosis occurred in 11 patients (10.78%). The difference was statistically significant (P = 0.001). Thrombosis was not associated with age, punctured limb, platelet count, or chemotherapy drugs (P > 0.05).
Conclusion: Blood vessel puncture was the main factor that affected PICC-related thrombosis in breast cancer chemotherapy. The basilic vein should be the primary choice for blood vessel puncture. Prolonged catheter retention does not increase the risk of thrombosis.
Keywords: breast cancer, peripherally inserted central catheter, thrombosis
Introduction
At present, a peripherally inserted central catheter (PICC) is routinely applied in the fields of tumor chemotherapy and long-term parenteral nutrition. It has the advantages of minimal trauma, safety, reliability, and long retention time. However, it also induces complications, and catheter-related venous thrombosis is one of its serious complications.1,2 Once thrombosis occurs, transfusion through the PICC catheter is stopped, anticoagulant and thrombolytic therapy are given, and chemotherapy time is delayed. It affects the long-term curative effect of breast cancer treatment and even endangers the patient’s life when it becomes severe. It has been reported that the incidence of PICC-related venous thrombosis in patients with a malignant tumor is 3.0%–71.9%,3–5 and the difference is great. This may be related to the heterogeneity of studies, including the types of tumors, the diagnostic techniques, and the methods of study.6,7 In order to prevent catheter-related thrombosis, it is necessary to investigate the risk factors of PICC-related deep vein thrombosis in patients with breast cancer. At present, most studies on PICC-related thrombosis in breast cancer have focused on the treatment and nursing for thrombosis.8,9 Designing a method to effectively reduce the incidence of PICC-related thrombosis in breast cancer is not possible using this study method. This study retrospectively analyzed the clinical data of 780 patients with breast cancer who received PICC chemotherapy from January 2014 to June 2015 in our hospital. The influencing factors of catheter-related thrombosis in patients were analyzed to provide a basis for clinical prevention and nursing intervention.
Methods
Design and Setting
Data of patients with breast cancer who were admitted to the breast department and underwent chemotherapy from January 2014 to June 2015 were retrospectively studied. The following information was collected for all patients: demographic and clinical data, admission diagnosis, thrombosis history, chemotherapy regime, catheter insertion date, vein and arm of insertion, the type of PICC, number of insertion attempts, location of the catheter tip, catheter-related thrombosis, and catheter retention time. Patients with previous thrombosis and coagulation disorders were excluded. All PICC catheters were inserted within three days prior to chemotherapy and screened ultrasonically to exclude the presence of thrombosis weekly. Informed consent was obtained from all patients. Our study plan was approved by the Ethics Committee of Union Hospital of Fujian Medical University.
Catheter Insertion and Follow-Up
All PICCs used for our study were 4F single lumen (Bard Access Systems and BD open-ended PICC). PICCs were inserted using a modified micro Seldinger technique by a professional PICC nursing team in a separate sterile ward to ensure all of the operations were carried out in a sterile environment. The basilic or cephalic veins were chosen according to the blood vessel assessment result. After PICC insertion, chest radiography was routinely carried out to identify the catheter tip position. The PICCs were flushed with 10 mL saline after placement, after each use, and once a week between chemotherapy. Doppler ultrasound was performed weekly to evaluate veins and diagnose PICC-related thrombosis. The veins we examined included cephalic veins, basilic veins, axillary veins, innominate veins, and subclavian veins. All catheters were removed after completion of intravenous therapy.
Diagnosis Criteria and Intervention for PICC-Related Thrombosis
PICC-associated thrombosis in the vein of insertion was diagnosed by Doppler ultrasound. Thrombosis was identified as positive when it included partial/complete thrombus in the veins of the arm, either inside or outside the catheter. Once PICC-related thrombosis was diagnosed, transfusion through the PICC catheter was stopped and was started through peripheral veins instead. Subcutaneous injection of 5000 U/12 hours of low-molecular-weight heparin was given, and 3 mg of warfarin was taken orally. Every 3–5 days, the dosage of warfarin was adjusted to allow the international normalized ratio to reach 1.5–2.5. Then, low-molecular-weight heparin was discontinued. Vascular Doppler ultrasound was conducted each week until ablation of the thrombi. Next, PICC chemotherapy was administered again, anticoagulation therapy was maintained until the end of chemotherapy, and the PICC catheter was removed.
Analysis of Risk Factor of Thrombosis
The possible factors that affect PICC-related thrombosis were considered in our study, including age, the limb punctured, the vein punctured, the catheter retention time, platelet count, chemotherapy drugs, and other related indicators.3–5 The patients with PICC-associated thrombosis were divided into the following groups based on these factors, and the rates of venous thrombosis were analyzed:
Age: <47 and ≥47
Puncture vein: basilic vein and middle cubital vein + cephalic vein
Puncture limbs: left side and right side
Keeping time of catheter: <92 d and ≥92 d
Platelet number: <200 ×109 and ≥200 ×109
Chemotherapeutic drugs: anthracycline and non-anthracycline
Statistical Analysis
Descriptive statistics for characterizing the study population and PICC-related thrombosis were used. The differences of PICC thrombosis rate in different groups of factors were evaluated with χ2 tests. Analyses were performed using SPSS 11.0 software, and all statistical tests were two-tailed. P < 0.05 was considered statistically significant.
Results
Incidence and Site of Upper Extremity Venous Thrombosis
All of the 780 patients with breast cancer enrolled in our study were female, with a median age of 47.0, ranging from 23 to 69. Among the 780 patients with breast cancer treated with PICC chemotherapy, PICC-catheter-related thrombosis was found in 36 patients, and the incidence was 4.62% (36/780). Among these 36 patients, PICC-catheter-related thrombosis was found in the axillary vein alone in 15 patients, in the subclavian vein in four patients, and at multiple sites of the axillary and subclavian vein in 17 patients. Thrombus finding time: thrombosis was found to occur within seven days of catheterization in three patients, between 7 and 30 days in 18 patients, between 30 and 92 days in 12 patients, and ≥92 days in three patients. Color Doppler ultrasound results revealed that within two weeks of anticoagulation therapy, the upper limb vein trunk of 24 patients was completely unobstructed. Within four weeks of anticoagulation therapy, 36 patients recovered completely. The catheter was successfully retained in all 36 patients until the end of chemotherapy. Warfarin anticoagulation therapy was maintained during this period, and no complications, such as the aggravation of thrombosis, pulmonary embolism, or hemorrhaging, occurred.
Risk Factor Analysis
According to related literature,3–5 possible factors that affect PICC-related thrombosis were collected, including age, the limb punctured, the vein punctured, the catheter retention time, platelet count, chemotherapy drugs, and related indicators (Table 1). Basilic vein puncture-induced thrombosis occurred in 25 patients (3.68%), and median cubital vein and cephalic vein puncture-induced thrombosis occurred in 11 patients (10.78%). The difference was statistically significant (P = 0.001). Thrombosis was found to occur within seven days of catheterization in three patients (8.33%), between 7–30 days in 18 patients (50%), between 30–92 days in 12 patients (33.3%), and ≥92 days in three patients (8.33%). The incidence of thrombosis did not increase with the prolonging of catheter retention time (P = 0.03). PICC-related thrombosis was not associated with age, the punctured limb, platelet count, or chemotherapy drugs (P > 0.05).
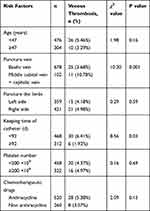 |
Table 1 Relationship Between Risk Factors and PICC-Associated Thrombosis |
Discussion
It has been reported that the incidence of PICC-related thrombosis is 3.0%–71.9%.3–5 The possible cause for the wide difference in reported incidences may include the study population (cancer or not), diagnosis measure (ultrasound or venography), diagnosis criteria, and screening population (only symptomatic or all). In this study, among the 780 patients with breast cancer who had PICC, 36 patients developed PICC-related venous thrombosis. The incidence was 4.62% (36/780), higher than the 3%. The reason may be that both symptomatic and asymptomatic patients with thrombosis were incorporated into the positive subjects after undergoing conventional PICC color Doppler ultrasound in this study. Different degrees of venous obstruction occurred according to the degrees of obstruction and the establishment of collateral circulation. Furthermore, there were no clinical symptoms in these mild patients, and severe patients presented with upper limb swelling, pain, skin cyanosis, and dysfunction. The three main causes of thrombosis are as follows: intimal injury or inflammation of the vein, slow blood flow, and blood hypercoagulability. If the venous thrombosis sheds off, pulmonary embolism might occur, which could endanger the life of patients. Therefore, one of the most important aspects of research is analyzing the risk factors for catheter-related thrombosis and the prevention of PICC-related thrombosis. The risk factors for PICC-related thrombosis may include two aspects as follows. (1) Catheter-related factors: catheter insertion method, catheterization vein, catheter diameter, catheter tip position, and catheter retention time. (2) Systemic disease factors: malignant tumors, the administration of chemotherapeutic drugs, obesity, and the preventive administration of anticoagulants.3–5,10,11
Effect of Vein Puncture on Thrombosis
Due to the specificity of breast cancer operations, patients undergoing chemotherapy often receive PICC catheterization at the elbow vein of the unaffected side after the operation; and the scope of options is relatively limited. This study revealed that basilic vein puncture-induced thrombosis occurred in 25 patients (3.68%). Puncture-induced thrombosis occurred in 11 patients (10.78%) who received PICC catheterization in other veins, including the median cubital vein and the cephalic vein. The incidence of puncture-related thrombosis of the median cubital vein and the cephalic vein was significantly higher than that of the basilic vein, consistent with contemporary studies.12,13 The reason may be related to the anatomical structure of the vessels. There are more puncture valves in cephalic veins and median elbow veins, so it is difficult to transport catheters when undergoing puncture in these veins. Repeated urethral catheter delivery may cause mechanical damage to the intima of blood vessels, which can easily induce venous thrombosis. Therefore, the basilic vein should be primarily chosen for PICC puncture for breast cancer chemotherapy.
Effect of Retention Time of Catheter on Thrombosis
It has usually been considered that the longer the retention time of the PICC catheter, the greater the risk of catheter-related thrombosis. However, our study revealed that PICC-related thrombosis occurred in 3 patients within 7 days of catheterization, between 7 and 30 days in 18 patients, between 30 and 92 days in 12 patients, and ≥92 days in 6 patients. The proportion of patients with a retention time less than the median value (30 days) was 50%, and less than 92 days was 83.3%, suggesting that 30 days after PICC catheterization can be considered as the time of high incidence of catheter-related thrombosis. It has been reported that the mean interval of thrombosis from PICC placement to the thrombosis forming ranged from 14 to 30 days,9,11,14 which also implies the importance of the first month after PICC insertion in the prevention of thrombosis. Thus, this period needs more attention.
Effect of Chemotherapeutic Drugs on Related Thrombosis
Recently, anthracycline (doxorubicin and epirubicin) and taxanes (paclitaxel and docetaxel) are the most commonly used drugs for the adjuvant chemotherapy of breast cancer.15 Chemotherapeutic drugs can directly damage vascular endothelial cells, resulting in patients developing thrombotic diseases. Due to these effects, chemotherapy has been widely considered as the risk factor for PICC-related thrombosis. However, there is little study about whether anthracycline and taxane regimens are associated with a greater risk of PICC-related thrombosis.16 In this study, the incidence of thrombosis induced by anthracycline-based regimens (5.38%) was higher than that induced by taxane-based regimens (3.07%), but the difference was not statistically significant (P = 0.15).
Our results indicated that prolonged catheter retention did not increase the risk of thrombosis. If no complication occurs, the PICC catheter can be maintained for the long term. There are many influencing factors for thrombosis. However, the data collected is limited, and the statistics of some influencing factors for thromboses, such as blood stasis and other factors, could not be conducted. This needs to be improved in future studies.
Conclusions
The incidence of peripherally inserted central venous catheter-related thrombosis was 4.62%, and most cases had significant symptoms. The vein puncture was found to be the key risk factor. Therefore, the basilic vein should be primarily chosen for blood vessel puncture.
Abbreviations
PICC, peripherally inserted central catheter.
Ethics Approval and Consent to Participate
This study was conducted in accordance with the Declaration of Helsinki. This study was conducted with approval from the Ethics Committee of Union Hospital of Fujian Medical University. A written informed consent was obtained from all participants.
Consent for Publication
A written consent for publication was obtained from every individual whose data are included in this manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Zhang X, Huang JJ, Xia Y, et al. High risk of deep vein thrombosis associated with peripherally inserted central catheters in lymphoma. Oncotarget. 2016;7:35404–35411. doi:10.18632/oncotarget.8639
2. Chopra V, Anand S, Krein SL, Chenoweth C, Saint S. Bloodstream infection, venous thrombosis, and peripherally inserted central catheters: reappraising the evidence. Am J Med. 2012;125:733–741. doi:10.1016/j.amjmed.2012.04.010
3. Evans RS, Sharp JH, Linford LH, et al. Risk of symptomatic DVT associated with peripherally inserted central catheters. Chest. 2010;138:803–810. doi:10.1378/chest.10-0154
4. Itkin M, Mondshein JI, Stavropoulos SW, Shlansky-Goldberg RD, Soulen MC, Trerotola SO. Peripherally inserted central catheter thrombosis-reverse tapered versus nontapered catheters: a randomized controlled study. J Vasc Interv Radiol. 2014;25:85–91. doi:10.1016/j.jvir.2013.10.009
5. Nash EF, Helm EJ, Stephenson A, Tullis E. Incidence of deep vein thrombosis associated with peripherally inserted central catheters in adults with cystic fibrosis. J Vasc Interv Radiol. 2009;20:347–351. doi:10.1016/j.jvir.2008.11.018
6. Fallouh N, McGuirk HM, Flanders SA, Chopra V. Peripherally inserted central catheter-associated deep vein thrombosis: a narrative review. Am J Med. 2015;128:722–738. doi:10.1016/j.amjmed.2015.01.027
7. Refaei M, Fernandes B, Brandwein J, Goodyear MD, Pokhrel A, Wu C. Incidence of catheter-related thrombosis in acute leukemia patients: a comparative, retrospective study of the safety of peripherally inserted vs centrally inserted central venous catheters. Ann Hematol. 2016;95:2057–2064. doi:10.1007/s00277-016-2798-4
8. Xing L, Adhikari VP, Liu H, et al. Diagnosis prevention and treatment for PICC-related upper extremity deep vein thrombosis in breast cancer patients. Asia Pac J Clin Oncol. 2012;8:e12–6. doi:10.1111/j.1743-7563.2011.01508.x
9. Kang J, Sun W, Li H, Ma E, Wang K, Chen W. Peripherally inserted central catheter-related vein thrombosis in breast cancer patients. J Vasc Access. 2016;17:67–71. doi:10.5301/jva.5000457
10. Pittiruti M, Hamilton H, Biffi R, MacFie J, Pertkiewicz M; ESPEN. ESPEN guidelines on parenteral nutrition: central venous catheters (access, care, diagnosis and therapy of complications). Clin Nutr. 2009;28:365–377. doi:10.1016/j.clnu.2009.03.015
11. Liu Y, Gao Y, Wei L, Chen W, Ma X, Song L. Peripherally inserted central catheter thrombosis incidence and risk factors in cancer patients: a double-center prospective investigation.Ther. Clin Risk Manag. 2015;11:153–160.
12. Liem TK, Yanit KE, Moseley SE, et al. Peripherally inserted central catheter usage patterns and associated symptomatic upper extremity venous thrombosis. J Vasc Surg. 2012;55:761–767. doi:10.1016/j.jvs.2011.10.005
13. Qiu XX, Guo Y, Fan HB, Shao J, Zhang XB. Incidence, risk factors and clinical outcomes of peripherally inserted central catheter spontaneous dislodgment in oncology patients: a prospective cohort study. Int J Nurs Stud. 2014;51:955–963. doi:10.1016/j.ijnurstu.2013.10.021
14. Ong B, Gibbs H, Catchpole I, Hetherington R, Harper J. Peripherally inserted central catheters and upper extremity deep vein thrombosis. Australas Radiol. 2006;50:451–454. doi:10.1111/j.1440-1673.2006.01623.x
15. Vandergrift JL, Niland JC, Theriault RL, et al. Time to adjuvant chemotherapy for breast cancer in National Comprehensive Cancer Network institutions. J Natl Cancer Inst. 2013;105:104–112. doi:10.1093/jnci/djs506
16. Bertoglio S, Faccini B, Lalli L, Cafiero F, Bruzzi P. Peripherally Inserted Central Catheters (PICCs) in cancer patients under chemotherapy: a prospective study on the incidence of complications and overall failures. J Surg Oncol. 2016;113:708–714. doi:10.1002/jso.24220
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
