Back to Journals » Journal of Pain Research » Volume 13
Retrospective Assessment of Salvage to 10 kHz Spinal Cord Stimulation (SCS) in Patients Who Failed Traditional SCS Therapy: RESCUE Study
Authors Kapural L , Sayed D , Kim B, Harstroem C, Deering J
Received 13 September 2020
Accepted for publication 17 October 2020
Published 10 November 2020 Volume 2020:13 Pages 2861—2867
DOI https://doi.org/10.2147/JPR.S281749
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Robert B. Raffa
Leonardo Kapural,1 Dawood Sayed,2 Brian Kim,2 Caroline Harstroem,1 James Deering1
1Carolinas Pain Institute, Winston-Salem, NC 27103, USA; 2Department of Anesthesiology, University of Kansas, Kansas City, KS, USA
Correspondence: Leonardo Kapural Email [email protected]
Background and Objectives: A randomized clinical trial demonstrated that 10 kHz SCS (10kHz-SCS) therapy is superior to traditional low-frequency SCS (LF-SCS) at 12- and 24-month clinical follow-ups and led to Food and Drug Administration (FDA) approval of the therapy. The results of the study led our practices to trial 10kHz-SCS in patients who had not maintained pain relief with LF-SCS therapy. Here, we report a large set of data from two clinical sites to assess if 10kHz-SCS is an effective salvage modality when LF-SCS fails.
Methods: We conducted a retrospective chart review of 120 patients across two clinical sites who had LF-SCS implants and were salvaged with 10kHz-SCS.
Results: Data were analyzed from 105 patients between 28 and 90 years old (median 60) with chronic pain for 13.6 years. The mean duration of LF-SCS therapy was 4.66± 3.9 years. The average Visual Analog Scale (VAS) decreased from 8.30± 1.4 (median of 8) cm to 3.32± 2.0 (median of 3) cm at 12 months and 3.36± 2.0 (median of 3) cm at the most recent clinic visit (p< 0.001) following salvage therapy. Pain relief of 50% or more was obtained in 85 out of 105 (81%) patients. Opioid usage decreased from 60.3± 77.1 mg to 32.1± 44.0 mg MSO4 equivalents (p = 0.001) at 12 months after salvage therapy.
Conclusion: Eighty-one percent of patient cases reviewed, where LF-SCS had failed, achieved > 50% pain relief with 10kHz-SCS, and almost all exhibited some clinical improvement. Therefore, 10kHz-SCS should be considered an appropriate option to rescue failed LF-SCS.
Keywords: spinal cord stimulation, high-frequency 10 kHz spinal cord stimulation, low-frequency spinal cord stimulation, neuromodulation, lower back pain, lumbosacral radiculopathy, post-laminectomy syndrome, failed back surgery syndrome
Introduction
Spinal cord stimulation (SCS) is considered a safe and efficacious strategy for treating select chronic refractory neuropathic, ischemic and other pain syndromes.1,2 According to the International Neuromodulation Society, SCS improved the quality of life for thousands of patients nationwide for decades, with close to 35,000 new devices implanted across the world annually.2 Despite the level of success of these systems, the rate of failure to accomplish long-term pain coverage still remains as high as 30%.3 Loss or lack of efficacy represents the most frequent reason for neurostimulator explant.4 The more commonly reported side effects documented amongst patients with these conventional low-frequency (40–1200 Hz) devices (LF-SCS) are unpleasant paresthesia or dysesthesia.2–4
High frequency SCS at 10 kHz (10 kHz SCS) is known to produce no paresthesias and provides superior pain relief with a favorable safety and efficacy profile.5–8 This therapy, therefore, may be considered a viable salvage option to conventional SCS systems.9–11
Patients who do not reach treatment goals with LF-SCS may experience a significant improvement in pain scores after the SCS system is changed to a 10 kHz SCS device.
Methods
One hundred and twenty salvage replacements of traditional low-frequency SCS systems were completed from June 2015 to July of 2019 at the Carolinas Pain Institute (Winston-Salem, NC) and Department of Anesthesiology, University of Kansas (Kansas City, KS). Patients were selected for procedure based on failure of conventional SCS to provide optimal pain relief and the patient’s desire to improve their outcomes. Some had associated discomfort from paresthesias, but that was not the primary reason to replace the device in any of the studied patients. Following the Forsyth Medical Center IRB and University of Kansas IRB study approval, patient data were collected from 105 of our electronic medical records. The patient consent to review their medical records was not required by the IRBs and a waiver was received, as patient data confidentiality was preserved during data collection. The study was conducted in compliance with the Declaration of Helsinki. Fifteen patients had incomplete records, so they were excluded. The data collected from these records included patient identifier, demographics, diagnosis, and variables related to their traditional low-frequency SCS system prior to the salvage therapy which is summarized in Table 1. Those 105 patients were followed for more than a year after the salvage therapy and had complete data sets. Electronic medical records were analyzed to determine degree of improvement in pain scores, and use of opioids before and after salvage therapy occurred.
 |
Table 1 Data Items Extracted from Medical Records |
Statistical Analysis
Data were summarized using descriptive statistics for continuous variables. We used Student’s t-tests and the Mann–Whitney rank sum test to determine whether the Visual Analog Scale (VAS) pain scores attained following replacement with the 10 kHz SCS system were significantly different than prior to salvage therapy. A similar analysis was carried out to determine changes in daily opioid use before and after salvage therapy. We calculated percentage of the patients who maintained >50% pain relief following this change. All analyses were completed using the program Sigma Plot for Windows (version 14.0, Carry, New York, USA)
Procedures Completed
All patients received salvage replacement from an ineffective traditional SCS device to a 10 kHz SCS system, or they had a new 10 kHz SCS system placed after the traditional SCS system was previously removed. All patients received either a surgical procedure to replace an existing generator to one that can deliver 10 kHz SCS pulse frequency, or they received both, a new generator and one or two octapolar leads. There were only 5 traditional paddle leads implanted, the remainder were percutaneous. We salvaged 96 patients with 16 contact leads, 4 with 8 contact leads, and 5 with a 32 contact system. Almost all traditional systems explanted (103) were full systems from one of the three traditional SCS manufacturers, while two patients had a combination of leads and generators supplied by different manufacturers. Implants/exchanges were completed under sterile conditions with fluoroscopic guidance while patients were under deep sedation as per International Neuromodulation Society Guidelines and manufacturer's recommendations. Anterior-posterior and lateral views were used to confirm a final placement of the leads. All implant procedures were completed as a part of our regular clinical practice.
Results
The 105 patients obtained from the database review are described in Table 2.
 |
Table 2 Demographic Summary |
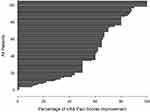
In addition to their primary diagnosis, patients had on average one or more sources of chronic pain (0–5; median 1). The majority of patients received their original, traditional SCS for a diagnosis of post-laminectomy syndrome (failed back surgery syndrome, FBSS) followed by complex regional pain syndrome (CRPS) type 1, peripheral neuropathy, neuralgia/CRPS type 2, chronic abdominal pain, neck and upper extremity pain, and degenerative disc disease (DDD) with low back pain. On average they used their traditional SCS for 4.66 ± 3.9 years (median 4 years). Average use of opioids at the last office visit before salvage replacement with 10 kHz SCS was 60.3 ± 77.2 mg in MSO4 equivalents (median 30). Details on patient disposition is shown in Figure 1. There was no correlation between age, gender, baseline opioid use (in MSO4 equivalents), or the amount of baseline pain, with pain relief achieved with the salvage therapy. The average Visual Analog Scale (VAS) score at the last clinical visit just before the salvage therapy was 8.3 ± 1.4 (median of 8) and decreased to 3.49 ± 2 (median of 4) at 1 month after the salvage, 3.65 ± 2.2 (median 4) six months after, 3.32±2 (median of 3) at 12 months and 3.36 ± 2 (median of 3) at the most recent patient visit (median of 2 years; range 1–4 years; Figure 2). At all of the follow up visits pain VAS was significantly lower than before the salvage therapy (p<0.001). In addition, there was no significant difference between VAS pain scores at 1, 6, or 12 months after the salvage and most recent patients visit, suggesting that the profound improvement in VAS pain scores was maintained long term. There were 85 out of 105 patients (81%) who improved with 50% or more pain relief (Figure 3) and 90% of the patients achieved at least 30% pain relief.
 |
Figure 1 Disposition of 120 consecutive patients who underwent change of their traditional SCS system to 10 kHz SCS. |
Opioid usage decreased from 60.3 ± 77.1 mg to 32.1 ± 44 mg MSO4 equivalents (p = 0.001) at 12 months after salvage (Figure 4).
Twenty-five patients used more than 90 mg MSO4 equivalents per day before salvage therapy. The average decrease of opioid use in that subgroup of patients was from 168.5 ± 89.7 to 77.8 ± 63.8 mg MSO4 equivalents. 10 patients (out of 25) decreased their overall opioid use per day to ≤ 90 mg MSO4.
Discussion
A pivotal pre-market approval trial (PMA) demonstrated superior improvements in pain relief, effective components of pain and functional outcomes with 10 kHz SCS compared to conventional 40–1200 Hz, paresthesia-based SCS with documented follow-up at one year,5 and extended follow-up at two years6 to demonstrate the durability of the outcomes. The large difference in effect size between the 10 kHz treatment group and the conventional control group suggests a different or additional mechanism of action(s) for 10 kHz stimulation.12 This is supported by multiple subsequent observational studies demonstrating clinical improvement with 10 kHz stimulation in patients who previously failed conventional SCS.9–11
A previously published case series demonstrated improvement with temporary lead placement and 10 kHz SCS in patients who failed previous traditional low frequency SCS implantation. However, this study presents a large case series where all patients underwent full SCS salvage replacement with 10 kHz SCS without a repeated trial.
In this study, only three patients showed no improvement and 90% sustained greater than 30% pain relief. Eighty-one percent of patients had more than 50% pain relief. To properly implement our salvage therapy and avoid an infection from externalized leads, we went directly to full explant of the traditional system and implant of the 10 kHz system during the same procedure. The high responder rate in this cohort suggests that trialing of 10 kHz for rescue purposes is unnecessary. There was no correlation between age, gender, amount of baseline opioid use, or amount of baseline pain with the degree of pain relief achieved after salvage with 10 kHz SCS therapy. A correlation may have been obscured by the variety of diagnosis for which the SCS system was used, additional sources of chronic pain (on average one more source of chronic pain per patient), and the wide standard deviation in opioid usage in the cohort.
We found that the decrease in opioid usage was not only profound after an effective 10 kHz SCS stimulation therapy was established, but also that such an effect could be maintained long term (Figure 4; 12 months follow-up), adding another powerful weapon in the war against opioid over usage in our country. Further, those patients with the highest opioid daily requirements (≥ 90 mg MSO4 per day) cut in half their daily opioid usage from 168.5 ± 89.7 to 77.8 ± 63.8 mg MSO4 equivalents, and 10 out of 25 decreased their usage below 90 mg of MSO4 equivalents. Although randomized prospective trials of 10 kHz SCS with reduction in opioid use at the primary outcome would be welcome to substantiate our data, other published studies on 10 kHz therapy have suggested the same.13,14
Previously, we attempted a similar therapeutic salvage tactic using the rate of 1000–1200 Hz and trialing various amplitudes in 105 consecutive patients who failed traditional, paresthesia-based SCS.15 Although initially we found a modest therapeutic response, we could not sustain significant pain relief at one year in most patients (98%) using such an approach.15 This study is in sharp contrast to our previous efforts at rescue and adds more evidence that frequencies higher than 5000 Hz may be required to provide additional pain relief as recently suggested in a randomized, sham controlled trial.16 Data were analyzed from 37 patients with back pain and lumbosacral radiculopathy without previous spinal surgery. Improvement in their pain scores was profound with 92% of this cohort achieving more than 50% pain relief. Although the outcomes were positive in both groups, comparing patients who underwent previous back surgery (listed as failed back and/or post-laminectomy syndrome patients) with those who had no previous spinal surgeries, the latter had the best outcomes with 10 kHz rescue for long-term pain relief. Such data are consistent with published European data showing profound relief of chronic back and leg pain when 10 kHz is used in patients with back pain who had no previous lumbar spinal surgery.8
To conclude, most of our patients (81%) who were considered failures of traditional SCS received >50% pain relief when rescued with 10 kHz SCS. In addition, clinical improvement resulted in the in the indirect benefit of decrease in opioid usage. SCS at 10 kHz should be considered a viable therapeutic option to rescue failed traditional SCS and provide in this population of patients long-term relief from chronic pain.
Acknowledgments
Authors acknowledge assistance in data collection by Dr. Tyler Concannon, MD and Dr. Tara Notarianni, DO.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
Drs Kapural and Sayed are paid consultants for Nevro Corp. This study was funded by an investigator initiated grant from Nevro to Drs Kapural and Sayed.
Dr Leonardo Kapural reports research grants from Nevro, Biotronik, Saluda Medical, Gimer and Neuros, personal fees from Nalu, Abbott and Medtronic, during the conduct of the study;and personal fees from Nevro, outside the submitted work.
Dr Dawood Sayed reports personal fees, consulting fees and sponsored research from Nevro, Abbott, Vertos,Medtronic, Flowonix, and Boston Scientific, and equity in SPR, PainTeq, and Vertos, outside the submitted work.
The authors report no other potential conflicts of interest for this work.
References
1. De La Cruz P, Fama C, Roth S, et al. Predictors of spinal cord stimulation success. Neuromodulation. 2015;18(7):599–602. doi:10.1111/ner.12325
2. Deer TR, Grider JS, Lamer TJ, et al. Mekhail NA systematic literature review of spine neurostimulation therapies for the treatment of pain. Pain Med. 2020;21(7):1421–1432. doi:10.1093/pm/pnz353
3. Dupré DA, Tomycz N, Whiting D, Oh M. Spinal cord stimulator explantation: motives for removal of surgically placed paddle systems. Pain Pract. 2018;18(4):500–504. doi:10.1111/papr.12639
4. Pope JE, Deer TR, Falowski S, et al. Multicenter retrospective study of neurostimulation with exit of therapy by explant. Neuromodulation. 2017;20(6):543–552. doi:10.1111/ner.12634
5. Kapural L, Yu C, Gliner B, et al.; Burgher AH for the SENZA Investigators. 10 kHz High frequency therapy is superior 1 to traditional low frequency spinal cord stimulation for the treatment of chronic back and leg pain: the SENZA-RCT randomized controlled trial. Anesthesiology. 2015;123:851–860. doi:10.1097/ALN.0000000000000774
6. Kapural L, Yu C, Gliner B, et al.; Burgher AH for the SENZA Investigators. Comparison of 10 kHz high frequency and traditional low frequency spinal cord stimulation for the treatment of chronic back and leg pain: 24-month results from a multicentre randomized controlled pivotal trial. Neurosurgery. 2016;79(5):667–677. doi:10.1227/NEU.0000000000001418
7. Van Buyten JP, Al-Kaisy A, Smet I, Palmisani S, Smith T. High-frequency spinal cord stimulation for the treatment of chronic back pain patients: results of a prospective multicenter European clinical study. Neuromodulation. 2013;16(1):59–65. doi:10.1111/ner.12006
8. Al-Kaisy A, Van Buyten JP, Kapural L, et al. Rotte A 10 kHz spinal cord stimulation for the treatment of non-surgical refractory back pain: subanalysis of pooled data from two prospective studies. Anaesthesia. 2020;75(6):775–784. doi:10.1111/anae.15036
9. Youn Y, Pilitsis JG. Successful use of high-frequency spinal cord stimulation following traditional treatment failure. Stereotact Funct Neurosurg. 2015;93:190–193. doi:10.1159/000380825
10. Ghosh PE, Gill JS, Simopoulos T. The evolving role of high-frequency SCS as salvage therapy in neuromodulation. Pain Pract. 2020;20(7):706–713. doi:10.1111/papr.12898
11. Gill JS, Asgerally A, Simopoulos T. High-frequency spinal cord stimulation at 10 kHz for the treatment of complex regional pain syndrome A case series of patients with or without previous SCS implant. Pain Pract. 2019;19(3):289–294. doi:10.1111/papr.12739
12. Lee KY, Bae C, Lee D, et al. Low-intensity, Kilohertz Frequency Spinal Cord Stimulation Differently Affects Excitatory and Inhibitory Neurons in the Rodent Superficial Dorsal Horn. Neuroscience. 2020;428:132–139. doi:10.1016/j.neuroscience.2019.12.031
13. Al-Kaisy A, Van Buyten JP, Carganillo R, et al. 10 kHz SCS therapy for chronic pain, effects on opioid usage: post hoc analysis data from two prospective studies. Sci Rep. 2019;9:11441. doi:10.1038/s41598-019-47792-3
14. Al-Kaisy A, Van Buyten JP, Amirdelfan K, et al. Opioid-sparing effects of 10 kHz spinal cord stimulation: a review of clinical evidence. Ann N Y Acad Sci. 2020;1462(1):53–64. doi:10.1111/nyas.14236
15. Kapural L, Harandi S. Long-term efficacy of 1-1.2 kHz subthreshold SCS following failed traditional SCS: a retrospective Case Series. Reg Anesth Pain Med. 2019;44:107–110. doi:10.1136/rapm-2018-000003
16. Al-Kaisy A, Palmisani S, Pang D, et al. Prospective, randomized, sham-control, double blind, crossover trial of subthreshold spinal cord stimulation at various Kilohertz frequencies in subjects suffering from failed back surgery syndrome (SCS frequency study). Neuromodulation. 2018;21(5):457–465. doi:10.1111/ner.12771
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.