Back to Journals » OncoTargets and Therapy » Volume 10
Response to crizotinib in a lung adenocarcinoma patient harboring a novel SLC34A2-ROS1 fusion variant
Authors Zhao Z, Song ZJ, Wang XW, Sun HF, Yang XM, Yuan Y, Yu P
Received 6 March 2017
Accepted for publication 13 June 2017
Published 21 August 2017 Volume 2017:10 Pages 4129—4133
DOI https://doi.org/10.2147/OTT.S136297
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Tohru Yamada
Video abstract presented by Zheng Zhao
Views: 1060
Zheng Zhao,1 Zhangjun Song,2 Xuwei Wang,3 Haifeng Sun,1 Xiaomin Yang,2 Yong Yuan,4 Pan Yu3
1Third Department of Medical Oncology, Shannxi Provincial Cancer Hospital, 2Breast Surgery Center, Shannxi Provincial Cancer Hospital, Xi’an, 3Marketing Department, Novogene Bioinformatics Institute, Beijing, 4Pathology Department, Shannxi Provincial Cancer Hospital, Xi’an, People’s Republic of China
Abstract: ROS1 fusion is a common genetic alteration in non-small-cell lung cancer. Crizotinib, an anaplastic lymphoma kinase inhibitor, shows efficacy in the treatment of lung cancer cases with ROS1 translocation. We report the response to crizotinib of a lung adenocarcinoma patient harboring a novel SLC34A2-ROS1 fusion variant, which was different from the two common SLC34A2-ROS1 fusion types reported in the literature. After crizotinib administration, overall recovery was good in this patient; the primary lesion was successfully treated, the lymph node metastases had disappeared, and the metabolism was normal.
Keywords: SLC34A2-ROS1 fusion, crizotinib, lung adenocarcinoma, next-generation sequencing
Introduction
ROS1 fusion, a common genetic alteration in non-small-cell lung cancer (NSCLC),1 is present in about 2.4% of lung adenocarcinoma cases. Several fusion types have been reported, including CD74, SLC34A2, EZR, LRIG3, SDC4, TPM3, FIG GOPC, CCDC6, KDEL R2 LIMA1, MSN, CLTC, TMEM106B, and TPD52L1.2–5 The application of reliable screening methods for gene rearrangement detection is key in identifying patients suitable for tumor-targeted therapy.5,6 To date, ROS1 fusions in NSCLC are most commonly diagnosed by florescence in situ hybridization (FISH) or amplification refractory mutation system-polymerase chain reaction (ARMS-PCR).6–10 However, ARMS-PCR and FISH are costly, less precise, and not suitable for detecting unknown significant mutations.11–16 Next-generation sequencing (NGS) is an optional method for identifying unknown genes with potential benefit.10,17–19 Crizotinib, a tyrosine kinase inhibitor targeting ALK and MET, was approved by the US Food and Drug Administration (FDA) on March 11, 2016, for the treatment of advanced metastatic ROS1-rearranged NSCLC. This report describes a patient successfully treated with crizotinib, in whom a novel SLC34A2-ROS1 fusion variant was identified. Written informed consent was obtained from the patient for the publication of this case report and any accompanying images. Approval from an ethics committee was not required due to this being a retrospective study and the information regarding the case being anonymous.
Case report
A 48-year-old female patient with back pain was admitted to the Shanxi Provincial Cancer Hospital; she was suspected with left breast cancer. Subsequently, lymph node metastasis of lung adenocarcinoma was considered in both sides of the neck and the left side of axillary lymph nodes after biopsy. She had 10 years of smoking history (1 pack/d) and no family history of genetic diseases. Biopsy samples were assessed for EGFR, ALK, and ROS1 gene mutations by ARMS-PCR (Amoy Diagnostics Co., Ltd, Xiamen, People’s Republic of China); however, all were negative. The patient received 2 cycles of chemotherapy (docetaxel 240 mg + cisplatin 240 mg). Because of progressive disease (PD), the treatment was discontinued on December 9, 2015. Positron emission tomography (PET)/computed tomographic (CT) imaging revealed that the tumor had developed multiple lymph node metastases. The chemotherapy regimen was replaced by 2 cycles of chemotherapy (pemetrexed 800 mg, intravenous [IV], d1; cisplatinum 20 mg IV, d1–5, bevacizumab 0.5 IV d1) and a combined 1 cycle of local radiotherapy (4,400 cGy/22f/5w). CT assessment showed stable disease in the patient, and palliative treatment was performed by pemetrexed plus cisplatin chemotherapy. However, the chemotherapeutic treatment was discontinued because of severe bone marrow suppression.
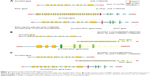
With no effective treatment at hand, biopsy samples were assessed using the NGS-483 gene panel (Novogene Bioinformatics Technology Co., Ltd, Beijing, People’s Republic of China). Biotinylated oligoprobes were designed along the no-repeat regions of 483 genes containing all the exons and some introns. The amplified exonic and intronic DNA was then sequenced on HiSeq X Ten sequencer (Illumina, San Diego, CA, USA) producing paired 150 bp reads. In the 483 genes, NGS revealed a new ROS1 fusion (SLC34A2-ROS1) and the TP53 p E132Q variation. Next, we assessed the causes of different detection results in the same samples. While analyzing ROS1 fusion sequence data, a novel SLC34A2-ROS1 fusion variant, different from the two common SLC34A2-ROS1 fusion types, was discovered (Figure 1). According to the sequencing data, the novel breakpoint was chr6:117653720, chr4:25678781. To verify the breakpoint, we respectively designed a primer pair based on the ROS1 gene’s breakpoint downstream and left to the SLC34A2 gene’s upstream breakpoint. Direct Sanger sequencing demonstrated that the 3′ untranslated region (3′UTR) of SLC34A2 in exon 13 was disrupted and inverted to connect a position of intronic_e32_e31 of ROS1 (Figure 2). The new breakpoint was not included in Catalogue Of Somatic Mutations In Cancer (COSMIC; http://cancer.sanger.ac.uk/cosmic) and was located in the SLC34A2 gene of the last transcription region (extra exon 13, as the 3′UTR region, was not involved in translation). Therefore, conventional detection methods cannot be used.
On March 11, 2016, the FDA formally approved crizotinib (Pfizer) for the treatment of patients with advanced (metastatic) NSCLC harboring ROS1 gene fusion. Would the patient with the novel SLC34A2-ROS1 fusion variant showed no response to crizotinib? We predicted the protein domain of the new fusion gene, and the ROS1 tyrosine kinase domain could not be destroyed.
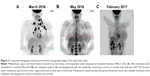
Combined with the above information, the female patient was started on treatment with crizotinib (250 mg, 2/d, d1–30) from March 24, 2016. After 2 weeks, the breast mass was reduced, and the patient reported relieved pain and improved quality of life. Two months later, according to PET/CT, most metastatic lymph nodes had disappeared, with the metabolism returning to normal. Eleven months later, the original irregular nodules in the basal segment of the right lung’s lower lobe were slightly narrowed, with reduced metabolism; the shape was uniform, with rough edges and no nodules or pleural adhesion. The lesion increase, which reflects the primary malignant lesion (pneumotype of the lower lobe of the lung), was slightly smaller and more active after treatment. The anterior mediastinum septum, the left lung, the upper and lower sides of the collarbone, and the left armpit had lymph node metastases. The lymph nodes of interstitial lesions violated the left vertebra of the neck; after treatment, the metastatic lymph nodes disappeared, with the metabolism returning to normal. In this case, the external pressure of lymph nodes, the left armpit, the cervical lymph and blood vessels, left upper extremities, left breast area edema, and swelling were relieved after treatment, and remained so. No new tumor metastases were found in other organs and bones in other parts of the body. The patient’s overall recovery was good; the primary lesion was relieved, the lymph node metastasis had disappeared, and the metabolism was normal (Figure 3).
Discussion
Crizotinib has been approved by FDA for the treatment of advanced metastatic NSCLC with ROS1 and ALK rearrangements.1,7,10,17 In the Phase I clinical trial of crizotinib, 50 NSCLC patients with ROS1 fusion were enrolled, including 49 cases detected by FISH and 1 by reverse transcriptase-polymerase chain reaction (RT-PCR). The overall response rate (ORR) was 72%, with a median duration of response (DOR) of 17.6 months and a median progression-free survival (PFS) of 19.2 months.3
The SLC34A2-ROS1 fusion variant is a common ROS1 fusion in NSCLC accounting for the second highest percentage.4,5 It is likely generated from an intra-chromosomal deletion and fusion, similar to EML4-ALK generation in NSCLC.1,8,13,15 NGS fortunately revealed a new ROS1 fusion (SLC34A2-ROS1) and TP53 p E132Q variation; ALK fusion mutation was not found in this sample. The novel SLC34A2-ROS1 fusion variant (3′UTR of SLC34A2 in exon 13 was disrupted and inverted to connect a position of intronic_e32_e31 of ROS1) differed from the two previously discovered SLC34A2-ROS1 fusion types (Figure 1). Based on sequencing results, the novel breakpoint was chr6:117653720, chr4:25678781. After the administration of crizotinib for 2 weeks, the breast mass was reduced, pain was relieved, and the quality of life improved. After 2 months, PET/CT indicated a progressive relief; 11 months later, overall recovery was good in this patient, with primary lesion successfully treated, no lymph node metastases, and normal metabolism. The patient also harbored a TP53 pE132Q variation. Would this also explain why crizotinib was effective in this patient? The tumor suppressor gene TP53 is the most frequently mutated in NSCLC. It encodes a 393-aa protein with three distinct domains. TP53p E132Q exists on the binding domain of TP53; however, it may not affect the function of proteins. In addition, this remains unclear as no relevant literature was found. These findings suggest that the novel SLC34A2-ROS1 fusion shows good response to crizotinib. ARMS-PCR is a fast and sensitive method for evaluating the expression of known ROS1 fusion variants,14,16 for which specific primers have been designed. In a previous study, the sensitivity and specificity of ARMS-PCR for detecting ROS1 fusion were 100% and 85.1%, respectively.10 There are two possible reasons why we failed to detect the ROS1 fusion in the patient’s sample: 1) only a small population of cancer cells might be carrying the ROS1 fusion and 2) this fusion might occur at the transcriptional level, with no translation. In this report, the rare ROS1 variants were not detected by ARMS-PCR, since ARMS-PCR is based on cDNA after mRNA transcription. The new breakpoint was not included in COSMIC and was located in the last transcription region of the SLC34A2 gene; therefore, it could not be detected by ARMS-PCR. In the 483 genes, NGS revealed the new ROS1 fusion (SLC34A2-ROS1); Direct Sanger sequencing demonstrated that the 3′UTR of SLC34A2 in exon 13 was disrupted and inverted to connect a position of intronic_e32_e31 of ROS1. For further confirmation, a FISH assay (Guangzhou Anpingping [LBP] Pharmaceutical Technology Co., Ltd) was performed to check for this fusion variant; however, the results were negative. Also, we analyzed the reasons for the difference of detection result between the two methods. The gene fusion process is very complex; it is possible that in the upstream area ROS1 missing part of the area, by parts of SLC34A2 gene insert this part again, without affecting the other areas in the FISH. The red probe can also be combined with a green probe, which is still negative when examined. A study shows that the same phenomenon exists in the ALK fusion.20 There is a complete possibility of a rearrangement is no separation of the red and green probes. NGS is an optional method for the detection of all forms of gene variation. Thus, the multi-platform technology of detection can maximally prolong patient survival. To monitor the expression of this fusion from diagnosis throughout the treatment, liquid biopsy-circulating tumor DNA was used with the NGS 483 gene panel. Circulating tumor DNA samples of the patient, in July 2016 and January 2017, respectively, were assessed by the NGS 483 gene panel; interestingly, the SLC34A2-ROS1 fusion variant had disappeared, in agreement with the PET/CT data.
Disclosure
Xuwei Wang and Pan Yu are employees of Novogene Bioinformatics Institute. The authors report no other conflicts of interest in this work.
References
Takeuchi K, Soda M, Togashi Y, et al. RET, ROS1 and ALK fusions in lung cancer. Nat Med. 2012;18(3):378–381. | ||
Bergethon K, Shaw AT, Ou SHI, et al. ROS1 rearrangements define a unique molecular class of lung cancers. J Clin Oncol. 2012;30(8):863–870. | ||
Shaw AT, Ou SH, Bang YJ, et al. Crizotinib in ROS1-rearranged non–small-cell lung cancer. N Engl J Med. 1971;372(7):1963–1971. | ||
Awad MM, Katayama R, Mctigue M, et al. Acquired resistance to crizotinib from a mutation in CD74–ROS1. N Engl J Med. 2013;368(25):2395–2401. | ||
Rimkunas VM, Crosby KE, Li D, et al. Analysis of receptor tyrosine kinase ROS1-positive tumors in non-small cell lung cancer: identification of a FIG-ROS1 fusion. Clin Cancer Res. 2012;18(16):4449–4457. | ||
Lee J, Lee SE, Kang SY, et al. Identification of ROS1 rearrangement in gastric adenocarcinoma. Cancer. 2013;119(9):1627–1635. | ||
Yoshida A, Kohno T, Tsuta K, et al. ROS1-rearranged lung cancer: a clinicopathologic and molecular study of 15 surgical cases. Am J Surg Pathol. 2013;37(4):554–562. | ||
Davies KD, Le AT, Theodoro MF, et al. Identifying and targeting ROS1 gene fusions in non-small cell lung cancer. Clin Cancer Res. 2012;18(17):4570–4579. | ||
Yoshida A, Tsuta K, Wakai S, et al. Immunohistochemical detection of ROS1 is useful for identifying ROS1 rearrangements in lung cancers. Mod Pathol. 2014;27(5):711–720. | ||
Shan L, Lian F, Guo L, et al. Detection of ROS1 gene rearrangement in lung adenocarcinoma: comparison of IHC, FISH and real-time RT-PCR. PLoS One. 2014;10(3):e0120422. | ||
Suehara Y, Arcila M, Wang L, et al. Identification of KIF5B-RET and GOPC-ROS1 fusions in lung adenocarcinomas through a comprehensive mRNA-based screen for tyrosine kinase fusions. Clin Cancer Res. 2012;18(24):6599–6608. | ||
Cai W, Li X, Su C, et al. ROS1 fusions in Chinese patients with non-small-cell lung cancer. Ann Oncol. 2013;24(7):1822–1827. | ||
Peraldo NC, Cavalloni G, Balsamo A, et al. Screening for the FIG-ROS1 fusion in biliary tract carcinomas by nested PCR. Genes Chromosomes Cancer. 2014;53(12):1033–1040. | ||
Pikor LA, Ramnarine VR, Lam S, Lam WL. Genetic alterations defining NSCLC subtypes and their therapeutic implications. Lung Cancer. 2013;82(2):179–189. | ||
Yamaguchi N, Lucenaaraujo AR, Nakayama S, et al. Dual ALK and EGFR inhibition targets a mechanism of acquired resistance to the tyrosine kinase inhibitor crizotinib in ALK rearranged lung cancer. Lung Cancer. 2014;83(1):37–43. | ||
Pan Y, Zhang Y, Li Y, et al. ALK, ROS1 and RET fusions in 1139 lung adenocarcinomas: a comprehensive study of common and fusion pattern-specific clinicopathologic, histologic and cytologic features. Lung Cancer. 2014;84(2):121–126. | ||
Lukas RV, Hasan Y, Nicholas MK, Salgia R. ROS1 rearranged non-small cell lung cancer brain metastases respond to low dose radiotherapy. J Clin Neurosci. 2015;22(12):1978–1979. | ||
Le X, Freed JA, Vanderlaan PA, et al. Detection of crizotinib-sensitive lung adenocarcinomas with MET, ALK, and ROS1 genomic alterations via comprehensive genomic profiling. Clin Lung Cancer. 2015;16(5):e105–e109. | ||
Boyle TA, Masago K, Ellison KE, Yatabe Y, Hirsch FR. ROS1 immunohistochemistry among major genotypes of non-small-cell lung cancer. Clin Lung Cancer. 2015;16(2):106–111. | ||
Ali SM, Hensing T, Schrock AB, et al. Comprehensive genomic profiling identifies a subset of crizotinib-responsive ALK-rearranged non-small cell lung cancer not detected by fluorescence in situ hybridization. Oncologist. 2016;21(6):762–770. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.