Back to Journals » Vascular Health and Risk Management » Volume 11
Reversal of statin-induced memory dysfunction by co-enzyme Q10: a case report
Authors Okeahialam B
Received 16 June 2015
Accepted for publication 15 September 2015
Published 6 November 2015 Volume 2015:11 Pages 579—581
DOI https://doi.org/10.2147/VHRM.S90551
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Daniel Duprez
Video abstract presented by author by Okeahialam.
Views: 5025
Basil N Okeahialam
Cardiology Sub-Unit 1, Department of Medicine, Jos University Teaching Hospital, Jos, Nigeria
Abstract: Statins are useful in the armamentarium of the clinician dealing with dyslipidemia, which increases cardiovascular morbi-mortality in hypertensive and diabetic patients among others. Dyslipidemia commonly exists as a comorbidity factor in the development of atherosclerotic cardiovascular disease. Use of statins is however associated with side effects which at times are so disabling as to interfere with activities of daily living. There are various ways of dealing with this, including use of more water-soluble varieties, intermittent dosing, or use of statin alternatives. Of late, use of co-enzyme Q10 has become acceptable for the muscle side effects. Only one report of any benefit on the rarely reported memory side effect was encountered by the author in the search of English medical literature. This is a report of a documented case of a Nigerian woman with history of statin intolerance in this case, memory dysfunction despite persisting dyslipidemia comorbidity. Her memory dysfunction side effect which interfered with activities of daily living and background muscle pain cleared when coenzyme Q10 was administered alongside low dose statin. Her lipid profile normalized and has remained normal. It is being recommended for use when statin side effects (muscle- and memory-related) impair quality of life and leave patient at dyslipidemia-induced cardiovascular morbi-mortality.
Keywords: statin, memory dysfunction, co-enzyme Q10, improvement
Introduction
Memory dysfunction, though said to be a rare occurrence,1 is a recognized side effect of statin therapy.2 Only one case has been reported in the medical literature from Nigeria.3 It tends to greatly limit normal functioning in an individual so afflicted, hence its potential for impairing quality of life. This side effect like others usually results in discontinuation of the culprit drug.4 In dyslipidemia, this results in persistence of cardiovascular morbidity in the individual(s) in question.
In the case of skeletal muscle side effect, co-enzyme Q10 (CoE Q10) has been reported to have an ameliorating effect,5 as well as vitamin D replacement.6 Only one report of such amelioration on the part of CoE Q10 for statin-induced memory dysfunction was encountered in a wide search of medical literature.7 No such report was encountered from sub-Saharan Africa. CoE Q10 has been used with statins in situations where the disease impairs mitochondrial bioenergetics with increased oxidative stress; as in some neurological diseases, with benefit.8 It has been shown to reverse age-related impairment in spatial learning while lowering protein oxidation, leading to the conclusion that it stands to ameliorate age-associated symptoms of cognitive deficit.9 Recently, one was compelled to try CoE Q10 supplementation in a patient previously reported3 for intolerance of statin manifesting with muscle and memory side effects – as her lipid profile worsened despite substitution with statin substitutes and adjuvants.10 The response was impressive. She experienced no muscle or memory side effect while on CoE Q10 supplementation and her lipid profile improved.
This case is being reported to suggest to clinicians who may encounter memory issues with statin that CoE Q10 supplementation may be worth the try.
Case report
Patient AA had been reported previously with memory dysfunction while on different statins.3 For the current report, ethical approval was not sought because the case did not meet institutional requirements and the Declaration of Helsinki was duly observed. As she remained dyslipidemic with a history of statin discontinuation, she was given fish oil in the form of Omega-3 1,000 mg daily in September 2011. This did not necessarily improve her memory. With her lipid profile standing at total cholesterol (TC) – 5.2 mmol/L, high density lipoprotein cholesterol (HDL-C) – 1.0 mmol/L, low density lipoprotein cholesterol (LDL-C) – 3.0 mmol/L, and triglyceride (TG) – 0.9 mmol/L in December 2011, she was given fenofibrate 160 mg daily. She reported to cause her muscle cramps like the statins prompting self-discontinuation and a repeat lipid profile. We could not assay for creatinine kinase, but her liver and kidneys were normal in structure and function. With lipid profile standing at TC – 5.7 mmol/L, HDL-C – 1.0 mmol/L, LDL-C – 3.2 mmol/L, and TG – 1.1 mmol/L in January 2012, she was advised to persevere on fenofibrate, because the symptom of muscle cramp was mild. Attempting to do this brought in addition vertigo and unsteady gait. Reporting this, she was asked to stop the drug and put on Stugeron brand of cinnarizine (15 mg twice daily) which controlled the symptoms.
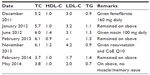
When her lipid profile was repeated late in June 2012, it stood at TC – 6.0 mmol/L, HDL-C – 1.4 mmol/L, LDL-C – 3.1 mmol/L, and TG – 1.3 mmol/L. She was consequently put on niacin 100 mg daily. With this, there was no complaint, but the lipid profile worsened over time to levels of TC – 6.1 mmol/L, HDL-C – 0.9 mmol/L, and TG – 1.3 mmol/L when repeated in February 2013. This prompted the addition of Lipitor brand of atorvastatin to the 100 mg daily of niacin. No sooner had she started this combination did disabling and unsettling memory lapses recur. Lipitor was dropped as a result. She was then asked to take a combination of low dose statin and CoE Q10 when she came in September 2013. Out of fear and frustration, she did not but returned in November 2013 with a lipid profile of TC – 6.1 mmol/L, HDL-C – 1.2 mmol/L, LDL-C – 4.2 mmol/L, and TG – 0.9 mmol/L. She was counseled and her fears dispelled. She then accepted to use rosuvastatin 5 mg at night and CoE Q10 capsules 100 mg daily. She returned in February 2014 to report that she had neither memory dysfunction nor muscle cramp, and her lipid profile now stood at TC – 3.7 mmol/L, HDL-C – 1.0 mmol/L, LDL-C – 1.7 mmol/L, and TG – 1.4 mmol/L. By her last appointment in May 2014 while still on the last treatment, she still remained well regarding her memory with a lipid profile of TC – 3.8 mmol/L, HDL-C – 1.0 mmol/L, LDL-C – 2.0 mmol/L, and TG – 0.7 mmol/L (Table 1).
Comments
The most common side effect reported with statins is myalgia.11 This is said to largely result from depletion of CoE Q10 caused by statins.12 Statins being HMG-CoA reductase inhibitors do this by blocking the mevalonate pathway.12 CoE Q10 is an antioxidant that stabilizes the cell membrane, and as a critical cofactor in the mitochondrial energy metabolism is responsible for regenerating ATP.13 Because it is found in every single cell in the body, its level could also fall in the brain cells as statins have been shown to target several tissues.14
Supplementation of CoE Q10 in statin-treated patients with myopathy has been shown to ameliorate such pains.5 This patient suffered myopathy with several statins as reported earlier.3 She eventually developed memory dysfunction which led to their discontinuation. She did not even tolerate fenofibrate, a statin alternative recommended in the face of persisting dyslipidemia for patients intolerant of statins.10 Interestingly, on another recommended alternative niacin, she had no side effect but the lipid parameters worsened. With the morbidity consequence of mixed dyslipidemia remaining and even worsening, she was put on low dose rosuvastatin 5 mg with 100 mg of CoE Q10. The choice of rosuvastatin was because of its water solubility and utilization of non-CYP3A4 metabolic pathways, which as posited in an earlier paper is a reasonable approach when statin intolerance is experienced.15 Her frustration with statins and the persisting dyslipidemia made us not give her any chance with only stains irrespective of the type. With CoE Q10 supplementation, she experienced no muscle discomfort and her memory remained unaffected.
CoE Q10 has been reported to improve cognitive function in depressed patients,16 and in both Alzheimer’s and Parkinson patients.17 It has been reported to improve statin-induced myopathy,5 as well as memory dysfunction.7 The nonappearance of both muscle and memory side effects in this patient when CoE Q10 was given with rosuvastatin albeit in low dose would tend to suggest that in her the possible depletion of CoE Q10 was affecting both muscle and brain energy metabolism. A limitation of this report is that a mini-mental state examination which would have objectively strengthened the link was not obtained.
Conclusion
Just as it is becoming acceptable to add CoE Q10 to reverse myopathy in patients on statins12 rather than on discontinuation, it may not be out of place to suggest considering CoE Q10 supplementation in other statin-induced side effects particularly and generally for cardiovascular diseases. As posited in a paper,18 mitochondrial oxidative defense integrity has a place in onset and severity of cardiovascular diseases. When in equilibrium such diseases are checked, but any disequilibrium results in disease manifestation. Further studies in this regard are called for, before it can become the standard of care.
Disclosure
The author reports no conflicts of interest in this work.
References
Rojas-Fernandez CH, Cameron JF. Is statin associated cognitive impairment clinically relevant? A narrative review and clinical recommendations. Ann Pharmacother. 2012;46(4):549–557. | |
Padala KP, Padala PR, Potter JF. Simvastatin induced decline in cognition. Ann Pharmacother. 2006;40(10):1880–1883. | |
Okeahialam BN, Isiguzoro IO. Statin related memory dysfunction in a Nigerian woman. A case report. Curr Drug Saf. 2012;7:33–34. | |
Brucket C, Haygen G, Dejager S, Yau C, Begand M. Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients – the PRIMO study. Cardiovasc Drugs Ther. 2005;19(6):403–414. | |
Caso G, Kelly P, McNurlan MA, Lawson WE. Effect of co-enzyme Q10 on myopathic symptoms in patients treated with statins. Am J Cardiol. 2007;99(10):1409–1412. | |
Morley JE. The cholesterol conundrum. J Am Geriatr Soc. 2011;59(10):1955–1956. | |
Langsjoen PH, Langsjoen JO, Langsjoen AM, Lucas LA. Treatment of statin adverse effects with supplemental co-enzyme Q10 and statin discontinuation. Biofactors. 2005;25(1–4):147–152. | |
Littarru GP, Tiano L. Clinical aspects of coenzyme Q10: an update. Curr Opin Clin Nutr Metab Care. 2005;8(6):641–646. | |
Shetty RA, Forster MJ, Sumien N. Co-enzyme Q10 supplementation reverses age-related impairment in spatial learning and lowers protein oxidation. Age (Dordo). 2013;35(5):1821–1834. | |
Sorrentino MJ. An update on statin alternatives and adjuncts. Clin Lipidol. 2012;7(6):721–730. | |
Nawarskas JJ. HMG-CoA reductase inhibitors and co-enzyme Q 10. Cardiol Rev. 2005;13(2):76–79. | |
Marcoff L, Thompson PD. The role of co-enzyme Q 10 in statin associated myopathy: a systematic review. J Am Col Cardiol. 2007;49(23):2231–2237. | |
Wyman M, Leonard M, Morledge T. Co-enzyme Q10: a therapy for hypertension and statin induced myalgia. Clin J Med. 2010;77(7):435–442. | |
Hargreaves IP, Duncan AJ, Heales SJ, Lend JM. The effect of HMG CoA reductase inhibitors on co-enzyme Q10: possible biochemical/clinical implication. Drug Saf. 2005;28(8):659–676. | |
DiNicolantonio JJ. Co-Q10 and L-carnitine for statin myalgia? Expert Rev Cardiovasc Ther. 2012;10(10):1325–1333. | |
Bragin V, Chemodanova M, Dzanofarova N, Bragin I, Czerniawski JL, Aliev G. Integrated treatment approach improves cognitive function in demented and critically depressed patients. Am J Alzheimers Dis Other Demen. 2005;20(1):21–26. | |
DiMauro S, Mancuso M. Mitochondrial diseases: therapeutic approaches. Biosci Rep. 2007;27:125–137. | |
Babato JC. Have no fear, MitoQ10 is here. Hypertension. 2009;54: 222–223. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.