Back to Journals » International Journal of General Medicine » Volume 13
Relationship Between Helicobacter pylori Infection, Serum Vitamin D3 Level and Spontaneous Abortion
Authors Ibrahim HA
Received 24 February 2020
Accepted for publication 2 July 2020
Published 28 July 2020 Volume 2020:13 Pages 469—476
DOI https://doi.org/10.2147/IJGM.S251075
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Hadeel Abdulelah Ibrahim
College of Nursing, University of Sulaimani, Kurdistan Region, Republic of Iraq
Correspondence: Hadeel Abdulelah Ibrahim
College of Nursing, University of Sulaimani, Kurdistan Region, Republic of Iraq
Tel +964(0)7703914402
Email [email protected]
Background: The effects of vitamin D3 (VD3) on pregnancy outcomes remain obscure. Thus, this study aims to investigate the relationship between maternal H. pylori infection, low VD3 level, and spontaneous abortion.
Methods: This research is conducted in Shahid Ali Qader Consultant Clinic in Sulaimaniyah city in which 100 women with a history of abortion and 100 women with no history of miscarriage were included. Serum detection of anti-H. pyloriIgG, IgA, and VD3 were done using enzyme-linked immunosorbent assay (ELISA) technique.
Results: The mean of VD3, H. pylori IgG, and IgAin patients and control group cases was determined and analyzed statistically.
Conclusion: H. pylori infection and VD3 play a significant role in early pregnancy loss. Blood group A and O are more prone to spontaneous abortion.
Keywords: miscarriage, bacterial infection, vitamin deficiency, antibody detection, ABO grouping
Introduction
The infection with Helicobacter pylori considered one of the most dreaded diseases in all age groups and both sexes, especially in undeveloped countries.1,2 The bacterium attacks the gastric wall and leads to severe acute infection or sometimes asymptomatic chronic infection. Recent researches confirmed that H. pylori infection is a leading cause of peptic ulcers and gastric carcinoma, especially in adult mature peoples.2 Additionally, studies reported a direct correlation between H. pylori infection and several extra-gastric diseases.3
Spontaneous abortion is the loss of a fetus before 20th weeks of pregnancy that mainly occurs due to fatal genetic problems, or other reasons (non-chromosomal reasons) such as infections and immunological dysfunction. There is evidence indicates that the local inflammation such as H. pylori infection may not be confined to the digestive tract, but it can spread to involve the extra- gastrointestinal tract, which may impact reproductive capacity negatively. VD3 deficiency and/or insufficiency could enhance the danger of several chronic sicknesses including immunological dysfunction.4,5 H. pylori infection is associated with several pathologies during pregnancy, such as pre-eclampsia and recurrent intrauterine death that is mainly characterized by placentation failure that leads to miscarriage cases.6
Vitamin D or cholecalciferol is an important hormone that has a potential effect on bone metabolism and mineral homeostasis.7,8 It has been found that the increased risk of first-trimester miscarriage is directly associated with low concentrations of VD3.9 However, the impact of VD3 on pregnancy outcomes remains unclear. Therefore, this study aims to find a relationship between maternal H. pylori infection, low VD3 level, and spontaneous abortion.
Methodology
This research is a case–control study, conducted in Shahid Ali Qader Consultant Clinic in Sulaimaniyah city for a period of 10 months starting from January 2019 to October 2019. In this study, 100 women were involved aged between 19 and 49 years old with 5 weeks to 20 weeks of pregnancy loss. Simultaneously, 100 women who have no history of miscarriage and not pregnant during data collection with previous normal delivery were included as a control group. Demographic data were collected including age, parity, number of abortions, dyspepsia, time of dyspepsia, and blood groups. Blood from all women of both groups was collected for the detection of anti-H. pylori IgG, IgA (1425-300 H. pylori IgG, 1625-300 H. pylori IgA, Germany), and VD3 (Bioactiva Diagnostica, BD200BA 25(OH) Vitamin D Kit, Germany) using enzyme-linked immune-sorbent assay (ELISA) technique, using Monobind Inc., USA. Both anti-H. pyloriIgG and IgA antibodies were previously endorsed in a huge series of H. pylori-positive and –negative individuals. Virtually, the existence of H. pylori-specific IgG antibody is the only biomarker for chronic infection with this micro-organism; thus, it is not a sign of acquisition of the infection, whereas specific IgA antibody is a more specific biomarker for a recently acquired infection with H. pylori.10
The normal value of H. pylori IgG is < 8.0 AU/mL and for IgA is < 1.2 Ndx. While VD3 levels are classified into 3 major groups according to the classification of the Institute of Medicine4 as follows: sufficient > 30 ng/mL; insufficient ranged between 20 and 30 ng/mL; and deficient < 20 ng/mL.
Written informed consent was obtained from the patients prior to enrollment in the study. All experiments were performed in accordance with the Declaration of Helsinki and approved by the College of Nursing, University of Sulaimani, Iraq.
Inclusion Criteria
Any women attended Shahid Ali Qader Consultant Clinic in Sulaimaniyah city with a spontaneous loss of a fetus from 5th to 20th weeks of pregnancy.
Exclusion Criteria
Any women with medical disorders such as diabetes, hypertension, bronchial asthma, chronic renal, liver or endocrine disorder, induced or septic abortion, history of trauma during the current pregnancy, uterine and cervical congenital anomalies, received medications that influence bone metabolism and VD3 and calcium intake during the 4th weeks before assessment. Also, blood diseases such as leukemia or thalassemia, malignant, or bone disease were excluded from this study.
Statistical Analysis
All data were analyzed using Excel and SPSS (Version 24) program. The statistical significance of the difference in mean between groups (cases and control) was assessed using an independent sample t-test while comparing several groups (more than 2 groups) determined using ANOVA test. Chi-square test was used to find an association between different qualitative variables as between maternal age groups or positivity of IgG and cases and control. P value considered a significant difference when (p ≤ 0.05) and highly significant differences when (p ≤ 0.01).
Results
The mean age of the examined subjects that ranged from 19 to 49 years old is 31.98 ± 6.83 and 35.01 ± 8.09for both cases and control groups, respectively, without significant difference (p > 0.05) (Table 1). The mean VD3 for both cases and controls is 9.72 ± 6.22 and 15.77 ± 9.06 ng/mL, respectively, with a highly significant difference (p ≤ 0.001) (Figure 1). A highly significant difference (p < 0.01) was noticed in the mean of H. pylori IgG and IgA between cases (9.44 ± 4.44 vs 6.19 ± 2.37) and control (1.09 ± 0.41 vs 0.54 ± 0.18) groups, respectively (Table 1).
 |
Figure 1 Shows CI % of vitamin D3 (ng/mL) for both cases and control groups which is highly significant (p ≤ 0.001) when compared. |
 |
Table 1 Age, Vitamin D3, and H. Pylori Antibody Levels of the Participants |
In this study, no significant difference was demonstrated between the age groups in both cases and control (p > 0.05), but the age of most women complained from abortion ranged between 30 and 39 years old. Out of 200 women, 52% and 17% were positive for H. pylori infection in cases and control groups, respectively, with a highly significant difference (p < 0.01) (Table 2).
 |
Table 2 Age distribution of the Study Population with the Percentage of H. Pylori Infection |
Additionally, the author revealed that 70% of women had an abortion at a gestational period of 5–10 weeks followed by 11–15 weeks (25%), then 16–20 weeks (5%) (Figure 2). On the other hand, it is observed that 67% of women had the 1st miscarriage, then 18% had 2nd abortion and 15% of women had more than 2 abortions (Figure 3).
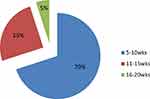 |
Figure 2 Shows the gestational period at the time of abortion in women of the case group (weeks). |
 |
Figure 3 Shows the percentage of abortions in examined subjects. |
The risk associated with H. pylori infection (IgG and IgA) such as age, blood group, dyspepsia, the number of abortions is shown in Table 3 in which no significant difference was demonstrated between the age groups and the H. pylori IgG and IgA levels (p > 0.05), while there is an increment in the H. pylori IgG level with increasing age and the highest level being found among the 40–49 years old group.
 |
Table 3 H. Pylori (IgG and IgA) Level with Selected Risk Factors Among Cases |
Furthermore, the author demonstrated that a higher mean of H. pylori IgG and IgA levels had blood group type A, followed by type O, then other blood groups with a highly significant difference (p < 0.01).Among, examined subjects, only 19 females were complained from dyspepsia more than 6 months with positive H. pylori infection (IgA) with significant differences (p < 0.01), while there is no significant relationship between female complained from dyspepsia > 6 months and H. pylori infection (IgG).
Simultaneously, there are highly significant differences (p < 0.01) between H. pylori infection (IgG and IgA) and many abortions. The higher mean of H. pylori IgG level (11.93 ± 5.44) noticed with more than 2 abortions and a higher mean of H. pylori IgA level (1.46 ± 0.50) noticed with 1 abortion only (Table 3).
Moreover, there are low serum VD3 in both control and case groups while there are no changes in the level of VD3 concerning age (p > 0.05). Besides, there was a significant difference (p < 0.05) in the mean of VD3 concentration between H. pylori-positive (9.91 ± 5.43 and 16.97 ± 5.2) and H. pylori-negative (6.91 ± 4.17 and 11.35 ± 7.28) in both control and cases, respectively (Table 4).
 |
Table 4 Vitamin D3 Level Among Control and Case Groups Concerning Age Distribution and H. Pylori Infection |
Finally, VD3 deficiency in women complained abortion is noticed in blood groups of types A and O (7.98 ± 3.36 and 7.63 ± 5.11), respectively, with a highly significant difference (p < 0.01). Also, VD3 deficiency revealed in women that complained of dyspepsia but without significant difference (p = 0.02). Lastly, VD3 deficiency was noticed more in women with more than 2 abortions (8.6 7 ± 5.97) than a woman with 2 abortions and 1 abortion (9.25 ± 6.56, 9.73 ± 6.10), respectively (Table 5).
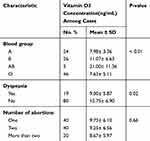 |
Table 5 Vitamin D3 Level with Selected Risk Factors Among Cases |
Discussion
To our knowledge, this is the first study investigating the relation of VD3 level and the existence of H. pylori with recurrent abortion in Iraq. The parental age, genetic, hormonal, immunological, and environmental factors are considered as the main causes of most miscarriages in humans. Instantly, it is confirmed that maternal age is the strongest known risk factor;11–13 for this reason, in this study, the population (patients and controls) were comparable and no significant difference between them excludes the age factor.
In the current study, a highly significant difference was noticed in the mean of H. pylori IgG and IgA between cases and control groups which is near to other studies revealed a statistically significant difference between cases and controls.14
Miscarriage is the most common problem in pregnancy, in which the major mechanisms responsible for pregnancy loss are not always clear. Hence, some efforts are being done to determine new factors like chronic infection with H. Pylori.15,16
Researchers suggested that H. pylori infection may have a role in the pathogenesis of different pregnancy-related ailments through various mechanisms such as pro-inflammatory cytokines release, oxidative stress, pre-eclampsia, and cross-reaction between specific anti-H. pylori antibodies and antigens localized in placental tissues. In most cases, H. pylori infection acquired before gestation, but it is extensively assumed that hormonal and immunological alteration arising during pregnancy could activate latent H. pylori with a negative impact not only on maternal health.17
In agreement with Shalabi et al, it was found that 52% of abortion cases are H. pylori seropositive, compared to 17% in control groups that are without a history of previous abortion, with a highly significant difference.14 H. pylori infection may appear as an asymptomatic syndrome and if left untreated, it progresses to a gastric disease especially during pregnancy. In opposite to our result, Golmahammadlou et al reported that there is no relation between H. pylori infection and miscarriage.18
Also, many studies have reported a high prevalence of H. pylori infection among healthy and non-healthy individuals worldwide.19,20
Additionally, the author reported that 70% of women had an abortion at the gestational period between 5 and 10 weeks. This finding is not agreed with Shalabi et al14 outcome. Also, it was revealed that 67% of women had an abortion in the first miscarriage; this finding is in line with Kirchner et al19 that concludes H. pylori infection associated with first miscarriage. On the other hand, Cardaropoli et al reported significantly higher H. pylori seropositive women with a miscarriage compared to controls, while the existence of maternal serum antibodies against H. pylori not occurred with repeated abortion.21 These findings suggest a relationship between H. pylori infection and implantation/placentation failure, possibly due to a cross-reaction between antibodies against H. pylori and placental tissue.6
No significant association between the age groups and the H. pylori (IgG and IgA) level was found, while there is an increment in the H. pylori IgG level with increasing age especially more than 40 years old. This finding is inconsistent with Shelbi et al who observed a statistically significant link between the increase in age and H. pylori infection.14
Moreover, the author showed a strong correlation between blood group types A and O with H. pylori infection (IgG and IgA) which is consistent with other studies22,23 in which they noticed that blood group type O is associated with increased risk of H. pylori infection compared to non-O blood group. The H antigen expressed in the duodenal mucosa by using type II oligosaccharide precursor and this antigen is not adapted to A or B antigens in blood group type O, which points to the fact that there is a positive correlation between this blood group and H. pylori infection.24,25 Additionally, the author noticed that blood group type B and AB patients were less prone to H. pylori infection and this finding is supported by previously conducted researches.26,27 While Ayalew et al28 showed no correlation between ABO blood group and H. pylori infection.
In this study, the author observed a statistically significant link between dyspepsia in patients with more than 6 months of pregnancy that has been diagnosed with positive H. pylori infection (IgA), but there is no link between female complained from dyspepsia > 6 months with H. pylori infection (IgG). These findings may be explained by local accumulation of CD8+ T cells, reducing CD4+ T cells, and augmented macrophages around the duodenal crypts persevered for months after the acute infectious, signifying impaired cessation of the inflammatory response even after sufficient removal of the infecting pathogen.29 H. pylori do not appear to invade the gastric mucosa, but rather release various toxins that ultimately exert tissue damage; among these are mucinase, phospholipase, neutrophil-activating protein A, heat shock protein 60, cytotoxin associated gene A protein (CagA), and vacuolating cytotoxin A (VacA).
The higher mean of H. pylori IgG level is reported with several abortions more than 2, and a higher mean of H. pylori IgA level was noticed with one abortion in this study. The IgG test is widely used in epidemiological studies and not affected by local changes in the stomach that could lead to false-negatives, this test cannot distinguish between current and past infections because H. pylori IgG persist even after the disappearance of this bacterium,30 thus the mucosal CagA-specific IgA antibodies are produced during the acute phase of gastric inflammation. The relevance of IgA testing remains controversial, and both IgA and IgG serum antibodies persist even if the H. pylori infection is eradicated. The role of antibody testing in differentiating active H. pylori infection from past infection and/or completion of therapy is therefore limited. Seropositivity to CagA has been broadly used to detect infections with virulent H. pylori strains.10,30,31
In this study, the mean VD3 among cases was more deficient with a highly significant difference than controls. Our result is supported by Hou et al who reported that serum VD3 levels are higher in women with normal pregnancies than in those with first trimester miscarriages, suggesting that VD3 deficiency is associated with pregnancy loss.32
VD3 plays an important immunological role at the fetal–maternal interface, as its role in the pathogenesis of recurrent spontaneous abortion has also begun to attract attention. It is the best indicator of nutritional status in clinical practice because of its long half-life and relative stability.4 A recent study conducted by Li et al reported that VD3 levels in the decidua are associated with inflammatory cytokine production, suggesting that VD3 may play a role in the etiology of recurrent spontaneous abortion.33
Our study agrees with Takaoka et al that found low serum VD3 levels among reproductive age in Japanese women,34 in addition to Masoumpour et al who observed that serum level of vitamin D did not decline with age.35 In contrast to these results, Nasri and Ardalan, and Bischof et al found a positive association of ages with VD3 level.36,37
Yang et al38 concluded that VD3 was a protective factor in H. pylori infection. Moreover, VD3 can improve the success rate of H. pylori eradication that supports our finding since were no changes in the level of VD3 about age (p > 0.05), but there was a significant difference in the mean of VD3 concentration between H. pylori-positive and H. pylori-negative in both groups (control and cases), in addition to other studies agree with our results.39–41 VD3 plays an important role in gastric mucosa homeostasis and host protection from H. pylori infection.42 VD3 may reduce the risk of infection through multiple mechanisms; by increasing innate immunity by modulating the production of AMPs and cytokine response. Moreover, VD3 helps in support and elevate the activity of monocytes and macrophages, that way contributing to a powerful systemic antimicrobial effect.43,44 VD3 is also known to regulate the expression of AMPs cathelicidin and β-defensin, which kill the bacteria. Although the effect of cathelicidin has been demonstrated only in macrophages infected with Mycobacterium tuberculosis, antibacterial action against Gram-negative and Gram-positive bacteria has also been reported. In the VD3-deficient state, the infected macrophage is unable to produce sufficient 1,25-(OH)D2 to upregulate the production of cathelicidin and β-defensin, thus rendering them unable to kill the H. pylori strains. Emerging evidence suggests that VD3 might be a good prophylactic and possibly therapeutic antimicrobial agent for the control and eradication of H. pylori.45,46
Highly significant associations between VD3 deficiency in women complained from abortion and blood group types A and O which in line with Al-Fartosi who concluded that a clear increase in several individuals for blood group A in-patients with repeated abortion, and this factor may need to consider in future investigations.47
Conclusion
The author concluded that H. pylori infection and VD3 play a significant role in early pregnancy loss, as well as blood group, type A and type O, which are more prone to spontaneous abortion in infected individuals. Also, VD3 levels in all women were very low, thus suggesting giving vitamin D to pregnant women according to the instructions of gynecologists.
Acknowledgment
The author appreciates the University of Sulaimani and Shahid Ali Qader Consultant clinic for providing the facilities and services for this research.
Disclosure
The author reports no conflicts of interest in this work.
References
1. Zullo A, Hassan C, De Francesco VD, et al. Helicobacter pylori and functional dyspepsia: an unsolved issue? World J Gastroenterol. 2014;20(27):8957–8963. doi:10.3748/wjg.v20.i27.8957
2. Suerbaum S, Michetti P. Helicobacter pylori infection. N Engl J Med. 2002;347(15):1175–1186. doi:10.1056/NEJMra020542
3. Banić M, Franceschi F, Babić Z, Gasbarrini A. Extragastric manifestations of Helicobacter pylori infection. Helicobacter. 2012;1(17 Suppl):49–55. doi:10.1111/j.1523-5378.2012.00983.x
4. Ammon Avalos L, Galindo C, Li DK. A systematic review to calculate background miscarriage rates using life table analysis. Birth Defects Res a Clin Mol Teratol. 2012;94(6):417–423. doi:10.1002/bdra.23014
5. Rossen LM, Ahrens KA, Branum AM. Trends in risk of pregnancy loss among US women, 1990–2011. Paediatr Perinat Epidemiol. 2018;32(1):19–29. doi:10.1111/ppe.12417
6. Franceschi F, Di Simone N, D’Ippolito S, et al. Antibodies anti-CagA cross-react with trophoblast cells: a risk factor for pre-eclampsia? Helicobacter. 2012;17(6):426–434. doi:10.1111/j.1523-5378.2012.00966.x
7. Tamblyn JA, Hewison M, Wagner CL, et al. Immunological role of vitamin D at the maternal-fetal interface. J Endocrinol. 2015;224(3):107–121. doi:10.1530/JOE-14-0642
8. Holick MF. Vitamin D deficiency. Ned Tijdschr Geneeskd. 2006;150:1315–1316.
9. Andersen LB, Jørgensen JS, Jensen TK, et al. Vitamin D insufficiency is associated with increased risk of first-trimester miscarriage in the odense child cohort. Am J Clin Nutr. 2015;102(3):633–638. doi:10.3945/ajcn.114.103655
10. Lanciers S, Despinasse B, Mehta DI, Blecker U. Increased susceptibility to Helicobacter pylori infection in pregnancy. Infect Dis Obstet Gynecol. 1999;7(4):195–198. doi:10.1002/(SICI)1098-0997(1999)7:4<195::AID-IDOG6>3.0.CO;2-R
11. Agenor A, Bhattacharya S. Infertility and miscarriage: common pathways in manifestation and management. Womens Health. 2015;11(4):527–541. doi:10.2217/whe.15.19
12. Garrido-Gimenez C, Alijotas-Reig J. Recurrent miscarriage: causes, evaluation and management. Postgrad Med J. 2015;91(1073):151–162. doi:10.1136/postgradmedj-2014-132672
13. Wilcox AJ, Morken NH, Weinberg CR, Håberg SE. Role of maternal age and pregnancy history in the risk of miscarriage: a prospective register-based study. Br Med J. 2019;364:l869. doi:10.1136/bmj.l42
14. Shalabi HM, Ali EH, Kotb Mohammed HM. Relationship between H. pylori and early pregnancy loss. Al-Azhar Assiut Med J. 2015;13(3):81.
15. Hajishafiha M, Ghasemi-Rad M, Memari A, et al. Effect of Helicobacter pylori infection on pregnancy rates and early pregnancy loss after intracytoplasmic sperm injection.. Int J Womens Health. 2011;3:329–335. doi:10.2147/IJWH.S24424
16. Malek M, Ghorbani R, Nameni M, et al. Association between Helicobacter pylori infection and first miscarriage. Koomosh. 2007;8(4):229–232.
17. Cardaropoli S, Rolfo A, Todros T. Helicobacter pylori and pregnancy-related disorders. World J Gastroenterol. 2014;20(3):654–664. doi:10.3748/wjg.v20.i3.654
18. Golmahammadlou S, Hagishafiha M, Karjooyan T, et al. Relationship between Helicobacter pylori infection and spontaneous abortion. Tehran Univ Med J. 2015;73(4):289–296.
19. Kirchner GI, Beil W, Bleck JS, Manns MP, Wagner S. Prevalence of Helicobacter pylori and occurrence of gastroduodenal lesions in patients with liver cirrhosis. Int J Clin Exp Med. 2011;4(1):26–31.
20. Al-Akwaa AM. Prevalence of Helicobacter pylori infection in a group of morbidly obese Saudi patients undergoing bariatric surgery: a preliminary report. Saudi J Gastroenterol. 2010;16(4):264–267. doi:10.4103/1319-3767.70610
21. Cardaropoli S, Piazzese A, Piccoli E, et al. Is Helicobacter pylori infection a risk factor for miscarriage? Placenta. 2013;34(9):A37–A38. doi:10.1016/j.placenta.2013.06.112
22. Valliani A, Khan F, Chagani B, et al. Factors associated with Helicobacter pylori infection, results from a developing country Pakistan. Asian Pac J Cancer Prev. 2013;14(1):53–56. doi:10.7314/APJCP.2013.14.1.53
23. Chakrani Z, Robinson K, Tay B. Association between ABO blood groups and Helicobacter pylori infection: a meta-analysis. Sci Rep. 2018;8(1):1. doi:10.1038/s41598-018-36006-x
24. Zhang L, Ren JW, Wong CCM, et al. Effects of cigarette smoke and its active components on ulcer formation and healing in the gastrointestinal mucosa. Curr Med Chem. 2012;19(1):63–69. doi:10.2174/092986712803413926
25. Zhong M, Zhang H, Reilly P, et al. ABO blood group as a model for platelet glycan modification in arterial thrombosis. Am Heart J. 2015;35:1570–1578.
26. Kanbay M, Gur G, Arslan H, et al. The relationship of ABO blood group, age, gender, smoking, and Helicobacter pylori infection. Dig Dis Sci. 2005;50(7):1214–1217. doi:10.1007/s10620-005-2762-y
27. Jaff MS. The relation between ABO blood groups and Helicobacter pylori infection in symptomatic patients. Clin Exp Gastroenterol. 2011;4:221–226. doi:10.2147/CEG.S23019
28. Ayalew F, Tilahun B, Taye B. Performance evaluation of laboratory professionals on malaria microscopy in Hawassa Town, Southern Ethiopia. BMC Res Notes. 2014;7(1):839. doi:10.1186/1756-0500-7-839
29. Marshall JK, Thabane M, Garg AX, et al. Walkerton health study investigators. Eight-year prognosis of postinfectious irritable bowel syndrome following waterborne bacterial dysentery. Gut. 2010;59(5):605–611. doi:10.1136/gut.2009.202234
30. Azevedo NF, Huntington J, Goodman KJ. The epidemiology of Helicobacter pylori and public health implications. Helicobacter. 2009;14(1):1–7. doi:10.1111/j.1523-5378.2009.00703.x
31. Bhat N, Gaensbauer J, Peek RM, et al. Local and systemic immune and inflammatory responses to Helicobacter pylori strains. Clin Diagn Lab Immunol. 2005;12(12):1393–1400. doi:10.1128/CDLI.12.12.1393-1400.2005
32. Hou W, Yan XT, Bai CM, et al. Decreased serum vitamin D levels in early spontaneous pregnancy loss. Eur J Clin Nutr. 2016;70(9):1004–1008. doi:10.1038/ejcn.2016.83
33. Li N, Wu HM, Hang F, et al. Women with recurrent spontaneous abortion have decreased 25(OH) vitamin D and VDR at the fetal-maternal interface. Braz J Med Biol Res. 2017;50(11):e6527. doi:10.1590/1414-431x20176527
34. Takaoka N, Nagao M, Umezawa M, et al. Distribution of serum 25-hydroxyvitamin D levels of reproductive age Japanese women. Nihon Koshu Eisei Zasshi. 2017;64(3):133–142. doi:10.11236/jph.64.3_133
35. Masoompour SM, Sadegholvaad A, Larijani B, Ranjbar Omrani G. Effects of age and renal function on vitamin D status in men. Arch Iran Med. 2008;11(4):377–381.
36. Nasri H, Ardalan MR. Association of serum vitamin D level with age in individuals with normal renal function. J Nephropharmacol. 2012;1(1):7–9.
37. Bischof MG, Heinze G, Vierhapper H. Vitamin D status and its relation to age and body mass index. Horm Res. 2006;66(5):211–215. doi:10.1159/000094932
38. Yang L, He X, Li L, Lu C. Effect of vitamin D on Helicobacter pylori infection and eradication: a meta-analysis. Helicobacter. 2019;24(5):e12655. doi:10.1111/hel.12655
39. MutSurmeli D, Surmeli ZG, Bahsi R, et al. Vitamin D deficiency and risk of Helicobacter pylori infection in older adults: a cross-sectional study. Aging Clin Exp Res. 2019;31(7):985–991. doi:10.1007/s40520-018-1039-1
40. Chen LW, Chien CY, Hsieh CW, et al. The associations between Helicobacter pylori infection, serum vitamin D, and metabolic syndrome: a community-based study. Medicine. 2016;95(18):e3616. doi:10.1097/MD.0000000000003616
41. Yildirim O, Yildirim T, Seckin Y, et al. The influence of vitamin D deficiency on eradication rates of Helicobacter pylori. Adv Clin Exp Med. 2017;26(9):1377–1381. doi:10.17219/acem/65430
42. Guo L, Chen W, Zhu H, et al. Helicobacter pylori induce increased expression of the vitamin d receptor in immune responses. Helicobacter. 2014;19(1):37–47. doi:10.1111/hel.12102
43. Youssef DA, Miller WT, El-Abbassi AM, et al. Antimicrobial implications of vitamin D. Dermatol Endocrinol. 2011;3(4):220–229. doi:10.4161/derm.3.4.15027
44. Veldman CM, Cantorna MT, DeLuca HF. Expression of 1,25-dihydroxyvitamin D(3) receptor in the immune system. Arch Biochem Biophys. 2000;374(2):334–338. doi:10.1006/abbi.1999.1605
45. Wang TT, Nestel FP, Bourdeau V, et al. Cutting edge: 1,25-dihydroxyvitamin D3 is a direct inducer of antimicrobial peptide gene expression. J Immunol. 2004;173(5):2909–2912. doi:10.4049/jimmunol.173.5.2909
46. El Shahawy MS, Hemida MH, Ibrahim El Metwaly I, Shady ZM. The effect of vitamin D deficiency on eradication rates of Helicobacter pylori infection. J Gastroenterol Hepatol. 2018;2(6):270–275.
47. Al-Fartosi KG. The association between ABO blood group and spontaneous abortion. Albasrah J Sci. 2008;26(1):1–10.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
