Back to Archived Journals » Journal of Neurorestoratology » Volume 2
Review of recently documented clinical neuroprotective and cellular treatment for spinal cord injury: an analysis of outcomes
Authors Saberi H, Derakhshanrad N, Yekaninejad MS
Received 7 August 2013
Accepted for publication 11 October 2013
Published 6 December 2013 Volume 2014:2 Pages 15—24
DOI https://doi.org/10.2147/JN.S49208
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Hooshang Saberi,1,2 Nazi Derakhshanrad,1 Mir Saeed Yekaninejad1,3
1Brain and Spinal Injuries Research Center, 2Department of Neurosurgery, Imam Khomeini Hospital, 3Department of Epidemiology and Biostatistics, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran
Abstract: Emergence of new neuroprotective treatments for spinal cord injury (SCI) in recent clinical studies is a matter of great scientific and public interest. Effect size and complication rates are of particular concern. In this study, a search was performed in the PubMed, Scopus, and Scholar Google search engines using the keywords “spinal cord injury”, “cell transplantation” and “neuroprotective”. Clinical studies published in the English language were included. Using the study inclusion criteria, 45 clinically relevant studies were found in which neurologic changes was descriptively reported. Of these, 20 studies with American Spinal Injury Association scale, and sensory and motor reports were included for quantitative review. Overall, these papers show an increment in the number of studies reported since the turn of the century, and recent endeavors in the field have accelerated. To compare newer and older studies, considering sufficient numbers of studies in each group, the year 2010 was identified as the turning point. The mean ± standard deviation change in motor score after treatment was 8.67±3.48 in reports before 2010, and this increased to 8.95±2.78 after 2010. Similarly, the mean change in score for light touch was 9.57±3.63 before 2010 and increased to 10.58±3.06 after 2010. The mean change in score for pinprick sensation improved from 8.36±2.82 before 2010 to 8.39±2.69 after 2010. Our study indicates that cellular and neuroprotective therapies are becoming more popular, and the mean neurologic effect size in terms of light touch shows an increment, paving the pathway for clinical applications to be established in the near future.
Keywords: spinal cord injury, cell transplantation, neuroprotective treatment
Introduction
Spinal cord injuries (SCIs) are amongst the most common and disabling neurologic disorders in modern society. Historically, the diagnosis of SCI and its prognosis goes back to Hippocrates, whose recommendation was that SCI is “an ailment not to be treated”. Final outcomes of established treatments for complete and/or incomplete SCI have well known limitations.1 For example, in patients with American Spinal Injury Association (ASIA) grade A impairment, the likelihood of neurologic autorecovery is in the range of 6%–13%.2 Pursuing these issues, numerous experimental models of SCI have been designed, and chemical and cellular interventions for neurological restoration have been conducted using these models and published over many years.3 These include a heterogeneous variety of SCI models, as well as a large number of drug and cellular interventions used for neurologic restoration,4 a thorough overview of which may not be possible in this paper. At the same time, meticulous surveys and assessments by ethical review boards have enabled small trials to be conducted in well known international centers5 with well developed guidelines.6–9
Meanwhile, a growing number of requests by SCI patients to participate voluntarily in clinical trials in the hope of neurologic improvement has been observed all over the world.5 On the other hand, information on the effect size of neurorestorative treatments, in terms of well established scores, has not been adequately documented. Taking into consideration that many of these patients may expect a perfect outcome from treatment, acceptable recruitment may also require explaining a rough estimate of possible outcomes8 to prospective volunteers. Historical progress regarding the effect size of the reported trials as well as trends over time is also an important issue that may realize the hope for more plausible, feasible, and cost-effective treatments. In this study, papers published on neurorestoration after SCI up until 2012 are reviewed and the outcomes are analyzed.
Methods
A search of the PubMed, Scopus, and Google Scholar databases for peer-reviewed papers published in the English language was performed using the keywords “spinal cord injury”, “cell transplantation”, and “neuroprotective”. A thorough survey and evaluation of 350 papers identified by two reviewers yielded 55 papers having clinical content correlated with bedside application of neuroprotective and/or cellular treatments for SCI published between 1966 and 2012. Review articles and/or nonoriginal papers were excluded, along with duplicate publications of the same original study and letters to the editor. Original papers reporting a definitive intervention in a stated number of patients were included in the analysis. In the case of two or three publications emanating from one study, the version with the longest follow-up and/or fullest reporting was included. All the included studies had been approved by local institutional review boards. Thereafter, of the 55 articles obtained, 45 original papers were included and analyzed as descriptive reports regarding cell types and/or drugs used, the geographic location of the study, publication year, sample size, chronicity of SCI, and level of the lesion in the neuraxis. Methodologic aspects, including randomization, blinding, and inclusion of a comparative design were also assessed. Route of cell delivery, cell type, duration of follow-up, frequency of assessment, inclusion and exclusion criteria, age range, and any associated combination therapies were also recorded. Neurologic and functional assessments, as well as objective paraclinical evaluations, were extracted from the studies.10–54 The findings were categorized and reported as being published before or after 2010. Finally, reported procedure-related complications were scrutinized and recorded. Case reports were excluded at this stage of the meta-analysis.
Using the aforementioned criteria (Table 1), of the 45 papers evaluated, 20 were studies that included quantitative ASIA scoring reports (from 1991 to 2012). These papers were investigated with regard to the magnitude of neurologic changes (ASIA motor, light touch, and pinprick sensation) after neuroprotective and/or cellular treatments. The exact duration of follow-up and reports of ASIA scores were extracted from papers with quantitative reporting.11,14,18,20,22,24,35,37,39–43,45–49,51,52
  | Table 1 Inclusion and exclusion criteria for selecting ASIA quantitative reports (20 studies) |
For the 20 studies selected, the magnitude of sensory (light touch, pinprick sensation) and motor changes based on the ASIA scoring system were analyzed. Mean changes and standard deviations were extracted. To compare the newer and older studies, considering sufficient numbers of studies in each group, the year 2010 was deemed to be the turning point. The studies were finally grouped as being before or after 2010. The degree of improvement in terms of this time point was compared and is reported. All numeric data are shown as the mean ± standard deviation. The independent-samples t-test was used to compare group means. A P-value less than 0.05 was considered to be statistically significant.
Results
A descriptive analysis was performed using the 45 eligible studies. In terms of lesion level, most of the studies included both cervical and thoracic patients (27 studies, 60%), while seven studies included cervical patients (15.6%), and the same number included only thoracic patients. Regarding sample size, 15 studies included more than 25 patients (33.3%), while in eight studies the patient number was less than five (17.8%). Duration of follow-up was most frequently between 6 and 18 months (45.9%, Table 2). The most common cell types used were mesenchymal stem cells (37.8%) and hematopoietic stem cells (13.3%). The relative frequencies of the various cell types and/or drugs used are shown in Table 3. Regarding route of cell or drug delivery, 15 studies used intramedullary injection (32.6%), while 12 (26.1%) used cell delivery via lumbar puncture (Table 4). In terms of SCI chronicity, 26 studies (59.1%) recruited patients with chronic SCI, seven recruited patients with acute SCI, four recruited patients with subacute SCI, and two studies were conducted using both acute and chronic cases. Four studies included both subacute and chronic cases, and one study covered all three phases (acute <1 week; 1 week ≤ subacute <6 months; chronic ≥6 months) after injury. One study did not mention the chronicity of SCI.54 A geographic distribution map of the reported studies is shown in Figure 1, based on the number of patients studied.
  | Table 2 Characteristics of the 45 studies analyzed |
  | Table 3 Frequency (relative) of various cellular and/or drug treatments for neurological neurorestoration in the studies analyzed |
  | Table 4 Frequency of various routes of treatment in the studies analyzed |
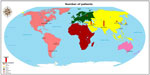  | Figure 1 Geographic distribution of the 45 studies included from around the world. |
The mean study sample size was 31.84 patients before 2010 and 38.10 patients from 2010 onwards. In terms of chronicity before 2010, 16 studies including chronic cases, five including acute cases, three including subacute and chronic cases, one including acute and chronic cases, and one covering all phases of chronicity were reported, while ten chronic, four subacute, two acute, one acute and chronic, and one subacute and chronic studies were reported after 2010.
In terms of cell types used, before 2010, eleven studies using mesenchymal stem cells, four using hematopoietic stem cells, two using embryonic stem cells, two using olfactory mucosa, one using olfactory ensheathing cells, one using macrophages, one using umbilical cord stem cells, one using fetal olfactory bulb tissue, one using a sural nerve graft, one using GM1 ganglioside, and one using thyrotropin-releasing hormone were reported. From 2010 onwards, six studies on mesenchymal stem cells, four on granulocyte colony-stimulating factor, two on hematopoietic stem cells, two on Schwann cells, two on fetal olfactory ensheathing cells, one on adipose mesenchymal stem cells, one on embryonic stem cells, and one on Cethrin® (BioAxone BioSciences Inc., Cambridge, MA, USA) were reported. The route of treatment delivery was intramedullary in eleven studies, intrathecal in seven, intravenous in three, and intra-arterial in two; from 2010 onwards, there were five intravenous studies, five intrathecal studies, four intramedullary studies, and one extradural study at the time of decompression. Of note, eight studies used more than one route of cell delivery.
In total, 20 studies were eligible for quantitative analysis of effect size. The studies were classified according to date of publication, ie, whether they were pre or post 2010. Nine studies were published before 2010 and eleven after 2010.
Before 2010, the mean change in ASIA motor score was 8.67±3.48 and increased to 8.95±2.78 after 2010. The mean change in score for light touch was 9.57±3.63 before 2010 and improved to 10.58±3.06 from 2010 onwards. The mean change in score for pinprick sensation improved from 8.36±2.82 before 2010 to 8.39±2.69 from 2010 onwards (P>0.20, Figure 2). None of the observed changes in motor, light touch, and pinprick scores were statistically significant. The mean overall improvement in motor scores was 9.19±2.60. The overall mean improvement in light touch was 9.61±2.55, while mean improvement in pinprick sensation was 8.19±2.24.
  | Figure 2 Comparison of ASIA score components before 2010 and thereafter in the 20 studies with a quantitative ASIA report. |
Regarding the effect of chronicity on outcome in terms of the ASIA scoring system, acute and subacute SCI was compared with chronic SCI, and the results are shown in Figure 3.
  | Figure 3 Comparison of mean ASIA score reports in terms of lesion chronicity in the 20 studies with a quantitative ASIA report. |
The most commonly reported complications were neuropathic pain (26.7%) and spasticity (13.3%). Less commonly reported untoward effects included neurologic deterioration, aseptic meningitis, encephalomyelitis, leakage of cerebrospinal fluid, fever, headache, skin rash, hepatic dysfunction, myelomalacia, and autonomic dysreflexia. Ten of the papers clearly stated their position regarding conflicts of interest, while ten papers did not.
Discussion
Neuroprotective treatments for clinical SCI have become very popular since 1991, and the frequency of these studies is progressively increasing.5 Despite the great diversity of cell types and/or drugs investigated for neurorestorative purposes in SCI trials, only hematopoietic cells, bone marrow stem cells, Schwann cells, and olfactory ensheathing cells have been reported to be safe for clinical use.55,56 The most commonly used have been mesenchymal stem cells followed by hematopoietic stem cells (Table 3) because they are easily available in generous numbers. No differences in safety have been found between these cell types. However, there is no documented clinical study comparing the various cell types (Table 5) in terms of efficacy. In our analysis, a rough comparison of the different reports showed greater changes in ASIA scores in studies using mesenchymal stem cells. Although there are a few studies on simultaneous transplantation of various cell types, our comparison did not support any superiority for this strategy as compared with single-cell treatments; however, well established assessment tools and teams may be necessary for such a comparison.
Diverse methods were used to assess the final outcome in these trials, most commonly the ASIA scoring system. Other modalities included functional tests (Functional Independence Measure, Functional Assessment Measure, Spinal Cord Independence Measure, Functional Rating Scale, Japanese Orthopaedic Association, Barthel Index, Walking Index of Spinal Cord Injury, International Spinal Cord Injury Scale), electrophysiologic tests (electromyography, nerve conduction studies, somatosensory evoked potentials, motor-evoked potentials), psychologic testing (Beck Depression Inventory), algometric measurements (visual analog scale), and testing for spasticity (Modified Ashworth Scale). The choice of testing method may depend on the outcome of interest.
Drug treatments for SCI are more promising, safer, less expensive, and more easily obtainable as compared with cell therapies. In our study, drugs commonly used have included granulocyte colony-stimulating factor, thyrotropin-releasing hormone, GM1 ganglioside, and Cethrin.21,22,37,47 The most commonly used of these has been granulocyte colony-stimulating factor due to its safety, regulatory approval, low complication rate, and availability.57 The conduct and design of randomized controlled trials for evaluation of drug effects seems to be much easier than for more invasive cellular treatments. Use of drugs may be less invasive and have lower complication rates. Also, as with other nonsurgical methods, drug treatment can be repeated if necessary, and patients remain able to participate in future studies without any risk of postoperative adhesions. Knowledge about adverse drug reactions may require further long-term follow-up. Further, administration of placebo may be more ethically plausible than carrying out sham surgery.
The most common route used for administration of cells and drugs has been intramedullary delivery followed by intrathecal delivery. Less invasive methods are preferred, and are becoming more popular, especially in studies reported from 2010 onwards, according to the present results.
Among the various reported series, there are only a few randomized clinical trials providing solid evidence on which to base clinical judgment. This is despite relatively large numbers of patients being included in some of the reported studies.2 This may reflect the difficulty of randomization when using cell therapy research methodologies, indicating a need to move towards better designed studies. The case series design is more common, but as we know, the level of evidence it provides is not sufficient to warrant definitive conclusions.
The Mackay-Sim group have reported an elegant randomized controlled study on the use of olfactory ensheathing cells for chronic SCI, that showed only sensory improvement in the zone of partial preservation in cases; however, magnetic resonance imaging showed extensive tissue loss in the thoracic spinal cord, this extent of tissue loss makes it a difficult case for neurorestoration.20 Nevertheless, this study did demonstrate the safety of this cell transplantation procedure.
Subjects with thoracic lesions have been preferred by many investigators because of the safety of performing cellular procedures at this level and the homogeneity of the study population. However, in our series, most of the investigators included both thoracic and cervical patients.9 It should be borne in mind that the likelihood of motor recovery in the zone of partial preservation is greater in patients with cervical lesions, although the procedure may carry safety risks.42 On the other hand, documentation of motor improvement in thoracic patients may be much more difficult because assessment of the intercostal motor nerves may require use of indirect methods, such as spirometry. Rehabilitation has been an important adjunct to cellular treatments, and almost all papers recommend adherence to a standard rehabilitation program along with cellular treatment.42 This was indeed practiced in almost all of the reviewed literature.
Interventions in the acute and subacute phase are believed to be associated with a better outcome regarding the glial scar formation phase in chronic cases.58 Since 2010, the numbers of acute and subacute interventions show an incremental trend. Of the papers reviewed, only 15.9% of studies were performed in acute cases. The better neurologic outcomes reported for acute and subacute lesions in these papers should be interpreted in the same way. Regarding timing of cell transplantation after SCI, the process of obtaining informed consent and cell processing may be time-consuming in the acute phase. General medical instability at this time may be a further impediment to cell transplantation. Also, injected cell suspensions may be lost through dural tears in the acute phase. Inflammation and hemorrhage in the acute phase, if severe, may also hinder the effectiveness of cell therapy. Again, the effect of autorecovery in the acute phase is more pronounced than in the chronic phase. On the other hand, undertaking revision surgery for cell transplantation following decompression and fixation may pose a problem in the acute stage. Further, cell processing centers may not be widely available during the acute stage. Denial of the irreversibility of the condition on the part of the patient during the acute phase of SCI also raises problems, largely regarding obtaining informed consent for cell transplantation.
All the above mentioned issues come together to delay the cell therapy procedure. On the other hand, these issues are less troublesome in the chronic phase, although emergence of the glial scar as a barrier to axonal sprouting poses a new problem. The optimum time for cell therapy in patients with minimal cord edema and scar formation remains unclear.
Regarding neurologic outcome, most of the studies report some improvement in pinprick sensation and light touch scores, but reports of motor improvement have been less frequent. However, almost all studies have reported the motor outcome, indicating its functional importance when compared with the sensory outcome (Table 5). From 2010 onwards, change in light touch score was improved compared with before 2010; however, this effect could be due to improvements in cell or drug preparation and/or technical advances.
The study population range has usually been in the order of 5–15 cases. This variation is the result of two considerations, ie, the larger the sample size the stronger would be the statistical evidence, and safety issues have limit the sizes of the studies, therefore primary phases (1 and 2) studies are smaller.
Conclusion
This paper highlights the multitude of attempts that have been made to test cell therapy, the reported adverse effects, as well as route of delivery. The choice of cell type as well as timing and route of delivery is an important decision in trials of treatment for SCI. Better designed studies are needed to produce generally accepted guidelines to make the best decisions in this regard.
Acknowledgments
The authors are grateful to the researchers who have published their work, thereby providing the material for this study. We also thank Rasoul Mansouri for preparing the international geographic plot of the studies.
Disclosure
The authors report no conflicts of interest concerning the materials or methods used in this study or its findings, and declare no competing financial interests.
References
Aidinoff E, Front L, Itzkovich M, et al. Expected spinal cord independence measure, third version, scores for various neurological levels after complete spinal cord lesions. Spinal Cord. 2011;49(8):893–896. | |
Harrop JS, Hashimoto R, Norvell D, et al. Evaluation of clinical experience using cell-based therapies in patients with spinal cord injury: a systematic review. J Neurosurg Spine. 2012;17(Suppl 1):230–246. | |
Kramer AS, Harvey AR, Plant GW, Hodgetts SI. Systematic review of induced pluripotent stem cell technology as a potential clinical therapy for spinal cord injury. Cell Transplant. 2013;22(4):571–617. | |
Firouzi M, Moshayedi P, Saberi H, et al. Transplantation of Schwann cells to subarachnoid space induces repair in contused rat spinal cord. Neurosci Lett. 2006;402(1–2):66–70. | |
Fehlings MG, Vawda R. Cellular treatments for spinal cord injury: the time is right for clinical trials. Neurotherapeutics. 2011;8(4):704–720. | |
Fawcett JW, Curt A, Steeves JD, et al. Guidelines for the conduct of clinical trials for spinal cord injury as developed by the ICCP panel: spontaneous recovery after spinal cord injury and statistical power needed for therapeutic clinical trials. Spinal Cord. 2007;45(3):190–205. | |
Lammertse D, Tuszynski MH, Steeves JD, et al. Guidelines for the conduct of clinical trials for spinal cord injury as developed by the ICCP panel: clinical trial design. Spinal Cord. 2007;45(3):232–242. | |
Steeves JD, Lammertse D, Curt A, et al. Guidelines for the conduct of clinical trials for spinal cord injury (SCI) as developed by the ICCP panel: clinical trial outcome measures. Spinal Cord. 2007;45(3):206–221. | |
Tuszynski MH, Steeves JD, Fawcett JW, et al. Guidelines for the conduct of clinical trials for spinal cord injury as developed by the ICCP panel: clinical trial inclusion/exclusion criteria and ethics. Spinal Cord. 2007;45(3):222–231. | |
Abdelaziz OS, Marie A, Abbas M, Ibrahim M, Gabr H. Feasibility, safety, and efficacy of directly transplanting autologous adult bone marrow stem cells in patients with chronic traumatic dorsal cord injury: a pilot clinical study. Neurosurg Q. 2010;20(3):216–226. | |
Adel N, Gabr H, Hamdy S, Afifi L, Mahmoud H. Stem cell therapy in chronic spinal cord injuries. Egyptian Journal of Neurology, Psychiatry and Neurosurgery. 2009;46(2):467–478. | |
Bhanot Y, Rao S, Ghosh D, Balaraju S, Radhika CR, Satish Kumar KV. Autologous mesenchymal stem cells in chronic spinal cord injury. Br J Neurosurg. 2011;25(4):516–522. | |
Callera F, de Melo CM. Magnetic resonance tracking of magnetically labeled autologous bone marrow CD34+ cells transplanted into the spinal cord via lumbar puncture technique in patients with chronic spinal cord injury: CD34+ cells’ migration into the injured site. Stem Cells Dev. 2007;16(3):461–466. | |
Chhabra HS, Lima C, Sachdeva S, et al. Autologous olfactory [corrected] mucosal transplant in chronic spinal cord injury: an Indian pilot study. Spinal Cord. 2009;47(12):887–895. | |
Kumar AA, Kumar SR, Narayanan R, Arul K, Baskaran M. Autologous bone marrow derived mononuclear cell therapy for spinal cord injury: a phase I/II clinical safety and primary efficacy data. Exp Clin Transplant. 2009;7(4):241–248. | |
Pal R, Venkataramana NK, Bansal A, et al. Ex vivo-expanded autologous bone marrow-derived mesenchymal stromal cells in human spinal cord injury/paraplegia: a pilot clinical study. Cytotherapy. 2009;11(7):897–911. | |
Mehta T, Feroz A, Thakkar U, Vanikar A, Shah V, Trivedi H. Subarachnoid placement of stem cells in neurological disorders. Transplant Proc. 2008;40(4):1145–1147. | |
Lima C, Escada P, Pratas-Vital J, et al. Olfactory mucosal autografts and rehabilitation for chronic traumatic spinal cord injury. Neurorehabil Neural Repair. 2009;24(1):10–22. | |
Cristante AF, Barros-Filho TE, Tatsui N, et al. Stem cells in the treatment of chronic spinal cord injury: evaluation of somatosensitive evoked potentials in 39 patients. Spinal Cord. 2009;47(10):733–738. | |
Mackay-Sim A, Feron F, Cochrane J, et al. Autologous olfactory ensheathing cell transplantation in human paraplegia: a 3-year clinical trial. Brain. 2008;131(Pt 9):2376–2386. | |
Pitts LH, Ross A, Chase GA, Faden AI. Treatment with thyrotropin-releasing hormone (TRH) in patients with traumatic spinal cord injuries. J Neurotrauma. 1995;12(3):235–243. | |
Geisler FH, Dorsey FC, Coleman WP. Recovery of motor function after spinal-cord injury – a randomized, placebo-controlled trial with GM-1 ganglioside. N Engl J Med. 1991;324(26):1829–1838. | |
Guest J, Herrera LP, Qian T. Rapid recovery of segmental neurological function in a tetraplegic patient following transplantation of fetal olfactory bulb-derived cells. Spinal Cord. 2006;44(3):135–142. | |
Geffner LF, Santacruz P, Izurieta M, et al. Administration of autologous bone marrow stem cells into spinal cord injury patients via multiple routes is safe and improves their quality of life: comprehensive case studies. Cell Transplant. 2008;17(12):1277–1293. | |
Ra JC, Shin IS, Kim SH, et al. Safety of intravenous infusion of human adipose tissue-derived mesenchymal stem cells in animals and humans. Stem Cells Dev. 2011;20(8):1297–1308. | |
Kang KS, Kim SW, Oh YH, et al. A 37-year-old spinal cord-injured female patient, transplanted of multipotent stem cells from human UC blood, with improved sensory perception and mobility, both functionally and morphologically: a case study. Cytotherapy. 2005;7(4):368–373. | |
Yoon SH, Shim YS, Park YH, et al. Complete spinal cord injury treatment using autologous bone marrow cell transplantation and bone marrow stimulation with granulocyte macrophage-colony stimulating factor: phase I/II clinical trial. Stem Cells. 2007;25(8):2066–2073. | |
Park HC, Shim YS, Ha Y, et al. Treatment of complete spinal cord injury patients by autologous bone marrow cell transplantation and administration of granulocyte-macrophage colony stimulating factor. Tissue Eng. 2005;11(5–6):913–922. | |
Park JH, Kim DY, Sung IY, et al. Long-term results of spinal cord injury therapy using mesenchymal stem cells derived from bone marrow in humans. Neurosurgery. 2011;70(5):1238–1247. | |
Moviglia GA, Fernandez Vina R, Brizuela JA, et al. Combined protocol of cell therapy for chronic spinal cord injury. Report on the electrical and functional recovery of two patients. Cytotherapy. 2006;8(3):202–209. | |
Moviglia GA, Varela G, Brizuela JA, et al. Case report on the clinical results of a combined cellular therapy for chronic spinal cord injured patients. Spinal Cord. 2009;47(6):499–503. | |
Deda H, Inci MC, Kurekci AE, et al. Treatment of chronic spinal cord injured patients with autologous bone marrow-derived hematopoietic stem cell transplantation: 1-year follow-up. Cytotherapy. 2008;10(6):565–574. | |
Frolov AA, Bryukhovetskiy AS. Effects of haematopoietic autologous stem cell transplantation to the chronically injured human spinal cord evaluated by motor and somatosensory evoked potentials methods. Cell Transplant. 2012;21 Suppl 1:S49–S55. | |
Rabinovich SS, Seledtsov VI, Poveschenko OV, et al. Transplantation treatment of spinal cord injury patients. Biomed Pharmacother. 2003;57(9):428–433. | |
Chernykh ER, Stupak VV, Muradov GM, et al. Application of autologous bone marrow stem cells in the therapy of spinal cord injury patients. Bull Exp Biol Med. 2007;143(4):543–547. | |
Seledtsova GV, Rabinovich SS, Belogorodtsev SN, Parlyuk OV, Seledtsov VI, Kozlov VA. Delayed results of transplantation of fetal neurogenic tissue in patients with consequences of spinal cord trauma. Bull Exp Biol Med. 2011;149(4):530–533. | |
Fehlings MG, Theodore N, Harrop J, et al. A phase I/IIa clinical trial of a recombinant Rho protein antagonist in acute spinal cord injury. J Neurotrauma. 2011;28(5):787–796. | |
Falci S, Holtz A, Akesson E, et al. Obliteration of a posttraumatic spinal cord cyst with solid human embryonic spinal cord grafts: first clinical attempt. J Neurotrauma. 1997;14(11):875–884. | |
Huang H, Xi H, Chen L, Zhang F, Liu Y. Long-term outcome of olfactory ensheathing cell therapy for patients with complete chronic spinal cord injury. Cell Transplant. 2012;21 Suppl 1:S23–S31. | |
Zhou XH, Ning GZ, Feng SQ, et al. Transplantation of autologous activated Schwann cells in the treatment of spinal cord injury: six cases, more than five years of follow-up. Cell Transplant. 2012;21 Suppl 1:S39–S47. | |
Wu J, Sun T, Ye C, Yao J, Zhu B, He H. Clinical observation of fetal olfactory ensheathing glia transplantation (OEGT) in patients with complete chronic spinal cord injury. Cell Transplant. 2012;21 Suppl 1:S33–S37. | |
Saberi H, Firouzi M, Habibi Z, et al. Safety of intramedullary Schwann cell transplantation for postrehabilitation spinal cord injuries: 2-year follow-up of 33 cases. J Neurosurg Spine. 2011;15(5):515–525. | |
Karamouzian S, Nematollahi-Mahani SN, Nakhaee N, Eskandary H. Clinical safety and primary efficacy of bone marrow mesenchymal cell transplantation in subacute spinal cord injured patients. Clin Neurol Neurosurg. 2012;114(7):935–939. | |
Saito F, Nakatani T, Iwase M, et al. Spinal cord injury treatment with intrathecal autologous bone marrow stromal cell transplantation: the first clinical trial case report. J Trauma. 2008;64(1):53–59. | |
Saito F, Nakatani T, Iwase M, et al. Administration of cultured autologous bone marrow stromal cells into cerebrospinal fluid in spinal injury patients: a pilot study. Restor Neurol Neurosci. 2012;30(2):127–136. | |
Sakuma T, Yamazaki M, Okawa A, et al. Neuroprotective therapy using granulocyte colony-stimulating factor for patients with worsening symptoms of compression myelopathy, Part 1: a phase I and IIa clinical trial. Eur Spine J. 2012;21(3):482–489. | |
Sakuma T, Yamazaki M, Okawa A, et al. Neuroprotective therapy using granulocyte colony-stimulating factor for patients with worsening symptoms of thoracic myelopathy: a multicenter prospective controlled trial. Spine (Phila Pa 1976). 2012;37(17):1475–1478. | |
Takahashi H, Yamazaki M, Okawa A, et al. Neuroprotective therapy using granulocyte colony-stimulating factor for acute spinal cord injury: a phase I/IIa clinical trial. Eur Spine J. 2012;21(12):2580–2587. | |
Kishk NA, Gabr H, Hamdy S, et al. Case control series of intrathecal autologous bone marrow mesenchymal stem cell therapy for chronic spinal cord injury. Neurorehabil Neural Repair. 2010;24(8):702–708. | |
Cheng H, Liao KK, Liao SF, Chuang TY, Shih YH. Spinal cord repair with acidic fibroblast growth factor as a treatment for a patient with chronic paraplegia. Spine (Phila Pa 1976). 2004;29(14):E284–E288. | |
Knoller N, Auerbach G, Fulga V, et al. Clinical experience using incubated autologous macrophages as a treatment for complete spinal cord injury: phase I study results. J Neurosurg Spine. 2005;3(3):173–181. | |
Sykova E, Homola A, Mazanec R, et al. Autologous bone marrow transplantation in patients with subacute and chronic spinal cord injury. Cell Transplant. 2006;15(8–9):675–687. | |
Hammadi AA, Marino A, Farhan S. Clinical response of 277 patients with spinal cord injury to stem cell therapy in Iraq. Int J Stem Cells. 2012;5(1):76–78. | |
Kato K, Yamazaki M, Okawa A, et al. Intravenous administration of granulocyte colony-stimulating factor for treating neuropathic pain associated with compression myelopathy: a phase I and IIa clinical trial. Eur Spine J. 2012;22(1):197–204. | |
Mackay-Sim A, St John JA. Olfactory ensheathing cells from the nose: clinical application in human spinal cord injuries. Exp Neurol. 2010;229(1):174–180. | |
Saberi H, Moshayedi P, Aghayan HR, et al. Treatment of chronic thoracic spinal cord injury patients with autologous Schwann cell transplantation: an interim report on safety considerations and possible outcomes. Neurosci Lett. 2008;443(1):46–50. | |
Rad ND, Saberi H, Yekaninejad MS, et al. Safety of granulocyte-colony stimulating factor (G-CSF) administration for post rehabilitated motor complete spinal cord injury patients: an open label, phase one study. Cell Transplant. August 29, 2013. [Epub ahead of print.] | |
Guest J, Harrop JS, Aarabi B, et al. Optimization of the decision-making process for the selection of therapeutics to undergo clinical testing for spinal cord injury in the North American Clinical Trials Network. J Neurosurg Spine. 2012;17(Suppl 1):94–101. |
 © 2013 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2013 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

