Back to Journals » Infection and Drug Resistance » Volume 11
Risk factors and prognosis of complicated urinary tract infections caused by Pseudomonas aeruginosa in hospitalized patients: a retrospective multicenter cohort study
Authors Gomila A, Carratalà J, Eliakim-Raz N, Shaw E , Wiegand I, Vallejo-Torres L, Gorostiza A , Vigo JM, Morris S, Stoddart M, Grier S , Vank C, Cuperus N, van den Heuvel L, Vuong C , MacGowan A, Leibovici L, Addy I, Pujol M
Received 30 August 2018
Accepted for publication 25 October 2018
Published 18 December 2018 Volume 2018:11 Pages 2571—2581
DOI https://doi.org/10.2147/IDR.S185753
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Aina Gomila,1,2 J Carratalà,1–3 N Eliakim-Raz,4 E Shaw,1,2 I Wiegand,5 L Vallejo-Torres,6 A Gorostiza,2 JM Vigo,7 S Morris,6 M Stoddart,8 S Grier,8 C Vank,5 N Cuperus,9 L Van den Heuvel,9 C Vuong,5 A MacGowan,8 L Leibovici,4 I Addy,5 M Pujol1,2
On behalf of COMBACTE MAGNET WP5 RESCUING Study Group and Study Sites
1Department of Infectious Diseases, Hospital Universitari de Bellvitge, Institut Català de la Salut (ICS-HUB), Spanish Network for Research in Infectious Diseases (REIPI RD12/0015), Instituto de Salud Carlos III (ISCIII), Madrid, Spain; 2Institut d’Investigació Biomèdica de Bellvitge (IDIBELL), Barcelona, Spain; 3Infectious Diseases Department, University of Barcelona, Barcelona, Spain; 4Department of Medicine E, Beilinson Hospital, Rabin Medical Center, Petah-Tiqva and Sackler Faculty of Medicine, Tel Aviv University, Israel; 5AiCuris Anti-infective Cures, Wuppertal, Germany; 6UCL Department of Applied Health Research, University College London, London, UK; 7Informatics Unit, Fundació Institut Català de Farmacologia, Barcelona, Spain; 8Department of Medical Microbiology, Southmead Hospital, North Bristol NHS Trust, Bristol, UK; 9Julius Center for Health Sciences and Primary Care, University Medical Center Utrecht, Utrecht, the Netherlands
Purpose: Complicated urinary tract infections (cUTIs) are among the most frequent health-care-associated infections. In patients with cUTI, Pseudomonas aeruginosa deserves special attention, since it can affect patients with serious underlying conditions. Our aim was to gain insight into the risk factors and prognosis of P. aeruginosa cUTIs in a scenario of increasing multidrug resistance (MDR).
Methods: This was a multinational, retrospective, observational study at 20 hospitals in south and southeastern Europe, Turkey, and Israel including consecutive patients with cUTI hospitalized between January 2013 and December 2014. A mixed-effect logistic regression model was performed to assess risk factors for P. aeruginosa and MDR P. aeruginosa cUTI.
Results: Of 1,007 episodes of cUTI, 97 (9.6%) were due to P. aeruginosa. Resistance rates of P. aeruginosa were: antipseudomonal cephalosporins 35 of 97 (36.1%), aminoglycosides 30 of 97 (30.9%), piperacillin–tazobactam 21 of 97 (21.6%), fluoroquinolones 43 of 97 (44.3%), and carbapenems 28 of 97 (28.8%). The MDR rate was 28 of 97 (28.8%). Independent risk factors for P. aeruginosa cUTI were male sex (OR 2.61, 95% CI 1.60–4.27), steroid therapy (OR 2.40, 95% CI 1.10–5.27), bedridden functional status (OR 1.79, 95% CI 0.99–3.25), antibiotic treatment within the previous 30 days (OR 2.34, 95% CI 1.38–3.94), indwelling urinary catheter (OR 2.41, 95% CI 1.43–4.08), and procedures that anatomically modified the urinary tract (OR 2.01, 95% CI 1.04–3.87). Independent risk factors for MDR P. aeruginosa cUTI were age (OR 0.96, 95% CI 0.93–0.99) and anatomical urinary tract modification (OR 4.75, 95% CI 1.06–21.26). Readmission was higher in P. aeruginosa cUTI patients than in other etiologies (23 of 97 [23.7%] vs 144 of 910 [15.8%], P=0.04), while 30-day mortality was not significantly different (seven of 97 [7.2%] vs 77 of 910 [8.5%], P=0.6).
Conclusion: Patients with P. aeruginosa cUTI had characteristically a serious baseline condition and manipulation of the urinary tract, although their mortality was not higher than that of patients with cUTI caused by other etiologies.
Keywords: health care-associated infections, complicated urinary tract infections, Pseudomonas aeruginosa, multidrug-resistance
Introduction
Urinary tract infections (UTIs) are among the most frequent infections in both the outpatient and inpatient setting. In the last European point-prevalence survey of health care-associated infections in acute-care hospitals from 2011 to 2012, UTIs were estimated to be the third-most common infection after surgical site infections and pneumonia, accounting for 19% of cases.1 Despite the application of specific prevention programs, rates seem to be increasing. In the US, 93,300 UTIs occurred in hospitalized patients in a 1-year period.2 Given the significant incidence of UTI, the impact of the disease on antibiotic consumption is noteworthy.3
Complicated UTIs (cUTIs), those occurring in immunocompromised patients or in patients with structural or functional urinary tract (UT) abnormalities, have been associated with high rates of treatment failure and serious complications, especially relapse and development of antibiotic resistance.4 These patients constitute a reservoir for the spread of multidrug-resistant (MDR) bacteria.
Other issues to consider are the growth of the elderly population and the increasing complexity of procedures and treatments in patients with cUTI, which entail severe immunosuppression and the insertion of foreign bodies and devices. In this situation, Gram-negative bacteria with high intrinsic resistance, particularly Pseudomonas aeruginosa, take on greater relevance.5,6 These microorganisms are associated with worse prognosis.7 MDR strains of P. aeruginosa, such as ST175 or ST111, have spread worldwide. They now pose a major challenge for clinicians, since few secondary treatment options remain active, and those available have side effects.8,9
Given the importance of cUTI in terms of prevalence and complexity of disease and the significant pathogenic role of P. aeruginosa in this context, it might be of interest to identify patients with cUTI who are at risk of infection by P. aeruginosa. This can help in the selection of empirical treatment in patients with a severe infection. The aim of this study was to gain insight into the risk factors for developing cUTI caused by P. aeruginosa and MDR P. aeruginosa among hospitalized patients and to assess the prognosis associated with these infections.
Methods
Study design
The COMBACTE-MAGNET WP5 RESCUING study was an international, multicenter, retrospective, observational cohort study involving hospitalized patients with cUTI between January 1, 2013, and December 31, 2014.10,11 This study conformed with the STROBE guidelines for reporting observational studies.12
Setting and patients
The study was conducted at 20 hospitals in Bulgaria, Greece, Hungary, Israel, Italy, Romania, Spain, and Turkey. At each hospital, all consecutive patients with a diagnosis of cUTI as the primary cause of hospitalization and patients hospitalized for another reason but who developed cUTI during their hospitalization were eligible for enrollment. The selection was done by searching the appropriate ICD-9 CM or ICD-10 CM codes at discharge (detailed in Supplementary materials), and 50–60 patients were included at each hospital until reaching the total estimated sample size of 1,000 patients.13,14
Inclusion criteria, based on the US Food and Drug Administration (FDA) guidance on cUTI,15 were patients with UTI and at least one of:
- indwelling urinary catheter, urinary retention (at least 100 mL of residual urine after voiding), neurogenic bladder, obstructive uropathy (eg, nephrolithiasis, fibrosis), renal impairment caused by intrinsic renal disease (estimated glomerular filtration rate <60 mL/min), renal transplantation, UT modifications, such as an ileal loop or pouch, and pyelonephritis and normal UT anatomy;
- at least one of the following signs or symptoms: chills or rigors associated with fever or hypothermia (temperature >38°C or <36°C), flank pain (pyelonephritis) or pelvic pain, dysuria, urinary frequency, or urinary urgency, and costovertebral angle tenderness on physical examination;
- urine culture with at least 105 colony-forming units/mL or greater of an uropathogen (no more than two species), or at least one blood culture growing possible uropathogens (no more than two species) with no other evident site of infection.
Exclusion criteria were age under 18 years, diagnosis of prostatitis according to FDA guidance, polymicrobial infections that included Candida spp., polymicrobial infections that included more than two bacterial species, and cUTI with Candida spp. as sole uropathogen.
Data collection
Data from eligible patients were collected from January 2015 until August 2016. For all patients, a standardized set of information was collected retrospectively from their electronic records and the hospital administration and laboratory systems. Data included patients’ demographic characteristics, comorbidities, place of acquisition of infection, signs and symptoms of infection, laboratory and microbiology tests, details of discharge, and outcome of infection, including death if applicable. The follow-up period was up to 2 months after hospital discharge. Patients without visits after discharge and not readmitted but not declared dead were considered “alive” for outcome purposes. For data collection, a controlled-access web-based electronic case report form was used. For confirmation of data quality, study sites were monitored and audited.
Definitions
Acquisition of cUTI in a medical care facility was considered if it was:
- hospital-acquired: if it started 48 hours or more after hospital admission;
- health care-associated: if it was detected at hospital admission or within the first 48 hours of hospitalization and met any of the following criteria: 1) receiving intravenous therapy, wound care, or specialized nursing care at home in the previous 30 days, 2) attendance at hospital or hemodialysis ward or administration of intravenous chemotherapy in the previous 30 days, 3) hospitalization for 2 or more days in the previous 90 days, 4) residence in a long-term care facility, 5) invasive urinary procedure performed in the previous 30 days, or 6) a long-term indwelling urethral catheter.
The following cUTI categories were used:
- UTI related to indwelling urinary catheterization, including long-term, short-term, or intermittent catheterization
- pyelonephritis, defined as sepsis, flank pain, or tenderness, and no other UT pathology.
- UTI related to anatomical UT modification, including any urinary diversion procedure, nephrostomy, stents, or renal transplants.
- UTI related to obstructive uropathy, including any obstruction intrinsic or extrinsic to the UT, such as lithiasis, tumor, ureteral herniation, and prostate hyperplasia
- UTI related to other events that did not fall under any other category, such as neurogenic bladder.
MDR P. aeruginosa was defined in accordance with the international expert proposal published by Magiorakos et al: insusceptibility to at least one agent in three or more antimicrobial categories (aminoglycosides, antipseudomonal carbapenems, antipseudomonal cephalosporins, antipseudomonal fluoroquinolones, antipseudomonal penicillins + β-lactamase inhibitors, monobactams, phosphonic acids, and polymyxins).16 Extensive drug resistance was defined as insusceptibility to at least one agent in all but two or fewer antimicrobial categories. Clinical and Laboratory Standards Institute or European Committee on Antimicrobial Susceptibility Testing guidelines were followed depending on each hospital protocol.
Steroid therapy was considered as the administration of a dose of ≥10 mg prednisolone or equivalent for 30 days or more prior to the diagnosis of cUTI. P. aeruginosa cUTI was defined as isolation of P. aeruginosa in urine cultures and/or blood cultures in accordance with the aforementioned inclusion and exclusion criteria. Adequate empirical antibiotic treatment was considered the empirical administration of at least one antimicrobial agent to which the P. aeruginosa isolate was susceptible according to microbiological sensitivity tests.
Primary outcome
The primary outcome was the analysis of independent predictive factors of P. aeruginosa cUTI and MDR P. aeruginosa cUTI. Secondary outcomes included comparison of evolution of P. aeruginosa cUTI and cUTI by other etiologies and evaluation of differences in mortality between cUTI caused by P. aeruginosa and MDR P. aeruginosa.
Statistical methods
Nominal categorical variables are presented as number of cases and percentage of the total. Ordinal categorical variables and continuous variables that did not follow a normal distribution are described as medians and interquartile ranges. Continuous variables with normal distribution are described as mean and SD. Normality of continuous variables was graphically tested through Q–Q plots and histograms. The χ2 or Fisher’s exact tests were used to compare categorical data and Student’s t-test or Mann–Whitney U test for continuous data, as appropriate.
After univariate analyses, a mixed-effect logistic regression multivariate analysis was estimated to assess association between P. aeruginosa cUTI and patient profiles, accounting for center effect. A mixed-effect logistic regression multivariate analysis was estimated to assess association between MDR P. aeruginosa cUTI and patient profile, accounting for country effect. Country instead of center was used as a cluster in the MDR P. aeruginosa model, due to the low number of events per center. Collinearity between variables was evaluated. Selection of model variables was based on minimization of the Akaike information criterion. The effect of center was assessed using intraclass correlations, which measured the proportion of total variance of the dependent variable explained by center or country effect. The higher the value, the more center or country effect. Results are given as ORs and 95% CIs. All tests were two-tailed, and P<0.05 was considered statistically significant. Data were managed and analyzed using R version 3.5.0 for Windows.
Ethics approval and informed consent
This study was approved by the Comitato Etico Provinciale di Modena and thereafter the local research ethics committee of each site. The processing of patients’ personal data was anonymized and complied with local data-protection legislations and with the European Union Data Protection Directive (95/46/EC). The study was conducted in accordance with the Declaration of Helsinki. As neither individual data were published nor any intervention was performed on patients, patient consent was waived by the ethics committee.
Results
Epidemiological characteristics of cUTI episodes
A total of 1,007 cUTI episodes from 981 patients were included. Differences between episodes of P. aeruginosa cUTI and episodes of cUTI caused by other etiologies are shown in Table 1. Differences between MDR P. aeruginosa cUTI and non-MDR P. aeruginosa cUTI episodes are shown in Supplementary materials.
Microbiological features of P. aeruginosa cUTI and cUTI due to other etiologies
Of all episodes, 97 were P. aeruginosa. Among these, 15 episodes (15.4%) had P. aeruginosa-positive blood cultures. Compared to other etiologies, P. aeruginosa cUTIs were more frequently polymicrobial (39.2% vs 9.7%, P<0.001). Associated microorganisms were mostly Enterococcus spp. (Table 2).
  | Table 2 Microbiological features of cUTI episodes with or without Pseudomonas aeruginosa Note: aComprised no more than two bacterial species. Abbreviation: cUTI, complicated urinary tract infection. |
Antibiotic-resistance profile of P. aeruginosa isolates
The resistance profile of the 97 P. aeruginosa isolates is shown in Table 3. Resistance rates to antipseudomonal cephalosporins were 35 of 97 (36.1%), to aminoglycosides 30 of 97 (30.9%), to piperacillin–tazobactam 21 of 97 (21.6%), to fluoroquinolones 43 of 97 (44.3%), and to carbapenems 28 of 97 (28.8%). The rate of MDR P. aeruginosa was 28 of 97 (28.8%) and of extensively drug-resistant P. aeruginosa 12 of 97 (12.3%).
  | Table 3 Antibiotic-resistance profile of Pseudomonas aeruginosa complicated urinary tractinfection episodes (n=97) |
Rates and risk factors for P. aeruginosa cUTI and MDR P. aeruginosa cUTI
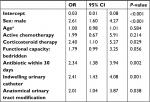
By mixed-effects logistic regression model accounting for center effect, male sex (OR 2.61, 95% CI 1.60–4.27), steroid therapy (OR 2.40, 95% CI 1.10–5.27), a bedridden functional capacity (OR 1.79, 95% CI 0.99–3.25), antibiotic treatment within 30 previous days of cUTI (OR 2.34, 95% CI 1.38–3.94), indwelling urinary catheter (OR 2.41, 95% CI 1.43–4.08), and anatomical UT modification (OR 2.01, 95% CI 1.04–3.87) as sources of infection were identified as independent risk factors for P. aeruginosa cUTI (Table 4). Figure 1 shows the differences in P. aeruginosa cUTI prevalence in the different participating centers.
  | Figure 1 Prevalence with 95% CIs of Pseudomonas aeruginosa complicated urinary tract-infection episodes by centers (A–R) and countries. |
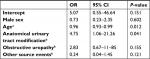
Factors independently associated with MDR P. aeruginosa cUTI compared to cUTI caused by non-MDR P. aeruginosa, accounting for country effect, were age (OR 0.96, 95% CI 0.93–0.99) and modification of the UT as source of cUTI (OR 4.75, 95% CI 1.06–21.26), as shown in Table 5. Figure 2 shows the prevalence of MDR P. aeruginosa cUTI in the eight participating countries.
  | Figure 2 Prevalence with 95% CIs of multidrug-resistant Pseudomonas aeruginosa complicated urinary tract infection by country. |
Outcomes
For readmission and mortality, a slightly higher 60-day readmission rate was found in P. aeruginosa cUTI compared to other etiologies (23/97 vs 144/910, P=0.04; Table 6). Thirty-day mortality did not significantly differ between cUTI caused by P. aeruginosa or by other etiologies; however, there were differences in mortality between non-MDR P. aeruginosa cUTI (six of 60, 10%) and MDR P. aeruginosa cUTI (one of 37, 2.7%), but they did not reach statistical significance (P=0.17; results not shown).
  | Table 6 Outcomes of episodes of Pseudomonas aeruginosa cUTI and episodes of cUTI caused by other etiologies Abbreviation: cUTI, complicated urinary tract infection. |
Discussion
In this international, multicenter, retrospective, observational study, we found that factors associated with P. aeruginosa were male sex, steroid therapy, low functional capacity, having had antibiotics and manipulation of the cUTI, either insertion of urinary catheter or surgical procedures that anatomically modify the UT. Of these, anatomical UT modification was a risk factor for MDR P. aeruginosa cUTI, as was, unexpectedly, young age. Interestingly, although hospital readmission was higher in patients with P. aeruginosa cUTI, mortality was not higher than other etiologies.
In our cohort, P. aeruginosa cUTIs were more frequently polymicrobial than cUTIs caused by other microorganisms. Interestingly, associated microorganisms in P. aeruginosa cUTI were more frequently Gram-positive, especially Enterococcus spp., than Gram-negative. The same association has been observed in other studies, and could be explained by an enhanced ability to adhere to urinary catheters through different factors and biofilm formation of Enterococcus spp. and P. aeruginosa.3,17–20
In regard to the antimicrobial resistance of P. aeruginosa isolates, high resistance to fluoroquinolones was documented, as previously observed in the European Antimicrobial Resistance Surveillance Network report.21 Significantly, 36% of P. aeruginosa strains were resistant to antipseudomonal cephalosporins. This tendency was also observed in a recent Turkish study among hospitalized men with P. aeruginosa cUTI.22 Piperacillin–tazobactam maintained a better resistance profile than antipseudomonal cephalosporins, maybe because it is less affected by certain AmpC β-lactamase variants and extended spectrum β-lactamases.23,24 Notably, 28.8% of P. aeruginosa isolates were resistant to carbapenems, a rate significantly higher than previously reported.22,25 In our study, the MDR rate of P. aeruginosa was 28.8%, slightly higher than in previous work; however, considering that our study focused on countries with a high incidence of MDR Gram-negative bacteria and that some of the studies mentioned were performed some years ago, this increase was expected.18,26
Importantly, rates of P. aeruginosa cUTI varied significantly among hospitals in the same country and among countries. This reflects the wide heterogeneity of patients and procedures performed in each hospital. Some of the risk factors observed in the present study, such as receiving corticosteroids or antibiotics, have been described by other authors, and reflect the opportunistic and nosocomial nature of infections due to P. aeruginosa.27,28 Previous exposure to antibiotics may be related to the effect of eradication of the competitive flora caused by nonantipseudomonal treatments.29 The indication of steroids and antibiotics should be carefully evaluated in every patient, given the high risk of P. aeruginosa infections.
As mentioned, P. aeruginosa can adhere to such surfaces as urinary catheters or foreign bodies inserted during urological surgery through production of adhesion factors and biofilm formation. This allows the microorganism to elude the immune system and the activity of antimicrobial agents, and persist in causing frequent relapses of infection.19 Therefore, the indication for insertion of an indwelling urinary catheter and its duration should also be periodically evaluated.
Interestingly, younger patients were at higher risk of having MDR P. aeruginosa cUTI than older patients. This was probably related to the fact that young patients are more likely to receive aggressive antimicrobial and surgical treatment than old patients and more frequently admitted to intensive-care units when complications arise, increasing the risk of acquiring a MDR isolate.30 Procedures that modified the UT represented a strong risk factor for MDR P. aeruginosa in our cohort.
P. aeruginosa cUTI was associated with a lower rate of adequate empirical treatment than cUTI by other etiologies. Nevertheless, a previous study in the same cohort showed that inadequate empirical antibiotic treatment was not independently associated with increased mortality.11 The lack of differences observed in mortality between P. aeruginosa and other etiologies was probably due to the low frequency of severe sepsis or septic shock and mortality in our cohort. Although many previous studies have observed worse outcomes of UTIs caused by P. aeruginosa, this was more frequently related to a more serious baseline status due to comorbidities than to the infection itself. For instance, one study of bacteremic UTIs showed that P. aeruginosa UTIs were associated with higher 30-day mortality than other etiologies, but this result did not remain significant after adjustment for confounding factors.31 Similar results have been observed in other work.18 The readmission rate was higher in patients with P. aeruginosa cUTIs, but this was mostly due to exacerbation of chronic baseline diseases: this observation stresses that these patients had worse baseline status. Interestingly, low mortality associated with MDR P. aeruginosa cUTI was observed. This was probably due to the small number of patients with MDR strains, but also because these strains have been associated with reduced fitness and virulence.8
Limitations
Our study has some limitations. The first is its retrospective design, which means that some patients’ data collected from medical records may have been missing. Second, the data come from hospitals in south and southeastern Europe, Turkey, and Israel, and may not be generalizable to other regions. The main strengths of this study are its large-scale, multicenter, and international nature and the fact that it addresses a frequent and important medical issue.
Conclusion
Our study reveals that resistance rates of P. aeruginosa cUTI isolates to antipseudomonal cephalosporins and carbapenems are especially worrisome in south and southeastern Europe, Turkey, and Israel. Risk factors for P. aeruginosa cUTI were related to more serious baseline condition and manipulation of the UT, although their mortality was not higher than that of patients with cUTI caused by other etiologies.
Abbreviations
UT, urinary tract; UTIs, urinary tract infections; cUTIs, complicated urinary tract infections; MDR, multidrug-resistance; GNB, Gram-negative bacteria; STROBE, Strengthening the Reporting of Observational studies in Epidemiology; ICD-9 (-10) CM, International Classification of Diseases-9 (-10) Clinical Modification; FDA, Food and Drug Admin-istration; CFU, colony-forming units; eCRF, electronic case report form; XDR, extensively drug-resistance; CLSI, Clinical and Laboratory Standards Institute; EUCAST, European Committee on Antimicrobial Susceptibility Testing; IQR, interquartile range; SD, standard deviation; ICC, Intra-class-correlation; OR, odds ratio; 95% CI, 95% confidence interval; EARS-Net, European Antimicrobial Resistance Surveillance Network.
Data sharing statement
The data sets used and analyzed during the current study are available from the corresponding author on reasonable request.
Author contributions
AG, LL, and MP conceived and designed the study. AG, JC, and MP were major contributors in writing the manuscript. ES, LL, IW, CVa, CVu, and IA participated in the design of the study and coordination and helped to draft the manuscript. AGor performed the statistical analysis of data. All authors read and approved the final manuscript. All authors contributed toward data analysis, drafting and revising the paper, gave final approval of the version to be published and agree to be accountable for all aspects of the work.
Acknowledgments
COMBACTE-MAGNET, RESCUING Study Group members: Tanya Babitch, Dora Tancheva, Rossitza Vatcheva-Dobrevska, Sotirios Tsiodras, Emmanuel Roilides, Istvan Várkonyi, Judit Bodnár, Aniko Farkas, Yael Zak-Doron, Yehuda Carmeli, Emanuele Durante Mangoni, Cristina Mussini, Nicola Petrosillo, Andrei Vata, Adriana Hristea, Julia Origüen, Jesus Rodriguez-Baño, Arzu Yetkin, and Nese Saltoglu. The research leading to these results was conducted as part of the COMBACTE-MAGNET consortium. For further information, please refer to www.combacte.com. The results of this study were presented at the 28th European Congress of Clinical Microbiology and Infectious Diseases (ECCMID), Madrid, April 21–24, 2018, and are available online. This research project receives support from the Innovative Medicines Initiative Joint Undertaking under grant agreement 115523|115620|115,737, resources of which are composed of financial contribution from the European Union Seventh Framework Programme (FP7/2007-2013) and European Federation of Pharmaceutical Industries and Association (EFPIA) companies in kind contribution.
Disclosure
IA, CVa, IW, and CVu belong to European Federation of Pharmaceutical Industries and Association (EFPIA) member companies in the Innovative Medicines Initiative Joint Undertaking (IMI JU), and costs related to their part in the research were carried by the respective company as in-kind contribution under the IMI JU scheme. The authors report no other conflicts of interest in this work.
References
European Centre for Disease Prevention and Control. Point prevalence survey of healthcare-associated infections and antimicrobial use in European acute care hospitals 2011–2012. Available from: https://ecdc.europa.eu/sites/portal/files/media/en/publications/Publications/healthcare-associated-infections-antimicrobial-use-PPS.pdf. Accessed March 14, 2018. | ||
Magill SS, Edwards JR, Bamberg W, et al. Multistate point-prevalence survey of health care-associated infections. N Engl J Med. 2014;370(13):1198–1208. | ||
Flores-Mireles AL, Walker JN, Caparon M, Hultgren SJ. Urinary tract infections: epidemiology, mechanisms of infection and treatment options. Nat Rev Microbiol. 2015;13(5):269–284. | ||
Pallett A, Hand K. Complicated urinary tract infections: practical solutions for the treatment of multiresistant gram-negative bacteria. J Antimicrob Chemother. 2010;65(Suppl 3):iii25–iii33. | ||
Bader MS, Hawboldt J, Brooks A. Management of complicated urinary tract infections in the era of antimicrobial resistance. Postgrad Med. 2010;122(6):7–15. | ||
Nicolle LE. AMMI Canada Guidelines Committee* ACG. Complicated urinary tract infection in adults. Can J Infect Dis Med Microbiol. 2005;16(6):349–360. | ||
Peña C, Cabot G, Gómez-Zorrilla S, et al. Influence of virulence genotype and resistance profile in the mortality of Pseudomonas aeruginosa bloodstream infections. Clin Infect Dis. 2015;60(4):539–548. | ||
Mulet X, Cabot G, Ocampo-Sosa AA, et al. Biological markers of Pseudomonas aeruginosa epidemic high-risk clones. Antimicrob Agents Chemother. 2013;57(11):5527–5535. | ||
Vasoo S, Barreto JN, Tosh PK. Emerging issues in gram-negative bacterial resistance: an update for the practicing clinician. Mayo Clin Proc. 2015;90(3):395–403. | ||
Shaw E, Addy I, Stoddart M, et al. Retrospective observational study to assess the clinical management and outcomes of hospitalised patients with complicated urinary tract infection in countries with high prevalence of multidrug resistant Gram-negative bacteria (RESCUING). BMJ Open. 2016;6(7):e011500. | ||
Eliakim-Raz N, Babitch T, Shaw E, et al. Risk factors for treatment failure and mortality among hospitalised patients with complicated urinary tract infection: a multicentre retrospective cohort study, RESCUING study group. Clin Infect Dis. Epub 2018 May 17. | ||
von Elm E, Altman DG, Egger M, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg. 2014;12(12):1495–1499. | ||
International Classification of Diseases, Ninth Revision, Clinical Modification, ICD-9-CM. Available from: https://www.cdc.gov/nchs/icd/icd9cm.htm. Accessed March 14, 2018. | ||
International Classification of Diseases, Tenth Revision, Clinical Modification, ICD-10-CM. Available from: https://www.cdc.gov/nchs/icd/icd10cm.htm. Accessed March 14, 2018. | ||
Tract CU. Guidance for Industry Complicated Urinary Tract Infections : Developing Drugs for Treatment Guidance for Industry Complicated Urinary Tract Infections: Developing Drugs for Treatment; 2012. Available from: https://www.fda.gov/downloads/Drugs/Guidances/ucm070981.pdf. Accessed June 4, 2018. | ||
Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268–281. | ||
Shigemura K, Arakawa S, Sakai Y, Kinoshita S, Tanaka K, Fujisawa M. Complicated urinary tract infection caused by Pseudomonas aeruginosa in a single institution (1999–2003). Int J Urol. 2006;13(5):538–542. | ||
Ferreiro LJ, Álvarez J, González L, et al. Pseudomonas aeruginosa urinary tract infections in hospitalized patients: mortality and prognostic factors. Plos One. 2018;12(5):e0178178. | ||
Mittal R, Aggarwal S, Sharma S, Chhibber S, Harjai K. Urinary tract infections caused by Pseudomonas aeruginosa: a minireview. J Infect Public Health. 2009;2(3):101–111. | ||
Tielen P, Narten M, Rosin N, et al. Genotypic and phenotypic characterization of Pseudomonas aeruginosa isolates from urinary tract infections. Int J Med Microbiol. 2011;301(4):282–292. | ||
Surveillance Report. Surveillance of antimicrobial resistance in Europe 2016. Available from: https://ecdc.europa.eu/sites/portal/files/documents/AMR-surveillance-Europe-2016.pdf. Accessed March 14, 2018. | ||
Ö K, Özger HS, Karas¸ahin EF, et al. Assessment of risk factors, treatment and hospital stay in complicated urinary tract infections in men caused by pseudomonas: a case-control study. J Urol Surg. 2017;44:48–52. | ||
Berrazeg M, Jeannot K, Ntsogo Enguéné VY, et al. Mutations in β-lactamase AmpC increase resistance of pseudomonas aeruginosa isolates to antipseudomonal cephalosporins. Antimicrob Agents Chemother. 2015;59(10):6248–6255. | ||
Laudy AE, Róg P, Smolin´ska-Król K, et al. Prevalence of ESBL-producing Pseudomonas aeruginosa isolates in Warsaw, Poland, detected by various phenotypic and genotypic methods. PLoS One. 2017;12(6):e0180121. | ||
Vazquez-Guillamet MC, Vazquez R, Micek ST, Kollef MH. Predicting resistance to piperacillin-tazobactam, cefepime and meropenem in septic patients with bloodstream infection due to gram-negative bacteria. Clin Infect Dis. 2017;65(10):1607–1614. | ||
Zilberberg MD, Shorr AF. Secular trends in gram-negative resistance among urinary tract infection hospitalizations in the United States, 2000-2009. Infect Control Hosp Epidemiol. 2013;34(9):940–946. | ||
Venier AG, Lavigne T, Jarno P, et al. Nosocomial urinary tract infection in the intensive care unit: when should Pseudomonas aeruginosa be suspected? Experience of the French national surveillance of nosocomial infections in the intensive care unit, Rea-Raisin. Clin Microbiol Infect. 2012;18(1):E13–E15. | ||
Schechner V, Nobre V, Kaye KS, et al. Gram-negative bacteremia upon hospital admission: when should Pseudomonas aeruginosa be suspected? Clin Infect Dis. 2009;48(5):580–586. | ||
Lange K, Buerger M, Stallmach A, Bruns T. Effects of antibiotics on gut microbiota. Dig Dis. 2016;34(3):260–268. | ||
Aloush V, Navon-Venezia S, Seigman-Igra Y, Cabili S, Carmeli Y. Multidrug-resistant Pseudomonas aeruginosa: risk factors and clinical impact. Antimicrob Agents Chemother. 2006;50(1):43–48. | ||
Horcajada JP, Shaw E, Padilla B, et al. Healthcare-associated, community-acquired and hospital-acquired bacteraemic urinary tract infections in hospitalized patients: a prospective multicentre cohort study in the era of antimicrobial resistance. Clin Microbiol Infect. 2013;19(10):962–968. |
Supplementary materials
Variable definitions
- Sepsis was defined as a diagnosis of systemic inflammatory response syndrome –SIRS- (at least two of the following: temperature > 38.3 or <36, respiratory rate > 20 min or paCO2 <32 mmHg (4.3 kPa)) in the presence of an infection.
- Severe sepsis was defined as sepsis together with evidence of organ dysfunction.
- Septic shock was defined as sepsis associated with hypotension (systolic blood pressure <90 mmHg or reduction of > 40 mmHg from baseline) or lactic acidosis (lactate > 4 mmol/L) despite adequate fluid resuscitation.
- Liver disease was defined as chronic liver failure, usually occurring in the context of cirrhosis. Mild liver disease could be classified as cirrhosis without portal hypertension, or chronic hepatitis. Moderate or severe liver disease as cirrhosis with portal hypertension.
- Chronic renal impairment was defined as an eGFR of 60 ml/min/1.732 (measured by the Cockroft-Gault equation) or less, or a creatinine of > 2.5mg/dL. Patients with renal replacement therapy were included. Patients with a functioning kidney transplant were not included (were recorded under transplantation).
  | Table S2 ICD9 data Notes: ICD9 CM code database http://icd9data.com/2015/volume1/580-629/default.htm. Abbreviation: ICD-9 CM, International Classification of Diseases-9 Clinical Modification. |
  | Table S3 ICD data Notes: ICD10 CM code database http://apps.who.int/classifications/icd10/browse/2015/en. Abbreviations: ICD-10 CM, International Classification of Diseases-10 Clinical Modification; NOS, not otherwise specified. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.